Abstract
Background
Fluoxetine (FLX) has been widely used as first-line treatment in cases of depression and other neuropsychiatric disorders. Although its safety has been approved, the use of FLX was associated with liver injury and chronic liver disease. Vinpocetine (Vinpo), a nootropic drug, possesses antioxidant and anti-inflammatory effects.
Objective
This study aimed to evaluate the protective effects of Vinpo on FLX-induced liver damage pointing to the role of peroxisome proliferator-activated receptor-gamma (PPAR-γ) and nuclear factor erythroid 2-related factor 2 (Nrf2).
Methods
Rats were randomized to four groups: control group, Vinpo group (20 mg/kg/day; orally), FLX group (10 mg/kg/day; orally), and Vinpo + FLX group.
Results
FLX-induced liver damage was evidenced through elevated liver function biomarkers and induced hepatic histopathological changes. Concurrent Vinpo treatment resulted in a significant decrease in hepatotoxicity biomarkers and histopathological alterations. FLX-induced oxidative stress and inflammation were attenuated by Vinpo. In addition, Vinpo attenuated the hepatic NRF2 and HO-1 levels and up-regulated PPAR-γ expression. Moreover, FLX elevated Bcl-2-associated X protein (Bax) mRNA expression and decreased B-cell lymphoma 2 (Bcl2) mRNA expression were markedly reversed by Vinpo.
Conclusion
Vinpo possesses ameliorative effects against FLX-induced liver injury in rats. This effect may be due to attenuation of oxidative stress and inflammation, in addition to upregulation of PPAR-γ expression.
Keywords
Introduction
Major depressive disorder (MDD) is a common illness that significantly diminishes the quality of life, and is currently the leading cause of disability worldwide. 1 FLX is a selective serotonin re-uptake inhibitor (SSRI), which is commonly prescribed for the treatment of several neurological disorders, such as MDD and anxiety. 2 It is extensively metabolized in the liver, where undergoes metabolic conversion to the active metabolite norfluoxetine among multiple other metabolites. 3 Several studies reported that fluoxetine and norfluoxetine induced-liver injury and oxidative stress in rats.4,5 Hepatic ischemia, necrosis, cholestasis, steatosis, portal zone inflammation, lobular inflammation, and hepatomegaly were reported with different doses of fluoxetine.4,6–8 Vinpo is a synthetic ethyl ester of vinca alkaloid vincamine, and it is widely prescribed as a cryoprotective agent in the treatment of memory disturbances, cognitive impairment, stroke, and dementia. 9 Vinpo is gaining interest from the academic and scientific community due to its higher safety profile and fewer side effects for long-term use. Vinpo has several pharmacological mechanisms of action: it inhibits cyclic nucleotide phosphodiesterase 1 (PDE1), 10 voltage-gated sodium channels and calcium channels.11,12 Moreover, It exerts an anti-inflammatory effect through inhibition of NF-κB-dependent inflammatory response and its downstream proinflammatory mediators such as TNF-α and IL-6. 13 Vinpo also reported to has antioxidant effects that account for its anti-inflammatory actions.14,15 Some researchers reported that Vinpo has hepatoprotective activity against hepatotoxicity due to its strong anti-oxidant and anti-inflammatory effects. Thus, the objective of this study was to evaluate the effects of Vinpo on FLX-liver injury in rats.
Materials and methods
Chemicals and drugs
The chemicals used were purchased from Sigma-Aldrich (St Louis, MO, USA) and were of high analytical grade. FLX (Philozac ®) was obtained as capsules (10 mg) from Amoun Pharmaceutical Company (Cairo, Egypt). Vinpo (Vinporal®) was obtained as tablets (5 mg) from Amriya Pharmaceutical Industries (Alexandria, Egypt).
Animals
Twenty-four male albino rats weighing approximately 150–180 g were obtained from the animal facility of The Nile Co. for Pharmaceuticals and Chemical Industries (Cairo, Egypt). The experimental animals were maintained under controlled environmental conditions of 20–25°C temperature, 55% humidity, and 12 h of a light/dark cycle and provided with regular feeding and free drinking. Animals were acclimatized for 1 week before any experimental procedures. The experimental procedures were performed in accordance with The Animal Ethics Committee of the Faculty of Pharmacy, Al-Azhar University, Egypt and Guide for the Care and Use of Laboratory Animals.
Experimental design
Rats were randomly divided into four groups (n = 6 rats per group). (1) Control group; rats received physiological saline orally via stomach tube. (2) Vinpo group; received vinpocetine (20 mg/kg/day; dissolved in physiological saline) orally via stomach tube for 7 days. The dosing and time of Vinpo used were selected based on previous studies.16–18 (3) FLX group; rats received fluoxetine (10 mg/kg/day; dissolved in physiological saline) orally via stomach tube for 7 days. The dosing and time of FLX used were chosen based on previous studies.4,5 (4) Vinpo + FLX group; rats received fluoxetine (10 mg/kg) and vinpocetine (20 mg/kg) orally via stomach tube for 7 days.
At the end of the experiment, blood samples were collected for determination of ALT, AST. Afterward, the animals were sacrificed and tissues liver of each rat were removed and cleaned. Each isolated liver was divided into three parts: the first part was fixed in 10% formalin solution for histopathological analysis. The second part was frozen immediately in liquid nitrogen and stored at −80°C for gene expression analysis. The third part was homogenized in saline; the homogenates were used for further biochemical analysis.
Assessment of liver functions and liver index
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels were assessed in the serum of rats using colorimetric kits (Abcam, UK), according to the manufacturer’s instructions. The liver index was calculated using the formula: (liver weight/total body weight) × 100.
Histopathological examination
Liver samples were fixed in 10% formalin, embedded in paraffin, cut into sections of 5 μm thickness, and stained with hematoxylin and eosin. 19
Determination of oxidative stress biomarkers
Hepatic homogenates were used for the estimation of GSH, SOD, CAT, and lipid peroxides expressed as malondialdehyde (MDA) using kits obtained from (Bio-diagnostic Co., Egypt). The procedures were conducted according to the manufacturer’s instructions and parameters were measured as provided by the manufacturer.
Assessment of the hepatic Nrf2/HO-1 pathway
Hepatic tissue homogenate was assayed for Nrf-2, and HO-1 levels using ELISA kits (My BioSource, USA), following the given manufacture instructions.
Determination of inflammatory cytokines, PPAR-γ and caspase-3
Hepatic tissue homogenate was assayed for NF-κB, TNF-α, and IL-6 quantification using ELISA kit obtained from (RandD Systems, Inc., USA) and (Cloud-clone corp., USA) respectively, caspase-3 content was assessed using ELISA kit obtained from (CUSABIO, USA). The level of PPAR-γ was determined using ELISA kit obtained from (My BioSource, USA). The manufacturer’s protocol was followed precisely according to instructions for each measured parameter and concentration samples were measured as provided by the manufacturer.
qRT-PCR analysis of PPAR-γ, Bax and Bcl-2 mRNA expression
Primer sequences used in real-time RT-PCR analysis.

Statistics
The data are presented as means ± SEM. GraphPad Prism software (version 5) was used for statistical analysis of data by one-way ANOVA followed by post hoc Tukey’s multiple comparison test. For all analyses, values of p < .05 were considered to be statistically significant.
Results
Vinpo administration for 7 days had no significant effect on all parameters assessed in the current study when compared to control rats.
Effect of Vinpo on liver index and hepatic functions in FLX-treated rats
Effect of Vinpo on liver index and hepatic functions in FLX-treated rats.
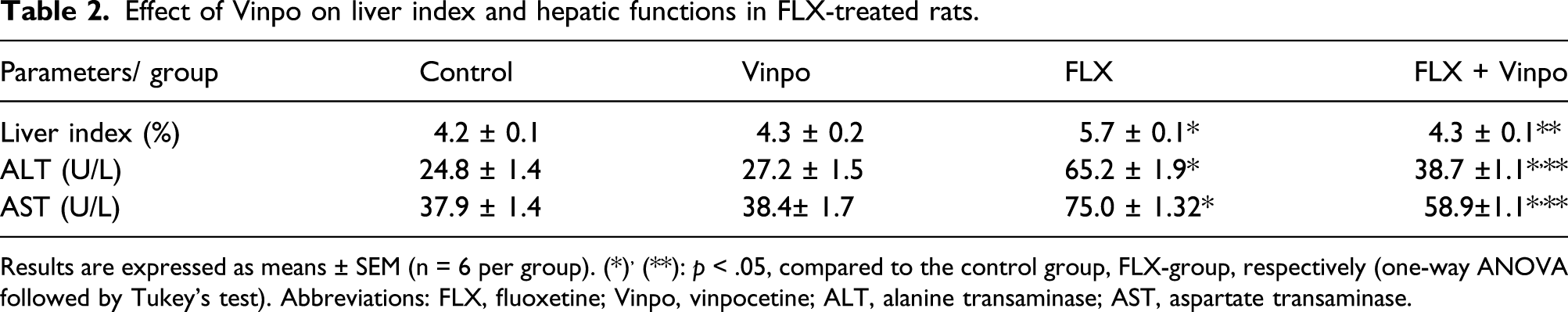
Results are expressed as means ± SEM (n = 6 per group). (*), (**): p < .05, compared to the control group, FLX-group, respectively (one-way ANOVA followed by Tukey's test). Abbreviations: FLX, fluoxetine; Vinpo, vinpocetine; ALT, alanine transaminase; AST, aspartate transaminase.
Effect of Vinpo on histopathological changes in FLX-treated rats
To further confirm the hepatoprotective effect of Vinpo, a histopathological study was performed. Microscopic examination of H&E-stained liver sections of control and Vinpo rats showed normal hepatic lobule structure and normal hepatocytes (Figure 1(a) and (b)) respectively. FLX administration for 7 consecutive days, induced histopathological alterations including; hepatocellular vacuolation, portal congestion (Figure 1(c)), mononuclear inflammatory cells infiltration, fibroplasia (Figure 1(d)), and vacuolar degeneration (Figure 1(e)). Concurrent treatment of Vinpo (Figure 1(f)) markedly prevented FLX-induced histopathological alterations in the liver of rats. Some mildly affected sections showed few vacuolated cells (Figure 1(g)). Effect of Vinpo on histological changes in liver of FLX-induced rats. Photomicrographs of H&E-stained liver sections of (a) control rats,(b) Vinpo rats, (c–e) FLX-induced rats showed hepatocellular vacuolation (arrow), congestion (bold arrow) (c), portal fibroplasia (bold arrow) (d) vacuolar degeneration in hepatocytes (arrow) and mononuclear inflammatory cells infiltration, (e) degeneration in hepatocytes with portal mononuclear inflammatory cells infiltration(arrow), (f–g) FLX-induced rats treated with Vinpo 20 mg/kg, (f) showing normal hepatic parenchyma, and (g) showing mild portal fibroplasia (arrow).
Effect of Vinpo on hepatic oxidative stress in FLX-treated in rats
Effect of Vinpo on hepatic oxidative stress parameters in FLX-treated rats.

Results are expressed as means ± SEM (n = 6 per group). (*), (**): p < .05, compared to the control group, FLX group, respectively (one-way ANOVA followed by Tukey's test). Abbreviations: FLX, fluoxetine; Vinpo, vinpocetine; GSH, glutathione; SOD, superoxide dismutase; CAT, catalase; MDA, malonaldehyde.
Effect of Vinpo on Nrf2/HO-1 pathway in FLX-treated rats
As illustrated in Figure 2 (a) and (b), FLX administration for 7 consecutive days significantly increased hepatic Nrf2 and HO-1 content by 161% and 223%, respectively, in comparison to the control group. Meanwhile, concurrent treatment of Vinpo significantly decreased hepatic Nrf2 and HO-1 content by 38% and 33%, respectively, in comparison to FLX-administered rats. Effect of Vinpo on hepatic Nrf2 (a) and HO-1 (b) levels in FLX-treated rats. Results are expressed as means ± SEM (n = 6 per group). (*), (**): p < .05, compared to the control group, FLX group, respectively (one-way ANOVA followed by Tukey's test). Abbreviations: FLX, fluoxetine; Vinpo, vinpocetine; Nrf2; nuclear factor erythroid 2-related factor 2; HO-1, heme oxygenase-1.
Effect of Vinpo on inflammation in FLX-treated rats
As illustrated in Figure 3(a)–(c), FLX administration for seven consecutive days significantly elevated hepatic levels of NF-κB, TNF-α and IL-6 by 143%,42%, and 76%, respectively, in comparison to the control group. Meanwhile, concurrent treatment of Vinpo significantly decreased the elevated of levels NF-κB, TNF- and IL-6 levels by 31%, 17%, and 23%, respectively, in comparison to FLX rats. These data proved that Vinpo can mitigate FLX-induced inflammation in rats. Effect of Vinpo on hepatic NF-κB (a), TNF-α (b) and IL-6 (c) levels in FLX-treated rats. Results are expressed as means ± SEM (n = 6 per group). (*), (**): p < .05, compared to the control group, FLX group, respectively (one-way ANOVA followed by Tukey's test). Abbreviations: FLX, fluoxetine; Vinpo, vinpocetine; NF-κB, nuclear factor kappa B; TNF-α, tumor necrosis factor alpha; IL-6, interleukin-6.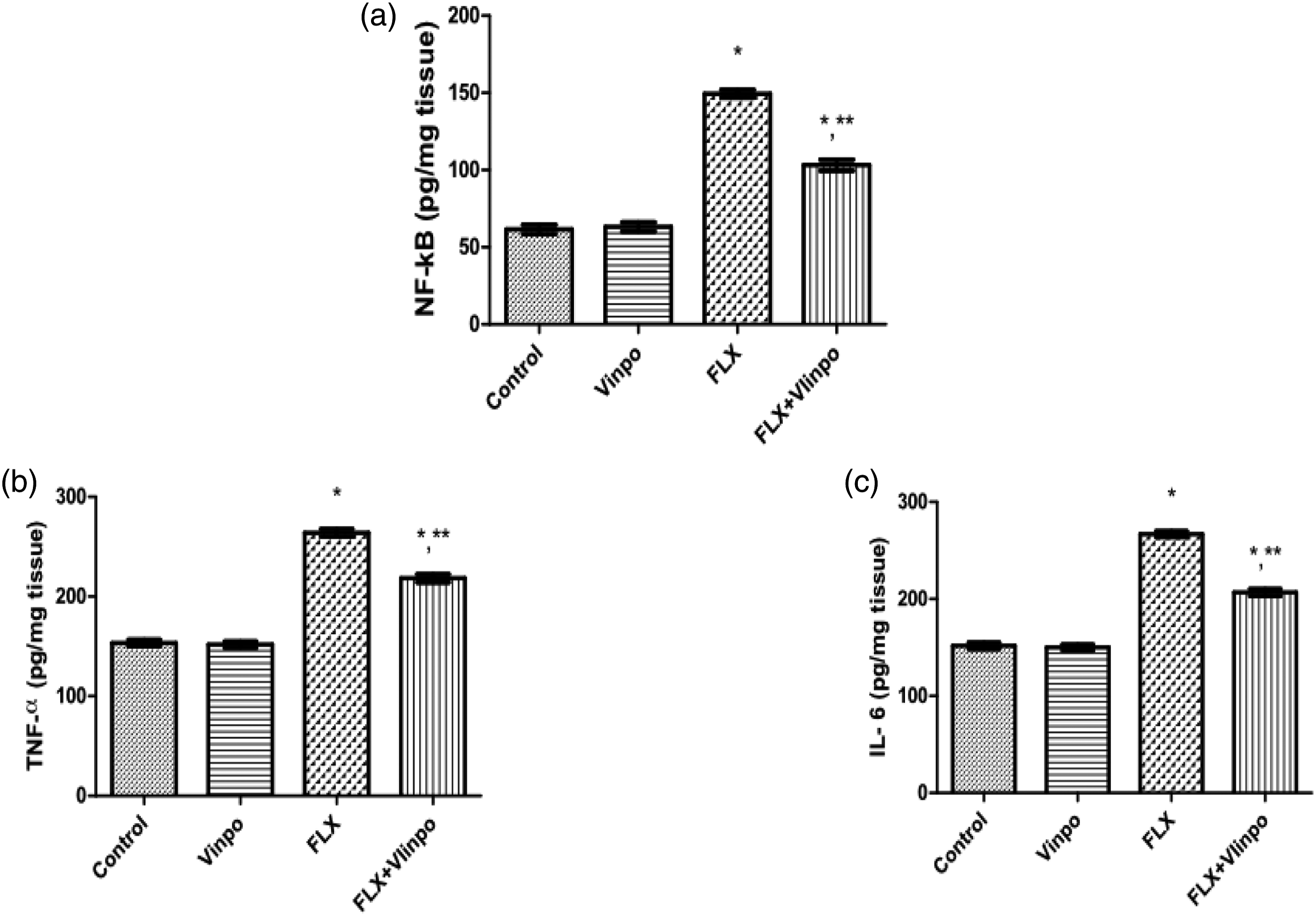
Effect of Vinpo on PPAR-γ mRNA and tissue level in FLX-treated rats
As illustrated in Figure 4(a) and (b), FLX administration for 7 consecutive days significantly decreased PPAR-γ mRNA expression by 72% and its tissue level by 60% in comparison to the control group. Administration of Vinpo ameliorated this effect, where PPAR- γ mRNA expression and tissue level were elevated by 3.6-fold and 75%, respectively, in comparison to FLX rats. Effect of Vinpo on PPAR-γ gene expression (a) and tissue level (b) in the liver tissue of FLX- treated rats. Quantitation of gene expression was done using real-time PCR. mRNA levels were normalized to that of β-actin in each group and expressed as relative quantification compared to the control group. Gene expression in the control group was defined as 1. Results are expressed as means ± SEM (n = 6 per group). (*), (**): p < .05, compared to the control group, FLX group, respectively (one-way ANOVA followed by Tukey's test). Abbreviations: FLX, fluoxetine; Vinpo, vinpocetine; PPAR-γ, peroxisome proliferator-activated receptor-gamma.
Effect of Vinpo on apoptosis in FLX-treated rats
As shown in Figure 5(a)–(c), FLX administration for 7 consecutive days showed a significant increase in hepatic caspase-3 activity by 2.5-fold in comparison to the control group. Moreover, FLX administration significantly elevated Bax mRNA expression by 4.1 fold and diminished Bcl-2 mRNA expression by 74%, as compared to the control group. Concurrent treatment of Vinpo showed a marked enhancement of apoptotic parameters. Vinpo significantly decreased caspase-3 activity by 48% and Bax mRNA expression by 110% with marked elevation of BCL-2 mRNA expression by 112% in comparison to FLX-administered rats. These data verified that Vinpo inhibited FLX -induced apoptosis in rats. Effect of Vinpo on caspase-3 level (a), Bax (b) and Bcl2 (c) gene expression in the liver tissue of FLX-treated rats. Quantitation of gene expression was done using real-time PCR. mRNA levels were normalized to that of β-actin in each group and expressed as relative quantification compared to the control group. Gene expression in the control group was defined as 1. Results are expressed as means ± SEM (n = 6 per group). (*), (**): p < .05, compared to the control group, FLX group, respectively (one-way ANOVA followed by Tukey's test). Abbreviations: FLX, fluoxetine; Vinpo, vinpocetine; Bax, Bcl-2-associated X protein B-cell lymphoma; Bcl2, B-cell lymphoma 2.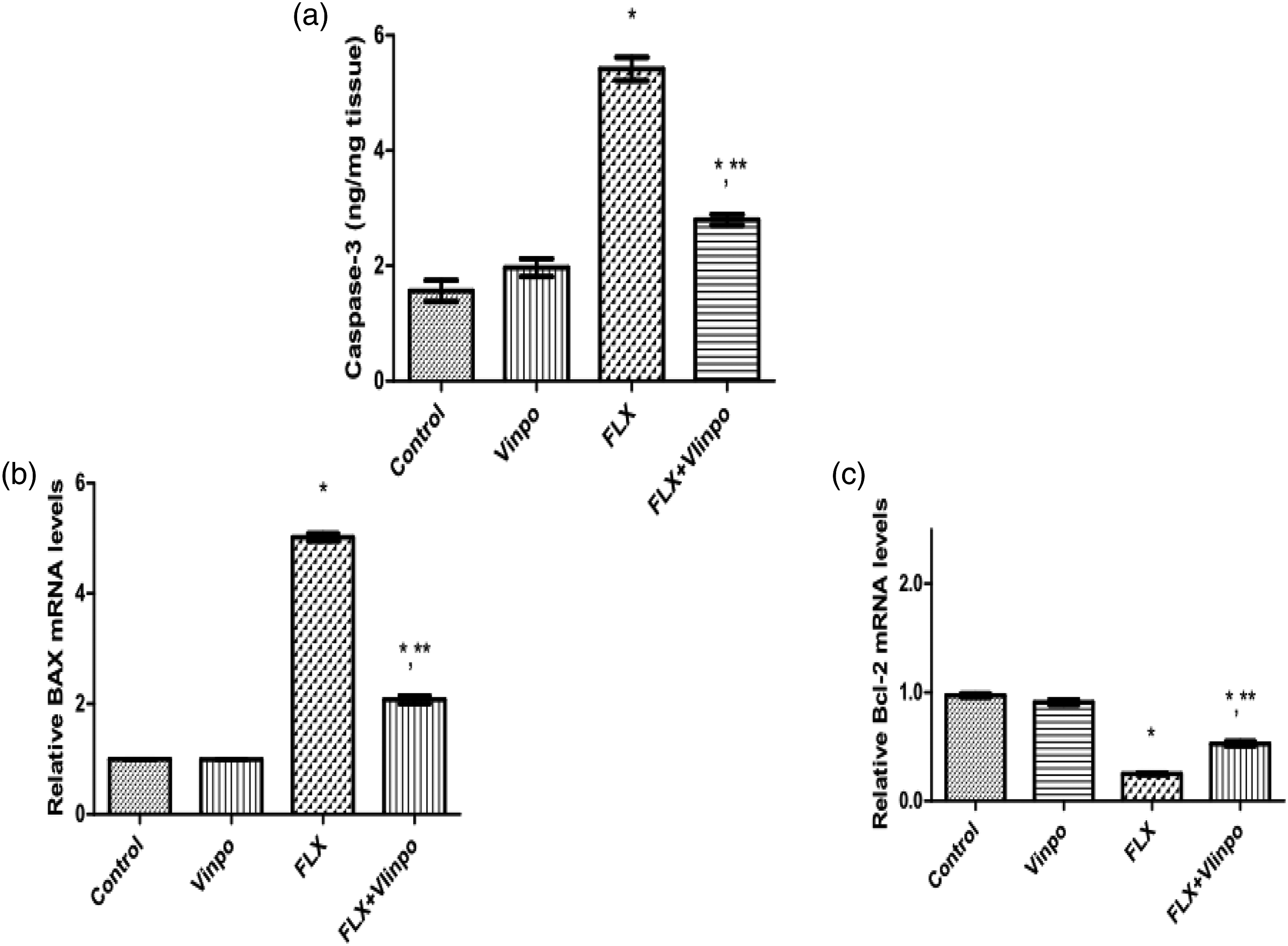
Discussion
Fluoxetine is, a commonly prescribed selective serotonin reuptake inhibitor antidepressant, used to treat disorders such as major depressive disorder, panic disorder, and bulimia nervosa. 4 Indeed, impaired liver function is seen in 0.5%–3% of patients treated with antidepressants. The molecular mechanism by which FLX induces hepatotoxicity is not fully understood, but clinical and experimental studies suggest oxidative stress, inflammatory process, and apoptosis have a role in this toxicity. 5 Vinpo, a nootropic drug, has antioxidant and anti-inflammatory properties. Hence, the current study was performed to evaluate the possible protective effect of Vinpo against FLX-induced liver injury and the underlying mechanisms.
Fluoxetine induced-liver injury is reflected by elevated liver function biomarkers, such as, serum ALT and AST, located in hepatocytes, which is released from damaged hepatocytes after cellular damage caused by FLX.20–22 These findings are consistent with several reports in which FLX caused changes in liver functions.4,5,7 Additionally, FLX caused a marked increase in the liver index (ratio of liver/body weight) which is can be explained by the accumulation of extracellular matrix (ECM) in the fibrotic tissues. 16 Moreover, FLX-induced hepatotoxicity in rats was confirmed by several histopathological changes, including inflammatory cell infiltrations, hepatocellular vacuolation with fibroplasia.4,5
Vinpo treatment significantly improved FLX-induced liver injury; including liver index, liver function biomarkers (AST and ALT), and histopathological changes. These findings indicate that Vinpo preserved hepatic functions and opposed FLX actions which come in agreement with several studies that reported the hepatoprotective effect of Vinpo.16,18,23,24
Oxidative stress plays an essential role in FLX-induced liver injury. Previous studies reported that FLX-induced hepatotoxicity has been associated with decreased anti-oxidant enzymes and increased reactive oxygen species (ROS) production which lead to damage of the cellular macromolecules, such as proteins, DNA, and lipids, producing lipid peroxidation.4,5,7 The end product of lipid peroxidation is MDA. It reacts with the cellular and tissue proteins or DNA to form adducts and induces bimolecular damage. Herein, this study showed marked oxidative stress in liver tissues of FLX-treated rats; including declined anti-oxidant defenses (GSH, SOD, and CAT) which were in parallel to the increased lipid peroxidation.5,22 Vinpo treatment markedly increased GSH, SOD, and CAT and decreased lipid peroxidation in the liver of FLX-treated rats. These results show the potential antioxidant effect of Vinpo. This antioxidant activity has been reported in multiple studies such as thioacetamide-induced liver fibrosis 16 and cisplatin‐induced liver injury. 23
Oxidative stress activates the Nrf2 pathway, which is considered the master regulator of a cellular defense mechanism against toxic insults. 25 In oxidative stress, Nrf2 stimulates the transcription of several enzymes like HO-1, 26 which has both antioxidant and anti-inflammatory actions.27,28 In the present study FLX increased hepatic Nrf2 and HO-1 levels which is associated with TBARS elevation. It was reported that the activation of the Nrf2/HO-1 pathway is a compensatory response to protect from ROS induced tissue injury in which hepatocytes are stimulated to produce a self-defense response against FLX by elevating the endogenous antioxidant levels. However, this defense mechanism failed to attenuate oxidative stress.29,30 Interestingly, Vinpo treatment ameliorated the levels of Nrf2 and HO-1 which is associated with TBARS decline in rat liver.16,18 These findings displayed the protective effect of Vinpo against FLX-induced hepatic oxidative stress. This effect could be attributed to the antioxidant effect of Vinpo. It may replace endogenous antioxidants to counteract FLX-induced oxidative stress, thus inhibiting Nrf2/HO-1 signaling activation.
It is noteworthy that oxidative stress plays an important role in the regulation of inflammation, which is triggers transcription factors such as NF-κB; releasing pro-inflammatory cytokines like TNF-α and IL-6.21,31 In accordance, the current work demonstrated that the significant increase in hepatic NF-κB, TNF-α and IL-6 levels in FLX group was attenuated by Vinpo treatment. Zhao et al. 2011, 9 has reported that Vinpo inhibits pro-inflammatory cytokines in microglia exposed to oxygen and glucose deprivation by reducing the expression of NF-κB.
PPAR-γ plays a vital role in modulating several cell functions such as adipogenesis and glucose homeostasis in addition to the regulation of inflammation. 32 Several studies have reported that PPAR-γ activation protects against drug-induced toxicity via activation of antioxidant and anti-inflammatory mechanisms.33–35 Moreover, it was reported that FLX down-regulated PPAR-γ expression in human adipose-derived Stem Cells (ASCs), the mechanism which contribute to inhibit the proliferation and adipogenic differentiation of ASCs and weight loss associated with FLX treatment.
In the same context, this study showed that FLX induced a significant down-regulation of liver PAR-γ gene expression and tissue level, an effect that was reversed by Vinpo treatment. The attenuated oxidative and inflammation by Vinpo treatment could be attributed to the up-regulated PPAR-γ. 20
Oxidative stress is the well-known initiator of apoptotic signaling which activates the mitochondrial pathway of apoptosis through upregulation of Bax and downregulation of Bcl-2.36,37 The balance between pro-apoptotic and anti-apoptotic Bcl-2 gene family is crucial for dictating the susceptibility of the cells to apoptosis. FLX treatment induced a significant decrease of antiapoptotic Bcl-2 protein and an increase of pro-apoptotic Bax, which provoked apoptosis of hepatocytes. Several studies reported that FLX-induced oxidative stress triggers the apoptotic pathway in hepatic tissues.5,38 This study showed that FLX mediated-apoptosis through enhanced Bax expression and caspase-3 activity while diminished Bcl-2 expression and this was attenuated by Vinpo treatment. Earlier studies has reported the anti‐apoptotic effect of Vinpo on drugs induced liver injury and various tissues.5,17,23 Overall these findings provided ample evidence that treating animals with Vinpo protect against FLX-induced liver damage.
Conclusion
Vinpo ameliorates FLX-induced hepatotoxicity by ameliorating oxidative stress, inflammation, and apoptosis. Further studies are needed to assess the utilization of Vinpo in the treatment of liver injury by FLX. Vinpo, a high safety profile drug, may serve as a new candidate drug to protect against FLX-induced hepatotoxicity. However, further investigations are needed to elucidate the effect of vinpocetine administration on the pharmacological actions of fluoxetine for treating neuropsychiatric disorders.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
