Abstract
Background:
Gastric cancer is a malignant tumor with high incidence rate and mortality rate.
Purpose:
In this study, we investigated the anti-cancer effect of alantolactone, a sesquiterpene lactone, on gastric cancer cell lines BGC-823 and SGC-7901.
Methods:
BGC-823 and SGC-7901 cells were treated with different concentrations of alantolactone, Hoechst 33258 staining, flow cytometry, wound healing assay, invasion assay, colony forming assay, quantative polymerase chain reaction, and western blot analysis were used to evaluate the anticancer activity of alantolactone to gastric cancer.
Results:
Alantolactone induced apoptosis of gastric cancer cells by regulating the expression of Bax, Bcl-2, and p53, which related to intrinsic apoptotic pathway, and suppressed colony formation, migration, and invasion by mediating the expression of matrix metalloproteinase (MMP)-2, MMP-7, and MMP-9. Cell signaling pathway analysis showed that alantolactone enhanced the phosphorylation of p38 and decreased the translocation of nucleus p65, suggesting that the apoptosis-promoting and migration-suppressing effect of alantolactone might at least partially rely on regulating p38 mitogen-activated protein kinase (p38MAPK) pathway and nuclear factor-κB (NF-κB) pathway.
Conclusions:
Alantolactone can be used as a potential therapeutic agent for treating gastric cancer.
Introduction
Gastric cancer is a malignant tumor of the digestive system with high incidence rate and mortality rate. In China, gastric cancer is ranked in the top five leading causes of cancer death. 1 The etiology of gastric cancer is multifactorial. In addition to the infection with Helicobacter pylori, the risk of gastric cancer is increased with excessive dietary salt and decreased with consumption of fresh fruits and vegetables. 2,3 Surgery is still the primary therapy for gastric cancer treatment. However, many patients undergoing radical resection of gastric cancer may recur locally or with distant metastases, therefore, the 5-year survival rate is disappointing. 4 Adjuvant chemotherapy can improve outcome of resectable gastric cancer and reduce the risk of death compared to surgery alone, 5 thus, it is urgent to find more effective therapeutic agents to improve survival rate against gastric cancer with lower side effects. In recent years, some bioactive compounds extracted from plants, such as asiatic acid derivative, 6 tubeimoside 1, 7 and crosolic acid, 8 have been demonstrated to induce apoptosis of gastric cancer cells. These natural components are considered to be useful sources of new drugs for gastric cancer therapy.
Alantolactone, a sesquiterpene lactone compound extracted from Inula helenium L., 9 and the chemical structure was shown in Figure 1(a). Studies have revealed that alantolactone possessed neuroprotective, 10 anti-bacterial, 11 anti-fungal, 12 anti-inflammatory, 13 and anti-proliferative 14 effects. Recently, the anticancer effects of alantolactone have been reported, which indicated that alantolactone could inhibit proliferation and induce apoptosis in various cancer, including lung cancer, 15 colon cancer, 16 liver cancer, 17,18 cervical cancer, 19 and breast cancer. 14 Researchers also found that alantolactone was able to suppress cell migration in breast cancer 14 and lung cancer. 20 Besides, alantolactone induced excess ROS accumulation and promoted oxidative DNA damage in human colorectal cancer cells. 21 Alantolactone has also been proved to be an inhibitor of cell cycle progression. 15 Therefore, alantolactone is seemed as a promising chemotherapeutic drug candidate for cancer therapy.

Chemical structure of alantolactone (a) and effect of alantolactone on cell viability of (b) BGC-823 and (c) SGC-7901 cells. Data were represented as (percentage of control) mean ± SD of three separate experiments. *p < 0.05 compared to their respective control cells.
Although there have been many attempts to study the effect of alantolactone on a variety of human cancer cell lines, currently, to the best of our knowledge, the potential anti-cancer activity and the molecular mechanism of alantolactone against gastric cancer cells are still unknown, meanwhile, different cancer cells exhibit different sensitivity to the same drug. Therefore, in the present study, the antitumor effects of alantolactone on gastric cancer cells were investigated by determining the ability of this compound to induce apoptosis, suppress colony formation, migration, and invasion of BGC-823 and SGC-7901 cells, and the possible mechanism on cell signaling pathway underlying these effects was elucidated. These findings will provide experimental basis for alantolactone to use as an anti-cancer agent.
Materials and methods
Materials
Alantolactone (purity, 99%) was purchased from Chengdu Must Biotechnology Co., Ltd, (Chengdu, China). Dulbecco’s Modified Eagle Medium (DMEM), RPMI 1640, and fetal bovine serum were obtained from Hyclone (Logan, Utah, USA). Penicillin, streptomycin, phosphate-buffered saline (PBS), acrylamide, 4% paraformaldehyde and bicinchoninic acid assay kit were purchased from Dingguo Changsheng Biotechnology Co., Ltd, (Beijing, China). 3-(4,5-Dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium (MTT) was obtained from Genview (Tallahasses, Florida, USA). Dimethylsulfoxide (DMSO) was provided by Sigma Chemical Co. (St Louis, Missouri, USA). Annexin V-fluorescein isothiocyanate (FITC) Apoptosis Detection Kit and Hoechst 33258 were obtained from Nanjing KeyGen Biotech Co., Ltd, (Nanjing, China). Sodium dodecyl sulfate (SDS) was provided by Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Antibodies against Bax, Bcl-2, p53, matrix metalloproteinase (MMP)-9, MMP-7, MMP-2, and β-actin were provided by Santa Cruz Biotechnology (Santa Cruz, California, USA). Plates (6 cm diameter) and Transwell chambers were purchased from Corning Life Sciences (Tewksbury, Massachusetts, USA). Non-fat dried milk was obtained from BD Biosciences (San Jose, California, USA). Trizol was purchased from Takara Shuzo Co., Ltd, (Kyoto, Japan). Enhanced chemiluminescence (ECL) was obtained from Beyotime Institute of Biotechnology (Shanghai, China). Antibodies against p38, phospho-p38 and nuclear factor-κB (NF-κB) were provided by Cell Signaling Technology (Boston, Massachusetts, USA). HRP-conjugated goat anti-rabbit IgG and HRP-conjugated goat anti-mouse IgG were purchased from Beijing Solarbio Science & Technology Co., Ltd, (Beijing, China).
Cell culture
Human gastric cancer cell lines (BGC-823 and SGC-7901) were provided by the China Medical University (Shenyang, China). Cell lines were respectively cultured in RPMI 1640 medium and high-glucose DMEM medium supplemented with 10% (v/v) fetal bovine serum, 100 U mL−1 penicillin, and 100 μg mL−1 streptomycin at 37°C in a humidified atmosphere with 5% carbon dioxide.
MTT assay
To measure cell viability, MTT assay was performed. 17 Cells were seeded at a density of 3000 cells/well in 96 well plates and cultured with alantolactone (10 and 20 μM) for 24 h. Then, cells were incubated with 20 μL MTT solution in each well for another 4 h. After the medium removed, the purple formazan crystals were dissolved with 150 μL DMSO by oscillation for 10 min, and the absorbance was evaluated at 570 nm using a microplate reader (Multiskan FC, Thermo Fisher Scientific, St Herblain, France).
Hoechst 33258 staining
Cells were washed with PBS and treated with buffer A twice for 5 min. Following incubated with 4% paraformaldehyde solution for 30 min and washed with buffer A, cells were stained with 30 μL Hoechst 33258 for 10 min. Cells were washed with PBS and mounted using 50% glycerol. Cells were observed under fluorescence microscope at 340 nm (20× magnification). Hoechst 33258 is a fluorescent probe taken up by nuclei, and normal cell shows uniformly light blue nuclei while apoptotic cell presents a bright blue fluorescence. 22,23
Annexin V FITC/PI double staining test
The procedures of Annexin V FITC and propidium iodide (PI) double staining were complied with the instructions of the commercial kit. In brief, after collected, washed, and resuspended in 500 μL binding buffer, cells were incubated in the solution at the presence of 5 μL Annexin V FITC and 5 μL PI staining for 15 min in dark. The apoptotic rate was detected within 1 h using a flow cytometry (FACSCalibur, BD Biosciences, San Jose, California, USA).
Wound healing assay
Wound healing assay was performed as described previously 24 to assess cell migration. Cells were seeded into 6 well plates and incubated for 24 h. After a wound was made in the center of each well, the medium containing alantolactone (10 and 20 μM) were added to each well. Cell migration was observed under a microscope, and the images (10× magnification) were captured at 24, 48, and 72 h.
Invasion assay
Transwell assay was applied to determine cell invasion, and the procedures were performed as previously described. 25 Cell suspension was added into the upper chamber of Transwell, and 600 μL medium with 10% fetal bovine serum and alantolactone were added into the lower chamber. After incubated for 24 h, the non-invaded cells on the upper side of the chamber were removed. The cells were fixed with methanol for 10 min, washed with PBS, and stained with 0.1% crystal violet for 15 min. After washing, the number of cells penetrated through the membrane was counted in five randomly chosen visual fields using a microscope (10× magnification).
Colony forming assay
Cells were plated at a density of 1 × 103 cells in each plate and cultured with different concentrations (10 and 20 μM) of alantolactone. The medium was replenished every 5 days until visible colonies were formed. After 14 days, colonies were fixed in 5 mL Carnoys fixative for 30 min and stained using 3 mL Giemsa solution for 10 min. After washing with water and drying out, the images were captured, and the cell colonies were counted.
Real-time polymerase chain reaction
To determine the mRNA expression of Bax, Bcl-2, p53, MMP-2, MMP-7, and MMP-9 in each group, real-time polymerase chain reaction (PCR) was performed. Total RNA of cells was extracted, and its concentration and purity were detected by measuring the absorbance ratio at 260/280 nm. Total RNA was converted to cDNA with the iScript™ cDNA Synthesis Kit, Bio-Rad Laboratories, Hercules, California, USA. mRNA expression analysis was performed using an Applied Biosystems 7500 real-time PCR system and software (Applied Biosystems, Carlsbad, California, USA), and the cycling conditions were as follows: 95°C for 30 s, 40 cycles of 95°C for 5 s, and 60°C for 30 s. PCR was done with specific primers: Bax: 5′-GGACGAACTGGACAGTAACATGG-3′ and 5′-GCAAAGTAGAAAAGGGCGACAAC-3′; Bcl-2: 5′-CACGCTGGGAGAACA-3′ and 5′-CTGGGAGGAGAAGATG-3′; p53: 5′-CTGCCCTCAACAAGATGTTTTG-3′ and 5′-CTATCTGAGCAGCGCTCATGG-3′; MMP-2: 5′-ACCGCGACAAGAAGTATGGC-3′ and 5′-CCACTTGCGGTCATCATCGT-3′; MMP-7: 5′-TACAGTGGGAACAGGCTCAGG-3′ and 5′-GGCACTCCACATCTGGGCT-3′; MMP-9: 5′-TGGGCTACGTGACCTATGACAT-3′ and 5′-GCCCAGCCCACCTCCACTCCTC-3′; and β-actin: 5′-GACGTGGACATCCGCAAAG-3′ and 5′-CTGGAAGGTGGACAGCGAGG-3′. Data were analyzed using the 2−ΔΔCt method relative to β-actin control.
Western blot analysis
Cells were harvested and lysed, and total protein extracts and nuclear extracts were prepared. The protein concentration of the extracts was measured, and equal proteins were separated by SDS-polyacrylamide gel via electrophoresis and transferred to polyvinylidene fluoride membranes. After blocked using 5% non-fat milk for 1 h, the blots were incubated with primary antibodies overnight at 4°C. The primary antibodies were used as follows: anti-Bax antibody, anti-Bcl-2 antibody, anti-p53 antibody, anti-MMP-9 antibody, anti-MMP-7 antibody, anti-MMP-2 antibody, and anti-β-actin antibody at a dilution ratio of 1:1000 and anti-p38 antibody, anti-phospho-p38 antibody, and anti-NF-κB antibody at a dilution ratio of 1:3000. The membranes were washed and incubated with the secondary antibodies, HRP-conjugated anti-rabbit IgG or HRP-conjugated anti-mouse IgG, at a dilution ratio of 1:5000 for 1 h. Finally, the blots were visualized using ECL. The relative protein levels were normalized using β-actin intensity.
Statistical analysis
All data were presented as the mean ± standard deviation (SD) from three separate experiments. Statistical analysis was carried out by one-way analysis of variance followed by least significant digit using SPSS 16.0 (SPSS, Inc., Chicago, Illinois, USA). The difference at p < 0.05 was considered as statistically significant.
Results
Effect of alantolactone on cell viability
MTT assay was performed to measure the inhibitory effect of alantolactone on cell proliferation of BGC-823 cells and SGC-7901 cells. The cell viability was significantly (p < 0.05) declined after treated with alantolactone in both cell lines in a concentration-dependent manner compared to control cells (Figure 1(b) and (c)).
Effect of alantolactone on apoptosis
After treatment with alantolactone for 24 h, the number of BGC-823 cells in each group was less and less with the increasing concentrations (10 and 20 μM) of alantolactone. Meanwhile, the fluorescence of cell treated with alantolactone showed brighter than that of control cells (Figure 2(a)). The similar phenomenon was found in SGC-7901 cells (Figure 2(b)).

Effect of alantolactone on apoptosis in (a) BGC-823 cells and (b) SGC-7901 cells shown by Hoechst 33258 staining using a fluorescence microscope. Representative photomicrographs were showed at ×20 magnification.
Apoptotic rates of cells were detected with Annexin V FITC and PI double staining method. The apoptotic rate of BGC-823 cells was significantly (p < 0.05) increased after treated with alantolactone for 24 h compared to that of untreated cells in a concentration-dependent manner (Figure 3(a) and (c)). The apoptotic rate was also significantly (p < 0.05) increased after treated with alantolactone in SGC-7901 cells (Figure 3(b) and (d)).

Effect of alantolactone on apoptotic rate of (a and c) BGC-823 and (b and d) SGC-7901 cells by Annexin V FITC and PI double staining using flow cytometry (a and b) flow cytometry scatter diagram obtained via Annexin V FITC and PI tests and (c and d) column bar graphs of apoptotic rate of each sample were shown. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to control cells. FITC: fluorescein isothiocyanate; PI: propidium iodide.
Effect of alantolactone on expression of Bax, Bcl-2, and p53
To explore the effects of alantolactone on the mRNA and protein expression of Bax, Bcl-2, and p53 in BGC-823 cells and SGC-7901 cells, real-time PCR and western blot analysis were carried out. As illustrated in Figure 4(a), treatment with alantolactone at concentrations of 10 and 20 μM for 24 h significantly (p < 0.05) increased the mRNA expression of Bax in BGC-823 and SGC-7901 cells. However, SGC-7901 cells had lower sensitivity to alantolactone than that of BGC-823 cells. Compared to untreated cells, cells treated with alantolactone (10 and 20 μM) for 24 h significantly (p < 0.05) decreased the mRNA expression of Bcl-2 and significantly (p < 0.05) increased the mRNA expression of p53 in both cell lines (Figure 4(b) and (c)). After treated with 10 μM alantolactone for 24 h, the protein expression of Bcl-2 significantly (p < 0.05) decreased and the protein expression of p53 significantly (p < 0.05) increased in both cell lines. In addition, under the same condition, the protein expression of Bax in SGC-7901 cells was also significantly (p < 0.05) increased, while it was not significant (p > 0.05) in changed BGC-823 cells. But 20 μM alantolactone significantly (p < 0.05) decreased the protein expression of Bcl-2 and significantly (p < 0.05) increased the protein expression of Bax and p53 in both cell lines compared to control cells (Figure 5(a) and (b)). It could be found that alantolactone regulated the expression of Bax, Bcl-2, and p53, although the sensitivity of these two cell lines to alantolactone were different.
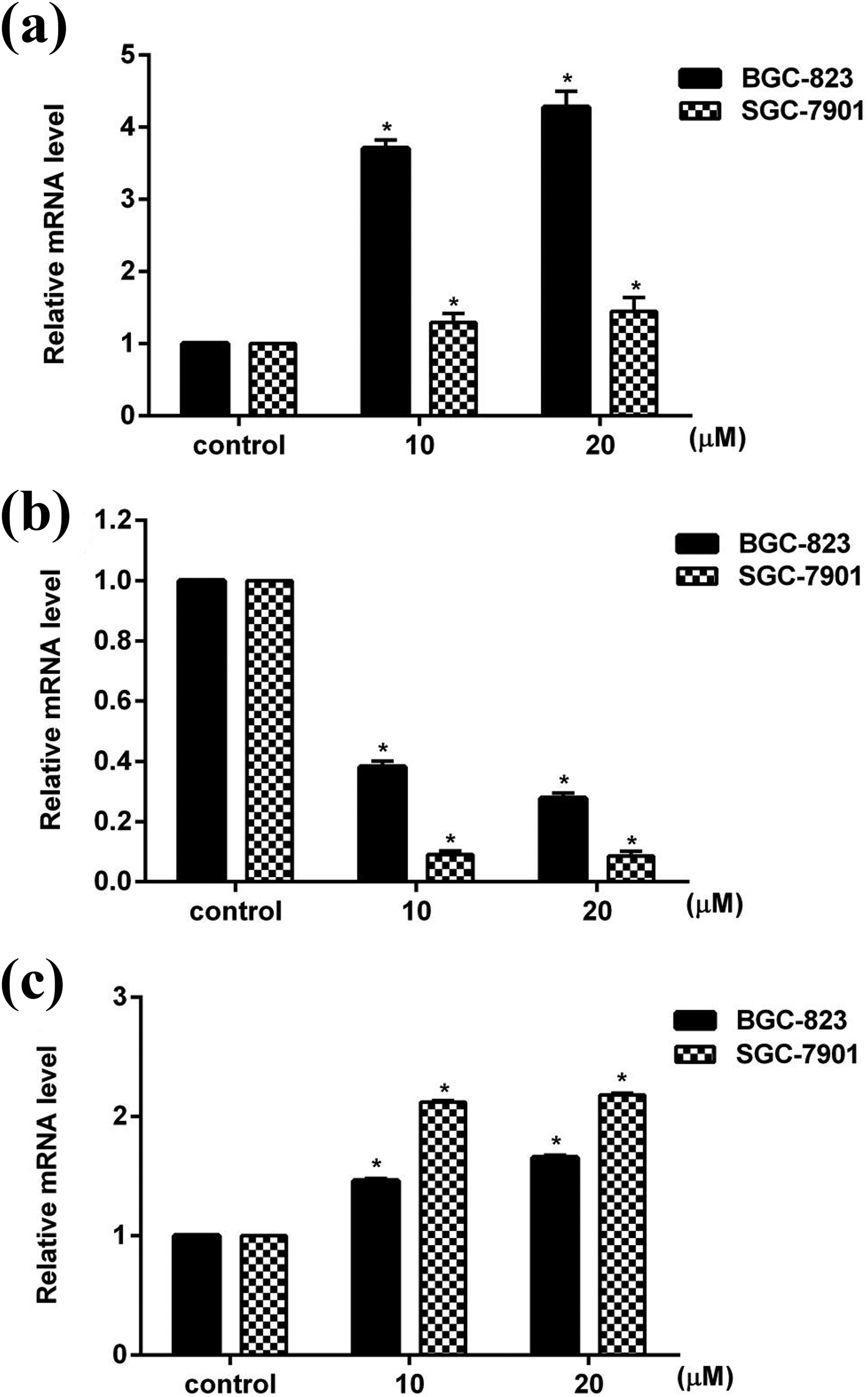
Effect of alantolactone on mRNA expression of (a) Bax, (b) Bcl-2, and (c) p53 in BGC-823 and SGC-7901 cells. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to their respective control cells.

Effect of alantolactone on protein expression of Bax, Bcl-, and p53 in (a) BGC-823 and (b) SGC-7901 cells. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to their respective control cells.
Effect of alantolactone on colony formation
To analyze the ability of colony formation in each group, the number of colonies was counted. Compared to untreated cells, the size of colonies in cells treated with alantolactone was smaller. The number of colonies decreased significantly (p < 0.05) at 10 and 20 μM alantolactone in both BGC-823 cells and SGC-7901 cells (Figure 6(a) to (d)).

Effect of alantolactone on colony formation in (a and c) BGC-823 and (b and d) SGC-7901 cells. (a and b) photomicrographs were shown, and (c and d) the number of colonies in each group was analyzed in BGC-823 and SGC-7901 cells. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to control cells.
Effect of alantolactone on migration and invasion
The effect of alantolactone on migration of gastric cancer cells was analyzed with wound healing assay. Compared to control group, alantolactone inhibited the migration of both BGC-823 cells and SGC-7901 cells (Figure 7(a) and (b)). The numbers of invasion cells were significantly (p < 0.05) declined at 10 and 20 μM alantolactone in both cell lines compared to respective control group, which showed in Figure 7(c) to (f).

Effect of alantolactone on migration and invasion in BGC-823 and SGC-7901 cells. Effect of alantolactone on migration in (a) BGC-823 cells and (b) SGC-7901 cells. Representative photomicrographs of wounds at 24, 48, and 72 h were showed at 10× magnification. Effect of alantolactone on invasion in (c and e) BGC-823 cells and (d and f) SGC-7901 cells. (c and d) Representative photomicrographs were showed at 10× magnification and (e and f) invasion cell number of each group was analyzed. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to control cells.
Effect of alantolactone on MMPs expression
The mRNA and protein expression of MMP-2, MMP-7, and MMP-9 in each group of BGC-823 and SGC-7901 cells were measured using real-time PCR and western blot assay. Figure 8(a) to (c) showed that the mRNA expression of MMPs in both cell lines were significantly (p < 0.05) downregulated after treated with alantolactone (10 and 20 μM) for 24 h. The protein expression of these MMPs was significantly (p < 0.05) declined at 20 μM alantolactone in BGC-823 cells. However, alantolactone at 10 μM could not significantly (p > 0.05) affect the expression of MMP-7, while significantly (p < 0.05) downregulated the protein expression of MMP-2 and MMP-9 in BGC-823 cells (Figure 9(a)). Similarly, 20 μM alantolactone significantly (p < 0.05) decreased the protein level of MMP-2, MMP-7, and MMP-9, but 10 μM alantolactone could not significantly (p < 0.05) affect them in SGC-7901 cells (Figure 9(b)).

Effect of alantolactone on mRNA expression of (a) MMP-2, (b) MMP-7, and (c) MMP-9 in BGC-823 and SGC-7901 cells. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to their respective control cells. MMP: matrix metalloproteinase.

Effect of alantolactone on protein expression of MMP-2, MMP-7, and MMP-9 in (a) BGC-823 and (b) SGC-7901 cells. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to their respective control cells. MMP: matrix metalloproteinase.
Effect of alantolactone on p38 mitogen-activated protein kinase (MAPK) pathway
To further explore the cell signaling pathways by which alantolactone induced gastric cancer cells damage, we measured the phosphorylation of p38 in each group. After treatment with 20 μM alantolactone for 24 h, the level of p-p38 was significantly (p < 0.05) increased compared to the control group of both cell lines, and alantolactone had no obvious (p > 0.05) effect on the p38 expression (Figure 10).
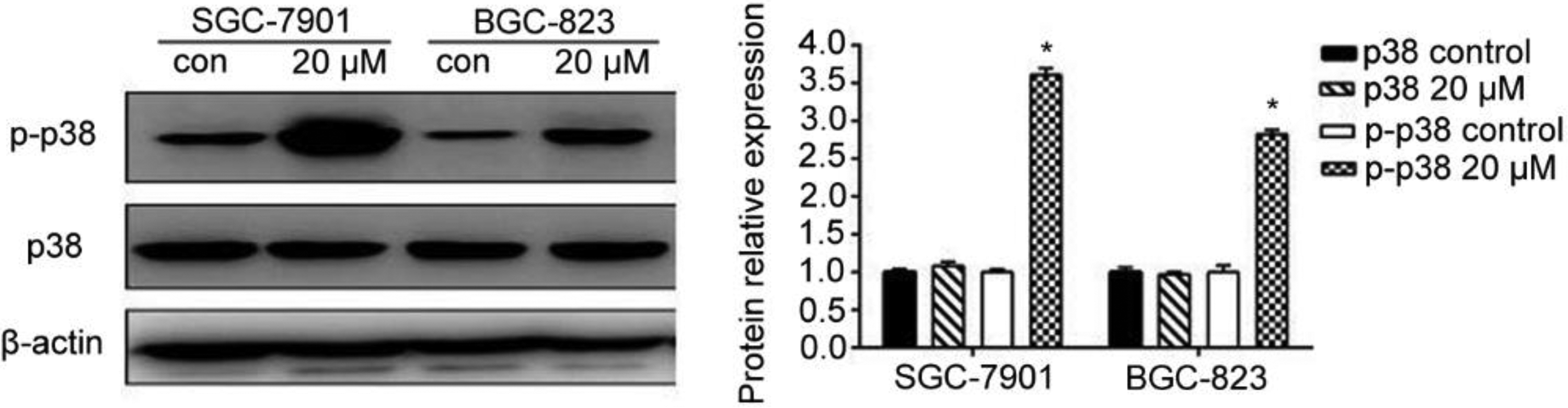
Effect of alantolactone on protein expression of p38 and p-p38 in BGC-823 and SGC-7901 cells using western blot assay. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to control cells.
Effect of alantolactone on NF-κB pathway
The expression and the translocation of p65 to the nuclei were measured in each group to explore the contribution of NF-κB pathway to the effect of alantolactone on gastric cancer cells. As shown in Figure 11, 20 μM alantolactone significantly (p < 0.05) downregulated p65 translocation into nucleus of BGC-823 and SGC-7901 cells. However, alantolactone at this concentration could not significantly (p > 0.05) affect p65 expression in total extract of both cell lines.

Effect of alantolactone on protein expression of total p65 and p65 in nucleus of BGC-823 and SGC-7901 cells. Data represented as mean ± SD of three separate experiments. *p < 0.05 compared to control cells.
Discussion
The balance between proliferation and apoptosis is important in tumor development. 26 MTT analysis was carried out to investigate whether alantolactone could reduce BGC-823 and SGC-7901 cells viability. Results showed that the viability of both cell lines decreased significantly after incubated with alantolactone, suggesting that treatment with alantolactone led to cell damage and inhibited proliferation of BGC-823 and SGC-7901 cells. The inhibition effect of alantolactone on gastric cancer cells was more effective than that on liver cancer cells, 17 which suggested that the sensitivity of different cancers to alantolactone was different, and alantolactone should be chosen for specific cancer treatment. Apoptosis, programmed cell death, is an intricate mechanism on responding to environmental stress by removing potentially dangerous cells, 27 which is associated to the pathogenesis of many diseases. Cancer is one of the diseases charactered by too little apoptosis. Therefore, inducing apoptosis is a common strategy for cancer treatment. 28 Apoptosis can be induced by alantolactone in various cancer cell lines. 15,17 In the present study, alantolactone significantly increased apoptotic rates in both BGC-823 and SGC-7901 cells. Compared with the apoptotic rates in other cancer cells, the ability of alantolactone to induce cells apoptosis in gastric cancer cells was more effective than that in liver cancer cells. 17
Apoptosis proceeds through extrinsic and intrinsic pathways in mammalian cells, and mitochondrion is an important organ in intrinsic apoptotic process. Mitochondrial apoptotic pathway is determined by the Bcl-2 family proteins in which Bax is one of pro-apoptotic proteins and Bcl-2 is one of anti-apoptotic proteins. 29 Alantolactone regulates Bcl-2 family proteins to induce cell apoptosis. 18 In this study, alantolactone significantly downregulated the expression of Bcl-2 and significantly upregulated the expression of Bax in both cell lines, which mean that alantolactone induced cell apoptosis which may partly be through mitochondrial apoptotic pathway. The protein p53 plays an important part in the cell evolvement from normal function to tumorigenesis, and it has been identified to be the common mutated tumor suppressor in tumorigenesis. 30,31 P53 is also associated to cell cycle arrest and apoptosis. Thus, some studies focused on the role of p53 in mediating cell apoptosis induced by chemotherapeutic agents. 30,32,33 In this study, we found that the level of p53 was significantly increased due to treatment with alantolactone, which further suggested that p53 was a key gene in the apoptosis of BGC 823 and SGC-7901 cells.
The lethal result of the major cancers is caused by the spread of cancer cells and the growth of tumors at distant tissues. To further assess the anti-cancer effect of alantolactone, colony formation, migration, and invasion were investigated with BGC 823 and SGC-7901 cells, which are related to progress of malignant tumors. The results showed that alantolactone significantly inhibited colonies formation, migration, and invasion of gastric cancer cells, which was consistent with the effects of alantolactone on breast cancer MCF-7 cells 14 and A549 lung adenocarcinoma cells. 20 MMPs are the major proteinases family 34 that have well identified roles in tumor progression, including regulating cell growth, differentiation, apoptosis, migration, invasion, and metastasis, and are related to adverse prognosis in patients with cancer. 35 –38 The dissemination of metastatic cancer cells makes the cancer fatal, and MMPs play an important role in cell migration. 36 MMPs are frequently overexpressed in human cancer, 37 therefore, they are regarded as the promising targets for cancer therapy. It has been reported that phenethyl isothiocyanate may inhibit the migration and invasion of GBM 8401 cells through reducing the expression of MMP-2, MMP-7, and MMP-9. 39 Thus, to explore the mechanisms of the inhibitive effect of alantolactone on migration and invasion of gastric cancer cells, we measured the expression of MMP-2, MMP-7, and MMP-9. In the present study, the expression of MMP-2, MMP-7, and MMP-9 decreased significantly after the treatment with alantolactone in both BGC 823 and SGC-7901 cells, suggesting that alantolactone inhibited migration and invasion which may partly be through modulating the expression of MMPs. Moreover, MMP-7 and MMP-9 regulate apoptosis, 37 thus, we speculated that alantolactone induced apoptosis in gastric cancer cells which may be via regulating MMPs expression.
The pathogenesis of cancer includes the change of signal transduction pathways. Therefore, the molecules of these abnormal signaling pathways provide druggable targets for cancer treatments. 40,41 MAPKs are signal transducing enzymes regulating a variety of physiological processes, including proliferation and apoptosis. MAPK signaling pathways consist of p38 mitogen-activated protein kinase (p38MAPK), c-Jun NH2-terminal kinase, and extracellular signal-regulated kinase. 42,43 The protein p38 can be activated by extracellular stimuli, thereby activating transcription factors and promoting cell apoptosis. 44 In addition, p38MAPK has been identified as the vital signal pathway to activate Bax after its translocation to mitochondria. 45 Besides apoptosis, migration and invasion of cancer cells can be regulated via p38MAPK signaling pathway. 46 In this study, phosphorylation of p38 was significantly upregulated by alantolactone treatment, which mean that the anti-cancer effect of alantolactone, inducing apoptosis and mitigating migration and invasion, on gastric cancer cells may partially via regulating p38MAPK pathway. MAPKs are closely related to some other signaling pathways, among which NF-κB (p65) is a downstream target of p38MAPK. 47,48 p65 has been regarded as an important factor in physiological and pathological processes, especially in cell proliferation and apoptosis, which is an important regulator in oncogenesis. 49 Normally, in most cell types, p65 is inactive in the cytoplasm and translocates to the nucleus when activated. 50 Various tumor promoters can activate NF-κB pathway, and the activated p65 blocks apoptosis and promotes proliferation. 51 Thus, p65 is considered to be an anti-apoptotic transcription factor. 49 In addition, it has been reported that the reduction of migration and invasion were observed when NF-κB pathway was inhibited. 50 Furthermore, the translocation of NF-κB from cytoplasm to nucleus is a major step that regulates MMPs transcription. 35 In our study, results showed that alantolactone significantly downregulated the level of p65 in nuclei of cancer cells. Therefore, we speculated that the effect of alantolactone on gastric cancer cell may partly via blocking the translocation of NF-κB from cytoplasm to nucleus.
In summary, our study demonstrated that alantolactone could inhibit gastric cancer in BGC-823 and SGC-7901 cells. The anti-cancer activity of alantolactone may depend on many mechanisms, including inhibition of proliferation, induction of apoptosis, as well as suppression of colonies formation, migration, and invasion, which might be associated with regulation of p38MAPK pathway, NF-κB pathway, and the expression of MMPs (Figure 12). Our study suggested that alantolactone was a promising anti-cancer drug candidate in gastric cancer therapy, and these findings expand the application of alantolactone and provide a candidate therapeutic agent for gastric cancer treatment. In future, the effects of alantolactone on gastric cancer will be explored with animal models.

Alantolactone induced apoptosis and suppressed migration of gastric cancer cells via regulating the expression of related genes, the underlying mechanism of which is regulating p38MAPK and NF-κB pathways.
Footnotes
Authors’ contribution
Yin He and Xiangyu Cao are co-first authors and they have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported by the National Natural Science Foundation of China (Grant No. 31770017), Cultivation Plan for Youth Agricultural Science and Technology Innovative Talents of Liaoning Province (Grant No. 2015013), Project Supported by Scientific Research Fund of Liaoning Provincial Education Department (Grant No. LQN201714), Startup Foundation for Doctors of Liaoning Province (Grant No. 20170520258), Innovation Team Project from the Education Department of Liaoning Province (Grant No. LT2015011), and Youth and middle-aged Science and Technology Innovative Talents of Shenyang City (Grant No. RC180240).
