Abstract
Introduction:
lncRNA DLEU2 has been indicated to act a crucial role in the bioprocess of cancer. But, the role and molecular mechanisms of lncRNA DLEU2 in gastric cancer (GC) remain unknown.
Methods:
The correlation of DLEU2 or miR-30a-5p with the clinical parameters and outcomes of GC patents was implemented by TCGA cohort. Cell viability and invasion were estimated by MTT and Transwell assays. The interplay between DLEU2 and miR-30a-5p was confirmed by a luciferase report assay. The effects of DLEU2 on miR-30a-5p or MMP2/9 were detected by qRT-PCR and Western blot.
Results:
We found that the enhanced expression of DLEU2 was linked to the pathological stage and poor survival in GC patients. Overexpressing DLEU2 prompted the cell proliferation and invasion of AGS cells, but silencing DLEU2 weakened these effects in MKN-28 cells. In addition, DLEU2 could negatively modulate and be bound with miR-30a-5p, which could counteract DLEU2-caused cell proliferation. High expression of miR-30a-5p was linked to a favorable survival in patients with GC.
Conclusion:
Our findings suggested that lncRNA DLEU2 could favor the growth of GC by downregulating miR-30a-5p.
Introduction
Though the incidence of gastric cancer (GC) is decreasing duo to the application of endoscopic treatment, it still harbors a higher mortality in China. 1 Aberrant expression of noncoding RNAs (ncRNAs) is involved in the progression of GC and represents the potential markers for GC patients.2,3
Accumulating data have indicated that deleted in lymphocytic leukemia 2 (DLEU2) as a long ncRNAs (lncRNA) acts a pivotal role in cancer. The expression levels of DLEU2 are elevated in multiple malignancies, such as non-small cell lung cancer (NSCLC),4,5 hepatocellular carcinoma (HCC), 6 esophageal cancer (EC),7,8 and pancreatic cancer (PC). 9 Increased expression of DLEU2 shows a relationship with tumor size, vascular invasion 6 and poor prognosis in NSCLC and EC.4,7–9 Functionally, it is indicated that DLEU2 encourages the tumor bio-behaviors of NSCLC and HCC cells,4–6 and silencing DLEU2 has the opposite effects.7,9
But, lncRNA DLEU2 is downregulated in acute myeloid leukemia (AML) and suppresses the proliferation and colony formation in AML and chronic lymphocytic leukemia (CLL).10,11 The function of DLEU2 in GC is still unclear. Herein, upregulation of DLEU2 was related to pathological stage and poor survival in GC patients. DLEU2 promotes the proliferation of GC cells by downregulating miR-30a-5p and indicates a worse prognosis in patient with GC.
Materials and methods
Clinical samples
The clinical information of GC patients including their survival time and status and DLEU2/miR-30a-5p expression levels from 367 cases of GC samples or 34 paired GC tissues were collected from The Cancer Genome Atlas (TCGA) database (https://genome-cancer.ucsc.edu). Five paired GC samples were stored in liquid nitrogen. Ethical approval for this study was obtained from the Ethics Committee of Shanghai Sixth People’s Hospital (YS-2017-003).
Materials
GC cell lines (AGS, HGC-27, MKN-28) were from the cell bank of Chinese Academy of Sciences. DLEU2 plasmids/pEX-3 and DLEU2 siRNA (si-SNHG8)/si-NC were from GenePharma (Shanghai, China).
Quantitative real-time PCR (qRT-PCR)
The GC cell lines (AGS, HGC-27, MKN-28) and GES-1 were stored in our laboratory. Total RNA was isolated by Trizol reagent (Invitrogen, USA). The cDNA was amplified by using a PrimeScript™ Reverse Transcription Kit (TakaRa, Japan) in an ABI 7500 System (Applied Biosystems; Thermo Fisher Scientific). The primer sequences of DLEU2 and miR-30a-5p were synthesized by Shanghai Sangon Biotech (Shanghai, China) and listed in Supplementary Table S1. GAPDH or U6 was used as the internal control. All qPCR reactions were performed in duplicate.
Western blot analysis
Western blot analysis was conducted as previously described. 3 Primary antibodies against MMP-2 (ab92536, Abcam, Cambridge, UK) and MMP9 (ab73734) were diluted according to the instructions and incubated overnight at 4°C.
SiRNA transfection
Plasmids mediated siRNA targeting DLEU2 (si-DLEU2, 5′-GGAGAAGGGAA ATAAGCTT-3′) were purchased from GenePharma (Shanghai, China) and the negative control (si-NC) was used as the control vector. MKN-28 cell line was planted in 6-well plates 24 h prior to si-DLEU2 transfection with 50% to 60% confluence, and then were transfected with Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacture instructions.
MTT and Transwell assays
They were conducted as previously described. 3 These studies were repeated at least three times.
Dual-luciferase gene reporter assay
WT or Mut DLEU2 3’ UTR vectors were established by annealing double-strand DNA and inserted into the PRL-TK vectors. GC cells were seeded into 24-well plates. After 24h incubation, luciferase report vector carrying WT or Mut DLEU2 3’UTR was co-transfected with miR-30a-5p mimic or inhibitor into AGS or MKN-28 cells. Luciferase activities were detected after the transfection for 48 h. This study was repeated at least three times.
Statistical analysis
Statistical analyses were conducted by SPSS 20.0 (IBM, SPSS, Chicago, IL, USA) and GraphPad Prism 7.0. Student’s
Results
Upregulation of DLEU2 was linked to a poor survival in GC patients
The expression of DLEU2, indicated by qRT-PCR was elevated in five paired GC tissue samples (Figure 1(a)). This result was validated to be upregulated in 367 unpaired and 34 paired GC tissue samples (Figure 1(b)). A cutoff value (1.364) of DLEU2 was acquired and divided the patients into high DLEU2 expression and low DLEU2 expression groups (Figure 1(c)). As shown in Supplementary Table S2, elevated expression of DLEU2 indicated a positive association with pathological stage in GC patients. Kaplan–Meier analysis showed that the patients with high DLEU2 expression possessed a poorer survival (Figure 1(d)). Univariate and Multivariate cox regression analysis unveiled DLEU2 as an independent prognostic factor of poor survival in GC (Supplementary Table S3).

Elevated expression of DLEU2 was an independent prognostic factor of poor survival in GC patients. (a) qRT-PCR analysis of the expression of DLEU2 in five paired GC tissue samples. (b) TCGA cohort showed a significantly elevated expression of DLEU2 in 367 unpaired and 34 paired GC tissues. (c) The cutoff value of DLEU2 was acquired by a ROC curve and divided the patients into DLEU2 high expression group and low DLEU2 expression groups. (d) Kaplan–Meier analysis of the association of high or low DLEU2 expression with overall survival in GC patients.
DLEU2 favored the proliferation of GC
The expression of DLEU2 in multiple GC cell lines was examined by qRT-PCR analysis, which indicated that DLEU2 possessed a higher expression in MKN-28 but a lower expression in AGS (Figure 2(a)). Thus, the overexpression efficiencies of DLEU2 plasmids in AGS cell line or the knockdown efficiency of si-DLEU2 in MKN-28 was defined by qRT-PCR (Figure 2(b)). Ectopic expression of DLEU2 promoted the viability and invasion in AGS, but knockdown of DLEU2 reduced these effects (Figure 2(c) and (d)). In addition, Western blot analysis indicated that ectopic DLEU2 expression upregulated MMP2/9, but knockdown of DLEU2 downregulated their expression (Figure 2(e)).

DLEU2 enhanced the proliferation and invasion of GC cells. (a) qRT-PCR analysis of the expression levels of DLEU2 in GC cell lines. (b) qRT-PCR analysis of the overexpression efficiencies of DLEU2 plasmids in AGS cell line or knockdown efficiency of si-DLEU2 in MKN-28 cell line. (c, d) MTT and Transwell analysis of the effects of DLEU2 overexpression or knockdown on cell proliferation and invasion in GC cells. (e) Western blot analysis of the effects of DLEU2 overexpression or knockdown on MMP2/9 expression in GC cells.
Upregulation of miR-30a-5p was linked to a better survival in GC
We then identified the DLEU2 specific binding with miRNAs, and found that 7 miRNAs (miR-30e-5p, miR-30c-5p, miR-30a-5p, miR-30b-5p, miR-30d-5p, miR-374a-5p, miR-374b-5p) might have the potential to bind with DLEU2. It was miR-30a-5p that had a dramatical decrease in 385 unpaired and 41 paired GC tissue samples (Figure 3(a)). TCGA cohort indicated that DLEU2 was negatively correlated with miR-30a-5p expression in 282 GC tissue samples (Figure 3(b)). A cutoff value (11.86) of miR-30a-5p was acquired in GC patients by a cutoff Finder (Figure 3(c)), and divided the patients into two groups (Figure 3(d)). The patients with high miR-30a-5p expression displayed a better survival in comparison with those with low miR-30a-5p expression (Figure 3(e)).
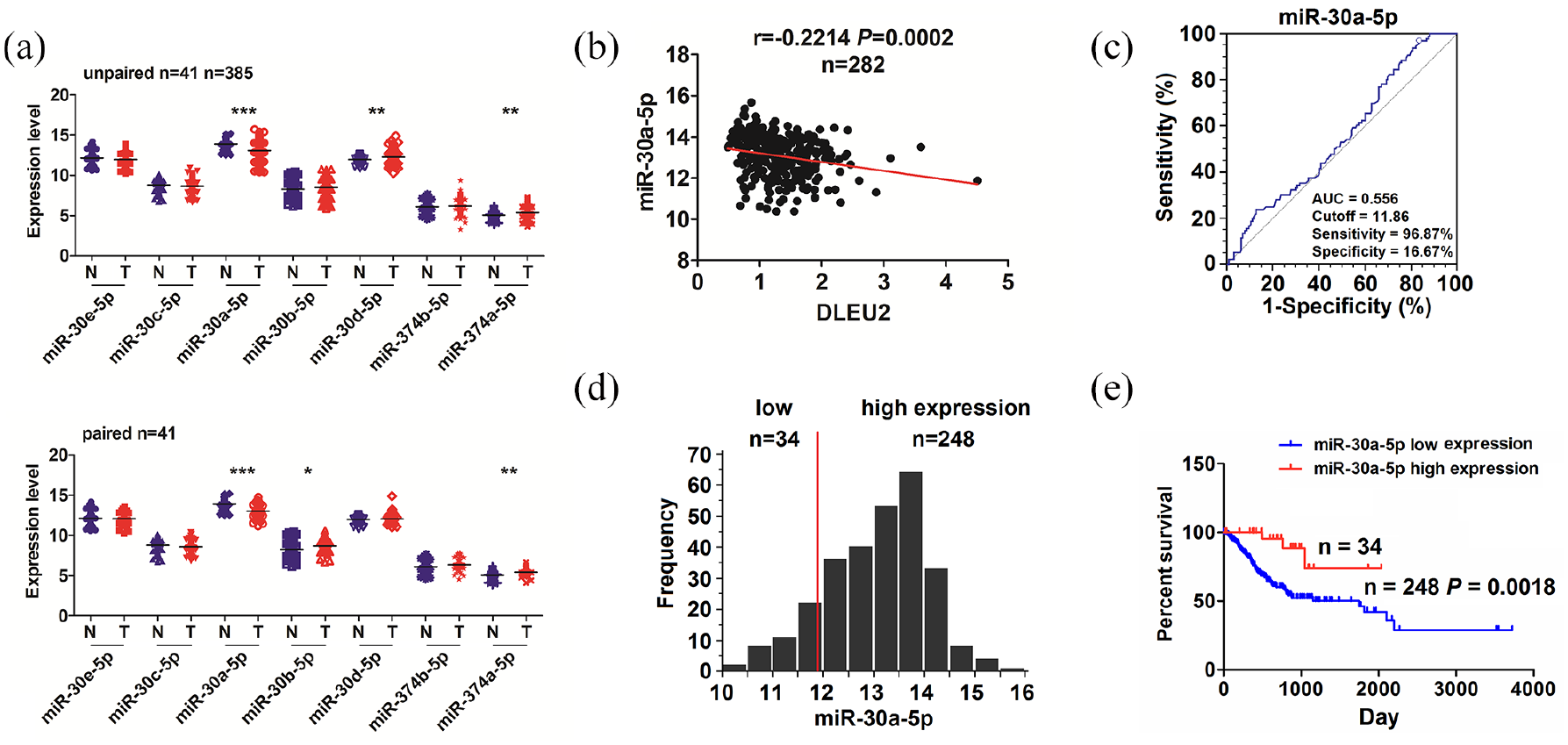
High expression of miR-30a-5p was associated with favorable survival in GC patients. (a) TCGA analysis of the expression levels of multiple miRNAs in 385 unpaired and 41 paired GC tissues. (b) Pearson correlation analysis of the correlation of DLEU2 with miR-30a-5p in GC tissues. (c, d) The cutoff value of miR-30a-5p was acquired by a ROC curve in GC, and divided the patients into miR-30a-5p high expression and low expression groups. (e) Kaplan–Meier analysis of the association of high or low miR-30a-5p expression with overall survival in GC patients.
DLEU2 negatively modulated miR-30a-5p expression in GC
The binding sites between miR-30a-5p and the WT or Mut DLEU2 3’UTR were provided in Figure 4(a). After WT or Mut DLEU2 3’UTR was co-transfected with miR-30a-5p into AGS or MKN-28 cells, miR-30a-5p lowered the luciferase activity of WT DLEU2 3’UTR, and its inhibitor reversed this effect (Figure 4(b)). qRT-PCR analysis indicated that overexpressing DLEU2 reduced the expression of miR-30a-5p, and silencing DLEU2 increased its expression (Figure 4(c)). In addition, after co-transfection with DLEU2 plasmids and miR-30a-5p mimic in AGS cells or si-DLEU2 and miR-30a-5p inhibitor in MKN-28 cells, MTT assay indicated that miR-30a-5p inhibited the cell viability and reversed DLEU2-induced cell proliferation in AGS cells, but miR-30a-5p inhibitor had the opposite effects (Figure 4(d)).

DLEU2 negatively regulated miR-30a-5p expression in GC cells. (a) The binding sites between miR-30a-5p and WT or Mut DLEU2 3’UTR. (b) The luciferase activity of WT DLEU2 3’UTR was reduced by miR-30a-5p mimic and increased by its inhibitor in AGS or MKN-28 cells. (c) qRT-PCR analysis of the effects of DLEU2 overexpression or knockdown on miR-30a-5p expression in AGS or MKN-28 cell line. (d) MTT analysis of the cell viability after co-transfection with DLEU2 plasmids and miR-30a-5p mimic in AGS cell line or si-DLEU2 and miR-30a-5p inhibitor in MKN-28 cell line.
Discussion
Accumulating evidence indicates that the elevated expression of DLEU2 is a frequent even in malignant tumors4–9 and associated with unfavorable prognosis in NSCLC and EC.4,8 However, the clinical significance of DLEU2 in GC is elusive. In coinciding with previous reports in other cancers,7–9 our findings showed that DLEU2 was upregulated in GC tissue samples. Elevated expression of DLEU2 was linked to pathological stage and worse survival in GC.
Previous reports showed that DLEU2 can act as an oncogene4–7 or tumor suppressive factor in cancers.10,11 In accordance with the studies,4–7 We found that ectopic expression of DLEU2 enhanced the growth of GC cells and knockdown of DLEU2 counteracted these effects. Moreover, DLEU2 can bind to EZH2 to favor HCC progression. 6 More knowledge have been shown about the regulation of DLEU2 on miRNAs in cancers.4,5,7,9 Herein, we identified DLEU2 specific binding with miR-30a-5p and found that DLEU2 could downregulate and bind to miR-542-3p in GC cells. DLEU2 also sponges miR-30a-5p/-30c-5p/-5e-5p to promote the progression of NSCLC and EC.4,5,7 These findings suggested that DLEU2 acted by regulating miR-30a-5p in GC.
It has been shown that miR-30a-5p is downregulated in prostate cancer, 12 osteosarcoma 13 gallbladder cancer, 14 breast cancer, 15 and inhibits the proliferation, migration and metastasis of these cancers.12–15 Besides, miR-30a-5p expression is decreased in GC and represses the proliferation of GC cells.16,17 In our study, downregulation of miR-30a-5p was associated with poor survival in GC patients, and miR-30a-5p counteracted DLEU2 induced cell proliferation in GC cells. In addition, more cohorts should be used for assessing the association of DLEU2 with the outcomes of the patients with gastric cancer, and the comprehensive role in vitro and in vivo and regulatory mechanisms of DLEU2 in gastric cancer need be further explored.
Conclusion
Our findings demonstrated that upregulated DLEU2 expression or downregulated miR-30a-5p expression was linked to a poor survival in GC patients. DLEU2 promoted the proliferation and invasion of GC cells by downregulating miR-30a-5p and represented a promising biomarker for GC.
Supplemental Material
Supplementary_Tables_1 – Supplemental material for lncRNA DLEU2 accelerates gastric cancer growth by downregulating miR-30a-5p
Supplemental material, Supplementary_Tables_1 for lncRNA DLEU2 accelerates gastric cancer growth by downregulating miR-30a-5p by Hua-Li Zhu and Jing Zou in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the ETHICS COMMITTEE of Shanghai Sixth People’s Hospital (YS-2017-003).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by General Project of Zhejiang Medical and Health science and Technology Plan (2019KY216).
Informed consent
Informed consent was not sought for the present study because the data were downloaded from the TCGA dataset.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
