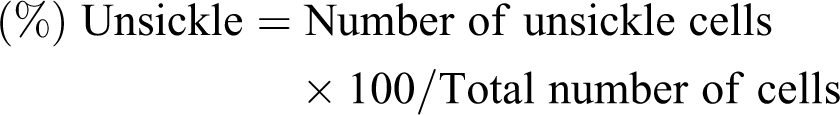Abstract
Sickle cell disease (SCD) is a medical condition caused by mutation in a single nucleotide in the β-globin gene. It is a health problem for people in sub-Saharan Africa, the Middle East and India. Orthodox drugs developed so far for SCD focus largely on symptomatic respite of pain and crisis mitigation. We investigated the antisickling effects of chrysin via modulation of deoxy-haemoglobin, 2,3-bisphosphoglycerate mutase, redox homeostasis and alteration of functional chemistry in human sickle erythrocytes. In silico and in vitro methods were adopted for the studies. Chrysin was docked against deoxy-haemoglobin and 2,3-bisphosphoglycerate mutase, with binding energies (−24.064 and −18.171 kcal/mol) and inhibition constant (K i ) of 0.990 µM and 0.993 µM at their active sites through strong hydrophobic and hydrogen bond interactions. Sickling was induced with 2% metabisulphite at 3 h. Chrysin was able to prevent sickling maximally at 2.5 µg/mL and reversed the same at 12.5 µg/mL, by 66.5% and 69.6%, respectively. Treatment with chrysin significantly (p < 0.05) re-established the integrity of erythrocytes membrane as evident from the observed percentage of haemolysis relative to induced erythrocytes. Chrysin also significantly (p < 0.05) prevented and reversed lipid peroxidation. Similarly, glutathione and catalase levels were observed to significantly (p < 0.05) increase with concomitant significant (p < 0.05) decrease in superoxide dismutase activity relative to untreated. From Fourier-transform infrared results, treatment with chrysin was able to favourably alter the functional chemistry, judging from the shifts and functional groups observed. Sickling-suppressive effects of chrysin may therefore be associated with sequestration of deoxy-haemoglobin, 2,3-bisphosphoglycerate mutase, alteration of redox homeostasis and functional chemistry of sickle erythrocytes.
Introduction
Sickle cell disease (SCD) is a medical condition caused by mutation in a single nucleotide in the β-globin gene resulting in transcription and translation to sickle-shaped haemoglobin (HbS) protein. 1 This alteration in the amino acid sequence of haemoglobin changes the isoelectric point of this protein, reduces the oxygen carrying capacity and makes HbS more prone to polymerization. 2 SCD is a health problem for people in sub-Saharan Africa, the Middle East and India. 3 About 20–30% of the Nigerian population are reported to be carriers of the sickle cell trait with 2–3% of the population suffering from SCD. Children with SCD experience periods of steady state interspersed by acute episodes characterized by chronic haemolytic anaemia and vaso-occlusive pain crisis. 4
The chronic haemolytic anaemia in SCD is caused by low oxygen levels in sickle haemoglobin, which makes HbS susceptible to deformation and polymerization. 5 The vaso-occlusive pain crisis is caused by inadequate oxygen supply induced damage to vascular endothelial cells resulting in altered vascular tone, vascular adhesion and receptor function. 6 The remarkable ability of haemoglobin to carry oxygen to tissues is dependent on the reversible binding of oxygen to Fe (II) haemoglobin, 7 which is altered in SCD resulting in increased generation of reactive oxygen species and subsequently oxidative stress. 8 The molecule 2,3-bisphosphoglycerate (2,3-BPG) synthesized from 1,3-bisphosphoglycerate by bisphosphoglycerate mutase is elevated in SCD. 9 2,3-Bisphosphoglycerate in normal erythrocytes binds to deoxy-haemoglobin and stabilizes it, thereby reducing its oxygen affinity. However in SCD, binding of 2,3-BPG to deoxy-HbS decreases its solubility, making HbS more prone to polymerization. 9 Thus, 2,3-BPG could serve as a therapeutic target in SCD, as agents that reduce the level and activity of 2,3-BPG in SCD patients have the potential to ameliorate the severity of SCD pathogenesis. 10
Orthodox drugs developed so far for sickle cell management focus largely on symptomatic respite of pain and crisis mitigation. Likewise, side effects related to orthodox drugs administration, availability and affordability especially in developing nations are some of the reported problems of major concern. 11 Chrysin (5,7-dihydroxyflavone) is a known antioxidant found in plants like Passiflora caerulea, Passiflora incarnata and Oroxylum indicum, honey and propolis, in addition to fruits and vegetables. 12 In recent studies from our research laboratory, the antioxidant, anticlastogenic and DNA-protective properties of chrysin were conveyed at in vitro and in vivo levels. 13 Chrysin is reported to have a high bioavailability which enhances its anti-inflammatory and protein stability effects that could be beneficial to SCD-associated oxidative stress, inflammation and decreased HbS stability. 14 Chrysin has also been reported to have enzyme inhibitory effects 15,16 but has not yet been studied on sickling-suppressive effects, thus necessitating this study. Accordingly, this study aimed at examining the antisickling effects of chrysin via modulation of deoxy-haemoglobin, 2,3-bisphosphoglycerate mutase, redox homeostasis and alteration of functional chemistry in human sickle erythrocytes for the first time. The findings from this study will go a long way in adding to the existing knowledge of exploring natural products in combating and managing SCD and its biochemical complications.
Materials and methods
Chemicals and reagents
Chrysin (Sigma Aldrich, St Louis, Missouri, USA) with molecular weight of 254.24 g/mol, 284–286 C melting point and 97% purity was purchased. All other chemicals/reagents used were of analytical grade until otherwise stated.
In silico studies
The structure of a ligand; chrysin (5281607) was retrieved from the major ligand database, PubChem. A PDB format of the target proteins, deoxygenated haemoglobin (PDB ID: 3WCU) and 2,3-bisphophoglycerate mutase (PDB ID: 3NFY), was obtained from the protein data bank. The proteins preparation and energy minimization were done using Discovery studio 2.5v. The CHARMm-based DOCKER program was used to score the interaction between the proteins and the ligand (chrysin) into the crystal structure of the receptors binding pockets. The PDB files of the best docked conformation were generated using Accerlrys discovery studio and were imported into Chimera 1.1v to view the hydrophobic and hydrophilic interactions. The binding energies and kinetic inhibition constants were equally calculated.
In vitro studies
Blood collection
About 4–5 mL blood was humanely obtained by venepuncture from each of seven confirmed people living with sickle cell anaemia (not in crises) at the Haematology Department of Ahmadu Bello University Teaching Hospital (ABUTH), Zaria, Kaduna State, Nigeria. They were aged between 19 and 32 years of both sexes and in reasonable stable condition. They were informed and provided their consents prior to the experiment. Blood was collected in sodium ethylenediaminetetraacetic acid bottles, and the content was carefully mixed by gently rolling the bottle. All experiments were carried out under the ethical approval of ABUTH Scientific and Health Research Ethics Committee (ABUTH/HREC/UG/6), in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All experiments were performed with fresh blood.
Mining of erythrocytes
The acquired homozygous HbS blood samples were spun at 3000 r/min for 10 min. The resultant plasma (supernatant) was cast-off to obtain erythrocytes which were suspended in phosphate buffer saline (PBS), spun and the supernatant discarded. This was continual thrice, after which the washed erythrocytes were resuspended in PBS and immediately used for investigation.
Induction of sickling, antisickling and osmotic fragility assays
Exactly 0.1 mL of the washed erythrocytes were mixed with 0.1 mL of freshly prepared 2% sodium metabisulphite in a clean test tube and thereafter incubated in water bath at 37°C for 3 h. A drop of the mixture was then smeared on a microscope slide, fixed with 95% methanol, dried and stained with 5% Giemsa (pH 7.2) for 30 min. 17 It was then examined under a microscope (100× magnification). Aliquots were taken at 30 min interval for up to 3 h. The counting of cells was carried out by viewing from different fields (five fields) across the slide. The numbers of both sickle and unsickle cells were counted and the percentages of cells unsickle were determined using the formula
For antisickling assay, chrysin was used at concentrations of 2.5, 5.0, 7.5, 10.0 and 12.5 µg/mL for both preventive and after induction approaches. For preventive, the cells were treated with the said concentrations before induction using 2% sodium metabisulphite and vice versa for after induction approach. Equally, osmotic fragility (haemolysis) test was conducted against chrysin after selecting the best concentration as previously described. 18,19 Subsequently, the same concentration with the best antisickling effect was also used for redox and Fourier-transform infrared (FTIR) analyses, respectively. However, it is worthy to mention that a single control was employed in this bioassay; that is, induced sickle cells with 2% sodium metabisulphite.
Evaluation of chrysin for redox-sensitive biomarkers in sickle erythrocytes
The best concentrations (2.5 µg/mL for preventive and 12.5 µg/mL for after induction studies) with the highest antisickling effect were incubated with 100 µL sickle erythrocytes for 3 h at 37°C as previously described in the above section using the method of Muhammad et al. 19 Thereafter, the cells were spun and the supernatant assayed for reduced glutathione (GSH), 20 lipid peroxidation, 21 superoxide dismutase (SOD) and catalase (CAT) levels. 22 , 23
FTIR spectroscopy analysis of treated sickle erythrocytes
The best concentrations (2.5 µg/mL for preventive and 12.5µg/mL for after induction studies) with the highest antisickling effect were incubated with 100 µL sickle erythrocytes for 3 h at 37°C as previously described in the above section, while adopting the methods of Erukainure et al. 11 After treatment, the samples were dehydrated under pressure and sandwiched between potassium bromide. They were then perused on FTIR spectrophotometer at room temperature (250–280°C) at 1000–3500 cm−1 spectral range. The peak heights were utilized in identifying the functional groups present by relating to the infrared spectroscopy correlation table.
Statistical analysis
The experiment was carried out and repeated at least thrice. The results were presented as mean ± standard deviation (SD) as the case may be, where sickle erythrocytes and chrysin were serving as dependent and independent variables, respectively. The data were analysed using Student’s t-test with the aid of Statistical Package for Social Sciences (SPSS) software (SPSS Inc., Chicago, Illinois, USA) Standard version 20.00. p < 0.05 was considered statistically significant for differences in mean.
Results
The results from in silico analysis showed chrysin’s ability to comfortably dock with strong binding affinity into the active site of the receptors, deoxygenated haemoglobin and 2,3-bisphosphoglycerate mutase (Tables 1 and 2), signifying a prospective impounded and inhibitory effect via hydrophilic and hydrophobic interactions (Figures 1 and 2).

3D and 2D binding interaction of chrysin with deoxygenated haemoglobin.

3D and 2D binding interactions of chrysin with 2, 3-bisphophoglycerate mutase.
The docking score (kcal/mol) and inhibition constant (µM) of chrysin at the binding site of deoxygenated haemoglobin.

The docking score (kcal/mol) and inhibition constant (µM) of chrysin at the binding site of 2,3-bisphosphoglycerate mutase.

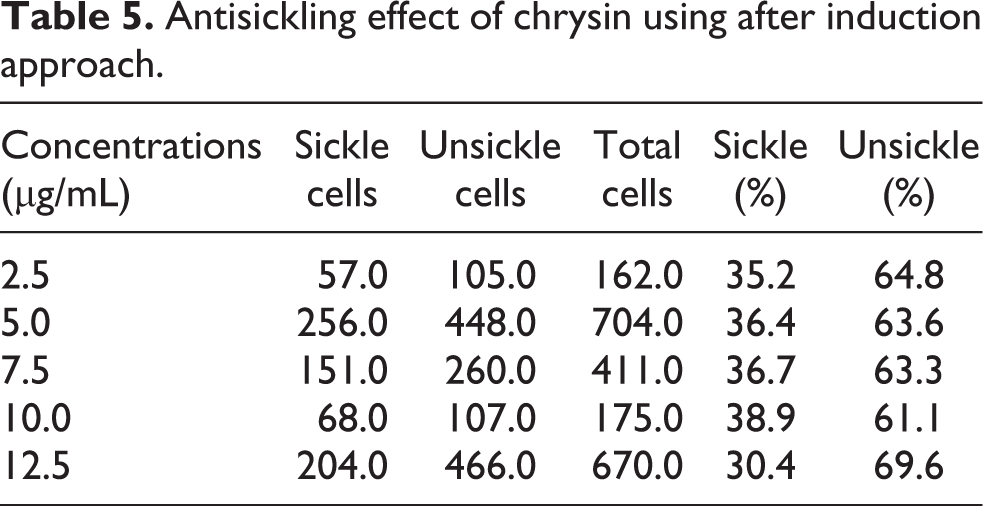
Taking a cue from the predictions at the docking levels that chrysin may in fact sequester deoxygenated haemoglobin and 2,3-bisphosphoglycerate mutase by means of strong chemical interactions, we proceeded with antisickling studies using preventive and after induction approaches. To realize this, 2% metabisulphite was in the first instance used to induce sickling for the reason that the said blood donors were not in crises as at the period of collection. As presented in Table 3, sickling was induced at 3 h by 73.2%. In essence, 3 h was employed for our supplementary assays. Fascinatingly, chrysin administration was able to prevent sickling hugely at 2.5 µg/mL (Table 4) and reversed the same at 12.5 µg/mL (Table 5), by 66.5% and 69.6%, one-to-one (Figure 3). Accordingly, the said concentrations were selected for additional assays because the detected sickling preventive and curative properties were not concentration-dependent. IC50 calculation is therefore not necessary.

Comparative photomicrograph of (a) induced, (b) preventive with 2.5µ/mL and (c) after induction with 12.5µ/mL chrysin. Stain: Giemsa, magnification ×100.
Percentage sickling after induction with 2% sodium metabisulphite.

Antisickling effect of chrysin using preventive approach.

Antisickling effect of chrysin using after induction approach.
Integrity of erythrocytes membrane is essential to antisickling properties. Looking at Figure 4, it can thus be seen that treatment with chrysin significantly (p < 0.05) re-established the integrity of erythrocytes membrane as evident from the observed haemolysis percentage relative to induced erythrocytes (without treatment). However, the effects were both preventive and reversal in nature (Figure 4).
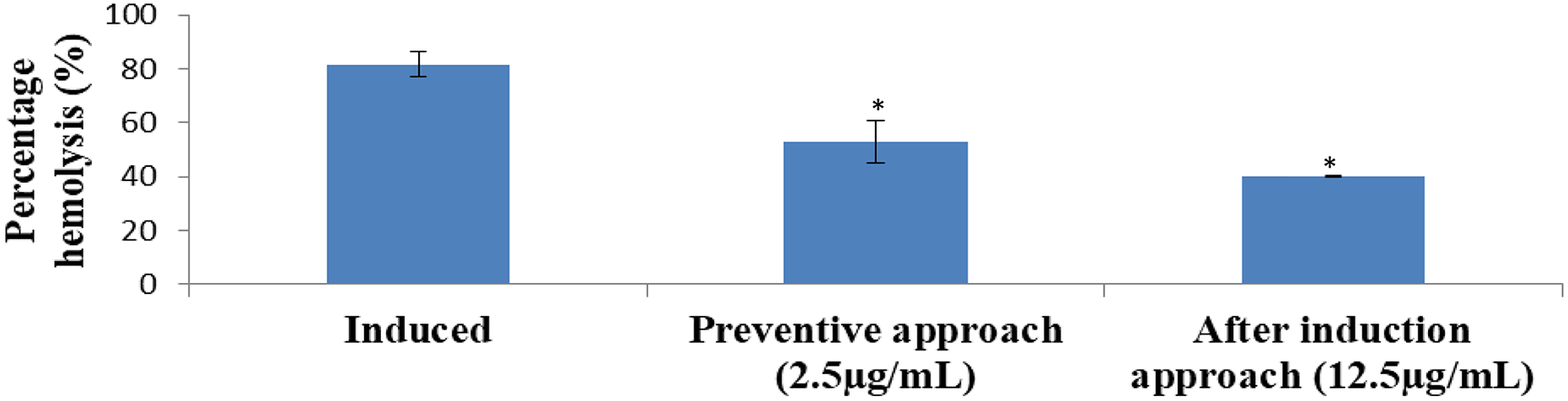
Membrane stabilizing effect of chrysin on 2% metabisulphite-induced sickling in human erythrocytes. *p < 0.05: statistically significant when compared with the induced.
Redox imbalance is one of the hallmarks of SCD, and as presented in Table 6, treatment with chrysin significantly (p < 0.05) prevented and reversed lipid peroxidation as is evident from the level of malondialdehyde (MDA) formed relative to induced erythrocytes. Similarly, GSH and CAT levels were observed to significantly (p < 0.05) increase with concomitant significant (p < 0.05) decrease in SOD activity relative to untreated, based on preventive and after induction approaches.
Effect of chrysin on redox-sensitive biomarkers on sickle erythrocytes induced with 2% metabisulphite.

MDA: malondialdehyde; GSH: glutathione; CAT: catalase; SOD: superoxide dismutase.
a Statistically significant (p < 0.05) when compared with the induced.
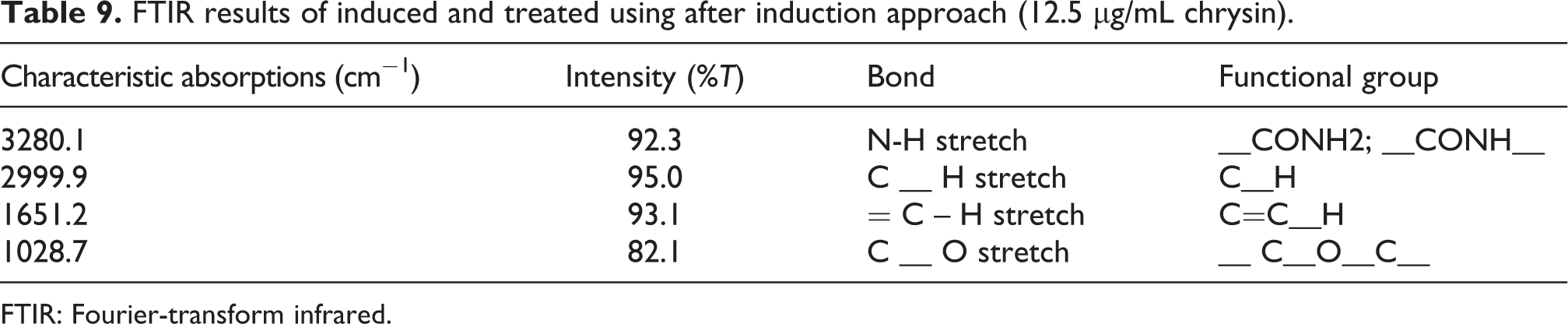
From the FTIR results (Tables 7, 8 and 9), it can be seen that treatment with chrysin was able to favourably alter the functional chemistry judging from shifts (bend and stretches; Figure 5) and functional groups observed relative to induced erythrocytes (Figure 5).

Comparative FTIR peaks between (a) induced, (b) preventive with 2.5µg/mL and (c) after induction with 12.5 µg/mL chrysin. FTIR: Fourier-transform infrared.
FTIR results of induced with 2% metabisulphite/untreated sickle erythrocytes.

FTIR: Fourier-transform infrared.
FTIR results of treated and induced using preventive approach (2.5 µg/mL chrysin).

FTIR: Fourier transform-infrared.
FTIR results of induced and treated using after induction approach (12.5 µg/mL chrysin).
FTIR: Fourier-transform infrared.
Discussion
SCD comprises a group of inborn haemolytic anaemia altogether characterized by the predominance of sickle HbS, which results from a single β-globin gene mutation, leading to an amino acid substitution (Glu→Val, βA→βS) and development of the abnormal HbS tetramer. 24 In deoxygenated states, HbS undergoes rapid intracellular polymerization, which denigrates the erythrocyte membrane and significantly modifies both its shape and flexibility while transporting oxygen to the body. This in turn affects the solubility of deoxygenated haemoglobin exacerbated by higher 2,3-bisphosphoglycerate mutase activity. 10 Similarly, stiff and ‘sickle’ red blood cells have a reduced life cycle and result in both acute and chronic haemolysis. This facilitates vaso-occlusion in which sickle erythrocytes and other circulating cells stick to the vascular endothelium and aggregate together to interrupt blood flow particularly in the small blood vessels, causing hypoxia to tissues and delicate organs. 24 –26
Orthodox drugs developed so far for sickle cell management focus largely on symptomatic respite of pain and crisis mitigation. As a sequel to this, scientists have so far philosophically had a rethink of a paradigm shift from synthetic drugs to medicinal plants, which are naturally endowed with bioactive compounds harbouring strong biological activities, including antisickling. 26 Chrysin is one such bioactive compound readily available within and around our microenvironment through consumptions of honey, fruits and vegetables. Likewise, side effects related to orthodox drugs administration, availability and affordability especially in developing nations are some the reported problems of foremost concern. 11 Consequently, this study has made an effort to examine the antisickling effects of chrysin via modulation of deoxy-haemoglobin, 2,3-bisphosphoglycerate mutase, redox homeostasis and alteration of functional chemistry in human sickle erythrocytes using in silico and in vitro models while advocating preventive and after induction approaches for the first time.
Molecular docking study carried out with chrysin on deoxy-haemoglobin and 2,3-bisphophoglycerate mutase indicated good interaction, which was found to effectively bind with the receptors by completely occupying their active sites. Higher levels of deoxy-haemoglobin and 2,3-bisphophoglycerate mutase have been implicated in SCD, 10,26,27 and therefore, targeting them via sequestration as clearly observed in our study could be a viable strategy in combating and managing sickle cell anaemia in addition to providing a clue on the possible antisickling potentials of chrysin (Figure 6). This can be scientifically supported from the facts obtained from our previous study that chrysin indeed had a strong affinity to proteins based on hydrophobic and hydrophilic interactions. 13 These findings agree with similar studies on other flavonoids like rutin and quercetin that exhibited strong affinities to deoxy-haemoglobin and 2,3-bisphophoglycerate mutase. 28,29 By implication, this might have revealed chrysin’s tendency to allosterically bind to haemoglobin with good binding affinity that may potentially alter oxygen’s affinity to haemoglobin towards oxygenation due to supportive potential inhibitory effects on 2,3-bisphophoglycerate mutase by chrysin.

Proposed antisickling mechanism of chrysin.
Interestingly, for the first time, we have been able to demonstrate in this study that chrysin indeed possesses sickling-suppressive property corroborating other findings on the antisickling activities of some flavonoids. 30 The observed antisickling activities were found to be associated with a favourable modulation of osmotic fragility, redox homeostasis and functional chemistry of erythrocytes as clearly seen from our repetitive experiments. This is in addition to the observed decrease on the number of sickle erythrocytes upon treatment with chrysin in vitro, which was a similar observation based on other studies with rutin and quercetin. 28,29
As a sequel to the observed increase in lipid peroxidation and objectionable effects on the part of enzymatic and non-enzymatic antioxidant system vis-à-vis higher haemolysis percentage due to sickling induction, it will be practically impossible to assume that the 3D structure of membrane lipid bilayer and associated macromolecules would not be affected, including their functional chemistry. To this end, the observed alterations of the functional chemistry may potentially be related to membrane stabilizing effects of chrysin in an induced versus treated erythrocytes by virtue of the fact that erythrocytes like other cells are composed of lipids, proteins and carbohydrates peripherally, integral or/and intracellularly. Chrysin has also been reported to have a high bioavailability which enhances its antioxidant effects by virtue of the observed decrease in lipid peroxidation with a concomitant increase in GSH, CAT and SOD, 13,14,31,32 anti-inflammatory and protein stability effects which could be advantageous in combating SCD-associated oxidative stress, inflammation and decreased HbS stability. 14 These may as well mitigate and possibly converse painful vaso-occlusive episodes characterized by endothelial dysfunction and activation of the inflammatory and coagulation pathway recently reported in SCD. 3,11,33 Flavonoids like rutin and quercetin have also been favourable reported with antisickling effects via modulation of sickling active molecules (haemoglobin and 2,3-bisphophoglycerate mutase), modulation of redox homeostasis and biochemical alteration in relation to functional chemistry of sickle erythrocytes. 28,29 Again, these further confirmed the observed antisickling properties of chrysin as clearly demonstrated from our current studies.
Conclusion
From our findings, it can thus be deduced that sickling-suppressive effects of chrysin may be associated with sequestration of deoxy-haemoglobin, 2,3-bisphosphoglycerate mutase, alteration of redox homeostasis and functional chemistry of sickle erythrocytes in vitro. Further researches are needed to quantify the levels of deoxy-haemoglobin, 2,3-bisphosphoglycerate mutase in vitro and also to possibly look at the in vivo contributions of chrysin in SCD models. Comparative studies are also needed for chrysin and hydroxyurea in relation to antisickling effects.
Footnotes
Acknowledgments
The authors are grateful to the advice and assistance of Mrs Zeenat Bello Kudan and Miss Funmi Audu E. from the Department of Biochemistry, Ahmadu Bello University, Zaria, towards the successful completion of this work. The authors equally appreciate Mrs Salamatu Sani of the Department of English and literary studies, Ahmadu Bello University, Zaria for improving the quality of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.