Abstract
Effects of flavonoids quercetin and chrysin on lipid peroxidation and histopathological changes in liver of diabetic mice were studied and compared with the antioxidant and reducing ability of quercetin and chrysin and their ability to chelate Fe2+ ions in vitro. Diabetes was induced in Swiss albino mice with a single intravenous injection of alloxan (75 mg kg−1). Two days after alloxan injection, flavonoid preparations (50 mg kg−1 per day) were given intraperitoneally for 7 days in diabetic mice. The lipid peroxidation was evaluated by measuring the malondialdehyde production using the 2-thiobarbituric acid test. Administration of quercetin and chrysin to diabetic mice resulted in a significant decrease in lipid peroxidation level in liver tissue. Treatment of diabetic mice with flavonoids solutions results in decreased number of vacuolated cells and degree of vacuolization of the liver tissue. The protective role of flavonoids against the reactive oxygen species–induced damages in diabetic mice gives a hope that they may exert similar protective action in humans.
Introduction
Flavonoids are a class of naturally occurring benzo-γ-pyrone derivatives found in fruits and vegetables as well as in food and beverages of plant origin, such as olive oil, tea, and red wine. 1 Individual differences within each subclass are determined by variation in number and arrangement of the hydroxyl groups as well as by the nature and extent of alkylation. They act in plants as antioxidants, antimicrobials, photoreceptors, visual attractors, feeding repellants, and for light screening. The dietary intake of flavonoids is considered important for preventing a wide variety of diseases, including cardiovascular disease, some forms of cancer, hepatic diseases, inflammation and diabetes. All of these disorders involve oxidative stress and lipid peroxidation damage. 2 –4 Most of the effects of flavonoids in prevention and therapy seem to be associated with their potency as antioxidants. The antioxidant activity of flavonoids has been considered to be via two possible ways of action, radical scavenging and metal chelating. Quercetin (3, 5, 7, 39, 49-pentahydroxyflavone), the most common flavonol in diet, 5 prevents oxidant injury and cell death 6 by several mechanisms, such as scavenging oxygen radicals, 7,8 protecting against lipid peroxidation, 9,10 and chelating metal ions. 11 Structural features of quercetin that have been associated with its antioxidant properties include a B-ring catechol group, a 2,3-unsaturated bond conjugated with a 4-oxo group in the C-ring, and functional hydroxyl groups. 12 Chrysin is one of many bioflavonoids with chemopreventive properties in cardiovascular disease and cancer. Chrysin, a dihydroxy flavone has a double bond between C-2 and C-3, and the ring B will be coplanar with the rings A and C due to conjugation. The mechanism of action as antioxidants seems to be associated with hydroxyl groups on A-ring, but the impact of the A-ring arrangement on antioxidant activity is not clear enough. 13
Diabetes mellitus is a disease initially characterized by a hyperglycaemia. The disease is progressive and possibly the world’s fastest growing metabolic disorder. Oxidative stress plays an important role in the aetiology of diabetes mellitus. 14,15 Reactive oxygen species (ROS) generated in diabetes causes oxidative damage, particularly to heart, kidney, eyes, nerves, liver, small and large blood vessels, immunological and gastrointestinal system. 14 –16
Hyperglycaemia not only generates more reactive oxygen metabolites but also attenuates antioxidative mechanisms through nonenzymatic glycosylation of antioxidant enzymes. 14 Hyperglycaemia directly increases hydrogen peroxide production, promotes glycosylation of circulating and cellular protein, and may initiate a series of auto-oxidative reactions that culminate in the formation and accumulation of advanced glycosylation end products (AGEs) in tissue proteins. 17,18 The AGEs have oxidizing potential and can promote tissue damage by free radicals. In addition, increased lipid peroxidation impairs membrane functions by decreasing membrane fluidity and changing the activity of membrane-bound enzymes and receptors. Its products, lipid radicals and lipid peroxides, are harmful to the cells in the body and associated with atherosclerosis and damage to brain, kidney, liver, and other tissues. 19
In this study, we examined quercetin and chrysin for their ability to inhibit lipid peroxidation and histopathological changes in liver caused by alloxan-induced diabetes in mice. Furthermore, the purpose of this work was to elucidate the relationship between differences in their antioxidant activity in vitro (to scavenge free radicals and chelate Fe2+ ions) and ability to inhibit lipid peroxidation in diabetic mice.
Material and methods
Animals
Male CBA inbred mice 2 to 3 months old, weighting 20 to 25 g, obtained from the Department of Animal Physiology, Faculty of Science, University of Zagreb, were used in this study. The animals were kept in individual cages during the experiment and at 12-h light:12-h dark cycle. They were fed a standard laboratory diet (4 RF 21, Mucedola, Settimo Milanese, Italy) and tap water ad libitum. Maintenance and care of all experimental animals were carried out according to the guidelines in force in the Republic of Croatia (Law on the Welfare of Animals, N.N. #19, 1999) and carried out in compliance with the Guide for the Care and Use of Laboratory Animals, DHHS Publ. # (NIH) 86-123. Study was approved by the ethical committee (Faculty of Science, University of Zagreb, Croatia).
Chemicals
Butylated hydroxyanisol (BHA), 2,2-diphenyl-1-picrylhydrazyl (DPPH), β-carotene, Folin–Ciocalteu reagent, linoleic acid, Tween-40 (polyoxyethylene sorbitan monopalmitate), ascorbic acid and gallic acid were purchased from Sigma-Aldrich Chemical Co. (USA). Quercetin (Quercetin dihydrate 98%) and chrysin (5,7-dihydroxyflavone) were purchased from Aldrich Ch. Co. Inc. Milwaukee Wisconsin, USA, and dissolved in saline solution with 0.5% v/v ethanol.
Experimental design
Forty mice were randomly divided into four groups, as follows:
Group (i): control animals (normal, non-diabetic animals); received 0.5 mL saline solution with 0.5% v/v ethanol per day by intraperitoneal (ip) injection for 7 days;
Group (ii): alloxan controls; injected intravenously (iv) with alloxan in a single dose of 75 mg kg−1 body weight; these served as the untreated diabetic group; received 0.5 mL saline solution with 0.5% v/v ethanol per day by ip injection for 7 days;
Group (iii): received chrysin solution ip in a daily dose of 50 mg kg−1 for 7 days starting 2 days after alloxan injection; these served as the chrysin-treated diabetic group.
Group (iv): received quercetin solution ip in a daily dose of 50 mg kg−1 for 7 days starting 2 days after alloxan injection; these served as the quercetin-treated diabetic group.
Four mice from each group were used for the experiments.
Induction of experimental diabetes and determination of serum glucose level
Diabetes was induced in Swiss albino mice by a single iv injection of alloxan monohydrate (75 mg kg−1) in total volume of 0.5 mL of freshly prepared saline solution. Blood glucose level was tested before alloxan injection and 48 h after treatment to monitor the immediate diabetogenesis. After 48 h, the animals with blood glucose level above 20 mmol L−1 were selected for the study (diabetic mice) and then treated with quercetin or chrysin. Blood glucose level was determined by test strips of blood glucose (Betachek Visual blood glucose test strips, Sydney, Australia).
Determination of antioxidant capacity of the extracts
The antioxidant activity of the extracts was evaluated using three different methods. Hence, the antioxidant activity was analyzed as DPPH free radical scavenging activity (RSA), reducing power and the capability to chelate iron (II) ions. DPPH RSA was calculated as the concentration that scavenges 50% of DPPH free radical and has thus RSA = 50% (EC50). Absorbance of the reaction mixture was read spectrophotometrically at 700 nm. Greater absorbance indicated greater reducing power. Chelating activity was expressed as ChEC50, the concentration that chelates 50% of Fe2+ ions present in the solution. Protocols are described in detail in a previous study. 20 Stat Fax 3200 (Awareness Technologies, USA) microplate reader was used for absorbance measurements in studies of antioxidant activity.
Liver harvesting
Four mice from each group were used on the ninth day after alloxan injection. Animals were killed by cervical dislocation. Subsequently the whole liver was harvested and the following variables were analyzed: determination of lipid peroxidation of liver cells and histopathological analysis of liver tissue.
Determination of lipid peroxidation
Samples of liver (100 mg mL−1 buffer) were homogenized in 50 mM phosphate buffer (pH 7.0), and then centrifuged at 15,000g for 15 min; the supernatant was used for biochemical analysis. The extent of lipid peroxidation was determined by the method of Ohkawa et al. 21 using tetramethoxypropane as an external standard. The level of lipid peroxides was expressed as nmoles of malondialdehyde (MDA) formed/mg protein. The protein concentration was determined by the method of Lowry et al. 22 using crystalline bovine serum albumin as standard.
Histopathological analysis
For histopathological analysis, liver tissue from diabetic control mice treated with physiological and ethanolic solution and diabetic mice treated with quercetin and chrysin was fixed in 10% neutral-buffered formalin for 24 h, dehydrated in a graded alcohol series, and after chloroform treatment embedded in Paraplast®. Deparaplasted 5–6 μm thick sections were stained with haematoxylin and eosin (HE) following standard protocol. Stained slides were examined under a light microscope (Nikon Eclipse E600) at 100, 200, 400 and 1000× magnification. Liver sections were examined for vacuolization, lymphocyte infiltrations, necrosis, and apoptosis. The percentage of apoptotic cells was determined by counting 200 cells in randomly selected microscopical fields of vision.
Photomicrographs were taken by digital camera (Nikon DMX1200) and Imaging Software Lucia G 4.80 (Laboratory Imaging Ltd., Prague, Czech Republic).
Statistical analyses
The experiments were performed in triplicate. The results were expressed as mean ± SD. Statistical comparisons were made using one-way ANOVA, followed by Dunnett’s post hoc test for multiple comparisons with the control. Statistical analyses were performed using the JMP V6 from SAS software (SAS Institute, Cary, North Carolina, USA). A value of p < 0.05 was considered to indicate statistical significance.
Results
Antioxidant capacity of the extracts
In this study, chrysin demonstrated notable antiradical activity (Table 1) albeit lower than the activity of BHA (p < 0.0001). On the other hand, quercetin was an extremely effective radical scavenger and its EC50 value was almost 20-fold lower than EC50 of BHA, indicating substantially greater RSA. To determine the reducing power of the investigated flavonoids, quercetin and chrysin solutions were compared to standard antioxidant

Reducing power of the extracts. Values are means ± SD (n = 4).
Radical scavenging activity (EC50) and metal chelating activity (ChEC50) of quercetin and chrysin.

STD: standard; BHA: butylated hydroxyanisol.
a Values are means ± SD (n = 3).
b BHA.
c EDTA.
*Statistically significant difference between sample and standard (**** p < 0.0001).
Effect of flavonoids on lipid peroxidation of liver
The principle of lipid peroxidation is the reaction of MDA, an end product of lipid peroxidation, with thiobarbituric acid (TBA) to form a pink chromogen. The levels of MDA in the liver of untreated diabetic animals and animals treated with solution of flavonoids are presented in Figure 2. The lipid peroxidation in liver tissue was notably decreased in animals treated by quercetin or chrysin (p < 0.001).

Levels of lipid peroxidation in liver and kidney of normal, untreated diabetic mice and diabetic mice treated with quercetin and chrysin. Values are means ± SD (n = 4). (Differences with untreated diabetic mice; **p < 0.01; Student's t test).
Histopatological analysis
In the liver of control diabetic mice treated with an ethanolic solution, the liver cells in half of the samples around the central veins were more vacuolated than the cells around Kiernan’s spaces (Figure 3(a)). Besides the vessels in some Kiernan’s spaces, smaller lymphocyte infiltrations were found. In the second half of the samples, almost all cells were quite vacuolated so there was no difference in the degree of vacuolization between cells in the central and peripheral part of the individual lobules (Figure 3(b)). In these samples, lymphocyte infiltrations were not found.

Liver tissue of diabetic mice stained with haematoxylin and eosin (HE). (a–b) Control mice treated with alcoholic solution. (a) In half of the samples, the liver cells around the central veins (cv) are more vacuolated than the cells around Kiernan’s spaces (Ks). (b) In the other half of the samples, there are no differences in the degree and distribution of vacuolated cells between center and periphery of lobules. Almost all the cells are considerably vacuolated. (c) In mice treated with alcoholic solution of chrysin, liver cells around the central vein (cv) are more vacuolated than the cells around Kiernan’s spaces (Ks), with a lower degree of cell vacuolization compared to diabetic control. (d–f) Mice treated with alcoholic solution of quercetin have fewer vacuolated cells with lower degree of vacuolization compared to diabetic control. With regard to arrangement and degree of vacuolization, there are individual differences: (d) poorly vacuolated cells around the central veins (cv) and non-vacuolated cells around Kiernan’s spaces (Ks), (e) cells with loose cytoplasm around the central veins (cv) and the few vacuolated cells around the Kiernan’s space (Ks) and (f) equal distribution of vacuolated cells throughout the lobules. Bar = 50 µm. Cv: central vein; Ks: Kiernan’s space; L-lymphocyte infiltration in the parenchyma.
In the liver of diabetic mice treated with ethanolic solution of chrysin, in most samples liver cells were less vacuolated compared to controls, but there were differences in their distribution. In one sample, more vacuolated liver cells were found around Kiernan’s spaces, while the other two samples showed strongly vacuolated cells around the central veins (Figure 3(c)). In all samples, a few small lymphocyte infiltrations were found in the parenchyma and besides the blood vessels in some Kiernan’s spaces.
The liver of diabetic mice treated with ethanolic solution of quercetin showed less vacuolated cells and lower degree of vacuolization in relation to diabetic control. However, regarding their distribution there were individual differences. In one sample, vacuolated cells were found around the central vein, while most cells in the area around Kiernan’s spaces were not vacuolated (Figure 3(d)). In the second sample, cells around central veins were slightly larger and had a loose cytoplasm, while a smaller number of cells around the Kiernan’s spaces were more vacuolated (Figure 3(e)). In the third sample, there was no difference in the level of the cell vacuolization between the central and peripheral part of the lobules (Figure 3(f)). In all samples, a few small lymphocyte infiltrations were found in the parenchyma (Figure 3(d)) and beside the vessels in some Kiernan’s spaces.
Apoptotic cells were also found in liver tissues in all the groups. The percentage of apoptotic cells per group is shown in Figure 4. Results show 15% less apoptotic cells in the liver of diabetic mice treated with ethanolic solution of chrysin and about 25% more apoptotic cells in the liver of diabetic mice treated with ethanolic solution of quercetin as compared to diabetic control.
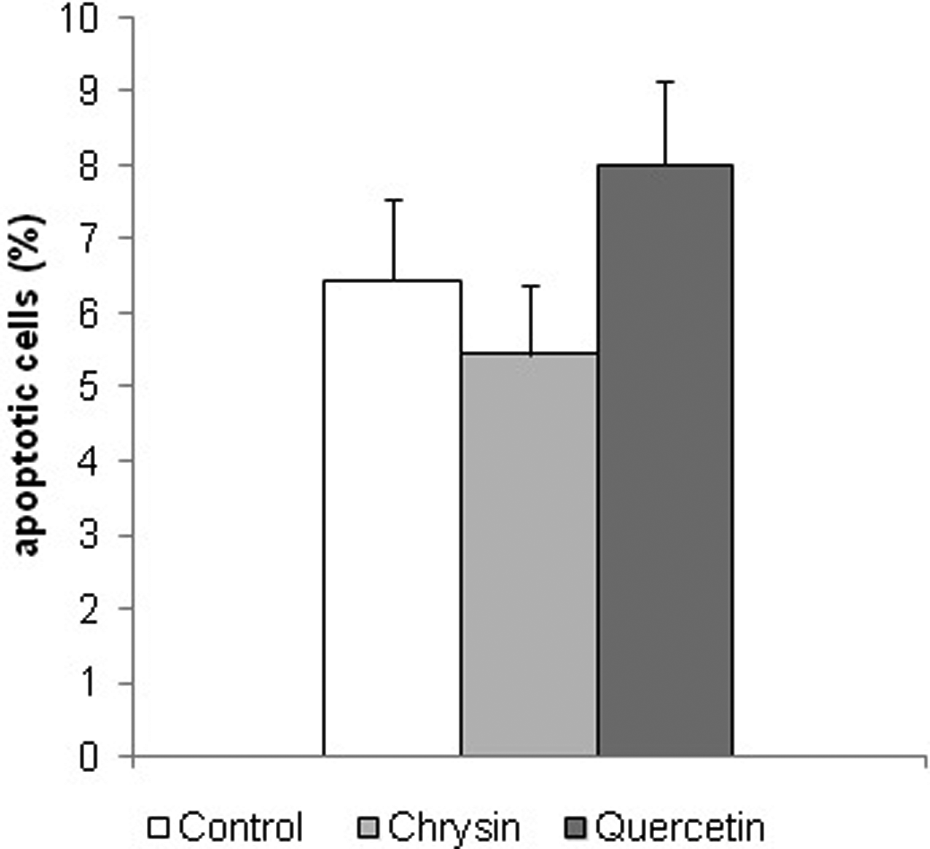
The percentage of apoptotic cells in the liver tissue of a control group of diabetic mice treated only with solvent for flavonoids (control) and diabetic mice treated with alcoholic solution of quercetin or chrysin, (50 mg kg−1). Results are presented as mean ± SD. There is no statistically significant difference between control and chrysin or quercetin treated diabetic mice (t test).
Discussion
Oxidative stress, a condition in which excessive formation of free oxygen radicals and ROS is greater than the ability of the organism to eliminate them, is an essential factor in the aetiology of diabetes. One of the organs most susceptible to oxidative stress in diabetes is liver, which plays a major role in the metabolism of carbohydrates and lipids and other substances. Since, under the pressure of oxidation–reduction imbalance, the body needs help, it is desirable to bring additional quantities of antioxidants into the diet.
The three main proposed mechanisms through which the antioxidants may realize their protective role are the hydrogen atom transfer, single electron transfer, and metal chelation. 23 Consequently, different trends between numerous antioxidant assays described in literature have been found. 24 It is, therefore, pertinent to use several assays instead of a single one to evaluate and compare the antioxidant activity in food and plant extracts. In this study, antioxidant activity of two flavonoids, quercetin and chrysin, was investigated using three assays that cover different aspects of antioxidant activity: DPPH assay estimates the reactivity of hydrogen-donating antioxidants with free radicals, while the reducing power of a compound estimates the ability of a substance, or an extract that contains it, to donate an electron, and thus cause the transformation of free radicals into a less reactive species. Chelating activity measures the ability of a substance to chelate Fe2+ ions that have a pivotal role in the generation of hydroxyl radicals in vivo. In this study, the DPPH antiradical activity of quercetin was higher than the activity of standard antioxidant, BHA. It was found that antiradical activity depends on the presence of a flavonol structure or free hydroxyl group at the C-4′ position. The effect of the 4′-hydroxyl is strongly modified by other structural features, such as the presence of free hydroxyls at C-3 and/or C-3′ and a C2−C3 double bond. 25,26 Since quercetin possesses all of these structural features, its high antiradical activity was expected.
It is known that both quercetin and chrysin can efficiently chelate free ferrous ions. 27 The 3-hydroxy-4-keto conformation together with 2,3-double bond and the catecholic B ring, which could be found in quercetin, is associated with a substantial iron chelation, while the 5-hydroxy-4-keto site, present in chrysin, is less efficient. However, it seems that pH can significantly influence the chelating activity of flavonoids, 28 thus in part accounting for the unexpectedly high chelating activity of chrysin observed in this study.
In this article, the hepatoprotective activities of bioflavonoids were determined in an alloxan-induced diabetic mice model. Our results show that dietary intervention with flavonoids reduces free-radical-induced lipid peroxidation and histopathological changes in liver. Our data of the antioxidant effects of flavonoids (Figure 2) match those of Abo-Salem et al.
29
who reported that propolis, natural material rich in flavonoids such as chrysin and quercetin, can attenuate alloxan-induced hepatotoxicity via decreasing lipid peroxidation. Although hepatoprotective effects of chrysin were observed in
Histological analysis of liver tissue of mice with alloxan-induced diabetes shows more or less vacuolated liver cells, a probable consequence of fat metabolism disorders. Besides vacuolated cells, the lymphocyte infiltrations were also observed suggesting inflammatory processes. Similarly, Ragavan and Krishnakumari 32 described cell vacuolization and focal necrosis in the liver of rats that received single ip alloxan dose of 120 mg kg−1, while Khalil et al. 33 with a higher dose of 150 mg kg−1 also described disorganization of liver parenchyma. Vinagre et al. 34 have found increased hepatocytes with vacuoles, dilated sinusoids and lymphocyte infiltrations in pericentral parts of lobules in diabetic mice treated with 70 mg kg−1 of streptozotocin. The same changes, but in the central part of the individual lobules and fibrosis, are described by Al-Rawi 35 after treatment of rats with a lower dose (50 mg kg−1) of streptozotocin, which is often used to induce diabetes.
Our results show a smaller amount of vacuolated cells with a lower degree of vacuolization in the liver of diabetic mice treated with ethanolic solutions of chrysin and quercetin compared to untreated diabetic mice. Furthermore, although not statistically significant, there is noticeable change in the number of apoptotic cells under the influence of investigated flavonoids. Chrysin treatment resulted in a reduced number of apoptotic cells, whereas treatment of diabetic mice with quercetin resulted in an increased number of apoptotic cells. Increased level of apoptosis in the liver of diabetic animals treated with quercetin is in compliance with the data shown by Iwao and Tsukamoto. 36 They provided evidence that quercetin inhibited DNA synthesis and induced apoptosis are associated with the increases in c-fos mRNA level and the upregulation of p21 mRNA as well as with protein expression during liver regeneration after liver damage. According to the same authors, p21 may play a dual role (contributory to or preventive of apoptotic cell death) depending on the particular stimulus, cell type, growth condition and cellular environment. Other authors 37,38 also suggest that flavonoids may induce cell survival or induce cell death through a cascade of different signaling pathways depending on the concentration. Thus, survival-signaling cascade in many cells involve phosphoinositide 3-kinase (PI-3-kinase), Akt kinase, and also cross-communication between PI-3-kinase and extracellular signal-regulated kinases (ERKs). Because a sustained activation of ERK1/2 is necessary for cell survival and cell proliferation, 37 the inhibition of ERK1/2 by quercetin could contribute to the increased occurrence of cell death. Although special attention must be given to flavonoid concentrations, taking into account the results of histopatological analysis and lipid peroxidation analysis, quercetin may be a potential preventive or therapeutic agent in diabetic mice, and further efforts to investigate these possibilities are needed.
These findings suggest that chrysin and quercetin have a positive effect on disorders of fat metabolism in diabetes.
In conclusion, the administration of natural antioxidants such as quercetin and chrysin mitigates alloxan-induced toxicity by attenuating hepatotoxicity induced by oxidative stress in a diabetic mouse model. The findings of the present study strongly suggest that oxidative stress has a pivotal role in the pathophysiology of diabetic hepatotoxicity. Naturally occurring flavonoids are able to attenuate diabetic hepatic damage, probably through their antioxidative action. Although diabetes cannot be cured, it can be managed very successfully. The best method of prevention and treatment of diabetic complications is efficient treatment of diabetes mellitus, preferably leading to the normalization of glucose and lipid blood levels. Usually this should include diet change. This study implies that flavonoids could be considered dietary supplements in the prevention or treatment of diabetic complications.
Footnotes
Conflict of Interest
The Authors declared no conflict of interest.
Funding
This work was supported by the Ministry of Sciences, Education and Sports of the Republic of Croatia project No. 119-0000000-1255.
