Abstract
The antioxidative effect of Monodora myristica seed acetone extract and its effect on chemical functional groups were investigated in sickled erythrocytes as well as molecular modeling of the antisickling potentials of its secondary metabolites. The extract was subjected to gas chromatography–mass spectrometry to identify the compounds present, which were then docked into the allosteric-binding site of deoxy-hemoglobin. The extract was incubated with sickled erythrocytes at 37°C for 6, 12, and 24 h and were subjected to antioxidative analysis for reduced glutathione (GSH), superoxide dismutase (SOD), catalase, and lipid peroxidation (LPO). Chemical functional group of the treated cells was analyzed via Fourier transform infrared spectroscopy (FTIR). The predominant compounds identified were 17-octadecynoic acid; oleic acid, androstan-3-one, 17-hydroxy-2-methyl- (2.beta.,5.beta.,17.beta.)-; estran-3-one, 17-(acetyloxy)-2-methyl-, (2.alpha., 5.alpha., 17.beta.), and (+)-3-carene, 10-(acetylmethyl)-. They all fitted well within the active site of Hb with good binding affinity, as evidenced by the negative CDocker interaction energies of their complexes ranging between −54.4 and −26.7 kcal/mol. Treatment with the extract exacerbated SOD and catalase activities as well as GSH level, while LPO was suppressed. This antioxidative activity was time and/or dose dependent, with 6 and 12 h incubation showing the optimum activity. FTIR analysis of the treated cells showed the presence of hydrophobic functional groups. The synergetic molecular interaction of the major compounds of the extract with the α-dimer of Hb depicts an antisickling effect of M. myristica acetone extract. This is accompanied by exacerbation of endogenous antioxidant enzymes activity and modification of the functional chemistry of the cells.
Introduction
Sickle cell disease (SCD) remains one of the most prevalent inherited hemoglobinopathy among Africans, with sub-Saharan Africa being the most hit. 1 It occurs due to a single mutation in the β-globin chain, causing valine to be substituted with glutamic acid at the sixth amino acid position, 2 thereby leading to the production of abnormal hemoglobin (HbS). Several studies have linked the pathogenesis of SCD to polymerization of HbS, which causes the erythrocyte to possess a crescent- or sickle-shape structure. 2,3 This triggers the release of hemoglobin (Hb) and iron into the plasma. The SCD is often associated with painful vaso-occlusive episodes characterized by endothelial dysfunction and activation of the inflammatory and coagulation pathway. 4 This has been attributed to repeated polymerization of HbS. The HbS undergoes recurrent auto-oxidation inducing the formation of methemoglobin and superoxide (O2−). 2,5 The formed O2− dismutates to hydrogen peroxide (H2O2), which accumulates and reacts with methemoglobin to yield hydroxyl radical (•OH). 6 These reactions cause an alteration in the reactive oxygen species and antioxidants balance. This imbalance has been described as oxidative stress and has been implicated in the secondary complications of SCD. 2,7
There have been increasing interest in the use of natural products in the treatment and management of several diseases. This paradigm shift can be explained by the high cost of most synthesized drugs coupled to the poor purchasing power of most Third World citizens. The side effects of these drugs is also a major concern to health practitioners. Several medicinal plants have been used in folk medicine for the treatment and management of SCD and its complications. 3,8 One of which is Monodora myristica. Commonly known as African nutmeg, M. myristica is a perennial plant of the Annanacea family found in the forests of West Africa. 9 The seeds are mostly used as flavoring condiment in food 10 and have been reported to be rich in dietary minerals. 11 Erukainure et al. 12 reported the ability of the seed extract to scavenge free radicals in vitro, which can be attributed to its reported phytochemicals constituents comprising alkaloids, glycosides, flavonoids, tannins, saponin, and steroids. 13 Essential oils from the seeds and leaves have been reported to contain α-phellandrene, α-pinene, myrcene, limonene, β-caryophyllene, and α-humulene. 14 The seed powder is used as stimulant for relieving constipation and control passive uterine hemorrhage after childbirth in women. 15 Nwozo et al. 16 reported the ability of the seed extracts to lower cholesterol and attenuate lipid peroxidation (LPO), thus indicating an antihyperlipidemic potency. Although Uwakwe and Nwaoguikpe 17 reported the antisickling properties of the seeds and their ability to reduce Fe3+ to Fe2+, its effect on chemical functional groups of sickled erythrocytes are still unknown.
Hence, the aims of the present study are to investigate the antioxidative effect of M. myristica seed extract in SCD and its effect on the chemical functional groups of sickle erythrocytes as well as molecular modeling of the antisickling potentials of its compounds.
Materials and methods
M. myristica seeds were purchased from a local market in Benin City, Nigeria. The seeds were sorted and air-dried. It was then dehulled and blended to dry powder before storage in air-tight container until further analysis.
The powdered sample (100 g) was defatted by soaking in n-hexane for 72 h. The hexane extract was decanted leaving behind the defatted sample, which was resoaked in acetone for 72 h. The acetone extract was decanted and concentrated in vacou at 20°C to yield 3.81 g concentrate which was then stored in vial bottles at −20°C until further analysis.
Gas chromatography–mass spectrometry (GC-MS) instrumentation
The extract was subjected to GC-MS analysis to identify the presence of secondary metabolites. Shimadzu GC-MS QP2010 ULTRA (Labstock Nigeria ltd) with column: Optima 5MS (5% diphenyl and 95% dimetylpolysiloxane); length: 30 m; thickness: 0.25 µm; ID: 0.25 mm; column flow: 1.34 mL/min; gas: helium was used for GC-MS analysis. Flow rate was set at 1.34 mL/min, and temperature of column maintained at 60°C for 2 min and then raised to 200°C (15°C/min) followed by 9 min at 280°C (5°C/min). The National Institute of Standards and Technology mass spectral program 2011 was used for mass spectral survey. The concentrations of the identified compounds were calculated using area normalization over flame ionization detector response method.
Molecular docking
In order to substantiate the antisickling effect of the acetone extract, we docked the five major compounds (17-octadecynoic acid (
The three-dimensional (3D) conformers of each metabolite (
To check the ability of docking protocol to reproduce experimental binding mode, the co-crystallized ligand (L35) was redocked into the binding groove of Hb using default parameters in CDocker. The computed root-mean-square deviation (RMSD) (all atoms) between the predicted pose of L35 and its X-ray structure was less than 1.5 Å and validated the docking procedure. Same docking procedure was subsequently used to dock our compounds (
Evaluation of extracts for redox activities in sickled erythrocytes
Blood collection
Blood of 4–5 mL was obtained by venipuncture from each of five confirmed male sicklers not in crises at the medical center, Federal Institute of Industrial Research, Lagos, Nigeria. They were of age between 21 and 24 years. They were notified and gave their consent prior to the experiment. Blood was collected in sodium ethylenediaminetetraacetic acid bottles, and the content thoroughly mixed by gently rolling the bottle.
All experiments were carried out under the ethical approval of the Federal Institute of Industrial Research, Lagos, Nigeria, in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Extraction of erythrocytes
The obtained homozygous HbS samples were centrifuged at 3000 r/min for 10 min in room temperature, the plasma was discarded to obtain the erythrocytes. The erythrocytes were suspended in phosphate buffer saline (PBS), centrifuged and the supernatant discarded. This was repeated thrice, after which the washed erythrocytes were resuspended in PBS and used for subsequent analysis.
Redox analysis
Different concentrations of aqueous extract (25, 50, 75, and 100 µg/mL) was incubated with 100 µL sickled erythrocytes for 6, 12, and 24 h at 37°C, as described by Muhammad et al. 20 After incubation, the cells were centrifuged and the supernatant assayed for reduced glutathione (GSH), 21 LPO, 22 SOD, 23 and catalase activities. 24
Fourier transform infrared (FTIR) spectroscopy analysis of treated sickled erythrocytes
The sample extract (100 µg/mL) was incubated with 100 µL sickled erythrocytes for 24 h at 37°C, as described by Muhammad et al. 20 After treatment, the samples were dried under pressure and sandwiched between potassium bromide (KBr) cells. They were then scanned on FTIR spectrophotometer at room temperature (25°C–28°C) at 300–4500 cm−1 spectral range. The peak heights were used in determining the functional group present by comparing to the IR spectroscopy correlation table.
Statistical analysis
Results were expressed as mean ± standard deviation. One-way analysis of variance was used in analyzing the differences between the groups with the aid of Statistical Package for Social Sciences (SPSS) software (standard version 10.0.1; SPSS Inc., Chicago, USA). The p values <0.05 were considered statistically significant for differences in mean using the least of significance difference.
Results
The GC-MS analysis of the acetone extract of M. myristica seeds revealed high concentrations of plant steroids notably androstan-3-one, 17-hydroxy-2-methyl- (2.beta.,5.beta.,17.beta.) and estran-3-one 17-(acetyloxy)-2-methyl- (2.alpha.,5.alpha.,17.beta.) as depicted in Figure 1 and Table 1. 17-Octadecynoic acid was the most predominant compound.

GC-MS spectra of acetone extract of Monodora myristica seeds. GC-MS: gas chromatography–mass spectrometry.
Identified compounds in acetone extract of Monodora myristica seeds.

Docking analysis revealed that all compounds fitted well within the active site of Hb with good binding affinity (Figure 2a to e), as evidenced by the negative CDIEs of their complexes ranging between −54.4 and −26.7 kcal/mol. The lowest negative CDIE obtained in the program indicates the strongest interaction of the ligand with the protein and vice versa. Accordingly, compound

Binding orientation of 17-octadecynoic acid (a), oleic acid (b), androstan-3-one, 17-hydroxy-2-methyl- (2.beta.,5.beta.,17.beta.) (c), estran-3-one, 17-(acetyloxy)-2-methyl (2.alpha.,5.alpha.,17.beta.) (d), and (+)-3-carene, 10-(acetylmethyl) (e) with human Hb (pdb id: 2D60). Ligands are depicted using blue lines, whereas the Hb amino acids are shown using line format. Hb: hemoglobin; PDB: protein data bank.
The visualization of the three-dimensional (3D) complexes (Figure 2a to e) further revealed the predominance of hydrophobic interactions in their host–guest relationship. Compound
The cyclic compound
Treatment with the extract led to significant (p < 0.05) increase in GSH level compared to the untreated sickled erythrocytes although the increase was inversed to increasing concentration, as depicted in Figure 3. The level was however observed to decrease with hours of incubations, with the least level observed after 24-h incubation. GSH level of sickle erythrocytes incubated with the standard antisickling drug (control) peaked after 12-h incubation.

GSH levels of untreated and treated sickled erythrocytes at different time intervals. Values are presented as mean + SD; n = 3. *Statistically significant compared to the untreated sickled erythrocytes (p < 0.05; SPSS for Windows, version 10.0.1). GSH: reduced glutathione; SD: standard deviation.
The SOD activities of the treated sickled erythrocytes (p < 0.05) increased significantly, as shown in Figure 4. The incubation time had little or no effect on the observed activity.

SOD activities of untreated and treated sickled erythrocytes at different time intervals. Values are presented as mean + SD; n = 3. *Statistically significant compared to the untreated sickled erythrocytes (p < 0.05; SPSS for Windows, version 10.0.1). SOD: superoxide dismutase; SD: standard deviation.
Treatment of sickle erythrocytes with the extract caused a significant (p < 0.05) increase in catalase activities, as shown in Figure 5. The activities were observed to increase with increasing concentration, indicating a dose-dependent effect. However, the increasing activities were inverse to incubation time, thus suggesting a time-dependent effect.

Catalase activities of untreated and treated sickled erythrocytes at different time intervals. Values are presented as mean + SD; n = 3. *Statistically significant compared to the untreated sickled erythrocytes (p < 0.05; SPSS for Windows, version 10.0.1). SD: standard deviation.
A significant decrease in malondialdehyde (MDA) levels was observed in sickle cells treated with the extract in a dose-dependent manner, as depicted in Figure 6. However, the levels increased significantly (p < 0.05) with increasing incubation time, peaking at 12-h incubation. Treatment with 25 µg/mL had the lowest MDA levels for all incubating time compared to the untreated sickled erythrocytes, suggesting it to be the most active. Decreased MDA levels indicates reduced LPO.
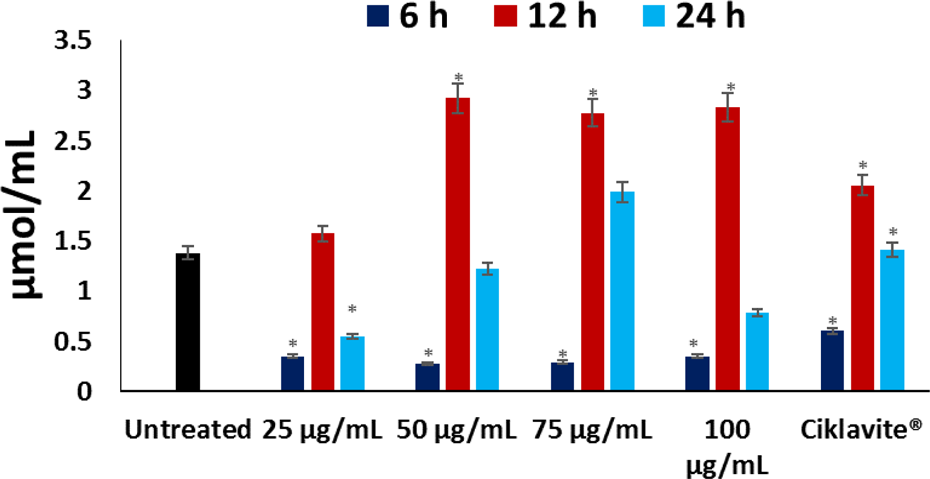
MDA levels of untreated and treated sickled erythrocytes at different time intervals. Values are presented as mean + SD; n = 3. *Statistically significant compared to the untreated sickled erythrocytes (p < 0.05; SPSS for Windows, version 10.0.1). MDA: malondialdehyde; SD: standard deviation.
The FTIR spectroscopy analysis of the untreated sickled erythrocytes revealed major stretches at 3435.17, 2065.73, 1634.44, and 543.05 cm−1, as depicted in Figure 7 and Table 2. Based on the IR spectroscopy library, these shifts were identified to represent –CONH2; –CONH–, C=C=N–, –NH2; –NH3 +, and C–Cl as the chemical functional groups. However, analysis of the treated cells showed 3283.25, 2925.46, 2854.45, 1633.97, 1539.48, 1454.58, 1239.83, and 1161.80 cm−1. These shifts based on the IR spectroscopy library indicates –CONH2; –CONH–, C–H; –CO2 H, =C–H, –NH2; –NH3 +, N–N=O, O–NO2, and –C–O–C– as the chemical functional groups present (Figure 7 and Table 3).

FTIR spectroscopy of untreated and treated sickled erythrocytes. FTIR: Fourier-transform infrared.
FTIR spectroscopy of sickle erythrocytes.

FTIR: Fourier-transform infrared.
FTIR spectroscopy of treated sickle erythrocytes.

FTIR: Fourier-transform infrared.
Discussion
The increasing paradigm shift from synthetic drugs to medicinal plants have been attributed to little or no side effects associated with their intake as well as their affordability especially in developing countries. The prevalence of SCD remains a burden in most developing African nations where there is little or no economic growth, making medical care unaffordable. From years immemorial, several medicinal plants have been used in the treatment and management of the disease. Although antisickling properties of some of these plants have been reported in the literatures, 25,26 no studies were done at deeper level targeting novel drug discovery. The choice of solvent used in the extraction of phytochemicals has been a major concern owing to toxic effects. Low acute and chronic toxicity as well as no carcinogenic effect have been associated with acetone ingestion and/or inhalation. 27 In this study, the effect of the acetone extract of M. myristica on redox imbalance in sickle erythrocytes as well as its functional chemistry was investigated, and the antisickling properties of identified secondary metabolites of the extract were molecularly modeled.
The motivation behind docking simulations was to check if the metabolites have tendency to bind with Hb and to predict their binding modes and key amino acids of Hb responsible for their host–guest relationship. The binding affinity of Hb for oxygen can be modulated using different synthetic allosteric effectors and has drawn the attention of researchers across the world for the development of drugs against several diseases including SCD.
28
Benzafibrate analogues such as L35, 2-[4-(3,5-dichlorophenylureido) phenoxy]-2-methylpropionic acid are potent Hb effectors that manipulate the oxygen binding affinity of Hb without switching protein to high-affinity state (T) through allosteric inhibition.
29,30
The docking studies revealed that all compounds (
GSH has long been recognized as an endogenous antioxidant and plays an important role in the maintenance of the cell redox potential, thus protecting the cell against oxidative stress. 30 Several studies have reported depleted levels of GSH in sickle erythrocytes compared to normal erythrocytes, owing to dysregulation of glutathione metabolism with concomitant depletion of erythrocyte glutamine level, 31,32 thus indicating an increased oxidative state in sickle erythrocytes. This corresponded to the low GSH level observed in the untreated erythrocytes (Figure 3). The significantly (p < 0.05) elevated levels after treatment indicates an antioxidant protective effect of the acetone extract against free radicals in sickled erythrocytes.
Owing to auto-oxidation, sickled erythrocytes increasingly generates significant amounts of O2 −, H2O2, and •OH, overwhelming the cells’ SOD and catalase antioxidant systems, 31 thereby causing an imbalance in the redox potential of the cell. This is reflected in the reduced SOD and catalase activities in the untreated sickled erythrocytes (Figures 4 and 5), implying an occurrence of oxidative stress. The SOD activities in SCD remains controversial, as some studies have implicated an increase as a defense response mechanism to the increased oxidative stress and may be responsible for H2O2 accumulation owing to O2 − dismutation. 31,33,34 This however contradicts the report by Schacter et al. 35 that decreased SOD activities is associated with SCD. Catalase neutralizes H2O2 to H2O and O2, and its reduced activity has been reported in SCD. 33,36 Thus, the observed significant (p < 0.05) increased activities of SOD and catalase (Figures 4 and 5) on treatment with M. myristica acetone extract further indicates its antioxidative potentials.
Elevated levels of MDA has been reported in SCD, indicating LPO. 32 This elevation has been attributed to the increased generation of O2 − with concomitant accumulation of H2O2. 2,35 The H2O2 reacts with methemoglobin which degrades Hb to release Fe3+. The H2O2 further reacts with Fe3+ via the Fenton reaction to produce •OH which attacks the membrane lipids, initiating a peroxidative reaction. 6 This corresponds to the high MDA level in the untreated sickle erythrocytes (Figure 6), indicating an occurrence of oxidative stress as also reflected by the depleted GSH and reduced antioxidant enzymes (Figures 3 to 5). The observed significantly (p < 0.05) decreased MDA level in the treated cells indicates an antiperoxidative effect of the extract. This observation correlates with the increased GSH levels and catalase activity, indicating an inverse relationship.
The above mentioned antioxidative properties can be attributed to the synergetic effect of the chemical constituents of the acetone extracts (Figure 1 and Table 1). Their medicinal properties are well-documented especially for plant steroids (androstan-3-one, 17-hydroxy-2-methyl, (2.beta.,5.beta.,17.beta.), estran-3-one 17-(acetyloxy)-2-methyl- (2.alpha.,5.alpha.,17.beta.), and terpenes ((+)-3-carene 10-(acetylmethyl)-) which are major constituents of most essential oils with reported antioxidant properties. 37,38 Erukainure et al. 39 reported that oleic acid is the major constituent of fatty acids that showed antioxidative effect in human breast cancer cells. The 17-octadecynoic acid which is the predominant constituent of the M. myristica acetone extract is a potent inhibitor of cytochrome P450 metabolism of arachidonic acid, 40,41 thus preventing an occurrence of LPO.
The FTIR analysis of the sickle erythrocytes revealed polar charged amines as the primary functional groups (Figure 7 and Table 2), indicating fewer negative surface charges than normal erythrocytes. The role of negative surface charge of erythrocytes on erythrocyte–erythrocyte interactions has been reported as well as its influence on interactions with the vascular endothelium and other circulating blood cells. 42,43 These polar charged functional groups may be responsible for the abnormal adherence of sickle erythrocytes to vascular endothelial cells, thus eliciting painful vaso-occlusive event. 44 The formation of hydrophobic functional groups (Figure 7 and Table 3) on treatment with the acetone extract depicts a change in the polar surface charge of the sickled erythrocytes. Thus implying a potential of the extract to suppress the occurrence of a vaso-occlusive episode by attenuating abnormal adherence of sickle erythrocytes to vascular endothelial cells.
Polymerization of sickled erythrocytes leads to an imbalance in redox homeostasis of the cells and alteration of the functional chemistry which in turn causes vaso-occlusion. Increased occurrence of vaso-occlusive episode also leads to the generation of free radicals, thus triggering a redox–vaso-occulation cycle. The acetone extract of M. myristica may thus bring about antisickling in SCD by (1) retaining disc shape in sickled erythrocytes with no effect on the T state, (2) suppression of oxidative stress by increasing the activities and level of endogenous antioxidant enzymes and GSH respectively, and (3) modification of sickled erythrocyte chemical functional groups which may suppress adherence, thus arresting the occurrence of a vaso-occlusive episode (Figure 8).

Proposed antisickling mechanism of Monodora myristica acetone extract.
Conclusion
The results of this study indicate that the antisickling activity of M. myristica acetone extract can be attributed to the synergetic molecular interaction of the major compounds of the extract with the α-dimer of Hb. This is accompanied by exacerbation of endogenous antioxidant enzyme activity and modification of the functional chemistry of the sickled cells.
Footnotes
Acknowledgments
The authors are grateful to the Centre for High-Performance Computing, an initiative supported by the Department of Science and Technology of South Africa, for usage of software and cluster support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
