Abstract
In the present investigation, the antitumor effect of ferruginol (FGL) in SK-Mel-28 human malignant melanoma cells was studied. To investigate the cytotoxic property of FGL, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay was used. Results revealed that prolonged treatment duration decreases the IC25, IC50, and IC75 concentrations of FGL. The cytotoxicity was further confirmed by lactate dehydrogenase assay. As evident from comet assay, FGL induces DNA damage in a dose-dependent manner. Annexin V and 7-ADD assays showed that FGL-induced DNA damage triggers apoptosis-mediated cell death as confirmed by caspase-3 activity assay. As seen through Western blotting, FGL increases phosphorylation of p38 and nuclear translocation of NF-κB. Further, it was observed that p38 phosphorylation is responsible for NF-κB translocation to the nucleus. Further, inhibition of p38 phosphorylation and translocation of NF-κB decrease caspase-3 activity. The above finding confirms that caspase-3 activation is mediated through P-p38 and nuclear translocation of NF-κB. The present findings indicate that FGL significantly suppresses the proliferation of SK-Mel-28 cells in a dose- and time-dependent manner through induction of apoptosis. Furthermore, FGL executes apoptosis through phosphorylation of key protein such as p38 and translocation of NF-κB into the nucleus.
Introduction
Malignant melanoma causes approximately 75% of the skin cancer-related deaths worldwide. Its frequency has augmented significantly in the recent times, owing to increased ultraviolet (UV) exposure and cosmetic usage. 1 Traditional chemotherapy drugs offer only very low accomplishment rates with an average survival rate of 8 ± 2 months for patients with stage IV melanoma. 2,3 Genetic studies have revealed that 50% of fast progressing melanomas contain a mutation in the gene that encodes B-Raf, which leads to constitutive activation of downstream signaling in the mitogen-activated protein kinase pathway. 4 The B-Raf V600E mutation is a hallmark for high-risk melanoma, associated with shortened patient survival rates and tumor drug resistance, 5,6 and B-Raf has emerged as validated target for melanoma intervention. B-Raf inhibitors like vemurafenib and dabrafenib show immense short time tumor repression. However, the tumor rapidly acquires chemoresistance to these drugs and disease recurrence within several months is commonly observed. Similarly, for patients with metastatic melanoma and who are not suitable for surgical resection, 5-(3,3-dimethyl-1-triazenyl)-1H-imidazole-4-carboxamide (DTIC) has been reported as a very potential chemotherapeutic agent. 7 However, in single-dose regimen, its overall response rate is very poor and the period of response is very low. Further, it has been reported that combination of DTIC with other chemotherapeutic agents like cisplatin, carmustine, and tamoxifen has led to greater response rates. 7,8 But unfortunately, this too has resulted in drastic adverse effect, affecting the quality of life. 8 Currently available immune treatment for melanoma includes the drugs that target immune checkpoint inhibitors (ICIs) such as cytotoxic T-lymphocyte-associated protein 4 and programmed cell death protein PD-1, and its ligand PD-L1. 9 Furthermore, these ICIs have shown noteworthy activity against a variety of tumors. On the other hand, it has been shown that a subpopulation of patients do not respond to ICIs or eventually develop resistant disease. 10 These limited treatment options indicate a need for novel and more effective anti-melanoma drugs with specific molecular targets.
Scientists are exploring the traditional and seemingly safe natural medicines for the anticancer therapeutics. Plant-based drugs including taxol, vincristine, vinblastine, topotecan, irinotecan, camptothecin, and podophyllotoxin have been successfully used for treatment in a variety of human tumors. 11 –13 These compounds have been used as prototypes for the design and development of future anticancer drugs. 14 As many of these drugs are associated with high adverse effects, there is an urge to validate drugs with minimal toxicity and higher efficacy. Consistent with this, diterpenoids have been shown to possess effective antiproliferative effect. Ferruginol (FGL), a bioactive principal isolated from the Chilean tree Persea nubigena and from the stem bark of Podocarpus andina (Podocarpaceae). It is an abietane diterpene present in plants belonging to the Podocarpaceae, Cupressaceae, Lamiaceae, and Verbenaceae families. This diterpene has shown promising biological activities, such as antifungal and antibacterial, 15 miticidal, 16 cardioactive, 17 antioxidative, 18 anti-plasmodial, 19 and anti-ulcerogenic 20 properties.
Numerous studies show that several signaling pathways are altered in melanoma pathogenesis; these include mitogen-activated protein kinases (MAPKs), JNK, and JAK-2/STAT-3. 21 –23 This creates opportunity to the scientist for developing the promising therapeutic agent to be used against melanoma cancer. We examined the probable effects of FGL in melanoma cancer cells. Here, we have reported for the first time the molecular mechanism by which FGL induces melanoma cell death. We specifically explored the effects of FGL on key molecular target p38 MAPK and the downstream effector Nuclear factor-κB (NF-κB) involved in apoptosis regulation. The modulating effects of FGL on these molecular targets in a dose-dependent manner were examined. The outcomes from this study would substantiate the role of FGL as an anti-melanoma drug candidate, for its future evaluation in preclinical studies.
Materials and methods
Chemicals and reagents
FGL (catalog no: T5330; 95% purity by high-performance liquid chromatography (HPLC)), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), dimethyl sulfoxide (DMSO), penicillin, and streptomycin were purchased from Sigma-Aldrich (St. Louis, Missouri, USA). Fetal bovine serum (10% heat-inactivated), 3.5% trypsin, and Dulbecco’s modified Eagle’s medium were purchased from Gibco (Carlsbad, California, USA).
Human malignant melanoma cell line (SK-Mel-28) was purchased from the Cell Institute, Chinese Academy of Sciences (Shanghai, China). The cells were cultured in Dulbecco’s modified Eagle’s medium comprising of 5% heat-inactivated fetal bovine serum, 100 U/ml penicillin, and 100 µg/ml streptomycin with 5% carbon dioxide at 37°C.
Cell cytotoxicity assay
In brief, human malignant melanoma (SK-Mel-28) cells, at a density of 1 × 105 cells/ml, were cultured in a 96-well chamber slide (10,000 cells/chamber) for 24 h before using them in the experiment. SK-Mel-28 cells were then treated with 0, 10, 20, 40, 80, 160, and 320 µM dose of FGL dissolved in DMSO (0.1%) for 24 and 48 h; 0 µM of FGL was served as control. Constant concentration of DMSO (10 µl of 0.1% DMSO per milliliter of medium) was added to per milliliter of medium to arrive desired concentration of FGL, including “0” µM of FGL. Subsequently, MTT was added into each well and then again incubated for 4 h before the supernatant was discarded. The formazan crystals formed were dissolved in DMSO and the absorbance values were measured using an automated microplate reader (BioTek Instruments, Inc., Winooski, Vermont, USA) at 570 nm. Cell viability was calculated based on percent change when compared with control cells (“0” µM of FGL). Cytotoxicity was represented as the concentration of FGL inhibiting cell proliferation by 25% (IC25 value), 50% (IC50 value), and 75% (IC75 value).
Lactate dehydrogenase assay
Lactate dehydrogenase (LDH) activity was assayed through commercially available kit from Stanbio Laboratory (Boerne, Texas, USA; catalog no: G428-86).
DNA damage (strand breaks) by comet assay
A sensitive and frequently used technique to quantify DNA damage within cells is comet assay. After the experimental period of incubation, the cells were harvested using trypsin digestion. The cells were washed in phosphate-buffered saline (PBS) by centrifugation at 80 × g for 2 min. Comet assay was performed and data were analyzed as explained in the literature. 24,25 In detail, the slides were pre-coated with 1% normal melting point (NMP) agarose. After the experimental period, cells were harvested through enzymatic digestion and counted (1 × 106/ml). The cell suspension was further diluted with 0.5% low-melting point agarose (1 in 100 dilution) and was layered (75 µl) on to the slide. The layer was covered with 1% NMP (50 µl) and the cells embedded in agarose were cooled down on ice to set. The slides were kept in lysis buffer (10 mM Tris-HCl pH 10 with 100 mM ethylenediaminetetraacetic acid (EDTA), 2.5 M sodium chloride, detergents, and 10% DMSO) for 1 h and were later neutralized with 0.4 M Tris pH 7.5. Electrophoresis (dark room) was done at 50 mA for 20 min with alkaline buffer pH 13 and the slides were observed using fluorescent microscope at 40 × objective after staining with ethidium bromide (10 mg/ml concentration). The comet images were captured under the microscope and processed using TriTek Comet Score Freeware™ v1.5.
Apoptosis assay through flow cytometry
Post experimental period cells were rinsed twice with ice-cold PBS, and the attached cells were harvested using trypsin-EDTA. Cells were stained with Annexin V-Cy3 and 7-AAD as explained by Thirunavukkarasu et al. 26
Terminal deoxynucleotidyl transferase dUTP nick-end labeling assay
The extent of DNA fragmentation in control and FGL-treated cells was analyzed by terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining using TACS 2 TdT-Fluor in situ apoptosis detection kit (Trevigen, Inc., Gaithersburg, Maryland, USA; catalog no: 3500-096-01). A randomly selected 10 different fields were observed for each condition.
Quantification of P-p38, NF-κB, and β-actin proteins by Western blot
Cells were treated with IC50 concentration of FGL for 24 h; 30 µg of protein was separated by 12% SDS-PAGE and transferred onto nitrocellulose membrane. For analysis of NF-κB expression, nuclear protein was extracted. The separated proteins were transferred to Immobilon-P membrane (Millipore, Bedford, Massachusetts, USA). The membranes were blocked for 1 h with 1.0% bovine serum albumin in tris-buffered saline (TBS). The membrane was incubated overnight with anti-P-p38 (Cell signaling; #9212) and anti-NF-κB (Cell signaling; #8242) antibodies (all at 1:1000 dilution). After washing with a mixture of tris-buffered saline (TBS) and Polysorbate 20 (TBST) (4×), the membranes were incubated with host-specific peroxidase-linked secondary antibodies (1:25,000) for 1 h. The immunoreactive protein detection was achieved using an enhanced chemiluminescence kit (Amersham Pharmacia Biotech Ltd, Little Chalfont, United Kingdom). To confirm equal protein loading, β-actin (Cell signaling; 4970S) was used as loading control. The band intensities were quantified using ImageJ software (NIH, Bethesda, Maryland, USA).
Assay of caspase-3 activity
Commercially available fluorescent assay kit (BD Biosciences-Clontech; catalog no: 556485) was utilized to estimate the caspase-3 activity. In detail, after the experimental period, cells were washed with ice-cold PBS and cells were collected using trypsin-EDTA. Cells were resuspended in 50 µl of chilled cell lysis buffer and cells were incubated on ice for 10 min. After incubation, cells were centrifuged at 10,000 × g for 1 min. The supernatant (cytosolic extract) was used to assay caspase-3 activity. Background reading from cell lysates and buffers were subtracted from the readings of experimental samples. Fold-increase in caspase-3 activity was determined by comparing FGL-treated cells with the level of the untreated control.
Statistical analysis
All data were expressed as mean ± standard deviation. The means of the different groups were compared using one-way analysis of variance Tukey’s post hoc test. Each experiment was performed at least three times unless otherwise mentioned. The p value <0.05 was considered to indicate a statistically significant difference.
Results
Cytotoxic effect of FGL on SK-Mel-28 human malignant melanoma cells
The cytotoxicity of the FGL was assayed through MTT assay using a range of concentrations including 0, 10, 20, 40, 80, 160, and 320 µM. The results point out that FGL is a potent cytotoxic agent showing dose-dependent growth inhibition of these cancer cells. The cytotoxic effects of the FGL were tested at 24- and 48-h incubation time and results revealed that the drug exerted time- and concentration-dependent anticancer effect. Figure 1(a) shows the cytotoxicity effect of FGL in SK-Mel-28 cells. It should be noted that the statistically significant cytotoxic activity of FGL was observed at 20 and 10 µM, respectively, for 24 and 48 h. We estimated that the concentrations of FGL for IC25, IC50, and IC75 were 36, 85, and 180 µM, respectively, at 24 h. Similarly, IC25, IC50, and IC75 concentrations were found to be 24, 55, and 103 µM, respectively, at 48 h. The above findings indicate that the drug concentration can be lowered to achieve similar efficacy with increased treatment duration.
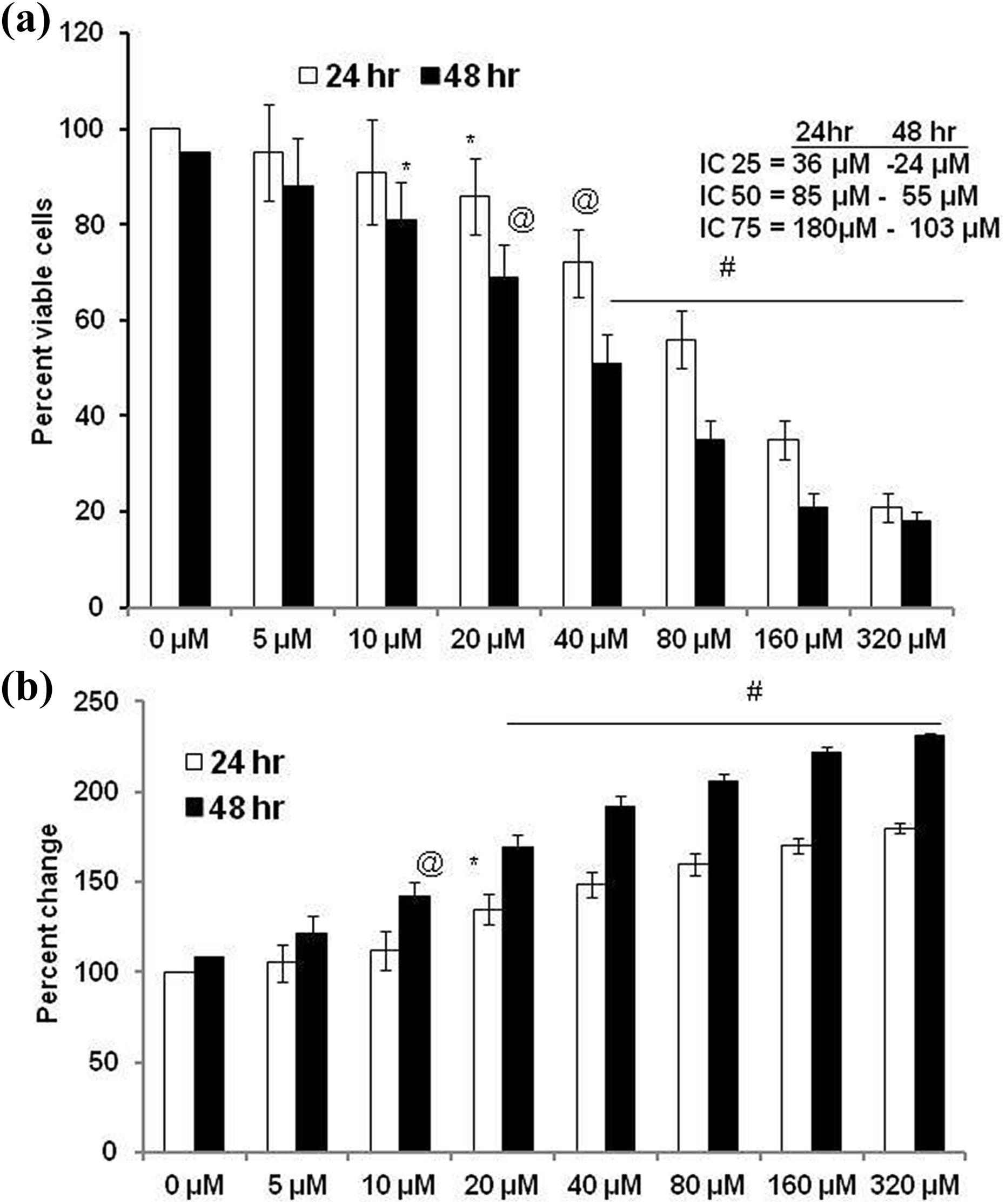
FGL induces concentration-dependent cytotoxicity in human malignant melanoma cells (SK-Mel-28). The cells were treated with 0, 5, 10, 20, 40, 80, 160, and 320 µM concentrations of FGL and then incubated for 24 and 48 h. (a) Cell cytotoxicity was assessed by MTT assay and (b) LDH released into the medium. Data are mentioned as mean ± standard deviation of three independent experiments. Differences were analyzed by one-way ANOVA Tukey’s post hoc test. *p < 0.05, @ p < 0.01, # p < 0.001: versus 0 µM (control). FGL: ferruginol; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; LDH: lactate dehydrogenase; ANOVA: analysis of variance.
Next, we analyzed the LDH activity in the culture supernatant (Figure 1(b)), to understand if the reduction in cell viability was due to plasma membrane damage. LDH is an enzyme located in the cytosol, which leaks from the cell upon membrane damage, hence the extent of LDH in the media is directly proportional to membrane damage and leakage, indicating cytotoxicity. Like MTT assay, LDH activity in the medium was elevated upon increasing the concentration of FGL. Statistically significant LDH activity was observed at 20 and 10 µM, respectively, at 24 and 48 h. We observed that increase in duration of treatment retained the cytotoxicity even upon lowering the concentration of FGL. Subsequently, for further experiments, we treated cells with IC25, IC50, and IC75 concentrations for 24 h alone.
FGL induced DNA damage on melanoma cells
Comet is an efficient technique to detect the DNA damage at cellular level. Cells were treated with IC25, IC50, and IC75 concentrations of FGL for 24 h and comet assay was performed; the results are shown in Figure 2. In the present study, we found that treatment of melanoma cells with IC25 concentration of FGL induces the statistically (p < 0.001) significant level of DNA damage as seen in increased tail length (Figure 2(a)), increased number of cells with comet (Figure 2(b)), and increased percent of DNA in tail regions (Figure 2(c)).

Effect of FGL on DNA damage. Cells were treated with IC25, IC50, and IC75 concentrations of FGL for 24 h. At the end of treatment, DNA damage was quantified through comet assay as explained in the “Materials and methods” section. (a) Tail length, (b) percent cells with comet, (c) percent DNA in comet, and (d) representative picture of control and FGL-treated cells. Values are mentioned as mean ± SEM. Experiments were repeated minimum thrice with duplicate each time. Differences were analyzed by one-way ANOVA Tukey’s post hoc test. The p value <0.05 was considered significant. # p < 0.001: versus control. FGL: ferruginol; SEM: standard error of mean; ANOVA: analysis of variance.
FGL induced apoptosis on melanoma cells
The results observed from comet assay inferred that FGL can induce the DNA damage. This might indicate that FGL-induced DNA damage leads to apoptosis or necrosis; hence, to further confirm the mode of DNA damage, flow cytometry assay (Figure 3(a) and Table 1) was performed. After the experimental period (24 h), cells were stained with 7-AAD and Annexin V-Cy3. 7-AAD is a membrane impermeable DNA-binding dye which could enter into the cells upon damage of plasma membrane. During the early stage of apoptosis, plasma membrane lipid such as phosphatidylserine translocates from inner leaflets of plasma membrane to outer leaflets, which can be detected by Annexin V-Cy3 staining. So, 7-AAD positivity indicates the necrosis, while Annexin V-Cy3 positivity indicates the early apoptosis and 7-AAD + Annexin V-Cy3 double positivity indicates late apoptosis. As seen in Figure 3(a), IC25 concentration increases (p < 0.001) significant number of Annexin V-Cy3 alone positive cells than that of control cells. Similarly, 7-AAD + Annexin V-Cy3 positive cells also increased from approximately 2% to 8%, whereas there is an increase in 7-AAD alone positive cells. In the same way, treatment of cells with IC50 and IC75 concentrations increases Annexin V-Cy3 alone positive cells (28%-IC50 and 36%-IC75) and 7-AAD + Annexin V-Cy3 positive cells (14%-IC50 and 30%-IC75), on the other hand there is no significant rise in 7-AAD alone positive cells. These findings indicate that FGL could induce apoptosis-mediated cell death in melanoma cells.

(a) Flow cytometry analysis. Representative experiment showing flow cytometric analysis in terms of percentage apoptosis in different experimental cells. The vertical drawn from x-axis shows 7-AAD intensity and horizontal drawn from y-axis shows Annexin V-Cy3 intensity. (a) Normal cells (both Annexin V-Cy3 and 7-AAD negative cells). (b) Early apoptotic cells (only Annexin V-Cy3 positive). (c) Necrotic cells (only 7-AAD positive cells). (d) Late apoptotic cells (both Annexin V-Cy3 and 7-AAD positive cells). Experiments were repeated twice with duplicate each time. (b) Active caspase-3 activity. Experiments were repeated minimum twice with triplicate each time. Data represented are mean ± SEM. Differences were analyzed by one-way ANOVA Tukey’s post hoc test. The p value <0.05 was considered significant. *p < 0.05, # p < 0.001: versus control. SEM: standard error of mean; ANOVA: analysis of variance.
Percent healthy, early apoptotic, late apoptotic, and necrotic cells in control and experimental conditions.a

FGL: ferruginol; SEM: standard error of mean; ANOVA: analysis of variance.
a Values are mentioned as mean ± SEM of two different experiments with duplicate. One-way ANOVA Tukey’s post hoc test was utilized to analyze the statistical difference. The p value <0.05 was considered significant.
b p < 0.001: versus control.
c p < 0.05: versus control.
Apoptosis could be initiated either via intrinsic or extrinsic mode, leading to the activation of effector caspases. Caspase-3 is a terminal effector caspase in the apoptosis, which is activated by both extrinsic and intrinsic pathways. In the present study, we found (Figure 3(b)) that FGL increases (p < 0.001) casepase-3 activity in treated cells than that of control cells. These findings confirm that FGL exhibits cytotoxicity in melanoma cells via apoptosis-mediated cell death. Further to confirm that FGL induced apoptosis, we have performed TUNEL assay. As seen in Figure 4(a) and (b), statistically significant (# p < 0.001) amount of TUNEL positive cells were observed in FGL (IC50)-treated cells.

TUNEL assay. Representative image (a) showing TUNEL positive cells in control and FGL-treated cells. Experiments were repeated thrice with duplicate each time. (b) Par diagram of TUNEL positive cells. Data represented are mean ± SEM. Differences were analyzed by one-way ANOVA Tukey’s post hoc test. The p value <0.05 was considered significant. # p < 0.001: versus control. TUNEL: terminal deoxynucleotidyl transferase dUTP nick-end labeling; FGL: ferruginol; SEM: standard error of mean; ANOVA: analysis of variance.
Involvement of MAPKs and NF-κB in FLG-induced cell death
In the present study, we found that P-p38 expression increased (p < 0.001) significantly (Figure 5(a) and (b)) in FGL-treated cells than that of control cells. We did not find any statistically significant modulation in other MAPKs such as P-ERK1 and P-JNK. In the present study, we found that FGL treatment increased the nuclear translocation of RELA in SK-Mel-28 cells. Significant level of translocation was observed in cells treated with IC25 concentration of FGL.

Western analysis for P-p38 and NF-κB expression. (a) Representative image (three identical experiments) showing immunoblotting of P-p38 or NF-κB and β-actin (as loading control). (b) Graphical representation of P-p38 or NF-κB/β-actin ratio. Data are represented as mean ± standard deviation. One-way ANOVA Tukey’s post hoc test was utilized to analyze the statistical difference. The p value <0.05 was considered significant. # p < 0.001: versus control. ANOVA: analysis of variance.
To understand P-p38 contribution in RELA translocation and vice versa, cells were pretreated with SB 203580, a p38 phosphorylation inhibitor, and ammonium pyrrolidine dithiocarbamate (PDTC), an antioxidant and RELA translocation inhibitor. Western analysis shows that (Figure 6(a) and (b)) inhibition of p38 phosphorylation could prevent the RELA translocation, on the other hand PDTC could not prevent the p38 phosphorylation. It is observed that FGL (Figure 6(b)) treatment causes nuclear translocation of RELA through P-p38. Further, it was observed that both the inhibitors could prevent FGL-induced melanoma cell death (Figure 6(c)).
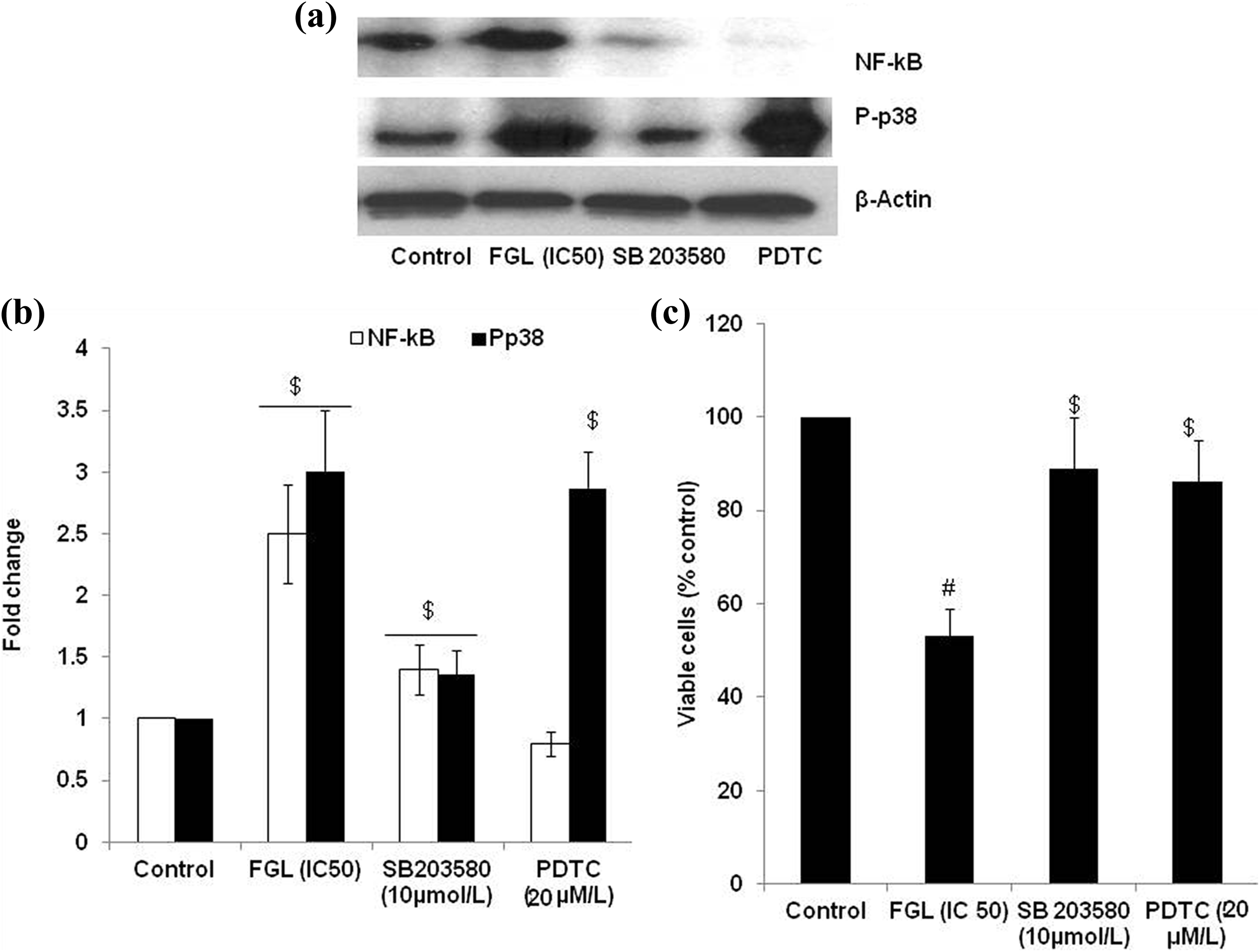
p38 Kinase inhibition and NF-κB translocation prevention in cell viability in FGL (IC50)-treated cells. Cells were pretreated with p38 kinase (SB203580; 10 µmol/l) and RELA translocation prevention (PDTC; 20 µM) for 30 min. FGL was then added and the incubation continued for 24 h. P-p38 and NF-κB Western blotting were then analyzed (a); fold change (b), and cell viability (c) (MTT assay). Values are mentioned as mean ± SD of three different experiments with triplicate. One-way ANOVA Tukey’s post hoc test was utilized to analyze the statistical difference. The p value <0.05 was considered significant. $ p < 0.01, # p < 0.001: versus control. FGL: ferruginol; PDTC: pyrrolidine dithiocarbamate; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; SD: standard deviation; ANOVA: analysis of variance.
Further to understand the P-p38 and NF-κB role in FGL-induced apoptosis, cells were pretreated with the abovementioned inhibitors. It was observed that inhibiting P-p38 and NF-κB could reduce caspase-3 activity in FGL-treated cells (Figure 7(a)). This finding indicated that FGL-induced apoptosis initiated through p38 phosphorylation leads to NF-κB translocation, further activating caspase-3 (Figure 7(b)).

p38 Kinase inhibition and NF-κB translocation prevention in active caspase-3 activity in FGL (IC50)-treated cells. Cells were pretreated with p38 kinase (SB203580; 10 µmol/l) and NF-κB translocation prevention (PDTC; 20 µM) for 30 min. FGL was then added and the incubation continued for 24 h. (a) Active caspase-3 assay was performed. (b) Schematic representation of proposed pathway. Values mentioned are mean ± SD of two different experiments performed in triplicate. One-way ANOVA Tukey’s post hoc test was utilized to analyze the statistical difference. The p value <0.05 was considered significant. $ p < 0.01, # p < 0.001: versus control; & p < 0.05: versus FGL (IC50 dose). FGL: ferruginol; PDTC: pyrrolidine dithiocarbamate; SD: standard deviation; ANOVA: analysis of variance.
Discussion
In the current clinical scenario, treatment choices for melanoma are limited, on the other hand approximately 75% of the skin cancer-related deaths are due to malignant melanoma. Currently, numerous investigators focus phytomedicine as a choice for chemotherapeutic/chemopreventive agent because of their minimal toxicity in normal cells. Aggravated in cell proliferation, aberrant skin lesions and metastasis are the common pathogenesis of malignant melanoma. Phytochemicals that could serve as cell cycle arrest, inducing apoptosis-mediated cell death, and oxidative status of cell could serve as potential agents against melanoma. Various phenolic compounds have shown to exhibit abovementioned properties. 11 –13 FGL is known for its antifungal, antioxidant properties and cytotoxicity against lung cancer cells, in addition to its cardioprotective property. In the present investigation, we evaluated its effects against (SK-Mel-28) melanoma cancer cells, one of the major lethal skin cancers in human.
It is known that number of drugs or lead molecules of existing drugs have been isolated from plants or natural sources, and many of them exhibit systemic toxicity. However, phenolics have been shown to exhibit minimal or nil systemic toxicity in human 27 and therefore may be established as vital targets for expansion of cancer chemotherapy. Further, phenolics are one of the universal ingredients in the human diet. Moreover, phenolics have been shown to exhibit chemopreventive effect against different cancers. They have been reported to execute anticancer activity by various mechanisms including cell cycle arrest, induction of apoptosis, carcinogen inactivation, suppression of angiogenesis, antioxidative capacity, and reversal of multidrug resistance. 28 –31
In the present investigation, our main aim was to determine the mode of anticancer effect of FGL in SK-Mel-28 human malignant melanoma cells. Its ability to induce apoptosis was correlated with studying its effects on MAPK activation and NF-κB translocation. The findings suggest that FGL inhibits cancer cell proliferation in SK-Mel-28 melanoma cells in a dose-dependent manner as well as time-dependent manner. FGL treatment also led to onset of DNA damage as visualized through comet assay, and the intensity of DNA damage increased with increasing concentration of the drug, indicating that FGL induces necrosis or apoptosis in a dose-dependent manner. To distinguish the mode of cell death, FGL-treated cells were stained with 7-AAD and Annexin V-Cy3. The increase in Annexin V-Cy3 positive and 7-AAD + Annexin V-Cy3 cells indicates that FGL causes apoptosis in SK-Mel-28 melanoma cells. Further, upon increasing the concentration (even IC75), we have not observed an increase in necrotic (only 7-AAD positive cells) cells. This observation indicates that even at higher concentration FGL induces apoptosis in SK-Mel-28 melanoma cells. Apoptosis could be mediated through death receptor activation (extrinsic) and mitochondrial (intrinsic) cytochrome-c release. In both cases, conversion of pro-caspase-3 to active caspase-3 is critical. FGL-induced apoptosis was confirmed through increased active caspase-3 in FGL-treated cells than that of control cells. Previous studies have shown that phenolics could induce apoptosis in various cancer cells. Recently, Ho et al. showed that FGL could induce apoptosis in lung cancer cells. 32 Similarly, Xiong et al. observed apoptosis-mediated cell death in human ovarian cancer cells. 33 So, our findings agree with previous literatures. 32 –36 Further, these results indicate that FGL induces caspase-dependent apoptosis and cell cycle arrest. 32 –35
p38 MAPK is a class of MAPKs which is responsive to stress stimuli, such as UV radiation, cytokines, osmotic shock, and heat shock. p38 MAPK activation through phosphorylation (P-p38) is involved in cell differentiation, autophagy, and apoptosis. 37 –39 NF-kB p65 subunit/RELA is a protein that in humans is encoded by the RELA gene. RELA as part of NF-κB heterodimer complex is retained in cytosol; upon activation, RELA is translocated into the nucleus. NF-κB is an important transcription factor complex and plays a vital role in cellular metabolism, differentiation, and chemotaxis. 40 –42 Western blotting assay shows that FGL increases P-p38 and RELA nuclear translocation in FGL-treated cells. Further, we also noticed that above changes are concentration dependent. P-p38 is one of the important members of MAPK family. Phosphorylation of p38 is a signaling mechanism where most of the flavonoids exert their effect. 43,44 In the current study, we also found that FGL could induce phosphorylation of p38. RELA is a transcription factor. As the prototypical heterodimer complex member of the NF-κB, together with p50, RELA/p65 interacts with various proteins in both the cytoplasm and the nucleus during the process of classical NF-κB activation and nuclear translocation. 45,46 In the inactive state, RELA/p50 complex is mainly sequestered by IκBα in the cytosol. In the present study, nuclear localization of RELA occurs in response to FGL exposure. Similarly, there is a nonclassical NF-κB activation pathway involving the proteolytic cleavage of p100 into p52 instead of p50. 47,48 This process does not require RELA, hence existence of nonclassical NF-κB activation pathway in response to FGL treatment will not be possible. After RELA nuclear localization due to FGL treatment, p50/RELA heterodimer would function as a transcription factor. This complex interacts with and activates different genes involved in various biological functions. Decreased caspase-3 activity in PDTC pretreated (prevents RELA translocation to nucleus) cells indicates that RELA translocation is necessary to induce apoptosis in FGL treatment.
In summary, the current study reports the anticancer and apoptotic effects of FGL in SK-Mel-28 human malignant melanoma cells. Apoptotic effect of the compound was mediated via p38 phosphorylation and translocation of RELA to nucleus. Moreover, the molecule did not exert necrosis as evident from 7-AAD and Annexin V-Cy3 staining. Taken together, the results indicate that FGL may be established as an important lead molecule.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
