Abstract
Chrysin (CHR) is a food-based bioactive ingredient whereas, sodium arsenite (SA) is one of the major contaminant in drinking water. When ingested, SA contributes to tissue damage due to bioactivation by S-adenosyl methionine (SAM)-dependent methyltransferase. Hence, the needs to nullify this effect by investigating the potentials of CHR on SA-induced genotoxicity in rats. The experiment was divided into two successive stages (ameliorative and preventive, curative studies) for 1 week. Rats were divided into four groups: distilled water, 10mg/kg SA, 10mg/kg CHR and co-administration. In stage 2, the experimental groups were given either CHR or SA for 1 week, and treated in reversed order for additional week. Lipid peroxidation, protein carbonyl and DNA fragmentation in liver, blood brain and bone marrow cells micronucleus were assayed for using standard protocols. Molecular docking of SAM-dependent methyltransferase in the presence of CHR was conducted. CHR significantly (
Introduction
Arsenic is a major environmental toxicant as well as a human carcinogen which is present in large amounts in the environment.
1,2
Arsenicals are found in the environment via industrial, agricultural processes as well as some medical applications,
3
and their consumption through contaminated water is prevalent in many areas of the world.
3,4
They are spontaneously absorbed through gastrointestinal tract after ingestion and distributed in the blood to the liver, kidney, spleen, lung and many other delicate organs.
5
Arsenic metabolism in the liver generates reactive oxygen species (ROS) which include dimethyl arsenic peroxyl ([(CH3)2AsOO]) and dimethyl arsenic radical [(CH3)2As].
6
–9
These reactions are catalysed by a cytosolic enzyme, methyltransferase, which uses
In recent years, there is a tremendous interest in the possible role of nutrition in prevention of oxidative stress-related diseases. 12 Chrysin (CHR) is a natural flavonoid, 15 biologically active compound extracted from many plants, honey 11,16 and propolis which were used as traditional medicine. 17 –19 CHR and its derivatives, in particular, have been reported to possess a broad range of biological activities, including anti-oxidation, anti-viral, anti-inflammatory and anticancer properties. 18,20,21 Previous work from our laboratory on acacia honey and propolis revealed a promising antioxidant property in sodium arsenite (SA)-induced oxidative damage. However, the work done was only on crude acacia honey, which necessitates the emergence of this idea to investigate the role of one of its bioactive component (CHR) and the possible mechanism of action in vivo. The findings from this work may provide an insight into the possible role of CHR in DNA repair or rather in genome protection.
Materials and methods
Chemicals/reagents
CHR (molecular weight, 254.24g/mol; melting point, 284–286°C; purity, 97%) was purchased from Sigma Aldrich St. Louis, Missouri, USA. SA (BDH Chemicals Ltd., Poole, England), thiobarbituric acid (Sigma Aldrich St. Louis, Missouri, USA), Giemsa (Sigma Aldrich St. Louis, Missouri, USA), phosphate buffer saline (PBS) (Sigma Aldrich St. Louis, Missouri, USA), 2, 4-dinitrophenylhydrazine (KEM light laboratories, Mumbai, India) were also purchased. All other chemicals and reagents used were of analytical grade.
Experimental animals and design
Thirty-five male Wistar albino rats (110–190 g) were used in the present study. They were allowed to adapt for 1 week under standard laboratory conditions of 12 h light–dark cycle before commencement of all experiments that lasted for 2 weeks. The rats were maintained in the animal house of Biochemistry Department, Ahmadu Bello University, Zaria, with protocol for the study approved by Ahmadu Bello University Animal Right Review Committee. They were randomly grouped into seven groups of five rats each. All the treatments were orally administered since arsenicals in the environment are majorly ingested through drinking water, and duration of exposure was selected based on our previous works. 3,8,22 However, it is paramount to note that the experiment was carried out in two stages using oral administrations for 2 weeks, the ameliorative stage (stage 1, first week) and the preventive and curative stage (stage 2, extended to second week) as shown in the experimental design below.
Stage 1 (Ameliorative Study) – sacrificed after first week
Stage 2 (Preventive and Curative Studies) – extended to second week
Group 1b: Distilled water Group 5: 10mg/kg bw CHR for one week followed by 10mg/kg bw SA for another week Group 6: 10mg/kg bw SA for one week followed by 10mg/kg bw CHR for another week
Collection of samples
After 24 h of last administration, the rats were kept without food overnight and sacrificed by decapitation following mild chloroform anaesthesia based on American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals, 2013 Edition, which reported that, any effect as a result of chloroform will be neutral since all the animals were subjected under same treatment.
29
About 2 to 3 mls of blood was collected by cardiac puncture. Equally, brain, liver and femurs were also collected. The tissues were homogenized in 1:10 of 0.1 M phosphate buffer. The supernatant (brain and liver) was collected after centrifugation at 3500×
Determination of lipid peroxidation
Lipid peroxidation was determined using thiobarbituric acid according to Ohkawa 30 with slight modification. Exactly 2 ml of 15% trichloroacetic acid (TCA) was added into a test tube, then 2 ml of thiobarbituric acid followed by 100 µl of the tissue homogenate. The mixture was incubated at 80oC in water bath for 30 minutes and allowed to cool for some time. This was followed by centrifugation at 1500 ×g for 10 minutes. Absorbance at 535 nm was taken using spectrophotometer and the amount of malondialdehyde (MDA) was calculated using a molar extinction coefficient of 1.56x10 5 /M/cm. The results were presented in nmol/L.
Determination of protein carbonyl
Carbonyl content was determined by a simplified alkaline method based on the reaction of protein carbonyls with dinitrophenylhydrazine forming dinitrophenylhydrazone, a yellow compound by Dixon. 31 A 400 µL portion of 2, 4 dinitrophenylhydrazine (10 mM in 2.5 M HCl) was added to 400 µL of sample and incubated in dark at room temperature for 10 min; 200 µL of NaOH (6 M) was then added and shaken mildly. Absorbance was read at 450 nm using microplate reader and molar absorption coefficient of 22,000 M-1 cm-1 was used to quantify the concentration of protein carbonyls. The results were presented in nmol/mg of protein.
DNA fragmentation assay
Quantitative DNA fragmentation assay was carried out according to the method of Sellins.
32
The tissue was broken using homogenizer in 0.1 M phosphate buffer (pH 7.4). The homogenate was centrifuged at several steps (200×

where
Bone marrow cells micronucleus assay
Presence of micronuclei was determined as described by Gonza. 33 Bone marrow cells were aspirated from femur and the smears was prepared on slides, fixed in methanol and stained with 5% Giemsa stain solution. The smear was analysed under oil immersion using light microscope (100 M). The slides were scored for the presence of micronucleated polychromatic erythrocytes (mPCEs) by examining a total of 2000 erythrocytes per animal; that is, 500 erythrocytes were examined per field in accordance with Organization for Economic Co-operation and Development (OECD) procedure 2014.
Molecular modelling of SAM-dependent methyltransferase
The structure of CHR (Figure 1) was sketched using ChemBioDraw program, imported into Discovery Studio 2.5v (Accelrys Inc., San Diego, California, USA) and optimized with a Chemistry at Harvard Macromolecular Mechanics (CHARMm) force field, while their partial charges were calculated via the Momany–Rone option
34,35
using smart minimizer algorithm. The X-ray diffraction crystal structure of human SAM-dependent methyltransferase
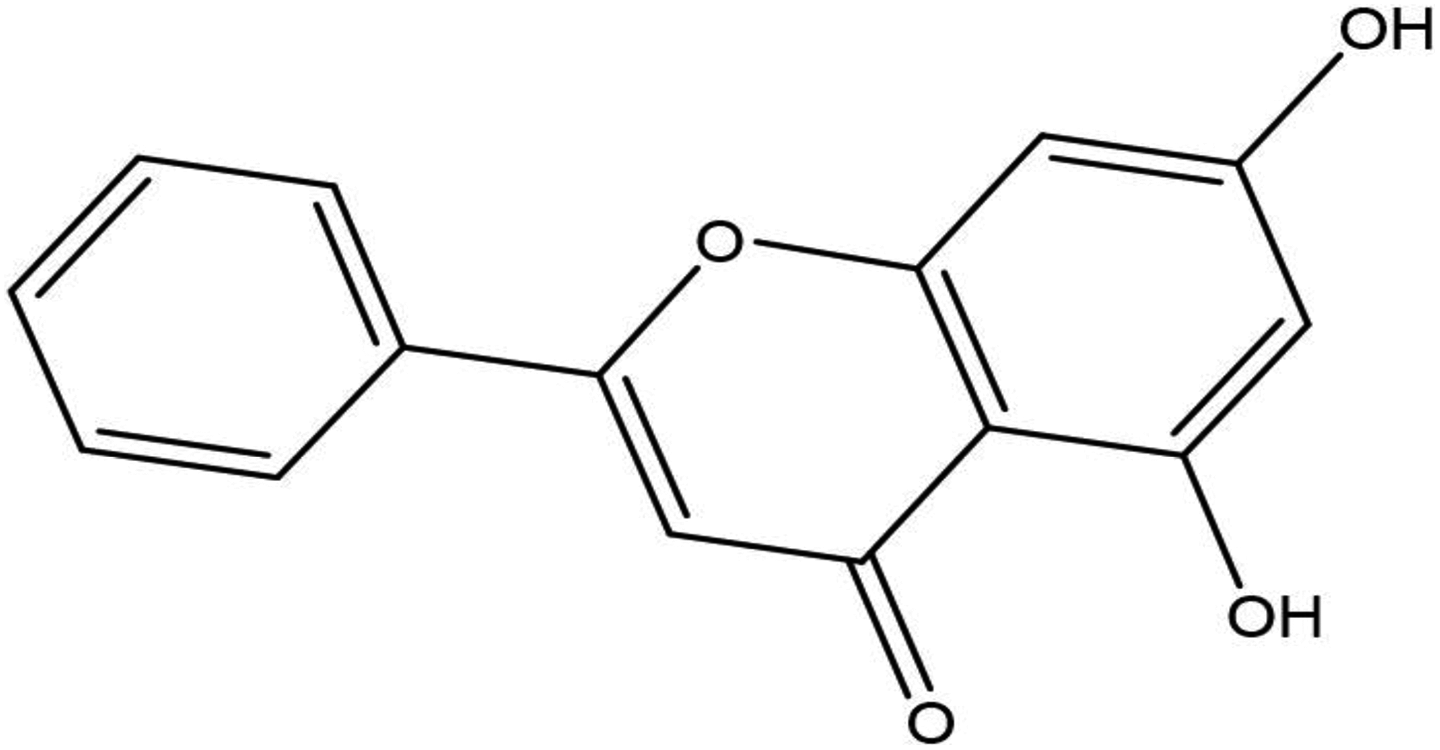
Structure of CHR. CHR: chrysin.
Statistical analysis
The experiment was carried-out in triplicate (independently) and the results were presented as mean ± SD. The data were analysed using one-way analysis of variance (ANOVA) with the aid of Statistical Package for Social Sciences (SPSS) software, SPSS Inc., Chicago, Standard Version 20.0.
Results
Effect of CHR on SA-induced lipid peroxidation
As shown in Tables 1 and 2, a significant (
Antioxidant and DNA protective effects of CHR against SA-induced damage to the liver, blood and brain of male Wister albino rats for 1-week treatment.
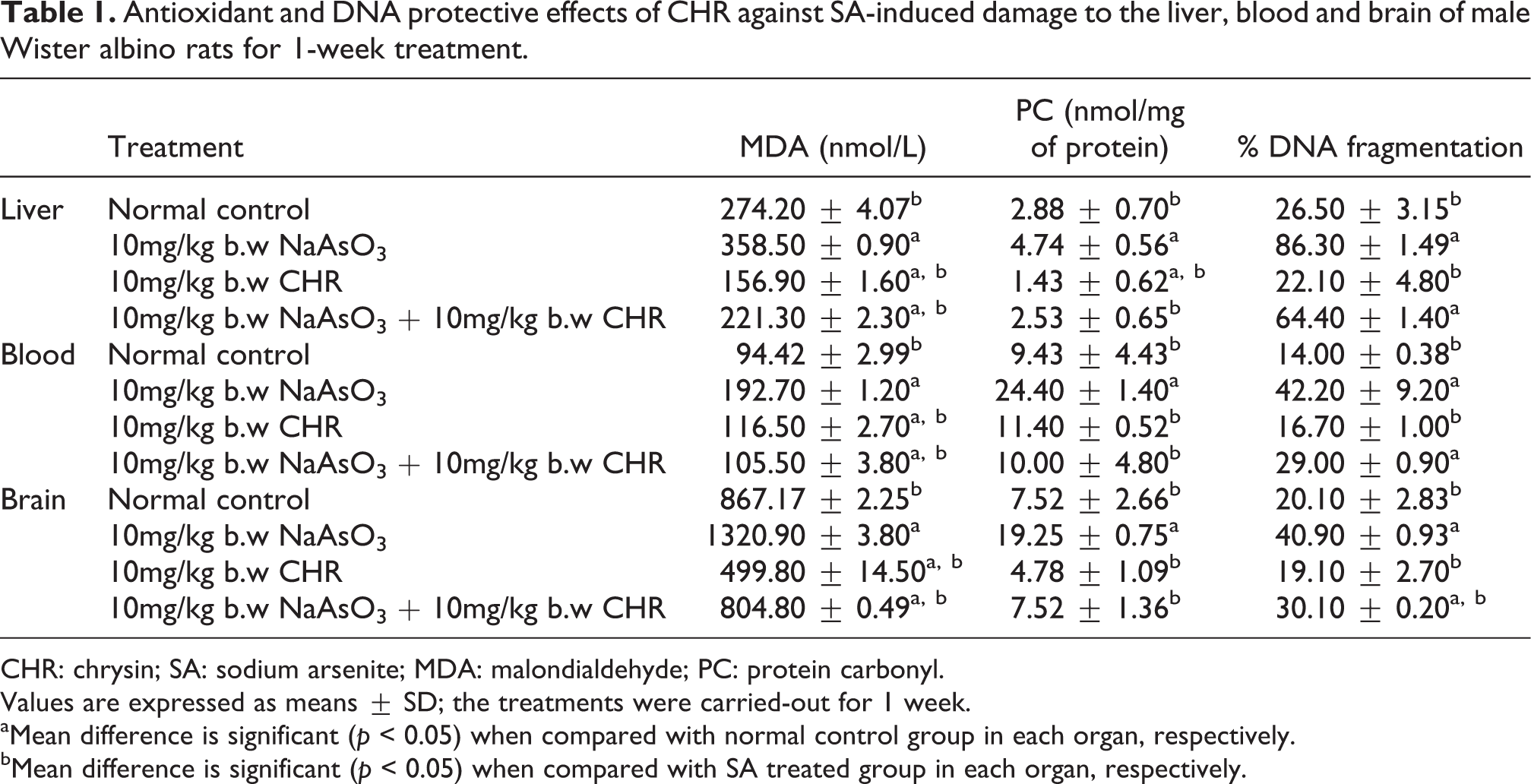
CHR: chrysin; SA: sodium arsenite; MDA: malondialdehyde; PC: protein carbonyl.
Values are expressed as means ± SD; the treatments were carried-out for 1 week.
aMean difference is significant (
bMean difference is significant (
Antioxidant and DNA protective effects of CHR against SA-induced damage to the liver, blood and brain of male Wister albino rats for 2-week treatment.

CHR: chrysin; SA: sodium arsenite; MDA: malondialdehyde; PC: protein carbonyl.
Values are expressed as means ± SD, CHR (first week) followed by SA (second week) and vice-versa to access the preventive and curative effect of CHR against arsenite-induced toxicity.
aMean difference is significant (
bMean difference is significant (
Effect of CHR on SA-induced protein damage
Tables 1 and 2 show a significant (
Effect of CHR on SA-induced DNA fragmentation
A significant (
Effect of CHR on SA-induced chromosomal damage
The clastogenic effect of SA was observed to be significantly (

Effect of CHR against SA-induced chromosomal damage in bone marrow cells of male Wistar albino rats. Values are expressed as Means ± SD; a Mean difference is significant (

Photomicrograph showing the presence of mPCEs in bone marrow cells of male Wistar albino rats. Magnification ×630; 5% Giemsa stain, arrow pointing mPCEs and ABCD indicating normal control,10mg/kg b.w SA, 10mg/kg b.w chrysin, 10mg/kg b.w chrysin + 10mg/kg b.w SA, respectively. The treatment was carried out for 1 week. mPCEs: micronucleated polychromatic erythrocytes; SA: sodium arsenite; b.w: body weight.

Photomicrograph showing the presence of mPCEs in bone marrow cells of male Wistar albino rats. Magnification ×630; 5% Giemsa stain, arrow pointing mPCEs and ABC indicating normal control, 10mg/kg b.w chrysin (first week) followed by 10mg/kg b.w SA (second week), 10mg/kg b.w SA (first week) followed with 10mg/kg b.w chrysin (second week) to access the preventive and curative effect of chrysin against arsenite-induced toxicity. mPCEs: micronucleated polychromatic erythrocytes; SA: sodium arsenite; b.w: body weight.
In silico inhibitory potentials of CHR on SAM-dependent methyltransferase
To validate the preceding observed effects of CHR from in vivo findings, we employed the use of molecular modelling strategies. To this end, CHR structure was docked into the binding site of 2B9E SAM-dependent methyltransferase and the optimal conformation was identified (Figure 5) via molecular modelling. The amino acids interacting with the ligand vis-à-vis the hydrophobicity were also determined (Figures 6(a) and 7(a)). Similarly, the two-dimensional view of the hydrogen bond and hydrophobic interactions of 2B9E-CHR complex were shown in Figures 6(b) and 7(b), respectively. It was revealed that the binding pocket of 2B9E-CHR complex was made of Lys240, Ser215, Asp212, Gln211, Leu210, Cys369, Asn239 of SAM-dependent methyltransferases. The binding energy of CHR was found to have a high binding affinity for the receptor 2B9E, which was given as −24.81 kcal/mol and predicted inhibition kinetic constant (Ki) of 0.959 µM.

Predicted structure of the best docking pose conformation of CHR on 2B9E protein. CHR: chrysin.

Illustration of the interacting amino acids within the active site of SAM-dependent methyltransferase 2B9. (a) Chrysin with surrounding amino acids of 2B9E. (b) Hydrogen bond interaction between chrysin and amino acid within 2B9E active site. SAM:

Illustration of hydrophobic interaction between CHR and 2B9E active site. (a) Chrysin with 4NEV (hydrophobicity surface) at the active binding site, in which the brown and blue regions represent oxygen and nitrogen atoms, respectively, whereas white regions indicate carbon or hydrogen atoms. (b) Two-dimensional illustration of the hydrophobic interaction between the binding modes of 2B9E-chrysin. CHR: chrysin; SAM:
Discussion
SA when ingested is bio-transformed to mono and dimethyl arsenic by SAM-dependent methyltransferase, which are more polar and easily excreted. 37 –39 However, when the level of dimethyl arsenic becomes high, it spontaneously reacts with superoxide radical to generate dimethyl arsenic peroxyl radical that can negatively affect the antioxidant system via a covalent binding to thiol group in proteins. This leads to generation of ROS that can directly affect protein and DNA or indirectly via lipid peroxidation, peptide crosslinking initiating chromosomal and DNA damage with fatal metabolic consequences. 8
SA was found to significantly (
Data from our study show a significant increase (
In this communication, SA led to significant (
Oxidative products of lipid peroxidation such as MDA, acrolein can initiate the formation of DNA adducts thereby exacerbating chromosomal breakage and micronuclei formation. Level of chromosomal damage has been reported in this study based on the frequency of micronuclei in PCEs. We have clearly demonstrated that SA treatment significantly (
Bearing in mind the metabolic role of SAM-dependent methyltransferase in facilitating the systemic toxicities associated with arsenicals, it can thus be hypothetically deduced that modulation of this enzyme could serve a viable strategy in combating arsenicals-induced toxicities. 37 –39 Based on the validation studies using in silico approach, CHR was able to comfortably dock into the active site of SAM-dependent methyltransferase via strong hydrogen bond and hydrophobic interactions. This of course agrees with the observations of Ramesh, 46 where strong hydrogen bonds were formed between the hydroxyl groups on CHR with polar amino acid residues in the binding pocket of some proteins in addition to the strong hydrophobic interactions observed. Inhibition of this enzyme by some bioactive food component such as CHR could be reasonable, thus decrease in generation of reactive methylated arsenic species responsible in arsenic-induced oxidative damage observed in this study. Although, in vivo inhibition analysis of CHR on this enzyme need to be confirmed. Overall, the in silico findings might further explain the observed ameliorative, preventive and curative roles of CHR against SA-induced chromosomal and DNA damage in the present study.
Conclusion
Findings from this study revealed that CHR possibly confers both ameliorative, preventive and curative role on SA-induced chromosomal and DNA damage in male Wistar albino rats, which might be due to inhibition of SAM-dependent methyltransferase. We, therefore, recommend further work to ascertain the effects of CHR on SAM-dependent methyltransferase in vivo.
Footnotes
Acknowledgements
We are grateful to the advice and assistance of Dr M Bisallah, Dr YY Pala from the Department of Veterinary Microbiology and Pathology and Mal Yau Dary from the Department of Pharmacology, Ahmadu Bello University, Zaria, towards the successful completion of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
