Abstract
Psoriasis is a common immune-mediated and genetic skin disease. Forkhead box M1 (FOXM1) is a member of FOX family that has been found to modulate skin disorders. However, its role in psoriasis remains unknown. Thus, we aimed to investigate the effect of FOXM1 on keratinocytes in response to tumor necrosis factor-α (TNF-α). The expression levels of FOXM1 in psoriasis tissues and normal skin tissues were examined using qRT-PCR and western blot. HaCaT cells were stimulated by TNF-α to mimic psoriasis in vitro. MTT assay was performed to assess cell proliferation. The caspase-3 activity and expression levels of bcl-2 and bax were determined to indicate cell apoptosis. The mRNA and secretion levels of IL-6, IL-23 and TGF-β were determined by qRT-PCR and ELISA, respectively. The NF-κB activation was assessed using western blot analysis. Our results demonstrated that FOXM1 was highly upregulated in psoriatic skin tissues and TNF-α-stimulated HaCaT cells. Knockdown of FOXM1 repressed cell proliferation of TNF-α-stimulated HaCaT cells. Knockdown of FOXM1 caused significant increases in caspase-3 activity, bax expression and decrease in bcl-2 expression in TNF-α-stimulated HaCaT cells. Moreover, FOXM1 knockdown also suppressed the TNF-α-induced production of IL-6, IL-23, and TGF-β in HaCaT cells. However, FOXM1 overexpression showed the opposite effect. Furthermore, the TNF-α-induced NF-κB activation was prevented by FOXM1 knockdown. Additionally, inhibition of NF-κB reversed the effects of FOXM1 on HaCaT cells. Taken together, these findings indicated that FOXM1 regulated cell proliferation, apoptosis and inflammation in TNF-α-induced HaCaT cells. The effects of FOXM1 were mediated by NF-κB pathway.
Introduction
Psoriasis is a common immune-mediated and genetic disease manifesting in the skin and/or joints with great physical and psychological burden. 1 Individuals with psoriasis are at an increased risk of developing other chronic and serious health diseases, such as psoriatic arthritis, cardiovascular disorders, anxiety, depression, non-alcoholic fatty liver disease, and lymphoma. 2 Therefore, psoriasis prevalence is an important consideration for WHO. In recent years, several high-quality evidence-based guidelines including topical therapies, phototherapy, and conventional and biological systemic therapies have been developed for the treatment of psoriasis. 3 However, patients with psoriasis also confront many challenges including high prevalence, chronicity, disfiguration, disability, and associated comorbidity. 4 Understanding the pathogenesis of psoriasis may help to manage this complex disease.
Forkhead box (Fox) proteins are evolutionarily conserved transcription factors that regulate diverse biological processes both during development and throughout adult life. 5 In humans, mutations or abnormal regulation of Fox genes are associated with various diseases such as cancers, metabolism diseases, and defects in immune regulation and function. 6 FOXM1, a member of Fox family, is a key target of both well-known and new classes of proteasome inhibitors. Recently, FOXM1 has been found to be involved in various diseases, including cancers, 7 pulmonary disease, 8 pulmonary arterial hypertension (PAH), 9 cardiac fibrosis, 10 asthma, 11 and osteoarthritis. 12 Moreover, FOXM1 was found to modulate skin disorders. FOXM1 expression is strongly reduced in keratinocytes isolated from old human subjects compared with young subjects. FOXM1 regulates proliferation, senescence and oxidative stress in keratinocytes. 13 UVB dose-dependently increases FOXM1 protein levels in human epidermal keratinocytes and thereby induces genomic instability, implying that FOXM1 may be an early molecular signal required for skin cancer initiation. 14 However, its role in psoriasis remains unknown.
Thus, in this study, we investigated the effect of FOXM1 on keratinocyte cell line HaCaT cells in response to tumor necrosis factor-α (TNF-α) stimulation. We found that FOXM1 had the capacity to regulate the cell proliferation, apoptosis and inflammation of TNF-α-induced HaCaT keratinocyte cells. Moreover, the effects were mediated by the NF-κB signaling pathway.
Materials and methods
Clinical samples collection
A total of 24 psoriasis tissues (15 male and 9 female, 17–52 years) and 17 normal skin tissues (10 male and 7 female, 15–41 years) were obtained from Dermatology Department, Affiliated Hospital of Shaoxing University, Shaoxing Municipal Hospital (Shaoxing). The tissue samples were snap frozen in liquid nitrogen and then stored at −80°C for the analysis of FOXM1 expression. Written informed consents were obtained from all participants. The study was approved by the Research Ethics Board of Affiliated Hospital of Shaoxing University, Shaoxing Municipal Hospital and performed in accordance with the declaration of Helsinki Principles.
Cell culture and treatment
Human keratinocyte cell line HaCaT cells (Procell Life Science and Technology Co. Ltd, Wuhan, China) were cultured in RPMI 1640 media supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin. The HaCaT cells were maintained at 37°C with 5% CO2. For the TNF-α-treated group, the HaCaT cells were stimulated with 10 ng/ml TNF-α (Sigma, St. Louis, MO, USA). For the PDTC-treated group, HaCaT cells were treated with 10 μμ PDTC (Sigma-Aldrich).
Cell transfection
To inhibit FOXM1, specific siRNA targeting FOXM1 (si-FOXM1; GenePharma, Shanghai, China) was transfected into HaCaT cells. The nonsilencing-siRNA (si-NC; GenePharma) was used as the negative control. The si-FOXM1 or si-NC was transiently transfected into HaCaT cells using Lipofectamine RNAiMAX (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. To overexpress FOXM1, the cDNA sequences encoding FOXM1 were sub-cloned into a pcDNA3.1 vector to generate a FOXM1 expression vector. The constructed plasmids were transfected into HaCaT cells using the Lipofectamine 3000 Transfection Reagent (Invitrogen) following the manufacturer’s manuals.
Cell proliferation assay
To measure cell proliferation, 4000 cells per well were plated in 96-well plate, and then incubated for 48 h at 37°C. At the indicated time points, 10 µl MTT solution (5 mg/ml) was added to each well and incubated for 4 h at 37°C. Then 150 µl dimethlsulfoxide was added to each well to dissolve the formazan crystals. Optical density (OD) value was measured at 490 nm using microplate reader (Thermo Fisher Scientific, Waltham, MA, USA).
Measurement of caspase-3 activity
The caspase-3 activity in HaCaT cells was measured by a caspase-3 fluorescent assay kit (NanJing Jiancheng Bioengineering Institute, NanJing, China) according to the manufacturer’s instructions. Briefly, whole lysates of HaCaT cells prepared by RIPA lysis buffer (NanJing Jiancheng Bioengineering Institute) were incubated with the caspase-3 substrate Ac-DEVD-pNA for 4 h at 37°C, followed by the measurement using a fluorescent spectrophotometer (Multiskan MK3, Thermo) with excitation at 380 nm and emission at 405 nm.
qRT-PCR assay
Total RNA was extracted from tissue samples and HaCaT cells using the High Pure RNA Isolation Kit (Roche, Penzberg, Germany) according to the manufacturer’s instructions. The quality of RNA sample was assessed using the RNA 6000 Nano Assay on the Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA). The cDNA was synthesized from an RNA template using Transcriptor First-Strand cDNA Synthesis Kit (Roche). QRT-PCR was performed on a RT-PCR system (Applied Biosystem, Foster, CA, USA) using SYBR Green Premix Ex Taq (TaKaRa, Dalian, China). Primers were synthesized by Sangon Biotech (Shanghai, China) and the following primers were used as follows: FOXM1, forward 5’-GGA GGA AAT GCC ACA CTT AGC G-3’, and reverse 5’-TAG GAC TTC TTG GGT CTT GGG GTG-3’, its product size is 131 bp; IL-6, forward 5’-CCT CCA GGA ACC CAG GTA TGA A-3’, and reverse 5’-TCA GGT GCC CCA GCT ACA TTA T-3’, its product size is 114 bp; IL-23, forward 5’-CGT ACT CCT GCT TGC TGA-3’, and reverse 5’-ATG CTA CCG AAT TTC GAC-3’, its product size is 125 bp; TGF-β, forward 5’-TAC TGG ACT CGA CCG AAT-3’, and reverse 5’-ACA GAA GGA GTG GCT AAG-3’, its product size is 107 bp; β-actin, forward 5’-TGT TGC CAT CAA TGA CCC CTT-3’, and reverse 5’-CTC CAC GAC GTA CTC AGC G-3’, its product size is 136 bp. β-actin was used as internal referee and relative gene expression was quantified by 2−ΔΔCt method. The PCR conditions comprised 35 cycles of denaturation at 94°C for 30 s, annealing at 54°C for 30 s, and polymerization at 72°C for 30 s.
Western blot analysis
Cells or tissues were homogenized and lysed with RIPA lysis buffer (100 mM NaCl, 50 mM Tris-HCl pH 7.5, 1% Triton X-100, 1 mM EDTA, 10 mM beta-glycerophosphate, 2 mM sodium vanadate and protease inhibitor). Protein concentration was measured using a BCA protein assay kit. The prepared lysates (40 μg) were separated by 12% SDS-PAGE and then the proteins were transferred to polyvinylidene difluoride (PVDF) membrane (Bio-Rad Laboratories, Hercules, CA, USA). The membranes were then blocked with TBST solution containing 5% skim milk for 1 h at room temperature to block the unoccupied binding sites, followed by an incubation with the primary antibodies against FOXM1, bax, bcl-2, p-p65, p65, IκBα, and β-actin (1:500; Abcam) at 4°C overnight. The membranes were then incubated in horseradish peroxidase (HRP)-conjugated goat anti-rabbit secondary antibody (1:3000; Abcam) at room temperature for 1 h. Finally, an ECL kit (Pierce Chemical, Rockford, IL, USA) was used to perform chemiluminescent detection.
Enzyme-linked immunosorbent assay (ELISA)
Cultured HaCaT cells were centrifuged for 10 min to prepare the culture supernatant. The contents of IL-6, IL-23, and TGF-β were detected by commercial ELISA Kits (Abcam, Cambridge, MA, USA) following the instructions. The absorbance at 450 nm was measured by a microplate reader (Thermo).
Statistical analysis
SPSS standard version 21.0 software (SPSS Inc., Chicago, IL) was used for the statistical analysis in the current study. Data were analyzed by unpaired Student’s t-test or one-way ANOVA followed by Tukey post hoc test and presented as mean ± SEM. P < 0.05 was considered statistically significant.
Results
FOXM1 was highly upregulated in psoriasis tissues and TNF-α-stimulated HaCaT keratinocyte cells
In the present study, we first investigated the expression of FOXM1 in 24 psoriasis tissues and 17 normal skin tissues using qRT-PCR and western blot. As shown in Figure 1(a) and (b), the FOXM1 expressions at both mRNA and protein levels were increased significantly in the psoriasis tissues as compared to that in the normal skin tissues. Moreover, the FOXM1 expression was also detected in HaCaT cells with or without TNF-α stimulation. We found that FOXM1 expression was significantly upregulated in HaCaT cells exposed to TNF-α stimulation (P = 0.000; Figure 1(c) and (d)).

FOXM1 expression in psoriasis tissues and TNF-α-stimulated HaCaT keratinocyte cells. (a) The mRNA expression levels of FOXM1 in psoriasis tissues and normal skin tissues were examined using qRT-PCR. *p < 0.05 relative to the normal skin tissues. (b) The protein expression levels of FOXM1 in psoriasis tissues and normal skin tissues were examined using western blot. N represents normal skin tissues, P represents psoriasis tissues. (c) The mRNA expression level of FOXM1 expression was detected using qRT-PCR in HaCaT cells with or without TNF-α (10 ng/ml) stimulation. (d) FOXM1 protein expression levels were detected using western blot in HaCaT cells with or without TNF-α (10 ng/ml) stimulation. #p < 0.05 relative to the control group.
Knockdown of FOXM1 repressed cell proliferation and promoted apoptosis in TNF-α-stimulated HaCaT keratinocyte cells
To confirm the role of FOXM1 in TNF-α-stimulated HaCaT cells, knockdown of FOXM1 was performed by transfecting HaCaT cells with si-FOXM1. After transfection with si-FOXM1, the protein expression level of FOXM1 was dramatically decreased in TNF-α-stimulated HaCaT cells (F = 63.604; P = 0.000; Figure 2(a) and (b)). Further experiment showed that knockdown of FOXM1 suppressed the TNF-α-induced cell proliferation of HaCaT cells (F = 34.209; P = 0.000; Figure 2(c)). Moreover, western blot results demonstrated that TNF-α treatment greatly decreased bax expression (F = 80.963; P = 0.000 and increased bcl-2 expression (F = 55.019; P = 0.000) in HaCaT cells, and these effects were reversed by FOXM1 knockdown (Figure 2(d) and (e)). Furthermore, the caspase-3 activity was inhibited in TNF-α-stimulated HaCaT cells, which was increased in si-FOXM1 transfected HaCaT cells (F = 81.708; P = 0.000; Figure 2(f)).

Knockdown of FOXM1 repressed cell proliferation and promoted apoptosis in TNF-α-stimulated HaCaT keratinocyte cells. (a) HaCaT cells were transfected with si-FOXM1 or si-NC in TNF-α-stimulated HaCaT cells. After 48 h post transfection, the protein levels of FOXM1 were detected by western blot. (b) Quantification analysis of FOXM1. (c) MTT assay was performed to assess cell proliferation. (d) Western blot was carried out to detect the expression levels of bax and bcl-2. (e) Quantification analysis of bax and bcl-2. (f) The caspase-3 activity was determined to indicate cell apoptosis. *p < 0.05.
Knockdown of FOXM1 inhibited TNF-α-induced inflammation in HaCaT keratinocyte cells
IL-6, IL-23, and TGF-β are three important inflammatory cytokines produced by keratinocytes, which are important for the immune regulation in the pathogenesis of psoriasis. We found that the mRNA levels of IL-6, IL-23, and TGF-β were induced by TNF-α in HaCaT cells, whereas the increased IL-6 (F = 73.130; P = 0.000), IL-23 (F = 92.544; P = 0.000), and TGF-β (F = 78.268; P = 0.000) mRNA levels were decreased si-FOXM1 transfected HaCaT cells (Figure 3(a)–(c)). Besides, ELISA results proved that the TNF-α-stimulated secretion of IL-6 (F = 26.422; P = 0.000), IL-23 (F = 47.339; P = 0.000), and TGF-β (F = 47.218; P = 0.000) were prevented by knockdown of FOXM1 (Figure 3(d)–(f)).

Knockdown of FOXM1 inhibited TNF-α-induced inflammation in HaCaT keratinocyte cells. After 48 h post transfection, the mRNA and secretion levels of IL-6, IL-23 and TGF-β were determined by qRT-PCR (a–c) and ELISA (d–f), respectively. *p < 0.05.
Overexpression of FOXM1 promoted cell proliferation and inhibited apoptosis in TNF-α-stimulated HaCaT keratinocyte cells
Subsequently, pcDNA3.1-FOXM1 was transfected into HaCaT cells to elevate the expression of FOXM1. As indicated in Figure 4(a) and (b), the protein expression level of FOXM1 was markedly increased by pcDNA3.1-FOXM1 in TNF-α-stimulated HaCaT cells (F = 75.203; P = 0.000). MTT assay revealed that overexpression of FOXM1 promoted the TNF-α-stimulated cell proliferation of HaCaT cells (F = 52.322; P = 0.000; Figure 4(c)). In addition, we found that the increased bcl-2 expression (F = 54.033; P = 0.000), as well as the decreased bax expression (F = 165.918; P = 0.000) and caspase-3 activity (F = 150.035; P = 0.000) in TNF-α-stimulated HaCaT cells were enhanced by FOXM1 overexpression (Figure 4(d)–(f)).

Overexpression of FOXM1 promoted cell proliferation and inhibited apoptosis in TNF-α-stimulated HaCaT keratinocyte cells. (a) HaCaT cells were transfected with pcDNA3.1-FOXM1 or pcDNA3.1 in TNF-α-stimulated HaCaT cells, followed by the detection of FOXM1 expression using western blot after 48 h post transfection. (b) Quantification analysis of FOXM1. (c) MTT assay was performed to assess cell proliferation. (d) Western blot was carried out to detect the expression levels of bax and bcl-2. (e) Quantification analysis of bax and bcl-2. (f) The caspase-3 activity was determined to indicate cell apoptosis. *p < 0.05.
Overexpression of FOXM1 promoted TNF-α-induced inflammation in HaCaT keratinocyte cells
Next, we examined the effect of FOXM1 overexpression on inflammatory response in TNF-α-stimulated HaCaT cells. The results indicated that TNF-α-induced increase in the mRNA levels of IL-6 (F = 63.922; P = 0.000), IL-23 (F = 97.615; P = 0.000) and TGF-β (F = 81.696; P = 0.000) were promoted by overexpression of FOXM1 (Figure 5(a)–(c)). Besides, FOXM1 overexpression also greatly increased the production of IL-6 (F = 43.896; P = 0.000), IL-23 (F = 89.365; P = 0.000) and TGF-β (F = 74.154; P = 0.000) in TNF-α-induced HaCaT cells (Figure 5(d)–(f)).

Overexpression of FOXM1 promoted TNF-α-induced inflammation in HaCaT keratinocyte cells. After transfection with pcDNA3.1-FOXM1 or pcDNA3.1, HaCaT cells were treated with or without TNF-α stimulation. The mRNA and secretion levels of IL-6, IL-23 and TGF-β were determined by qRT-PCR (a-c) and ELISA (d-f), respectively. *p < 0.05 relative to the control group; #p < 0.05 relative to the TNF-α + pcDNA3.1 group.
Knockdown of FOXM1 inhibited TNF-α-induced NF-κB activation in HaCaT keratinocyte cells
NF-κB activation has been implicated in the inflammatory response. Then we evaluated the effect of FOXM1 knockdown on NF-κB activation in HaCaT cells. Western blot analysis proved that TNF-α induction significantly increased the level of p-p65 and reduced the expression of IκBα. However, FOXM1 knockdown attenuated the changes in the expression levels of IκBα (F = 63.792; P = 0.000) and p-p65 (F = 102.058; P = 0.000; Figure 6(a)–(c)), implying that FOXM1 knockdown prevented TNF-α-induced NF-κB activation.
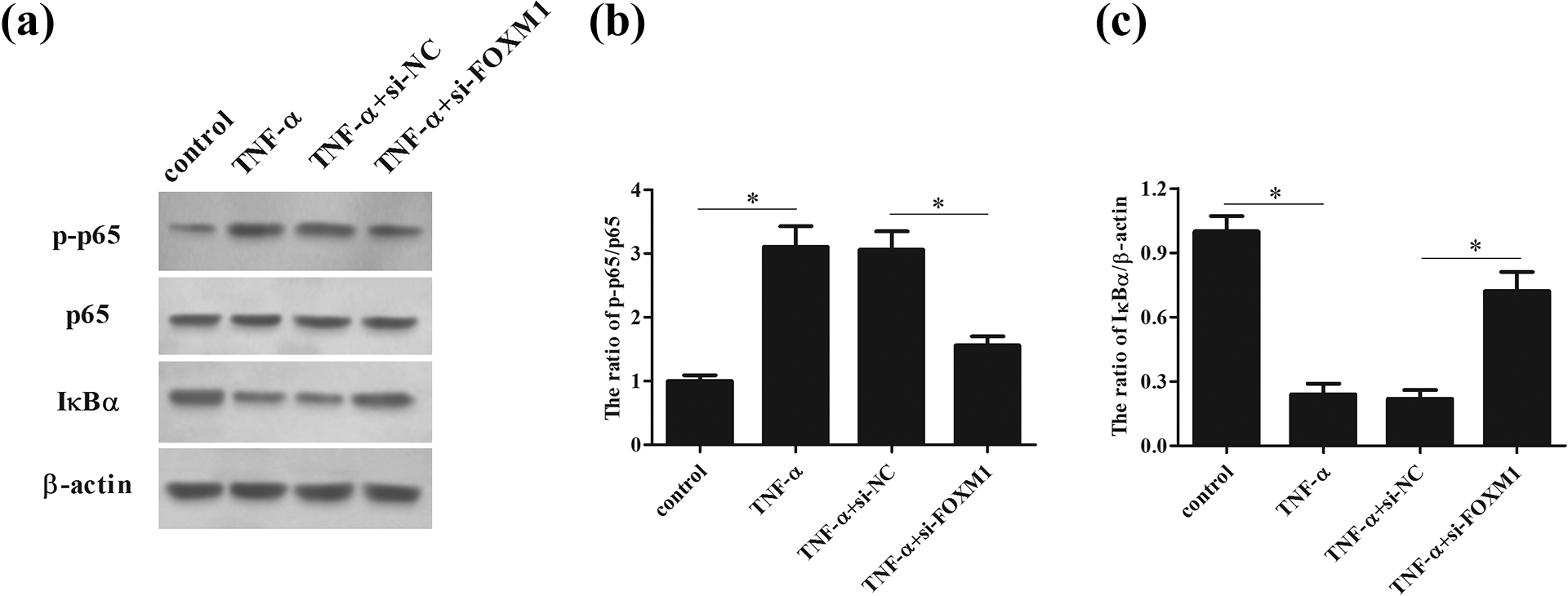
Knockdown of FOXM1 inhibited TNF-α-induced NF-κB activation in HaCaT keratinocyte cells. After 48 h post transfection with si-FOXM1 or si-NC, HaCaT cells were treated with or without TNF-α. (a) The NF-κB activation was evaluated by detecting the expression levels of p-p65, p65 and IκBα using western blot. (b) The ratio of p-p65/p65. (c) Quantification analysis of IκBα. *p < 0.05.
Overexpression of FOXM1 promoted TNF-α-induced NF-κB activation in HaCaT keratinocyte cells
In addition, the effect of FOXM1 overexpression on the NF-κB activation in TNF-α-induced HaCaT cells was also assessed by western blot analysis. As shown in Figure 7(a)–(c), FOXM1 overexpression markedly upregulated the level of p-p65 (F = 67.988; P = 0.000) and down-regulated IκBα expression (F = 145.992; P = 0.000) in TNF-α-stimulated HaCaT cells, which suggested that FOXM1 overexpression enhanced the TNF-α-induced NF-κB activation.
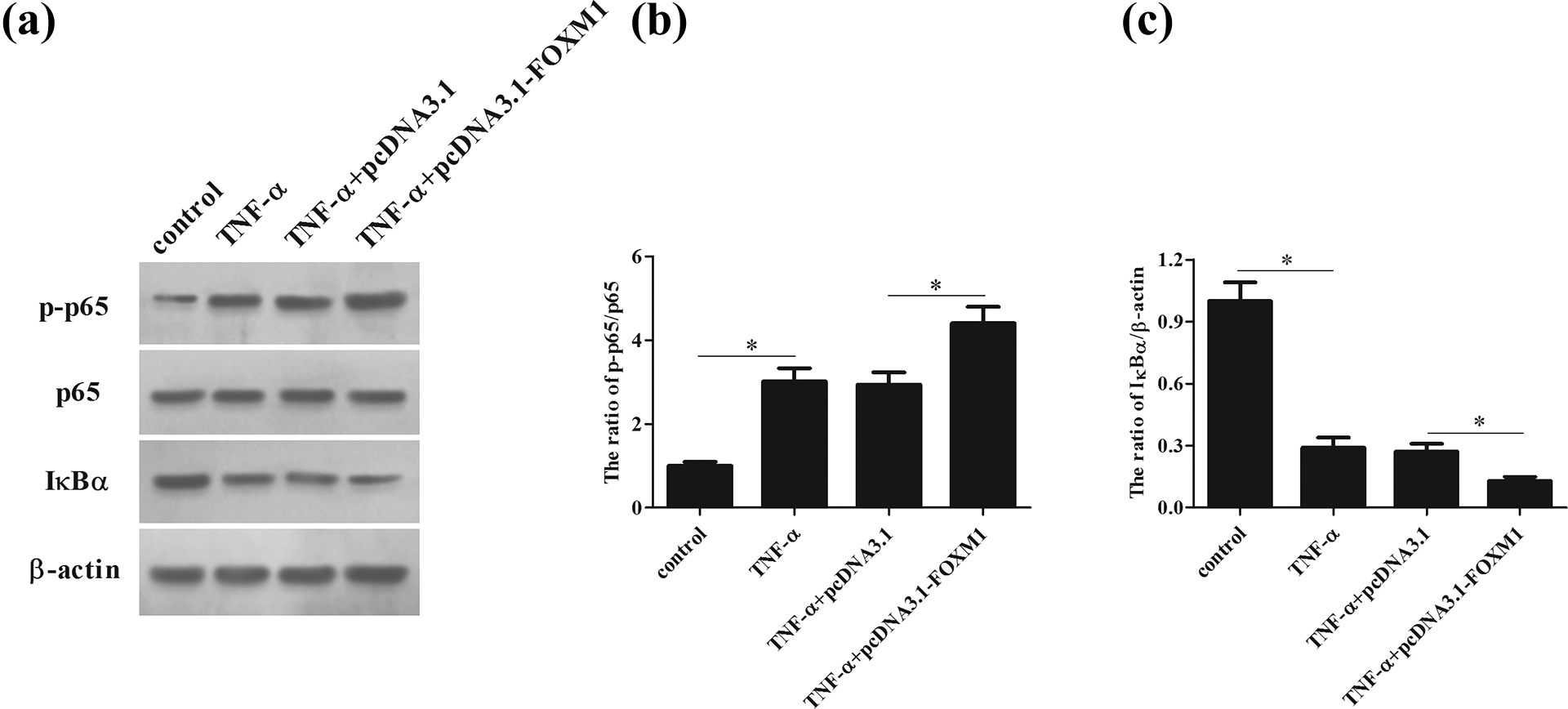
Overexpression of FOXM1 promoted TNF-α-induced NF-κB activation in HaCaT keratinocyte cells. HaCaT cells were treated with or without TNF-α after 48 h post transfection with pcDNA3.1-FOXM1 or pcDNA3.1. (a) The expression levels of p-p65, p65 and IκBα were evaluated by western blot. (b) The ratio of p-p65/p65. (c) Quantification analysis of IκBα. *p < 0.05.
PDTC reversed the effects of FOXM1 on cell proliferation, apoptosis and inflammation in HaCaT keratinocyte cells
To confirm that NF-κB pathway contributes to the regulation of the FOXM1-mediated effect in HaCaT cells, we detected the effect of PDTC on the FOXM1-mediated effect in TNF-α-stimulated HaCaT cells. We found that PDTC treatment reversed the induction effect of FOXM1 on cell proliferation of TNF-α-induced HaCaT cells (F = 43.084; P = 0.000; Figure 8(a)). The inhibitory effect of FOXM1 on cell apoptosis in TNF-α-induced HaCaT cells was also reversed by PDTC treatment, as evidenced by increased caspase-3 activity (F = 147.404; P = 0.000; Figure 8(b)). Additionally, the FOXM1 promoted the productions of IL-6 (F = 37.756; P = 0.000), IL-23 (F = 68.272; P = 0.000) and TGF-β (F = 87.707; P = 0.000) in TNF-α-stimulated HaCaT cells were also prevented by PDTC (Figure 8(c)–(e)).

PDTC reversed the effects of FOXM1 on TNF-α-induced HaCaT keratinocyte cells. After 48 h post transfection with pcDNA3.1-FOXM1 or pcDNA3.1, HaCaT cells were treated with 10 μμ PDTC and/or TNF-α. (a) MTT assay was performed to assess cell proliferation. (b) The caspase-3 activity was determined to indicate cell apoptosis. (c–e) The secretion levels of IL-6, IL-23 and TGF-β were determined by ELISA. *p < 0.05 relative to the control group, #p < 0.05 relative to the TNF-α group, &p < 0.05 relative to the TNF-α + pcDNA3.1-FOXM1 group.
Discussion
Psoriasis is a common, chronic and relapsing autoimmune and inflammatory skin disease, which is histologically characterized by increased epidermal thickness, elongated papillae, and a moderate inflammatory infiltrate composed of lymphocytes, macrophages and neutrophils. 15,16 Although the pathogenesis of psoriasis is still an enigma, keratinocytes had been long considered to be related to the pathogenesis of psoriasis. 17 Dysfunctionally increased proliferation, and altered differentiation and apoptosis of the keratinocytes lead to epidermal hyperplasia in psoriasis. 18 In the current study, we investigated the effect of FOXM1 on the proliferation and apoptosis of HaCaT keratinocyte cells. Our results proved that knockdown of FOXM1 repressed cell proliferation and promoted apoptosis, while FOXM1 overexpression promoted cell proliferation and inhibited apoptosis in TNF-α-stimulated HaCaT keratinocyte cells. These results indicated that FOXM1 regulated the altered proliferation and apoptosis of keratinocytes.
There are some hints that the immune system participates in the etiology and pathogenesis of psoriasis. 19,20 Many researchers have reported that cells existed in psoriatic lesions consist of activated T cells, macrophages, monocytes and polymorphonuclear cells. In addition to these cells, many kinds of cytokines have been identified in tissue extracts of patients with psoriasis, including TNF-α, IFN-γ, IL-1α, IL-1β, IL-6, IL-8, VEGF, and TGF-α 21 In recent years, it is evidenced that psoriasis is a Th17-type helper T cells (Th17 cells) mediated autoimmune disease. 22 Th17 cells are the third subset of the effecter T helper cells, which have an important role in host defense and are potent inducers of autoimmunity and tissue inflammation. 23 IL-6 together with TGF-β, produced by keratinocytes, elicit the differentiation of naive CD4+ T cells into Th17 cells. 24 –26 Moreover, IL-23, which is produced by dendritic cells and keratinocytes in psoriatic lesions, also promotes activation of Th17 cells. 27 The activation of Th17 cells produce IL-17A and IL-22, which are reported to have greatest expression in lesional psoriatic skin. 28,29 Therefore, we evaluated the effect of FOXM1 on the production of IL-6, IL-23 and TGF-β in keratinocytes. We found that knockdown of FOXM1 inhibited TNF-α-induced production of IL-6, IL-23 and TGF-β in HaCaT keratinocyte cells, which might contribute to the prevention of Th17 cells activation.
NF-κB is a transcription factor that has been considered to impact on many cellular phenomena, such as inflammation, immune responses, proliferation, apoptosis, tumor progression, and differentiation. 30,31 Increasing evidence has confirmed the participation of NF-κB signaling in the pathogenesis of psoriasis. 32 NF-κB has been hypothesized to connect the altered keratinocytes and immune cells behaviors through its effects on cellular proliferation, differentiation and apoptosis, as well as cytokine and chemokine production. 33 Compounds that specifically target NF-κB signaling may be developed as novel therapeutics for psoriasis. Previous studies have demonstrated that FOXM1 has the ability to regulate NF-κB signaling. FOXM1 has a crucial role in the development and progression of human gastric cancer through regulation of NF-κB signaling pathway. 34 FOXM1 silencing attenuates inflammatory response in human osteoarthritis chondrocytes via NF-κB signaling pathway. 12 Our results proved that NF-κB mediated the effects of FOXM1 on cell proliferation, apoptosis and inflammation in HaCaT keratinocyte cells.
In summary, this study demonstrated that FOXM1 expression was upregulated in psoriasis tissues. FOXM1 regulated the cell proliferation, apoptosis and inflammation in TNF-α-induced HaCaT keratinocyte cells, which were mediated by NF-κB signaling pathway. Our results provide new insight of FOXM1 and may contribute to the development of target for psoriasis treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
