Abstract
In this study, to investigate the effect of overexpression of miR-146a on autophagy of hippocampal neurons in rats with intracerebral hemorrhage (ICH), 72 Sprague-Dawley rats were randomly divided into the sham, ICH, miR-146a agomir, and miR-146a agomir control groups. The ICH model was constructed by injection of collagenase VII. The apoptosis of hippocampal neurons was measured by TUNEL assay. The levels of LC3 and Beclin 1 were analyzed by immunohistochemistry. Mitochondrial autophagy was examined by transmission electron microscopy. The levels of LC3A, LC3B, Beclin 1, Bax, Bcl-2, and cleaved caspase 3 were examined by Western blot. Western blot was also used to evaluate the expression of nuclear factor κB signaling pathway-related factors. To examine the effect of autophagy inhibitor (3-methyladenine (3-MA)) on miR-146a-regulated apoptotic protein expression, 30 rats were further divided into the sham, ICH, miR-146a agomir, 3-MA, and miR-146a + 3-MA groups. The levels of Bax, Bcl-2, and cleaved caspase 3 were examined by Western blot. Compared with the sham group, the nerve function scores, brain water content, the percentage of apoptotic cells, and the expression levels of LC3, Beclin 1, Bax, cleaved caspase 3, and p-P65 in the hippocampus of rats in the ICH group were all significantly increased (p < 0.05), whereas the expression levels of miR-146a, Bcl-2, and p-IκBα were markedly decreased (p < 0.05). Mitochondrial autophagy was also evident. Furthermore, compared with the ICH group, the results of the abovementioned tests in the miR-146a agomir group were reversed. The overexpression of miR-146a inhibited the autophagy of hippocampal neurons in rats with ICH.
Introduction
Intracerebral hemorrhage (ICH) is associated with a high rate of morbidity and mortality. Statistics indicate that 15% of stroke cases in developed countries are due to ICH and its counterpart in developing countries is 50% or more. 1,2 The rate of mortality among patients with ICH is 40–50%, and in more than half of cases, death occurs within 2 days following ICH. In addition, 75% of survivors are unable to live independently after 1 year, and only 30% of patients survive after 5 years. 3,4 The blood circulation around the cerebral ischemia is interrupted following ICH, the oxygen supply to nerve cells is impaired, which then leads to cerebral hypoxia, associated with autophagy. 5 In addition, it has been demonstrated that excessive autophagy in the short term may be involved in the brain damage induced by endoplasmic reticulum stress. 6 The identification of strategies which may be used to protect against secondary nerve injury following cerebral hemorrhage by autophagy and to reduce the apoptosis of nerve cells has become a research hotspot.
miRNAs are evolutionarily highly conserved small RNAs without a coding ability, which can bind to the noncoding domain of the 3-terminal end of mRNAs to affect their expression. The miR-146 family has two members, miR-146a and miR-146b, whose coding genes are located on chromosomes 5q34 and 10q24, respectively. miR-146 is highly conserved, differing only in the two nucleotide positions at the 3′ end, and its family members have the same seed region; therefore, they have the same targeted genes. 7,8 Previous studies demonstrated that the overexpression of miR-146a contributed to the dysfunction of cerebral microvascular cells in rats with ICH. 9 miR-146a can alleviate inflammation in psoriasis and can restrain the proliferation of epidermal cells to reduce the infiltration of neutrophils. 10,11 miR-146a is one of the miRNAs expressed most abundantly in the human central nervous system and has been proved to be expressed in a number of nervous system diseases. However, the role of miR-146a in ICH has not yet been specifically studied.
Nuclear factor κB (NF-κB) proteins are key regulatory proteins associated with inflammation and immunity. They are composed of NF-κB/Rel family and exist in the cytoplasm, in the form of homologous or heterodimer proteins. IκBs phosphorylated by IKK kinase can activate NF-κB under pathological stimulation, such as inflammation and stress, which can lead to NF-κB entering the nucleus and mediating the expression of related genes, participating in the regulation of the inflammatory response, cell proliferation, and apoptosis. 12 Extensive studies have demonstrated the role of NF-κB transcription in the promotion and progression of several tumors, in which NF-κB constitutive activation and/or overexpression are common clinical features. 13 Through the analysis of autophagy in patients with intractable heart failure, autophagy has been extensively detected in end-stage heart failure and results in secondary cell death. The occurrence of secondary cell death of autophagy exceeds that of apoptosis. Conversely, the activation of the receptor interacting protein 1 (RIP1)/NF-κB pathway has been shown to be associated with cell survival. 14 Recent studies have indicated that NF-κB is activated following ICH 15,16 ; however, whether its activation is associated with autophagy remains to be determined.
This study aimed to examine the effects of miR-146a on the autophagy and apoptosis of hippocampal neurons of rats with ICH.
Materials and methods
Animals
Male healthy Sprague-Dawley rats (8–10 weeks old) of specific pathogen-free grade (n = 102), weighing approximately 240 ± 20 g, were obtained from Jinan Pengyue Experimental Animal Breeding Co., Ltd (China; Animal Production License No. SCXK (Lu) 2014-0007). All rats were maintained under standard conditions (temperature, 23 ± 2°C; air humidity, 55 ± 5%; and a 12-h day/12-h night cycle) and had free access to food and water. The animal study protocol was in line with the National Institutes of Health (NIH Publication No. 85-23, revised 1996) and approved by the Institutional Animal Care and Use Committee of Qingdao Women’s and Children’s Hospital.
Construction of ICH model
The ICH model was constructed by an injection of 0.2 U/µl collagenase type VII, according to previously published data. 17 The rats were randomly divided into two groups, the sham group (n = 18) and the operation group (n = 54). The rats were subjected to preoperative fasting for 12 h and were anesthetized by an injection of 3% pentobarbital sodium into the cavum abdominis (50 mg/kg). 6 The rats were then placed in the prone position on the WDT-V-type stereotaxis instrument. According to the stereotactic guidance, the position of the needle insertion was determined as follows: A square bone window approximately 2 × 2 mm2 was opened in the injection point (the frontal fontanel as origin, 1.4 mm backward, and 3.2 mm to the right of sagittal suture). The needle was then inserted downward approximately 5.6 mm to reach the globus pallidus and collagenase VII (2 μl (0.20 U/μl)) was injected slowly (C2799; Sigma, Shanghai, China). The microinjector was slowly pulled out, and the foramen was sealed by bone wax. After the operation, the skin were sterilized and stitched. The rats in the sham group were prepared as those in the ICH group and were injected with a 2 μl amount of normal saline into the right striatum. All operations were performed under sterile conditions. Rats were fed in cages after the operation and had access to food and water.
Subgrouping and administration
The rats with ICH were randomly divided into the ICH group, miR-146a agomir group, and agomir control group (n = 18 per group; RiboBio Biotechnology, Guangzhou, China). From the second day after modeling, the rats were fixed on the WDT-V-type stereotaxis instrument following anesthesia and were administered an intracerebroventricular injection (0.8 cm after the anterior fontanel, 1.5 cm to the right of raphe, and 2.5 cm to the ventral side of cerebral cortex). The rats in the sham and ICH groups were treated with 10 μl of normal saline. The rats in the miR-146a agomir and agomir control groups were treated with 2 mg/kg of miR-146a agomir and agomir control groups (dissolution in 10 μl amount of normal saline), respectively. 9 The rats were injected every 2 days. After modeling on the seventh day, the rats were anesthetized with 3% pentobarbital sodium (50 mg/kg) and euthanized.
Nerve function scores
After the seventh day of modeling, the rats were subjected to the nerve function scores to evaluate the level of the function defect of the nervous system with the adoption of a five-mark scoring method described by Berderson et al.. 18 The scoring criteria are presented in Table 1. Higher scores indicate more severe neurological deficits.
Scoring criteria.

RT-qPCR
In each group, six rats were used and anesthetized by an injection of 3% pentobarbital sodium (50 mg/kg) and then euthanized. The brains were removed and placed on ice. Part of the hippocampus was frozen in liquid nitrogen for use in Western blot analysis, and the other part was homogenized after grinding and centrifugation under 4°C (1204 × g, 15 min). Total RNA was isolated using TRIzol reagent (15596018; Thermo Fisher Scientific, Shanghai, China). The ratio of OD260/OD280 between 1.8 and 2.0 indicates that the purity of RNA was qualified. The RNA was reversed transcribed using the TaqMan™ MicroRNA Reverse Transcription kit (4366596; Thermo Fisher Scientific). Reverse transcription polymerase chain reaction (RT-PCR) was carried out on Mastercycler® nexus X2 (Eppendorf), using the following program: 35 cycles of 95°C for 15 s, 60°C for 60 s, and 72°C for 40 s. The 2−ΔΔCt method was used to quantify the data. U6 mRNA was employed as a reference to calculate the expression. 19 The primer sequences were as follows (Shanghai Biotechnology Service Co., Ltd, China): miR-146a forward, 5′-TGAGAACTGAATTCCATGGGT-3′ and reverse, 5′-TATGGCACTGGTAGAATTCACT-3′; and U6 forward, 5′-GACCTCTATGCCAACACAGT-3′ and reverse, 5′-AGTACTTGCGCTCAGGAGGA-3′.
Measurement of brain water content
Six rats were used from each group and were anesthetized by an injection of 3% pentobarbital sodium (50 mg/kg) and were then euthanized. The brain water content was assayed by dry/wet weight method. Brain tissues were obtained, and the meninges, lower brainstem, and cerebellum were removed. The remaining tissues were placed in a weighing bottle and weighted by an electronic balance and were then placed in electrothermal oven to a constant weight for 24 h. The brain water content was calculated as follows: Brain water content (%) = (wet weight − dry weight)/wet weight × 100%. The brain water content was applied to evaluate the degree of brain edema.
TUNEL staining
The left and right part of hippocampi of the remaining six rats were obtained and fixed in 4% paraformaldehyde for 24 h. The tissues were dehydrated in an ascending series of ethanol, cleared, and embedded in paraffin. Paraffin-embedded sections of the hippocampus were prepared with a thickness of 5 μm. Apoptosis was detected using the terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling assay (TUNEL) by apoptosis kit after the paraffin-embedded sections were conventionally dewaxed in water (ZK-8005; Beijing Zhongshan Jinqiao Biotechnology Co., Ltd, China). Five visual fields were randomly selected using a 400-fold optical microscope (type BX50; Olympus, Japan). Those with brown or yellow particles and morphological features of apoptotic cells were identified as apoptotic cells. The apoptotic index (AI) was used to evaluate the level of apoptosis. AI = (positive cells/total cells) × 100%.
Immunohistochemistry
Paraffin-embedded tissue samples were sliced, dried, dewaxed in xylene, and dehydrated in an ascending series of ethanol. For inactivation, 3% H2O2 methanol solution was added for 20 min, citrate buffer (pH6.0) was added to heat for antigen retrieval for 10 min, and 5% BSA were used for blocking for 20 min. Rabbit anti-rat LC3 (1:500, orb157781; Biorbyt) and Beclin 1 (1:500, orb378577; Biorbyt) were added, then incubated overnight at 4°C. After rewashing, the samples were incubated for 1 h with the secondary antibody, horseradish peroxidase-labeled goat against-rabit IgG (1:1000, ABIN101988; antibodies-online, Aachen, Germany), and then colored by DAB (DA1010; Solarbio, Beijing, China). The samples were then dried and mounted by entellan. Five fields for each section of animals were randomly selected and an optical microscope at 400× magnification (CX22, Olympus) was used to observe the cells. Aperio ImageScope software (version 11.1) was used to count the number of cells, and the results were expressed as the percentage of positive cells. Positive reactions were defined as those showing in brown signals in the cell cytoplasm. The frequency of positive cells was defined as the percentage of positive cells.
Observation of mitochondrial autophagy by transmission electron microscopy
The hippocampus was immobilized in 2.5% glutaraldehyde solution for 4 h, washed with PBS, then immobilized with 1% osmium acid for 1.5 h, washed with distilled water, dehydrated with a gradient series of ethanol and acetone, and embedded with epoxy resin, ultrathin sliced and electron stained. The morphological changes of the mitochondria were observed using a transmission electron microscope (Hitachi H-7500, Japan) and photographed.
Western blot analysis
The hippocampus was homogenized, centrifuged, and the supernatant was collected (1003 × g, 10 min). The BCA kit (PC0020; Solarbio) was used to detect the protein concentrations. The 1:1 ratio of 40-μg protein samples with 10% sodium dodecyl sulfate (SDS) gel buffer was mixed and denatured at 95°C for 5 min, then transferred to a polyvinylidene difluoride membrane under 25 V for 30 min (YA1701; Solarbio). The mixture was sealed with Tris buffer solution tween (TBST) solution with 5% nonfat milk powder at 4°C for 1 h. Rabbit anti-rat LC3A (1:1000, orb378164; Biorbyt), LC3B (1:1000, orb382715; Biorbyt), Beclin 1 (1:500, orb227780; Biorbyt), Bax (1:500, orb224426; Biorbyt), Bcl-2 (1:500, orb228150; Biorbyt), cleaved caspase 3 (1:500, orb126608; Biorbyt), P65 (1:500, orb229138; Biorbyt), p-P65 (1:500, orb304662; Biorbyt), IκBα (1:500, orb223182; Biorbyt), p-IκBα (1:500, orb223035; Biorbyt), and β-actin (1:2000, orb178392; Biorbyt) polyclonal antibodies were diluted in TBST solution with 3% bovine serum albumin overnight at 4°C. Horseradish peroxidase-labeled goat against rabbit IgG (1:1000, ABIN101988; antibodies-online) was used for incubation for 1 h after rewarming, and enhanced chemiluminescence was used for visualization for 3–5 min after washing. Protein expression was normalized to β-actin. Gray scale scanning and quantification of the protein bands were performed using Image J software (NIH).
To further investigate the effects of autophagy inhibitor (3-methyladenine (3-MA); Selleck, Shanghai, China) on miR-146-regulated apoptotic protein expression (Bcl-2, Bax, cleaved caspase 3), 3-MA (400 nmol/µl) was injected into ICH rats. 16 The groups included the sham group, ICH group, miR-146a agomir group, 3-MA group, and miR-146a agomir + 3-MA group. There were six rats in each group. The expression of Bcl-2, Bax, and cleaved caspase 3 was examined by Western blot analysis.
Statistical analysis
SPSS 19.0 software was used to analyze the data, and the results are expressed as the means ± standard deviation (SD). Analysis of variance was used to analyze data from multiple groups followed by the least significant difference test. The normality of data was analyzed using Levene’s test. A value of p < 0.05 was considered to indicate a statistically significant difference.
Results
miR-146a overexpression improves nerve function scores
As shown in Figure 1(a), no nerve function was observed in the sham group. The scores in the miR-146a agomir group were decreased compared with those of the ICH group (p < 0.05). As shown in Figure 1(b), the expression of miR-146a was lower in the operation group than in the sham group (p < 0.05). miR-146a expression was increased in the miR-146a agomir group in comparison to the ICH group (p < 0.05).

(a) Nerve function scores. (b) The expression of miR-146a was examined by RT-qPCR. *p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group. ICH: intracerebral hemorrhage.
miR-146a overexpression decreases brain water content
As shown in Figure 2, the brain water content in the ICH group was higher than that in the sham group (p < 0.05). The content in the miR-146a agomir group was decreased in contrast to the ICH group (p < 0.05).

Measurement of brain water content. *p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group. ICH: intracerebral hemorrhage.
miR-146a overexpression decreases apoptosis in the hippocampus
As shown in Figure 3, the percentage of apoptotic cells in the ICH group was increased compared with that in the sham group (p < 0.05), and that in the miR-146a agomir group was decreased in comparison with the ICH group (p < 0.05).

Detection of apoptosis of hippocampal neurons by TUNEL assay (×400 magnification): (a) TUNEL staining and (b) AI (%).*p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group. ICH: intracerebral hemorrhage.
Expression of LC3 and Beclin 1 measured by immunohistochemistry
The positive signals of LC3 proteins were located in the cytoplasm, exhibiting yellow to brown-yellow color, and the positive signals of Beclin 1 proteins were located in the cytoplasm, exhibiting light yellow or dark-brown color as shown in Figure 4. The LC3- and Beclin 1 protein-positive cells were scattered in the region of CA1 in hippocampal neurons in the sham group. In the ICH group, the LC3- and Beclin 1 protein-positive cells were more abundant than those in the sham group (p < 0.05). The expression was decreased in the miR-146a agomir group compared with the ICH group (p < 0.05).
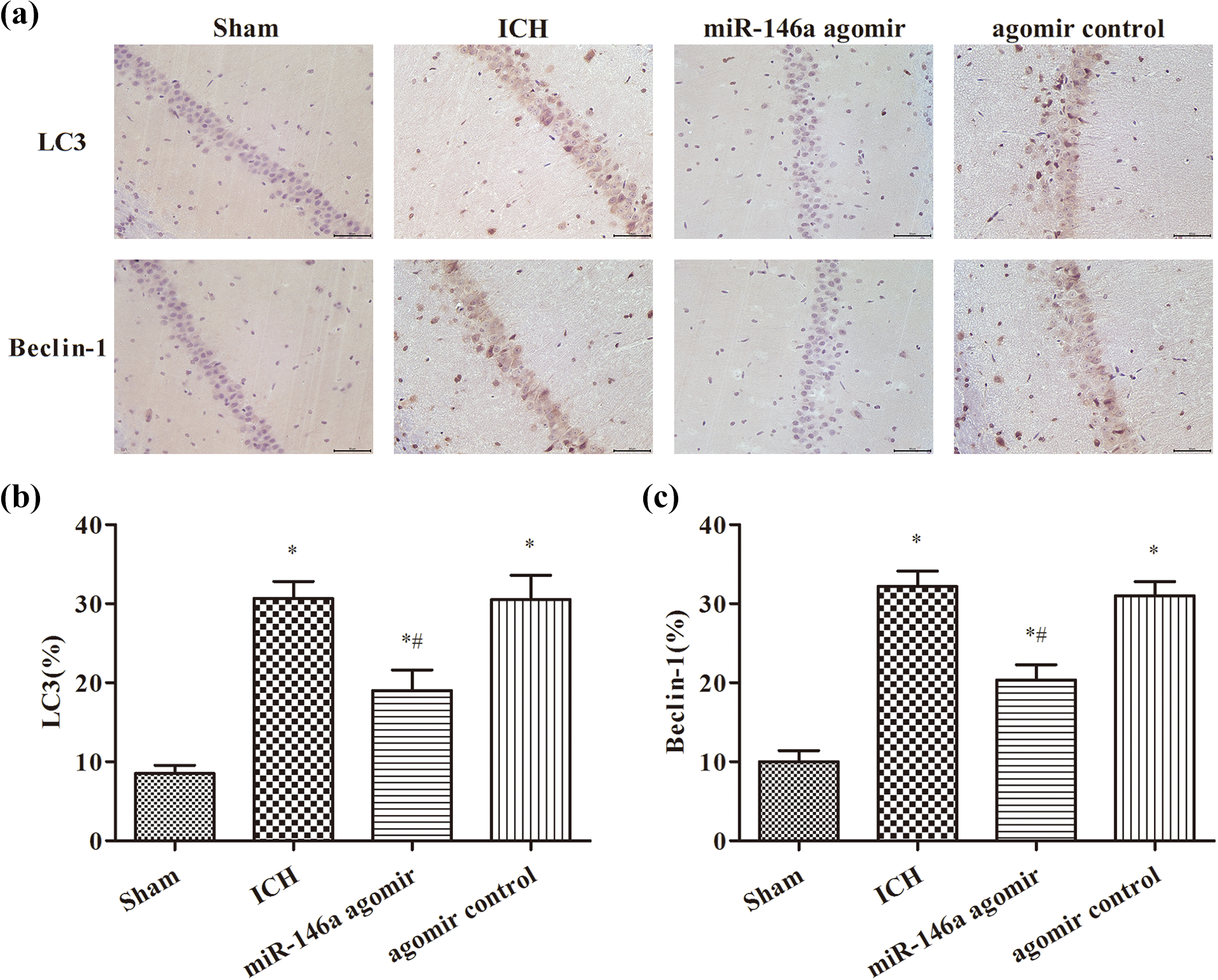
Measurement of the expression of LC3 and Beclin 1 by immunohistochemistry (×400 magnification): (a) immunohistochemical staining results, (b) LC3 (%), and (C) Beclin 1(%). *p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group. ICH: intracerebral hemorrhage.
Observation of mitochondrial autophagy by transmission electron microscopy
As shown in Figure 5, the size and distribution of the mitochondria were uniform, elliptic or round, and the membranes were clear without the occurrence of vacuolization in the sham group. The size and distribution of the mitochondria were altered and the mitochondria were swollen, the cristae were broken, matrix density was decreased, vacuolization had occurred, the mitochondrial structure was encapsulated by a bilayer, and multilayer membranes were clear in the ICH group. Mitochondrial autophagy was alleviated in the miR-146a agomir group.
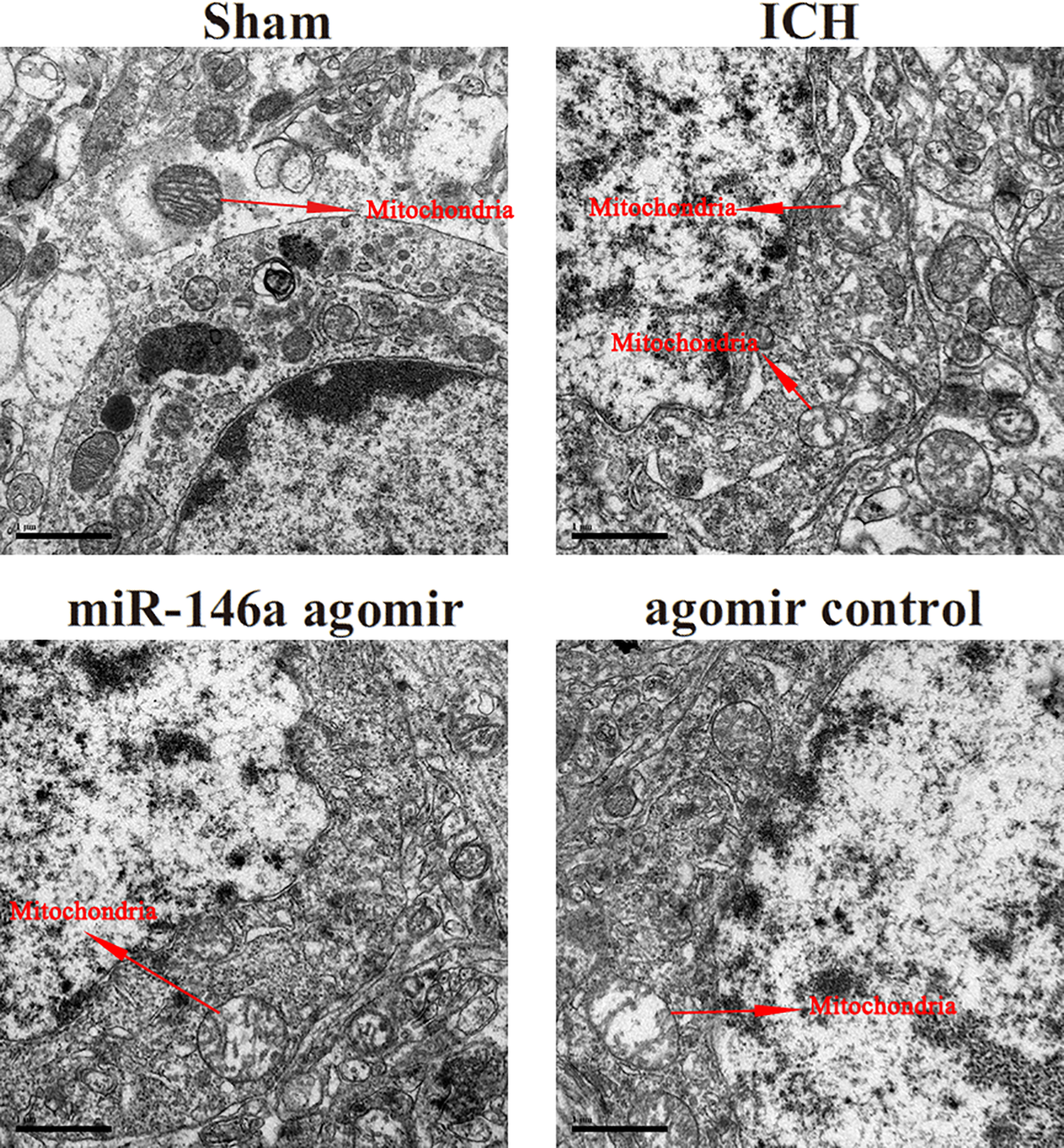
Observation of mitochondrial autophagy by transmission electron microscopy (×25,000 magnification): (a) sham, (b) ICH, (c) miR-146a agomir, and (d) agomir control. ICH: intracerebral hemorrhage.
miR-146a overexpression decreased the expression of apoptosis- and autophagy-related proteins
The expression of LC3A, LC3B, and Beclin 1 was higher in the ICH group than in the sham group (p < 0.05), as shown in Figure 6. The expression was decreased in the miR-146a agomir group compared with the ICH group (p < 0.05). The expression of Bax and cleaved caspase 3 was increased, and the expression of Bcl-2 in the ICH group was decreased compared with the sham group (p < 0.05). The expression of Bax and cleaved caspase 3 was decreased and the expression of Bcl-2 in the miR-146a agomir group was increased compared to the ICH group (p < 0.05).

Expression of apoptosis- and autophagy-related proteins detected by Western blot analysis: (a) Western blot analysis results, (b) LC3A, (c) LC3B, (d) Beclin 1, (e) Bax, (f) Bcl-2, and (g) cleaved caspase 3. *p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group. ICH: intracerebral hemorrhage.
miR-146a overexpression controls the activation of NF-κB signaling
The expression of p-P65 was increased and that of p-IκBα was decreased in the ICH group compared with the sham group, as shown in Figure 7 (p < 0.05). The expression of p-P65 was decreased and that of p-IκBα was increased in the miR-146a agomir group in contrast to the ICH group (p < 0.05).

Detection of the expression of NF-κB signaling pathway-related proteins by Western blot analysis: (a) Western blot analysis results, (b) p-P65/ P65, and (c) p-IκBα/IκBα. *p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group. ICH: intracerebral hemorrhage.
miR-146a overexpression inhibits apoptosis-related proteins via the regulation of autophagy
To further investigate the effects of autophagy inhibitor (3-MA) on miR-146-regulated apoptotic protein expression, 3-MA (400 nmol/µl) was injected into ICH rats and the results are shown in Figure 8. Compared with the sham group, the Bax, cleaved caspase 3 protein levels in the hippocampus of rats in the ICH group were markedly increased (p < 0.05). When compared to the ICH group, the expression of Bax and cleaved caspase 3 was significantly decreased, while the expression of Bcl-2 was markedly increased in the miR-146a and 3-MA groups (p < 0.05). The expression of Bax and cleaved caspase 3 was further decreased, while the expression of Bcl-2 was significantly increased by intervention with miR-146a agomir and 3-MA.
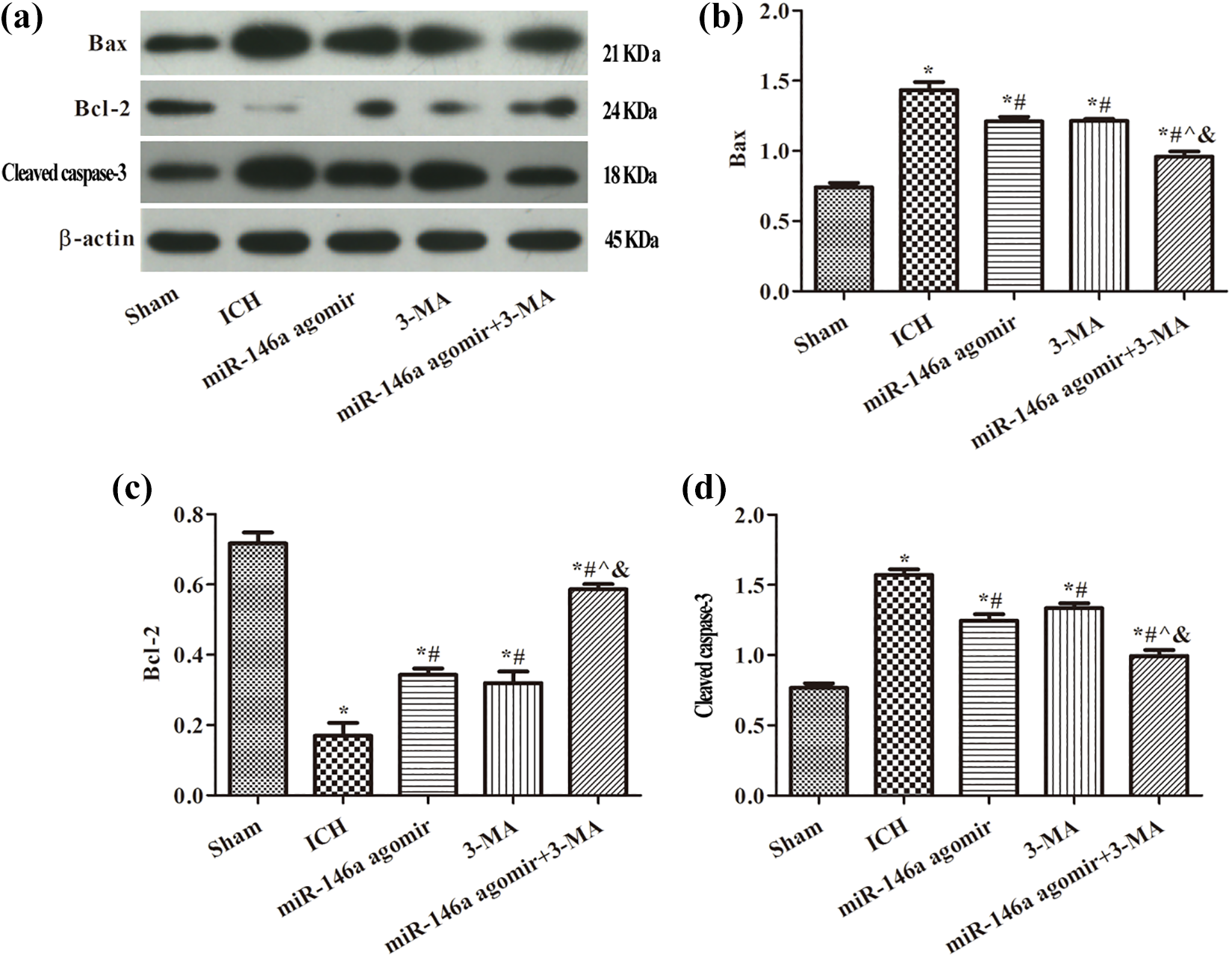
Expression of apoptosis-related proteins detected by Western blot analysis: (a) Western blot analysis results, (b) Bax, (c) Bcl-2, and (d) cleaved caspase 3. *p < 0.05, compared with the sham group; # p < 0.05, compared with the ICH group; ^ p < 0.05, compared with the miR-146a agomir group; & p < 0.05, compared with the 3-MA group. ICH: intracerebral hemorrhage.
Discussion
miR-146a is one of the most abundantly expressed miRNAs in the human central nervous system and has been proved to be expressed in a number of nervous system diseases. Studies have demonstrated that miR-146a is closely related to ischemic stroke, Alzheimer’s disease (AD), and nervous system autoimmune disease. 20 –22 Hu et al. found that the expression of miR-146a was decreased in the serum of patients with AD and increased in patients with mild cognitive impairment, indicating that miR-146a played a dynamic role in the occurrence and development of AD. 23 In this study, the expression of miR-146a was decreased in the ICH group; however, the nerve function scores were increased and the water content was decreased following miR-146a agomir treatment. This study revealed that the overexpression of miR-146a improved brain injury in rats with ICH, which was consistent with the findings of a previous study. 23
Secondary nerve injury following ICH is the main factor that affects the quality of life of patients with ICH. Therefore, the prevention of nerve injury is crucial in improving the prognosis and quality of life of patients with ICH. Neuronal apoptosis is an important factor leading to neuronal injury following ICH. Cerebral edema and the apoptosis of neuronal cells occur within 3 h after ICH. 24,25 Bcl-2 and Bax are major regulators of apoptosis, which play a crucial role in the regulation of apoptosis in brain injury diseases, including ICH 25 and hypoxic ischemic reperfusion (I/R). 26 As a pro-apoptotic member of the Bcl-2 family, Bax translocates from the cytoplasm to the mitochondrial outer membrane, which results in caspase 3 activation. 26 When the ratio of Bcl-2/Bax is decreased, apoptosis is induced, while the opposite occurs when it is increased. 24 Consistent with previous reports, the percentage of apoptotic cells was increased, and the Bcl-2/Bax ratio was reduced with the aggravation of brain injury in this study. Following treatment with miR-146a agomir, the number of apoptotic cells significantly decreased and the ratio of Bcl-2/Bax markedly increased in the brain tissue. These findings suggested that miR-146a protected nerve cells from apoptosis.
Excessive autophagy can lead to cell death by destroying the normal structure of cells. LC3 exists in the autophagosome membrane, which is closely related to the number of autophagosomes. 27 Beclin 1 is expressed in a number of domains, including the cortex, hippocampus, and cerebellum. The binding with Beclin 1 and BH-3Bcl-2 affects the activity of Beclin 1. 28 When normal intracellular homeostasis is destroyed by external pressure or injury, the decrease of Beclin 1 binding with Bcl-2 can lead to autophagy; the interaction of these proteins can control the transformation between autophagy and apoptosis. 29,30 In this study, it was found that the autophagosome was encapsulated by a bilayer or multilayer membrane structure in the ICH group, while autophagy was alleviated following miR-146a agomir treatment. The expression of LC3A, LC3B, and Beclin 1 was also increased in the miR-146a agomir group. To determine whether miR-146a takes part in apoptosis via the regulation of autophagy in rats with ICH, an autophagy inhibitor (3-MA) was used to observe the expression of Bcl-2/Bax and cleaved caspase 3. An obvious regulation of apoptosis was observed between the miR-146a agomir and miR-146a agomir + 3-MA groups. Through these data, it was concluded that miR-146a inhibited the apoptosis of neurons via the regulation autophagy in ICH.
The NF-κB pathway is associated with inflammation and immunity. The phosphorylation of IκBs can activate NF-κB signaling, which can lead to NF-κB entering the nucleus and participating in the regulation of the inflammatory response. 12 The activation of NF-κB can lead to apoptosis in ICH. 15,31 The activation of NF-κB after ICH was proved in this study. The overexpression of miR-146a inhibited the activation of the NF-κB signaling pathway. However, the mechanism of miR-146a promoting the improvement of brain injury is not clear. In addition, the mechanisms of miR-146a, autophagy, and inflammation need to be further studied to better understand the mechanism of ICH.
Conclusion
In conclusion, this study revealed that the overexpression of miR-146a attenuated brain injury in rats with ICH. Treatment with miR-146a agomir protected the nerve cells from apoptosis and autophagy following ICH. This study suggested that the overexpression of miR-146a inhibited the apoptosis of hippocampal neurons of rats with ICH by regulating autophagy.
Footnotes
Author contribution
SH and JJ equally contributed to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shandong Province, China (Nos. ZR2019MH124 and ZR2019PH119).
