Abstract
Gastric ulcer (GU) is the most common health concern that occurs due to alcohol consumption, smoking and physiological stress. Ethanol-induced GU in animal model resembles the pathophysiology of human ulcer. The present study was designed to investigate the cytoprotective and anti-inflammatory properties of tert-butylhydroquinone (tBHQ), a nuclear factor erythroid 2-related factor 2 (Nrf2) activator, against gastric mucosal damage induced by acute exposure of ethanol (5 ml/kg). The intervention of tBHQ (25 and 50 mg/kg, per os (po)) and omeprazole (20 mg/kg, po) was done for 10 consecutive days. Omeprazole was chosen as a standard drug because it is prescribed for the treatment of GU. Pretreatment of tBHQ decreased gastric mucosal lesion, ulcer index, apoptotic cells and lipid peroxidation level induced by ethanol. Furthermore, the intervention of tBHQ increased gastric mucosa integrity, pH, reduced glutathione, collagen and mucus-producing goblet cells. Intervention of tBHQ increased the expression of antioxidant markers such as Nrf2, haeme oxygenase-1 and catalase and decreased the expressions of inflammatory markers such as nuclear factor kappa-light-chain-enhancer of activated B cells and cyclooxygenase-2. The cytoprotective potential of tBHQ against gastric mucosal damage might be due to its ability to enhance cellular antioxidants and anti-inflammatory responses.
Keywords
Introduction
Gastric ulcer (GU) is the most common disease with a prevalence of 20–60 per 100,000 population and accounts for 5–10% mortality worldwide. 1 Long-term occurrence with GU or duodenal ulcer has maximum risk of stomach cancer. 2 It has been reported that GU is more common in patients with liver cirrhosis, type 2 diabetes mellitus and mental health disorders. 3 –5 GU occurs due to the presence of several factors such as acid, pepsin, bile acids, Helicobacter pylori, ethanol and non-steroidal anti-inflammatory drugs (NSAIDs) that interfere with defensive factors such as tight junctions between epithelial cells, microvascular blood circulation, bicarbonate secretion, availability of prostaglandins and nitric oxide. 6 GU can be induced by pharmacological, physiological and surgical operations in several animal species. However, the most commonly used experimental models are rodents because a large number of animals are required to generate reproducible data on the efficacy of new anti-ulcer compounds. The rats are preferred over the other species and selected as the model of choice for ulcer induction because the stomach of the rat is anatomically and functionally analogous to the human stomach and can be divided into two parts: the upper non-glandular, non-secretory portion and the lower glandular secretory portion. 7 Numerous experimental models have been reported to evaluate the chemical entity possessing the anti-ulcer activity. The most frequently used models are pyloric ligation, ethanol, NSAIDs and stress-induced GU. 8 –11 The ethanol-induced acute GU model is one of the widely used experimental models. 7 Moreover, the individuals taking ethanol are at higher risk of ulcer. 12 Ethanol-induced GU model has the advantage over other GU models, resembles numerous features of acute human peptic ulcer condition. 13 Ethanol digests the mucosal layer and exposes the mucosa to the proteolytic and hydrolytic actions of hydrochloric acid and pepsin. 14 Ethanol-induced microvascular injuries by reducing blood flow, increasing the production of reactive oxygen species (ROS) and proinflammatory cytokines hence, reduced the cellular antioxidant levels. 13
Tert-butylhydroquinone (tBHQ) is a phenolic antioxidant and possess significant cytoprotective and antioxidant properties. 15 It has been reported as a selective nuclear factor erythroid 2-related factor 2 (Nrf2) activator in human keratinocytes and mouse testis model. 16,17 It showed anti-apoptotic and cytoprotective properties in the pressure overload-induced cardiac dysfunction in mice model. 15 tBHQ at the dose ranging from 20 mg/kg to 150 mg/kg exhibited antioxidant and anti-inflammatory properties in diabetes-induced atherosclerosis mice model. 18 Intervention of tBHQ exhibited protective properties in various experimental models of traumatic brain injury, cognitive performance, Alzheimer’s disease and nephrotoxicity. 19 –22 Treatment of tBHQ attenuated ethanol-induced apoptosis in H9c2 cardiomyocyte in vitro. 23 Biotransformation of tBHQ results in the formation of reactive metabolite, tert-butylbenzoquinone, which covalently binds to cysteine residue and changes the Kelch-like erythroid cell-derived protein with Cap ‘N’ Collar homology (ECH)-associated protein 1 conformation. Thereby, Nrf2 translocated to the nucleus and increases the expressions of antioxidant enzymes. 24 It has already been reported that activation of Nrf2 mitigates the GU in ischaemia/reperfused rat model via restoration of antioxidant levels. 25 The activation of Nrf2 protected the gastrointestinal tract in NSAID-induced GU and inflammatory bowel disease by decreasing the production of ROS. 26 So far there is no direct report available on the anti-ulcer as well as antioxidant properties of tBHQ against ethanol-induced acute peptic ulcer in the experimental model. The objective of the present study is to evaluate the cytoprotective properties of tBHQ against ethanol-induced acute GU model in Sprague–Dawley (SD) rats.
Methods
Chemicals
Omeprazole (CAS#73590-58-6, >98%), t-BHQ (CAS#1948-33-0, 97%), corn oil (CAS#8001-30-7, vehicle grade), thiobarbituric acid, 5,5′-dithiobis (2-nitro benzoic acid), haematoxylin and eosin (H&E), sodium dodecyl sulphate (SDS), acrylamide and ammonium persulphate were purchased from Sigma Aldrich (St Louis, Missouri, USA); absolute ethanol (99.9%) was obtained from Chong Yu Hi-Tech Chemicals (China).
Experimental animals
All the experimental protocols were approved by the Institutional Animal Ethics Committee (IAEC), of the institute against approval no IAEC/18/26 dated 14 July 2018. Male SD rats (180 ± 20) were procured from Central Animal Facility, NIPER, S.A.S. Nagar, Punjab, and acclimatized under standard conditions of temperature (22 ± 2°C), relative humidity (45–65%) and 12-h light/12-h dark cycles with free access to standard pellet diet and purified water ad libitum. The experiments were conducted as per the Committee for the Purpose of Control and Supervision of Experimentation on Animals guidelines.
Experimental design
Animals were divided into five different groups at random (Figure 1). Group 1 (vehicle control, n = 3), normal rats administered with corn oil for 10 days and denoted as ‘control’; group 2 (ulcer group, n = 4), rats were administered with normal saline for 10 days and on the 10th day after 1 h of saline, ethanol (5 ml/kg, po (oral)) was administered and denoted as ‘ulcer control (UC)’; group 3 (low dose tBHQ + UC, n = 4), rats were administered with tBHQ (25 mg/kg, per os (po)) for 10 days and on the 10th day after 1 h of the last dose, ethanol (5 ml/kg) was administered and denoted as ‘tBHQ25 + UC’; group 4 (high dose tBHQ + UC, n = 4), rats were administered with tBHQ (50 mg/kg, po) for 10 days and on the 10th day after 1 h of the last dose, ethanol (5 ml/kg, po) was administered and denoted as ‘tBHQ50+UC’; group 5 (omeprazole 20 mg/kg + UC, n = 3), rats were administered with omeprazole 20 mg/kg for 10 days and on the 10th day after 1 h of the last dose, ethanol (5 ml/kg) was administered and denoted as ‘omeprazole20 + UC’. tBHQ was dissolved in corn oil and omeprazole was suspended in 1% sodium carboxymethyl cellulose (weight/volume (w/v)). Both the intervention was administered orally at 2 ml/kg po. Animals were allowed to fast (but provided free access to water) for 24 h in metabolic cages to repudiate coprophagy in order to eliminate the interference of faecal matter in the induction of GU.

Schematic presentation of experimental design. Group 1 (control) received corn oil, group 2 (ulcer control; UC) received saline, group 3 (tBHQ25 + UC) and group 4 (tBHQ50+UC) received tBHQ at the dose of 25 and 50 mg/kg/day in corn oil, respectively, for 10 consecutive days. Group 5(Omeprazole20 + UC) received omeprazole at the dose of 20 mg/kg/day in 1% sodium carboxymethyl cellulose. All the groups, except group 1, received intragastric ethanol (5 ml/kg) at 10th day after 1 h of last intervention dose and rats were killed 1 h after ethanol administration. tBHQ: tert-butylhydroquinone; CMC: carboxymethyl cellulose; UC: ulcer control.
Rationality of dose selection
Ethanol (5 ml/kg) have damaging effects in the stomach, which readily penetrates and digest the gastric wall by proteolytic and hydrolytic actions, generate ROS and reduce blood circulation, increase apoptosis. 27 The dose of ethanol (5 ml/kg) was selected for the induction of GU in rats based on the previous literature. 28 The dose range 25–50 mg/kg of tBHQ was selected based on its cytoprotective effect in hypoxic–ischaemic brain damage in rats and athero-protection in diabetic mice. 29,30 Omeprazole (20 mg/kg, po), suppresses stomach acid secretion by specific inhibition of the H+/K+-ATPase system, was selected as a standard treatment in the present study. 31
Tissue collection and preparation
The animals were euthanized by decapitation after 1-h ethanol administration, stomach of each animal were removed, opened along the greater curvature and washed in chilled phosphate buffer saline to remove blood clots. Gastric contents were collected for the determination of gastric pH. The stomach tissues were blotted dry, fixed on paraffin cardboard and photographs were captured for the determination of ulcer area. Each stomach tissue was divided into two equal parts, right half and left half. Right half from all animals were immediately immersed in 10% (volume/volume (v/v)) formal saline for histopathological and immunohistochemical evaluations. The other left half was homogenized and evaluated for biochemical and immunoblotting parameters.
Evaluation of gastric mucosal lesions and percentage of inhibition
The severity of gastric mucosal erosions was evaluated and scored according to the ulcer score scale described by Zhang et al. 32 (0): no erosion, (*): pinpoint erosions, (**): ulcer erosion size <1 mm, (***): ulcer erosion between 1 and 5 small erosion and (****): ulcer erosion size >2 mm. The percentage of inhibition of gastric erosion was calculated by using following formula: [(UIcontrol − UItreated)/UIcontrol] × 100, where UI represents ulcer index.
Evaluation of gastric pH
The gastric content was collected, centrifuged at 1000 r/min for 10 min at 4°C, and the supernatant was used for the measurement of pH using a digital pH meter (PB-11; Sartorius, Sartorius AG, Göttingen, Germany) as suggested by Bigoniya and Singh. 33
Evaluation of gastric biochemical parameters
Evaluation of gastric MDA activity
The malondialdehyde (MDA) level in the stomach was measured according to the method previously described. 34 The stomach was homogenized in phosphate buffer (pH 7.4, 3 mM ethylenediaminetetraacetic acid), centrifuged at 10,000 r/min and supernatants were further used for MDA estimation. The supernatant (50 μl) was incubated with 0.8% thiobarbituric acid (375 μl), 8.1% SDS, 50 μl) and 20% acetic acid (AA, 375 μl) for 1 h at 95 C, and absorbance was determined spectrophotometrically at 532 nm. Known concentrations of MDA standard (1,1,3,3-tetramethoxy propane) were used for the generation of the standard curve and expressed as micrometres of MDA per milligram of protein.
Evaluation of gastric of reduced glutathione level
The supernatant (0.25 ml) from the previously homogenized tissue was mixed with 5% sulphosalicylic acid and kept in ice for 30 min for the precipitation of protein and centrifuged at 10,000 r/min for 10 min at 4 C. The supernatant (200 μl) was mixed with 5,5′-dithiobis (2-nitro benzoic acid) and incubated at 37 C for 10 min. Product absorbance was measured at 412 nm and calculated using known concentrations of reduced glutathione (GSH) for the generation of the standard curve. The results were expressed as micrometres of reduced GSH per milligram of protein. 35
Evaluation of gastric cellular damage
Histopathological examination
Stomach tissue was fixed in buffered formalin solution (10% v/v) and embedded in paraffin. Sections were cut (5 μm) using microtome (Leica RM2145, Germany), mounted on Mayer’s albumin-coated glass slides and dried overnight. Sections were deparaffinized in xylene, rehydrated with alcohol, stained with H&E, mounted with dibutylphthalate polystyrene xylene (DPX) and examined under microscope. 36 Special staining for collagen fibres was performed by picrosirius red (PSR) and pentachrome staining. Goblet cells were stained with alcian blue periodic acid–Schiff’s base (PAS) stain. Briefly, deparaffinized and rehydrated sections were acidified with AA (3%), followed by incubated with alcian blue solution (1% in 3% AA w/v, pH 2.5). Furthermore, slides were treated with periodic acid (0.5%) and Schiff’s reagent. Processed sections were DPX mounted and examined under the microscope. The % collagen and goblet cells areas were quantified using ‘Image J’ software (Version 1.44p NIH, USA).
Evaluation of gastric apoptotic cells by TUNEL assay
The experiment was performed according to manufacturer protocol using terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling (TUNEL) assay kit (Calbiochem, Oncogene Research Products, La Jolla, California, USA). Images were acquired using a charged coupled device camera and TUNEL-positive cells were counted using image analysis software ‘Isis’ (Carl Zeiss, AxioImager M1, Germany)
Immunohistochemical analyses of phospho-nuclear factor kappa light chain enhancer of activated B cells p65, COX-2 and HO-1 in gastric tissue
Immunohistochemistry (IHC) was performed by using impress excel staining kit (cat# MP-7601; Vector Laboratories, Burlingame, California, USA) according to the manufacturer’s protocol. Briefly, the sections were deparaffinized, hydrated and antigen retrieval was done by boiling at 95 C in citrate buffer (10 mM, pH 6) for 30 min. The sections were incubated overnight with primary antibodies of anti-cyclooxygenase-2 (COX-2; sc-796), anti-p-NF-κB p65 (sc-33039) and haeme oxygenase-1 (HO-1; sc-10789) at 1:50 dilution in tris-buffered saline (TBS). Furthermore, sections were incubated with secondary antibody and counterstained with haematoxylin. The % immunopositive areas were quantified using ‘Image J’ software. 37
Quantification of protein expression by Western blotting
The stomach samples were homogenized in radioimmunoprecipitation assay buffer and resolved on SDS-polyacrylamide gel electrophoresis. The resolved protein transferred onto polyvinylidene difluoride/nitrocellulose membrane. After blocking with (3%) bovine serum albumin, the membrane was incubated with anti-catalase (CAT, sc-50508), and anti-Nrf2 (sc-722) polyclonal rabbit primary antibody, followed by incubation with horseradish peroxidase-conjugated secondary antibody. Enhanced chemiluminescence solution and X-ray films were used for the detection of the luminescent signal. Densitometric analyses were quantified by Image J software. 38
Statistical analyses
All results were expressed as mean ± standard error of the mean for each animal group. Graph pad prism 7 (Statistical Software, California corporation) was used for statistical analyses. Analysis of variance was used for multiple comparisons. p Values ≤ 0.05 was considered significant.
Results
Effect of tBHQ on morphometric parameters
The ethanol exposed ulcer group showed a significant increase in stomach weight (p < 0.05) and organ coefficient (p < 0.001) as compared to the control group (Figure 2(a) and (b)). Pretreatment with tBHQ at both the doses 25 and 50 mg/kg and omeprazole 20 mg/kg for 10 days showed a significant decrease in stomach weight and reduction in organ coefficient as compared to per se GU group.

Effect of tBHQ on body weight (a) and organ coefficient (b). All the values are expressed as mean ± SEM (n = 3–4). *p < 0.05 and ***p < 0.001 versus control; @ p < 0.05, @@ p < 0.01 and @@@ p < 0.001 versus UC. tBHQ: tert-butylhydroquinone; SEM: standard error of the mean; UC: ulcer control.
Effect of tBHQ on ulcer area and percentage of inhibition
Oral administration of absolute ethanol led to severe (Figure 3) gastric lesions with extensive visible hemorrhagic erosion and infiltration of inflammatory cells results in high UI as compared to the control group. Pretreatment of tBHQ 25 and 50 mg/kg significantly reduced the UI compared to per se ulcer group. Anti-ulcer drug omeprazole 20 mg/kg also significantly decreased UI, when compared to the GU group (Figure 4). Pretreatment with tBHQ 25 and 50 mg/kg and omeprazole 20 mg/kg showed significant (65.57%, 77.04% and 66.12%, respectively) percent inhibition as compared to per se UC.

Representative photographs illustrating the morphology of rat stomach at the time of necropsy. Control group ((a) to (c)) showed the animals without any visible lesions. The ulcer group ((d) to (f)) showed moderate to severe lesion in the gastric mucosa as compared to the control group. Low dose tBHQ 25 mg/kg ((g) to (i)), high dose 50 mg/kg ((j) to (l)) and omeprazole 20 mg/kg (m to o) groups showed mild lesions. White arrows indicate the ulcer erosion. tBHQ: tert-butylhydroquinone.
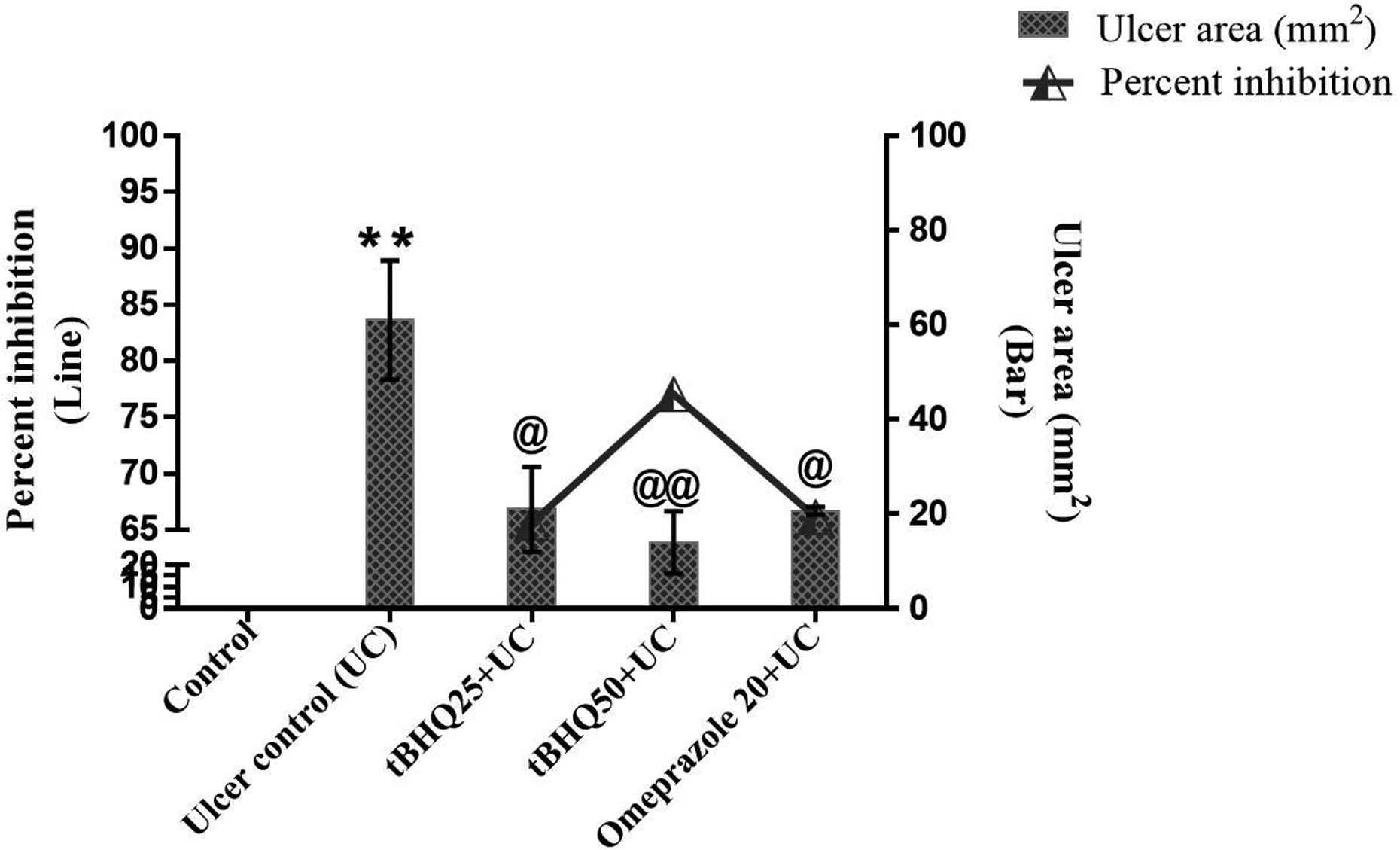
Effect of tBHQ on ulcer index and per cent inhibition. All the values are expressed as mean ± SEM (n = 3–4). **p < 0.01 versus control; @ p < .05 and @@ p < .01 versus UC. tBHQ: tert-butylhydroquinone; SEM: standard error of the mean; UC: ulcer control.
Effect of tBHQ on GU-induced pH
Administration of absolute ethanol significantly decreased gastric pH (p < 0.01) as compared to the control group (Figure 5). Pretreatment with tBHQ 25 and 50 mg/kg showed significant increases in gastric pH (p < 0.05 and p < 0.01, respectively) as compared to per se ulcer group, whereas pre-administration of omeprazole 20 mg/kg for 10 days showed significant increase in gastric pH (p < 0.05) as compared to GU group.
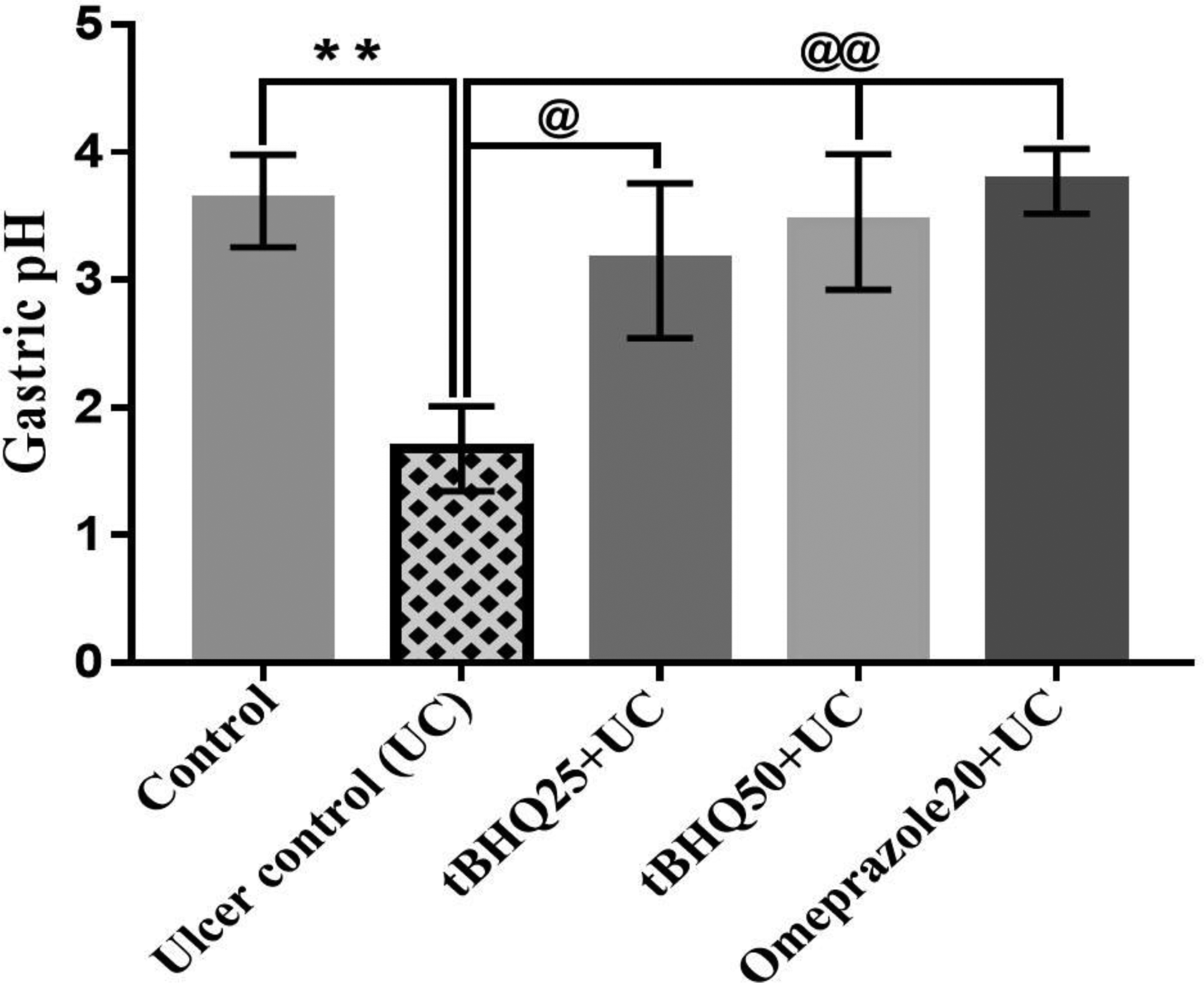
Effect of tBHQ on gastric pH. All the values are expressed as mean ± SEM (n = 3). **p < 0.01 versus control; @ p < 0.05 and @@ p < 0.01 versus UC. tBHQ: tert-butylhydroquinone; SEM: standard error of the mean; UC: ulcer control.
Effect of tBHQ on MDA and GSH in GU condition
GU-induced by ethanol was associated with a significant increase in MDA (p < 0.001) and decrease in reduced GSH (p < 0.01) as compared to the control group. Treatment with tBHQ 25 and 50 mg/kg showed significant decrease in MDA (p < 0.001) and no effect on the GSH level (Figure 6(a) and (b)). Treatment of omeprazole 20 mg/kg showed significant decrease in MDA (p < 0.001) and increase (p < 0.01) in GSH level.

Effect of tBHQ on MDA (a) and GSH (b) level. All the values are expressed as mean ± SEM (n = 3). **p < 0.01 and ***p < 0.001versus control; @@ p < 0.01 and @@@ p < 0.001 versus UC. tBHQ: tert-butylhydroquinone; MDA: malondialdehyde; GSH: glutathione; SEM: standard error of the mean; UC: ulcer control.
Effect of tBHQ on GU evaluated by histopathology
Stomach of ethanol-exposed ulcer group showed loss of epithelium integrity and glandular structure with severe mucosal and submucosal damage, oedema of submucosal cell, necrosis and inflammatory response as compared to the control group (Figure 7(a)). Conserved mucosal integrity and reduction in tissue damage were observed with the treatment of tBHQ- and omeprazole-treated groups. Ethanol-exposed rats showed a significant decrease in the amount of collagen (red stain in yellow background) as compared to the control group in PSR and pentachrome staining (Figure 7(b) and (d)). Significant restoration in the amount of collagen was observed in tBHQ and omeprazole pretreated groups. In the ulcer group, a number of mucus-producing goblet cells were significantly decreased as compared to the control group (Figure 7(c)). Pretreatment of tBHQ at both the doses and omeprazole significantly increased the number of goblet cells as compared to per se ulcer group.

Representative photomicrographs showing stomach sections stained with H&E (a); PSR staining (b); alcian blue-PAS staining (c) and pentachrome staining (d): In H&E staining, ulcer group was observed with the loss of epithelial and glandular structures (black arrows) in mucosal (mu) and submucosal (subm) region as compared to the control. tBHQ-and omeprazole-treated groups preserved the integrity (yellow arrows) of gastric mucosa as compared to the ulcer group. In PSR and pentachrome staining, the ulcer group was observed with less collagen content (black arrows) deposition in mu, subm and muscularis (ml) region as compared to the control group. tBHQ- and omeprazole-treated groups showed restoration of collagen content (blue arrows) as compared to the ulcer group. In alcian blue PAS staining, the ulcer group was observed with less goblet cells (black arrows) in mu region as compared to control. tBHQ- and omeprazole-treated groups showed restoration of goblet cells (yellow arrows) in mu region as compared to the ulcer group. All the values are expressed as mean ± SEM (n = 3). *p < 0.05 and ***p < 0.001 versus control; @ p < 0.05, @@ p < 0.01 and @@@ p < 0.001 versus UC. H&E: haematoxylin and eosin; PSR: picrosirius red; PAS: periodic acid-Schiff’s base; tBHQ: tert-butylhydroquinone; SEM: standard error of the mean; UC: ulcer control.

(a to f) Representative photomicrographs showing COX-2 immunohistochemical staining (magnification 200×): Immunopositive areas are brown in colour. Higher magnification is inserted in the respective panels, in which, black arrows indicate immunopositive cells. All the values are expressed as mean ± SEM (n = 3). ***p < 0.001 versus control; @@@ p < 0.001 versus UC. COX-2: cyclooxygenase-2; SEM: standard error of the mean; UC: ulcer control.

(a to f) Representative photomicrographs showing p-NF-κB p65 immunohistochemical staining (magnification 200×): Immunopositive areas are brown in colour. Higher magnification is inserted in the respective panels, in which, black arrows indicate immunopositive cells. All the values are expressed as mean ± SEM (n = 3). ***p < 0.001 versus control; @@@ p < 0.001 versus UC. UC: ulcer control; SEM: standard error of the mean.

(a to f) Representative photomicrographs showing HO-1 immunohistochemical staining (magnification 200×): Immunopositive areas are brown in colour. Higher magnification is inserted in the respective panels, in which, black arrows indicate immunopositive cells. All the values are expressed as mean ± SEM (n = 3). ***p < 0.001 versus control; @@@ p < 0.001 versus UC. HO-1: haeme oxygenase-1; UC: ulcer control; SEM: standard error of the mean.
Effect of tBHQ on GU-induced expression of COX-2, p-NF-κB p65 and HO-1 proteins as evaluated by IHC
The GU group showed a significant increase (p < 0.001) in the expression of COX-2, p-NF-κB p65 and HO-1 (Figures 8 -10). Pretreatment with tBHQ at both the doses and omeprazole showed significant (p < 0.001) decrease in the expression of COX-2 and p-NF-κB p65. A significant (p < 0.001) increase was observed in the expression of HO-1 in the groups pretreated with tBHQ (at both the doses) and omeprazole as compared to the ulcer group.
Effect of tBHQ on apoptosis induced by GU
Administration of ethanol significantly increased apoptotic cell (p < 0.001) in the ulcer group as compared to the control group (Figure 11). Pretreatment with tBHQ at both the doses and omeprazole 20 mg/kg significantly decreased the number of apoptotic cells (p < 0.001) as compared to the ulcer group.

(a to f) Representative photomicrographs showing the TUNEL-positive cells in stomach tissue sections. The given images are the merged 4′,6-diamidino-2-phenylindole (DAPI) + fluorescein isothiocyanate (FITC) pictures. Control group ((a) to (c)) showed the animals without any visible lesions. The ulcer group (b) showed moderate to severe TUNEL positive in the gastric mucosa as compared to control group. Low dose tBHQ 25 mg/kg (c), high dose 50 mg/kg (d) and omeprazole 20 mg/kg (e) groups showed TUNEL positive compared to UC group. White arrows indicate TUNEL positive (apoptotic) cells. All the values are expressed as mean ± SEM (n = 3). ***p < 0.001 versus control; @@@ p < 0.001 versus UC. TUNEL: terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labelling; tBHQ: tert-butylhydroquinone; UC: ulcer control; SEM: standard error of the mean.
Effect of tBHQ on the expressions of Nrf2 and CAT in GU
Ethanol-exposed ulcer group significantly decreased the expression of Nrf2 and CAT (p < 0.001) as compared to the control group and tBHQ at both the doses, significantly (p < 0.001) increased the Nrf2 expression as compared to the ulcer control group. Whereas, tBHQ at both the doses significantly (tBHQ 25 mg/kg, p < 0.01 and tBHQ 50 mg/kg, p < 0.001) increased CAT expressions as compared to the ulcer control group (Figure 12).
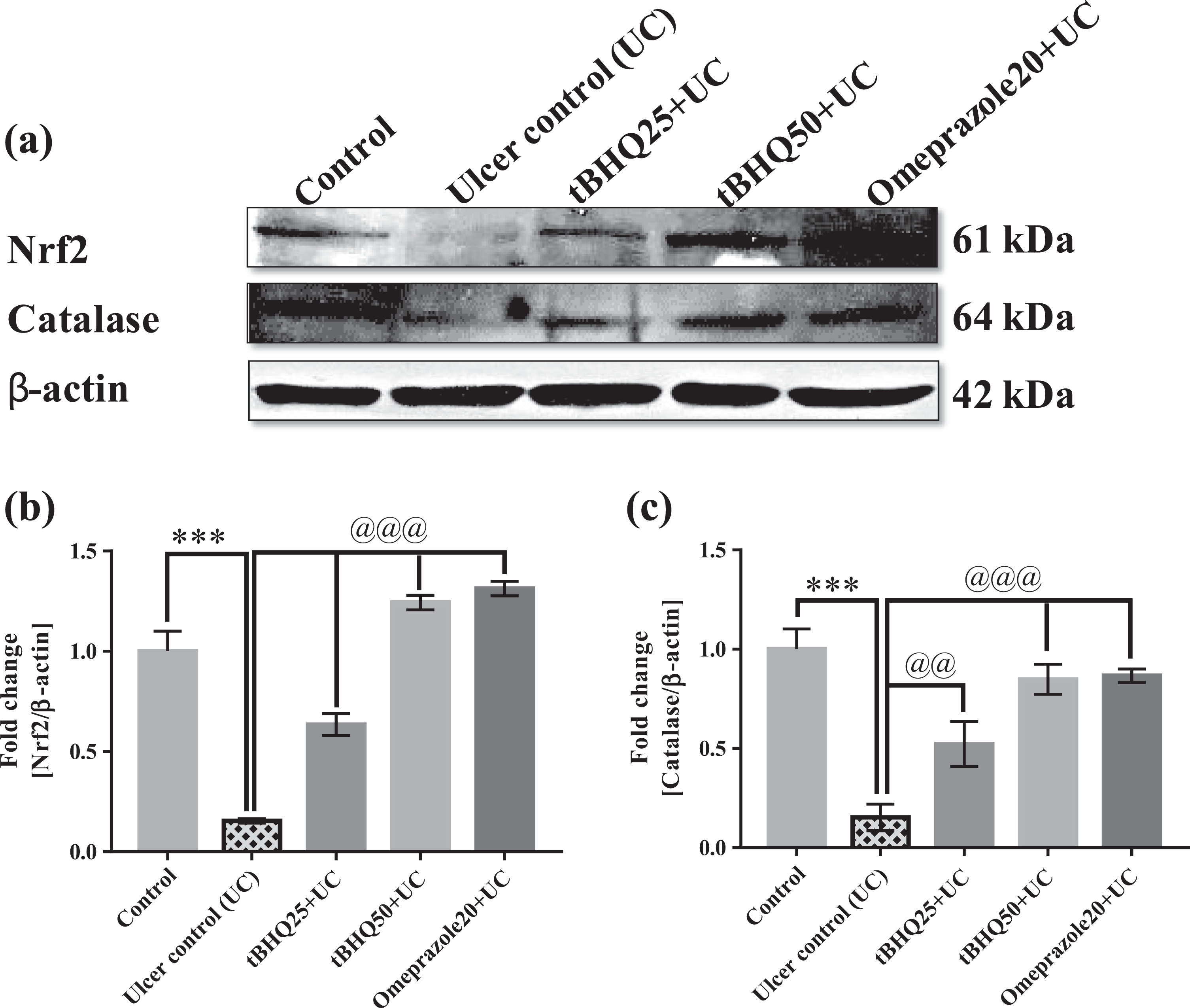
(a to c) Representative immunoblots of Nrf2 and CAT and their quantification stomach. All the values are expressed as mean ± SEM (n = 3). ***p < 0.001 versus control, @@ p < 0.01 and @@@ p < 0.001 versus UC. Nrf2: nuclear factor erythroid 2-related factor 2; CAT: catalase; UC: ulcer control; SEM: standard error of the mean.
Discussion
GU occurs due to imbalance between endogenous aggressive (increased secretion of hydrochloric acid and other proteolytic enzyme such as pepsin, increase in the number of H. pylori) and defensive factors (decreased mucus secretion and prostaglandin production and reduced endothelial blood flow). 13 For the first time, we have reported the cytoprotective, anti-ulcer, antioxidant, anti-inflammatory and anti-apoptotic action of tBHQ against ethanol-induced GU in SD rats. Ethanol exposure produces gastric lesion by penetrating and digesting the gastric wall due to its proteolytic and hydrolytic action as well as by endothelial cell damage due to reduction in blood circulation. 13 In the present study, ethanol-exposed rats were observed with increased stomach weight and stomach coefficient, while pretreatment with tBHQ at both the doses ameliorated stomach weight and stomach coefficient. It has previously reported that acute exposure of ethanol leads to the increase in stomach weight and stomach coefficient, which was due to haemorrhage, oedema, necrosis and inflammation. 39 Moreover, no significant change in the body weight was observed, which might be due to the acute exposure of ethanol. In the present study, a significant decrease in gastric juice pH was observed in per se ethanol-exposed stomach in comparison to control stomach and pre-administration of tBHQ at both the doses and omeprazole 20 mg/kg significantly increased the pH in comparison to per se ulcer group. It has previously reported that acute exposure of ethanol decreased gastric juice pH as compared to the control group by increasing acid secretion. 40 Administration of absolute ethanol showed significant increase in UI, while pre-administration of tBHQ at both the doses showed significant reduction in UI. Ismail et al. reported that administration of ethanol produced linear haemorrhagic lesions, extensive submucosal oedema, mucosal friability, inflammatory cells infiltration and epithelial cell loss into the gastric lumen. 41,42 So, it could be concluded that the number of erosions is directly proportional to the severity of GU and amelioration by the intervention of protectant can decrease the same. Moreover, the % inhibition of ulcer was 65.57%, 77.04% and 66.12% with tBHQ 25 and 50 mg/kg and omeprazole 20 mg/kg treatment, respectively.
In the present study, administration of absolute ethanol significantly increased the MDA and decreased the reduced GSH levels in the stomach as compared to the control group (Figure 13). Pre-administration of tBHQ and omeprazole significantly reduced MDA level as compared to the ulcer group. Moreover, tBHQ pretreatment showed no change in GSH level, whereas pretreatment with omeprazole showed a significant increase in GSH level as compared to the GU group. Liu et al. reported that administration of absolute ethanol produces ROS that further stimulates lipid peroxidation in gastric tissue and causes the severe damage of gastric mucosal layers. 43

Schematic presentation illustrating the possible pathways of GU induction and targets of ulcer protection by tBHQ. GU was induced by ingestion of ethanol that metabolize into acetaldehyde and increased the production of ROS. ROS increased the MDA and decreased reduce GSH, CAT and resulted mucosal damage. Ethanol also caused the digestion of mucosal epithelial layer and collagen, which contributed in GU. Pretreatment of tBHQ and its active metabolite tBBQ prevented the GU through cytoprotective actions and the reduction in oxidative stress. GU: gastric ulcer; tBHQ: tert-butylhydroquinone; ROS: reactive oxygen species; MAD: malondialdehyde; GSH: glutathione; CAT: catalase; tBBQ: tert-butylbenzoquinone.
In the present study, H&E staining revealed the severe damage in the mucosal and submucosal layer, presence of inflammatory cells, vascular oedema, gastric erosion and reduced epithelium integrity in ethanol-exposed group as compared to the control group. Whereas, tBHQ at both the doses and omeprazole restored the epithelial, mucosal and submucosal integrity in the stomach as compared to per se ethanol-exposed group. It has previously been reported that ethanol administration showed typical histological changes that include the loss of gastric mucosa, epithelium integrity, vascular congestion, submucosal oedema and necrosis as well as an inflammatory response due to neutrophil and eosinophil infiltration. 44 Hajrezaie et al. reported that ethanol exposure digested the circular muscles in the stomach that led to the decrease in the integrity of gastric epithelial layers, mucosal thickness and destruction of mucosal glands. 45 Moreover, PSR and pentachrome staining were performed to evaluate the amount of collagen, which provides the strength and maintains the integrity of mucosal, submucosal and muscular layers of the stomach. Exposure of ethanol showed a significant reduction in collagen levels, while pretreatment with tBHQ and omeprazole significantly restored collagen levels in the gastric tissue. It has been well reported that ethanol directly dissolves the collagen fibre, which have a protective role against GU. 46 Liu et al. reported the decrease in the amount of collagen after the acute exposure of ethanol in the ulcer group. 47 Furthermore, PAS alician blue staining revealed a significant reduction in the number of goblet cells in ethanol-exposed stomach in comparison to the control stomach. While pretreatment with tBHQ at both the doses and omeprazole showed increase in the number of goblet cells. Goblets cells release the mucus that protects the mucosal layer of the stomach against damaging effects of hydrochloric acid and pepsin. Namulema et al. reported the reduction in the number of goblet cells in ethanol-exposed rats. 48
IHC staining revealed the increased expression of phospho-NF-κB p65 and COX-2 in ethanol-exposed stomach in comparison to control. While pre-administration of tBHQ at both the doses significantly reduced the expression of p-NF-κB p65 and COX-2 in comparison to the ulcer group. The transcription factor NF-κB is the major inflammatory mediator that further facilitates and activates the pathways producing inflammatory cytokines. 49,50 Previously, van den Berg et al. reported the massive inflammatory cell infiltration in the stomach of rat exposed to ethanol. 51 COX exists in two isoforms, COX-1 and COX-2, and the expression of COX-2 increases in inflammatory conditions such as GU. 52 Positive COX-2 immunolocalization has been found in various cell types of the stomach such as mesenchymal inflammatory cells, endothelial cells, surface epithelial cells and parietal cells. Salman et al. reported the expression of COX-2 dominates at the site of inflammation in ethanol-induced GU. 27 In the present study, administration of ethanol significantly increased the per cent of apoptotic (TUNEL-positive) cells in the gastric mucosa and intervention of tBHQ reduced number of apoptotic cells in comparison to the ethanol-exposed GU group. Liu and Cho reported the exposure of ethanol produced mucosal damage due to necrotic and apoptotic effects. 53 Transcription factor, Nrf2 plays a critical role in the defensive mechanism of the cell by maintaining the antioxidants status in the cell. Nrf2 has been reported to inhibit pro-inflammatory signalling and reduces inflammation by downregulating NF-κB. 26 The downstream effector of Nrf2 signalling includes the antioxidant enzyme HO-1, CAT and reduced GSH. In the present study, immunoblotting showed a significant decrease in the expression of Nrf2 and CAT in ethanol-exposed ulcer group as compared to control. Whereas, pretreatment with tBHQ at both the doses and omeprazole increased the expression of Nrf2, which further facilitated the increase in the expression of HO-1 and CAT in comparison to the ulcer group. It has also been reported that HO-1 possesses protective roles against ethanol-induced ulcer in rats through the involvement of Nrf2/HO-1 pathway. 32
Altogether, the present study provides significant evidence related to the potent anti-ulcer effects of tBHQ against ethanol-induced gastric mucosal injury. tBHQ exhibited cytoprotective effects mediated by the activation of transcription factor Nrf2 and its downstream HO-1 and CAT, restoration of collagen and goblet cells, reduction of apoptosis and downregulation of NF-κB. The prospective use of tBHQ as a protective agent against ulcer could be a viable strategy to ameliorate gastric mucosal injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Pharmaceutical Education and Research (NIPER), S.A.S. Nagar, Punjab, India, for carrying out the present experimentation.
