Abstract
Introduction
Whether and how mesoderm posterior 1 (MESP1) plays a role in the proliferation of gastric cancer cells remain unclear.
Methods
The expression of MESP1 was compared in 48 human gastric cancer tissues and adjacent normal tissues. Knockdown of MESP1 was performed to investigate the role of MESP1 in the proliferation and apoptosis of BGC-823 and MGC-803 gastric cancer cells. Knockdown of alternative reading frame (ARF) was performed to study the role of ARF in the inhibitory effect of MESP1 knockdown on cell proliferation in gastric cancer cells. Mouse subcutaneous xenograft tumor model bearing BGC-823 cells was used to investigate the role of MESP1 in the growth of gastric tumor in vivo. The effect of seven active ingredients from T. terrestris on MESP1 expression was tested. The anti-cancer effect of diosgenin was confirmed in gastric cancer cells. MESP1 dependence of the anti-cancer effect of diosgenin was confirmed by MESP1 knockdown.
Results
MESP1 was highly expressed in human gastric cancer tissues (p < 0.05). MESP1 knockdown induced apoptosis and up-regulated the expression of ARF in gastric cancer cells (p < 0.05). Knockdown of ARF attenuated the anti-cancer effect of MESP1 knockdown (p < 0.05). In addition, MESP1 knockdown also suppressed tumor growth in vivo (p < 0.05). Diosgenin inhibits both mRNA and protein expression of MESP1 (p < 0.05). MESP1 knockdown attenuated the anti-cancer effect of diosgenin (p < 0.05).
Conclusions
MESP1 promotes the proliferation of gastric cancer cells via inhibiting ARF expression. Diosgenin exerts anti-cancer effect through inhibiting MESP1 expression in gastric cancer cells.
Introduction
Gastric cancer (GC) is one of the most common cancer types worldwide. 1 The therapeutic strategies for advanced GC remain unsatisfying and limited. Therefore, it is still imperative to fully elucidate the mechanisms underlying GC progression. Mesoderm posterior 1 (MESP1) belongs to the family of basic helix-loop-helix transcription factors, which transiently expresses in nascent mesoderm of mice at E6.5-E7.0. It is a master regulator of mesendoderm development, leading to formation of organs such as heart. 2 MESP1 could bind to canonical E-box motif (CACGTG) to trigger the expression of a cascade of lineage-specific transcription factors. 3 Functional loss of MESP1 in mouse embryos develops cardiac malformation, leading to embryonic lethality at E10.5. 4 In another study, MESP1 combined with ETS2 could transdifferentiate human dermal fibroblasts into cardiac progenitors. 5 Besides, MESP1 could also regulate hematopoietic and skeletal myogenic differentiation. 6 Despite that MESP1 has been established to play a critical role in the tumorigenesis of non-small cell lung cancer (NSCLC), 7 broad evidences demonstrating a role of MESP1 played in cancer biology are still not available. Thus, whether MESP1 is involved in the pathological process of GC and could be a therapeutical target for GC remain unclear.
Alternative reading frame (ARF), encoded by the CDKN2a locus, is one of the most frequently mutated or deleted tumor suppressors in human cancer. 8 The major function of ARF is to stabilize and activate p53, resulting in cellular senescence or apoptosis.9, 10 Importantly, ectopic MESP1 expression cooperates with loss of tumor suppressor ARF to promote the progression of NSCLC. 7 In GC, ARF also plays an important role,11, 12 but the relationship among MESP1, ARF, and GC remains unknown.
Tribulus terrestris (TT) is a plant that belongs to the family of Zygophyllaceae, and it is used individually as a single therapeutic agent or as a prime or subordinate component of many compound formulations and food supplements (health food) in China. The dry fruits of TT, as a traditional herb medicine (commonly named “Ji Li”), have been used for more than thousands of years in China within the context of protecting the liver, activating blood circulation, improving eyesight, and relieving itching (Chinese Pharmacopoeia, 2015 version). Modern studies have shown that TT has various pharmacological effects including anti-inflammatory, 13 anti-oxidant, 14 anti-bacterial, 15 anti-aging, 16 and anti-tumor activities.17,18 The main active ingredients of T. terrestris include diosgenin, ruscogenin, hecogenin, gitogenin, tribuloside, astragalin, and harmine; 19 however, which ingredient exerts the anti-tumor activity and its underlying mechanisms remain unclear.
In current study, we have employed human GC samples and GC cells lines to demonstrate that MESP1 promotes the proliferation of GC cells via inhibiting the expression of ARF. Diosgenin inhibits MESP1 expression, leading to suppressed proliferation of GC cells via inducing ARF expression. These findings provide a potential approach for the treatment of GC.
Materials and methods
Agents and cell culture
Fetal bovine serum (FBS) was purchased from Gibco Company (Grand Island, USA). Roswell Park Memorial Institute (RPMI)-1640 medium was obtained from KeyGEN BioTECH (Nanjing, China). Penicillin, streptomycin, Hoechst 33342, and MTT were purchased from Beyotime Biotech (Nanjing, China). Annexin V/PI staining kit was purchased from Vazyme Biotech (Nanjing, China). Diosgenin, ruscogenin, hecogenin, gitogenin, tribuloside, astragalin, and harmine compounds (purity ≥ 98%) were purchased from Sigma-Aldrich (St Louis, USA).
BGC-823 and MGC-803 human GC cell lines were purchased from American Type Culture Collection and cultured in RPMI-1640 medium containing 10% FBS, 1% penicillin (100 U/mL) and streptomycin (100 U/mL). The cells were maintained at 37°C, in a humidified incubator with 5% CO2 and in mid-log phase were used.
Human GC samples
The characteristics of patients.

Quantitative real-time polymerase chain reaction (RT-qPCR)
Primer sequences for RT-qPCR.

Immunohistochemical study
Thick sections (3 μm) of human GC tissues and corresponding pericarcinomatous tissues, shNC and shMESP1 xenograft tumor tissues were fixed in 4% paraformaldehyde, followed by deparaffinization, rehydration, and antigen retrieval. Sections were blocked in PBS with 10% goat serum (Gibco, USA), incubated with described antibodies and horseradish peroxidase (HRP) tagged goat anti-rabbit as a secondary antibody. After washing, samples were incubated in diaminobenzidine (Sigma-Aldrich, USA) for 5 min and counterstained in hematoxylin. Tissue slides were scored in a blinded fashion. No staining was observed with a negative control rabbit anti-IgG antibody. The images were measured blindly by one observer using Image-Pro Plus (Media Cybernetics, Silver Spring, USA).
Western blot
Cell lysates were prepared in radio immunoprecipitation assay lysis buffer buffer (20 mM Tris-HCl, pH 7.5, 0.1% (w/v) sodium lauryl sulfate, 0.5% (w/v) sodium deoxycholate, 135 mM NaCl, 1% (v/v) Triton X-100, 10% (v/v) glycerol, 2 mM ethylene diamine tetraacetic acid), supplemented with protease inhibitor and phosphatase inhibitor (Beyotime, Jiangsu, China). The total protein concentration was determined using the Bradford-based Bio-Rad assay (Hercules, CA, USA) and quantified using a BioTek SynergyMx (Winooski, VT, USA). Cell lysates in Sodium dodecyl sulfate (SDS)-sample buffer were incubated for 15 min at 95°C and equal amounts of total protein were analyzed by SDS-polyacrylamide gel electrophoresis and western blot. The antibodies used in this study are rabbit anti-MESP1, rabbit anti-p-mTOR (Ser2448), rabbit anti-p-p70S6k (Ser371), and rabbit anti-ARF antibodies and were purchased from Cell Signaling Technology (Cell Signaling Technology, Danvers, USA). Anti-rabbit IgG, HRP-linked antibody, rabbit anti-mTOR, rabbit anti-p70S6k, rabbit anti-Caspase 3, rabbit anti-Caspase 9, rabbit anti-PARP, and rabbit anti-GAPDH were obtained from ABclonal (Wuhan, China). All primary antibodies were used at 1:1000, and all secondary antibodies were used at 1:10, 000.
MESP1 knockdown and transfection
siRNA sequences.

MTT assay
BGC-823 and MGC-803 cells were seeded into 96-well plate, and each well contains about 1000 cells. Before the end of the experiment, 20 μL MTT (5 mg/mL) was added to each well for 4 h incubation. Then, the liquid in each well was excluded, and 150 μL DMSO was added to each well. The absorption value at the wavelength of 490 nm was measured, and the inhibition rate was calculated according to the absorption value.
Cell proliferation assay
3 × 104 cells were plated in 12-well culture plates. Cell number was counted using automatic counter (Countstar) every 24 h to assess cell growth.
Clone formation assay
After establishment of BGC-823 and MGC-803 cells with stable MESP1 knockdown, 1000 cells within five passages were seeded to six-well culture plates. After culture for 14 days, the cells were stained with crystal violet solution (Beyotime, Jiangsu, China). The number of colonies was then counted macroscopically. Clone numbers were quantified by ImageJ.
Cell cycle and apoptosis analysis kit
After establishment of BGC-823 and MGC-803 cells with stable MESP1 knockdown, cell apoptosis and cell cycle were analyzed by the Annexin V-FITC Apoptosis Detection kit (Vazyme Biotech, Jiangsu, China) in accordance with the manufacturer’s instructions. Cells were Annexin V/PI double stained to detect apoptosis. Cells were PI stained to detect cell cycle. Flow cytometric analysis was performed immediately after supravital staining. Data acquisition and analysis were performed in a Becton Dickinson Fluorescence activated Cell Sorting-Calibur fow cytometer using the Cell Quest software (Franklin Lakes).
Hoechst 33342 staining
Hoechst 33342 staining of cells was performed according to the manufacture’s instruction, and apoptotic cells were identified on the basis of morphological changes in nuclear assembly involving chromatin condensation and fragmentation. Morphological changes were quantified by ImageJ.
ARF siRNAs
siRNA sequences.

In vivo tumorigenicity
In vivo tumorigenicity experiment was carried out according to previous studies.20,21 Female athymic BALB/c nude mice (5–6 weeks old) with body masses ranging from 18 to 22 g were supplied by the Shanghai Institute of Material Medica, Chinese Academy of Sciences. For subcutaneous (S.C) injection of GC cells, sub-confluent BGC-823 cells were collected in serum-free medium (1 × 106 cells/100 μL). Then, the cell suspension was injected subcutaneously into mice in one flank (n = 5–6). All assays were repeated at least three times. Animal care and surgery protocols were approved by the Ethics Committee for Animal Experiments of Bengbu Medical College (Permission Number: 2020-033). All animals were treated appropriately and used in a scientifically valid and ethical manner.
Statistical analysis
Data are the mean ± SD of triplicate independent experiments performed in a parallel manner unless otherwise indicated. Statistically significant differences (one-way analysis of variance (ANOVAs) followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software according to previous studies.22,23 A value of p < 0.05 was considered significant, and values of p < 0.01, p < 0.001 and p < 0.0001 were considered highly significant, ns meant no significance.
Results
MESP1 is highly expressed in human GC tissues
In order to investigate whether MESP1 expression is correlated with GC, the mRNA expression of MESP1 in human GC tissues was analyzed. As shown in Figure 1(a), the mRNA level of MESP1 was significantly higher in GC tissues compared with pericarcinomatous tissues. Next, immunohistochemical staining and western blot were used to analyze MESP1 protein expression at different pathological stages of GC. The results showed that MESP1 was also up-regulated at protein level in GC tissues and the expression was positively correlated with the pathological stages of GC (Figure 1(b) and (c)). Taken together, these results indicate that MESP1 is highly expressed in human GC tissues. MESP1 was highly expressed in human gastric cancer tissues. (a) Total RNA samples from 48 human gastric cancer tissues and corresponding pericarcinomatous tissues were harvested. The mRNA expression of MESP1 was detected by RT-qPCR (n = 48). (b) Immunohistochemical staining was performed to detect MESP1 expression in gastric cancer tissues with different pathological stages. Representative images were captured. Original magnification, ×200(n = 3). (c) Western blot was used to detect MESP1 expression in gastric cancer tissues with different pathological stages. GAPDH was used as loading control (n = 3). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. **p < 0.01; ***p < 0.001; ****p < 0.0001.
Knockdown of MESP1 inhibits the proliferation of gastric cancer cells
In order to study the role of MESP1 in the progression of GC, MESP1 knockdown was performed in BGC-823 and MGC-803 GC cells using lentiviral vector. The knockdown efficiency at protein level is shown in Figure 2(a). MESP1 knockdown decreased the cell viability of BGC-823 and MGC-803 GC cells (Figure 2(b)). The proliferation of BGC-823 and MGC-803 GC cells was also suppressed by MESP1 knockdown (Figure 2(c) and (d)). The inhibitory effect of MESP1 knockdown on the proliferation of BGC-823 and MGC-803 GC cells was also verified by siRNA-mediated MESP1 knockdown (Supplemental Figure 1A–C). In addition, suppressed mTOR-p70S6k signaling axis demonstrated the inhibitory effect of MESP1 knockdown on the proliferation of BGC-823 and MGC-803 GC cells at molecular level (Figure 2(e)). In summary, these findings suggest that knockdown of MESP1 could inhibit the proliferation of GC cells. Knockdown of MESP1 inhibited the proliferation of BGC-823 and MGC-803 gastric cancer cells. (a) BGC-823 and MGC-803 gastric cancer cells were transfected with MESP1 shRNA lentivirus and negative control (NC) lentivirus for 72 h, protein samples were harvested and the expression of MESP1 was analyzed by western blot (n = 3). (b) MTT assay was used to detect the effect of MESP1 knockdown on cell viability in BGC-823 and MGC-803 gastric cancer cells (n = 3). (c) Proliferation assay was used to analyze the effect of MESP1 knockdown on cell proliferation of BGC-823 and MGC-803 gastric cancer cells (n = 3). (d) 1000 MESP1 knockdown or NC cells were seeded into six well plates for 14 days. The cells were stained with crystal violet solution and representative images were captured (n = 3). (e) Western blot was used to detect the expression of indicated proteins. GAPDH was used as loading control (n = 3). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. **p < 0.01; ***p < 0.001; ****p < 0.0001.
Knockdown of MESP1 induces apoptosis in gastric cancer cells
Apoptosis is a common manner of death of cancer cells.
24
The result of Annexin V/PI staining and Hoechst 33342 staining showed that MESP1 knockdown induced apoptosis in both BGC-823 and MGC-803 GC cells (Figure 3(a) and (b)). To investigate whether MESP1 knockdown could influence cell cycle, cells were stained with PI followed by flow cytometry analysis. The result showed that MESP1 knockdown did not have obvious effect on cell cycle in BGC-823 and MGC-803 GC cells (Supplemental Figure 2). Moreover, the cleavage of Caspase 3, Caspase 9, and PARP increased, suggesting that MESP1 induced apoptosis of BGC-823 and MGC-803 GC cells at molecular level (Figure 3(c)). Collectively, these results indicate that MESP1 knockdown induces apoptosis in GC cells. Knockdown of MESP1 induced apoptosis in BGC-823 and MGC-803 gastric cancer cells. (a) Cells were stained with Annexin/PI. Apoptotic cells were detected by flow cytometry in BGC-823 and MGC-803 gastric cancer cells after MESP1 knockdown (n = 3). (b) The effect of MESP1 knockdown on cell apoptosis was detected by Hoechst 33342 staining. Representative morphological images of nucleus were captured. Original magnification, ×200 (n = 3). (c) Western blot was used to detect the expression of indicated proteins. GAPDH was used as loading control (n = 3). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. **p < 0.01; ***p < 0.001.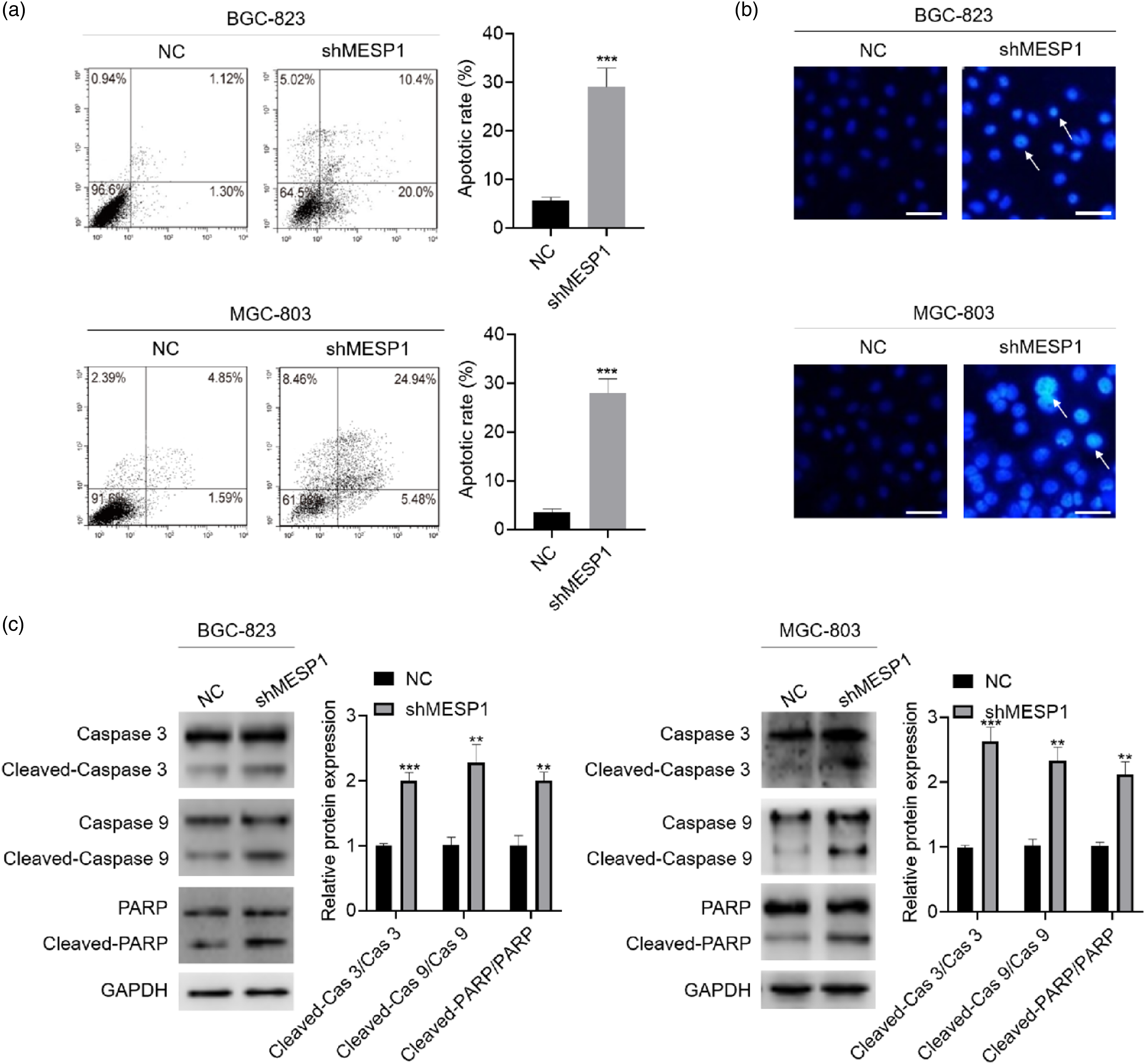
MESP1 promotes the proliferation of gastric cancer via down-regulation of ARF
Based on the previous study, MESP1 promotes the tumorigenesis of NSCLC via inhibiting the expression of the tumor suppressor, ARF.
7
Therefore, we investigate whether MESP1 promoted the proliferation of GC cells via inhibiting ARF expression. As shown in Figure 4(a), knockdown of MESP1 up-regulated ARF expression in both BGC-823 and MGC-803 GC cells (Figure 4(a)). The expression of ARF was also negatively correlated with the pathological progress of human GC (Figure 4(b)). These findings suggest that ARF is correlated with GC. In order to investigate the role of ARF in MESP1 knockdown-caused inhibitory effect on cell proliferation in GC, ARF knockdown was performed using siRNAs targeting ARF. Among three tested siRNAs, siARF#2 and siARF#3 showed better efficiency on ARF knockdown in both BGC-823 and MGC-803 GC cells (Figure 4(c)). Thus, siARF#2 and siARF#3 were chosen to address the following experiments. Flow cytometry was performed to study the effect of ARF knockdown on MESP1 knockdown-induced apoptosis in BGC-823 and MGC-803 GC cells; the result showed that ARF knockdown significantly attenuated the pro-apoptotic effect of MESP1 knockdown (Figure 4(d)). These results demonstrate that MESP1 suppresses ARF expression, and knockdown of ARF could attenuate the pro-apoptotic effect of MESP1 knockdown in GC cells. Furthermore, ARF knockdown weakened the inhibitory effect of MESP1 knockdown on cell viability in BGC-823 and MGC-803 GC cells (Figure 5(a)). The inhibitory effect of MESP1 knockdown on cell proliferation in BGC-823 and MGC-803 GC cells was also attenuated by ARF knockdown (Figure 5(b) and (c)). Consistently, ARF knockdown also attenuated the inhibitory effect of MESP1 knockdown on mTOR-p70S6k signaling axis (Figure 5(d)). In conclusion, these results demonstrate that MESP1 knockdown inhibits the proliferation of GC cells via increasing ARF expression. MESP1 knockdown induced apoptosis via up-regulating ARF expression in BGC-823 and MGC-803 gastric cancer cells. (a) ARF expression after MESP1 knockdown was detected by western blot in BGC-823 and MGC-803 gastric cancer cells (n = 3). (b) Western blot was used to detect ARF expression in gastric cancer tissues with different pathological stages. GAPDH was used as loading control (n = 3). (c) ARF knockdown was performed using specific siRNAs targeting ARF. BGC-823 and MGC-803 gastric cancer cells were transfected with ARF siRNAs with Lipofectamine 3000 transfection reagent for 48 h, protein samples were collected, and ARF knockdown efficiency was analyzed by western blot (n = 3). (d) ARF knockdown was performed in BGC-823 and MGC-803 gastric cancer cells with MESP1 knockdown. siARF#2 and siARF#3 were transfected with Lipofectamine 3000 transfection reagent for 48 h. Then cells were stained with Annexin/PI. Flow cytometry was used to detect apoptotic cells (n = 3). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. *p < 0.05; **p < 0.01; ***p < 0.001. Knockdown of MESP1 inhibited the proliferation of BGC-823 and MGC-803 gastric cancer cells via up-regulating ARF expression. ARF knockdown was performed using siARF#2 and siARF#3 in BGC-823 and MGC-803 gastric with MESP1 knockdown. The cells were subjected with following experiments. (a) Cell viability was measured by MTT assay (n = 3). (b) Cell proliferation assay. ARF knockdown was performed using siARF#3 in BGC-823 and MGC-803 gastric with MESP1 knockdown. The cells were subjected with following experiments (n = 3). (c) Clone formation assay (n = 3). (d) Western blot was used to detect the expression of indicated proteins. GAPDH was used as loading control (n = 3). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. *p < 0.05; **p < 0.01; ***p < 0.001. ns meant no significance.

MESP1 knockdown inhibits tumor growth in vivo
Next, we employed BGC-823 xenograft model to investigate the inhibitory effect of MESP1 knockdown on tumor growth in vivo. Knockdown of MESP1 significantly suppressed tumor growth in vivo (Figure 6(a)–(c) and Table 5). Immunohistochemical staining and western bolt showed that MESP1 expression was lower and ARF expression was higher in shMESP1 group (Figure 6(d) and (e)). The mTOR-p70S6k axis was also inhibited by MESP1 knockdown in vivo (Figure 6(e)). In summary, these results indicate that MESP1 knockdown inhibits the growth of gastric tumors in vivo. Knockdown of MESP1 inhibited the growth of gastric tumors in vivo. Sub-confluent MESP1 knockdown BGC-823 cells and NC BGC-823 cells were collected in serum-free medium (1 × 106 cells/100 μL). Then, the cell suspension was injected subcutaneously into mice in one flank (n = 5–6). (a) Tumor size was measured every 2 days for 2 weeks (n = 5–6). (b) The picture of tumors was captured (n = 5–6). (c) Tumor weight. (d) Immunohistochemical staining was performed to detect the expression of MESP1 and ARF in tumors collected from mice. Representative images were captured (n = 3). (e) Western blot was used to detect the expression of indicated proteins. GAPDH was used as loading control (n = 2). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. **p < 0.01; ***p < 0.001; ****p < 0.0001. Tumor size and weight in day 14.

Diosgenin inhibits the proliferation of gastric cancer cells via inhibiting MESP1 expression in gastric cancer cells
To investigate whether there is an ingredient of T. terrestris which could perform anti-cancer effect via inhibiting MESP1, a small-scale screening on targeting MESP1 mRNA expression was performed. As shown in Figure 7(a), among seven tested active ingredients, diosgenin showed desirable inhibitory effect on MESP1 mRNA expression in both BGC-823 and MGC-803 GC cells. In addition, diosgenin inhibited both mRNA and protein expression of MESP1 in a concentration-dependent manner in these two cell lines (Figure 7(b) and (c)). Consistently, diosgenin also induced ARF1 protein expression in a concentration-dependent manner in BGC-823 and MGC-803 GC cells (Figure 7(c)). Diosgenin was reported to possess an anti-cancer effect on a series of cancer types.25,26 Consistent with previous studies, diosgenin inhibited the proliferation of both BGC-823 and MGC-803 GC cells in our study (Figure 7(d)). To confirm whether diosgenin performed anti-cancer effect on BGC-823 and MGC-803 GC cells through inhibiting MESP1 expression, the anti-proliferation effect of diosgenin was checked in MESP1 knockdown BGC-823 and MGC-803 GC cells. The results showed that diosgenin could suppress the proliferation of GC cells through inhibiting MESP1 expression. Diosgenin inhibited the proliferation of gastric cancer cells via inhibiting MESP1 expression in gastric cancer cells. (a) BGC-823 and MGC-803 gastric cancer cells were treated with 10 μM indicated active ingredients from TT for 24 h. Total RNA samples were harvested and the mRNA expression of MESP1 was checked by RT-qPCR (n = 3). (b) BGC-823 and MGC-803 gastric cancer cells were treated with 5, 10, 20 μM diosgenin for 24 h. Total RNA samples were harvested and the mRNA expression of MESP1 was checked by RT-qPCR (n = 3). (c) BGC-823 and MGC-803 gastric cancer cells were treated with 5, 10, 20 μM diosgenin for 24 h. Total protein samples were harvested, and the protein expression of MESP1 and ARF1 was checked by western blot (n = 3). (d) BGC-823 and MGC-803 gastric cancer cells were treated with 5, 10, 20 μM diosgenin for 24 h; cell viability was measured by MTT assay (n = 3). (e) BGC-823 and MGC-803 gastric cancer cells were previously transfected with MESP1 shRNA lentivirus and negative control lentivirus for 72 h; then, cells were treated with 10 μM diosgenin for another 24 h. The cell viability was checked by MTT assay (n = 3). Statistically significant differences (one-way ANOVAs followed by Bonferroni’s Multiple Comparison Test) were determined using GraphPad Prism 8 software. **p < 0.01; ***p < 0.001; ****p < 0.0001.
Discussion
Despite the increasing knowledge and understanding of GC, effective treatment of GC is still urgently needed. MESP1 is critical for NSCLC; 7 however, its role in GC remains unclear. In the present study, we found that MESP1 was highly expressed in human GC tissues and the expression of MESP1 was positively correlated with the pathological stages of GC. MESP1 promoted the proliferation of GC cells via inhibiting ARF expression. Importantly, diosgenin from TT exerts anti-cancer effect against GC cells via inhibiting MESP1 expression. Thus, our study suggests that MESP1 could be a potential therapeutic target and diosgenin could be a potential natural product for the treatment of GC.
MESP1 reportedly played important roles in the development of heart,6,27–29 and it was rarely reported in tumors. MESP1 was first found to play a critical role in NSCLC; 7 however, its role in GC was not clear. Thus, the expression of MESP1 in human GC tissues was analyzed by RT-qPCR to explore the correlation between MESP1 expression and GC. Immunohistochemical staining and western blot were then used to analyze MESP1 expression in different pathological stages. MESP1 knockdown was performed to investigate the role of MESP1 in the proliferation of GC cells. Apoptosis is a common reason for inhibited proliferation of cancer cells. 24 Hence, we analyzed the effect of MESP1 on cell apoptosis in GC cells. As expected, MESP1 knockdown significantly induced apoptosis in GCs. Mechanistically, MESP1 knockdown induced apoptosis and inhibited proliferation in GC cells via up-regulating ARF expression. ARF, a tumor suppressor, could exert anti-cancer effect via stabilizing p53.30,31 p53, is a critical regulator of cell apoptosis. 32 Moreover, p53 could inhibit mTOR activity in a Sestrin1/Sestrin2-AMPK-TSC2 axis-dependent manner. 33 Thus, it could be speculated that MESP1 knockdown-induced apoptosis and inhibition of mTOR activity was mediated by p53. Thus, given the powerful anti-cancer function of ARF and p53, it is in accordance with expectation that genetic inhibition of MESP1 displayed obvious anti-cancer effect in vivo.
Active ingredients from TT have been reported to battle against cancers by several previous studies.17,34,35 However, which ingredient exert the main function and the underling mechanisms have not been fully understood. We first identified MESP1 as a therapeutic target for GC and screened main active ingredients from TT based on the inhibitory effect of these ingredients on MESP1 expression. Diosgenin was previously reported to suppress tumor growth or metastasis of prostate cancer, 25 glioblastoma, 36 breast cancer, 37 cholangiocarcinoma, 38 and pancreatic cancer. 39 However, mechanisms involved in the anti-cancer effect of diosgenin against different cancer type are distinct and may not imitable in GC. Our data demonstrate that MESP1 as a mediator of diosgenin’s anti-cancer effect, advancing the mechanisms of diosgenin in cancer therapy. Nonetheless, the mechanism through which diosgenin suppressed MESP1 expression remains unclear. It was reported that transforming growth factor-β (TGF-β) and Wnt signaling was correlated with MESP1 in cardiomyocytes. 40 Particularly, diosgenin could also modulate TGF-β and Wnt signaling in various cell types,41–43 leading to the concern that diosgenin may regulate MESP1 expression via modulating TGF-β and Wnt signaling.
Besides, our data (Supplemental Figure 3) also showed that MESP1 knockdown inhibited the migration of BGC-823 and MGC-803 GC cells, suggesting that MESP1 may possess a role in regulating the migration of GC cells. Up to now, there have been no reports of the regulation of MESP1 on the migration of cancer cells. Thus, this might be the first report to uncover the role of MESP1 in regulating the migration of cancer cells. Nonetheless, a potential regulation of MESP1 on migration could also be traced in literature. MESP1 induces features of epithelial-mesenchymal transition (EMT) in differentiating embryonic stem cells (ESCs) and promotes development of mesoderm precursors of the cardiovascular lineage.28,44 Consistently, MESP1 also plays a vital role in vascular progenitor cells. MESP1 knockdown cells are less likely to undergo EMT during vascular progenitor differentiation. 45 As EMT is a precondition of the migration and metastasis of cancer cells, 46 it is not difficult to speculate that MESP1 might play a role in the migration and metastasis of GC cells. However, the underlying regulatory mechanisms of MESP1 in the migration of GC cells were not mentioned in our present study. This is an attractive study and will be further studied in our future studies.
There are some limitations of our study. The mechanism through which MESP1 inhibits ARF expression has not been investigated. In addition, searching for a small molecular specifically targeting MESP1 is another urgent thing for further study. Virtual screening and small molecule compound library screening will be performed in our future studies.
In conclusion, the present study demonstrates that MESP1 promotes the proliferation of GC cells via inhibiting ARF expression. Diosgenin inhibits the proliferation of GC cells through down-regulation of MESP1.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211053292 – Supplemental Material for Diosgenin inhibits the proliferation of gastric cancer cells via inducing mesoderm posterior 1 down-regulation-mediated alternative reading frame expression
Supplemental Material, sj-pdf-1-het-10.1177_09603271211053292 for Diosgenin inhibits the proliferation of gastric cancer cells via inducing mesoderm posterior 1 down-regulation-mediated alternative reading frame expression by Lin Gu, Hailun Zheng, Rui Zhao, Xiaojing Zhang and Qizhi Wang in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. This research project was financially supported by Key Natural Science Project of Bengbu Medical College (Grant No. BYKY2019085ZD) and the Natural Science Key Program of College and University of Anhui Province (Grant No.KJ2019A0361).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
