Abstract
In order to study the effects of chitinase-like protein YKL-40 on proliferation, apoptosis, and migration of human bronchial epithelial cell line (BEAS-2B), and the underlying mechanisms, we cultured BEAS-2B alone or with different concentrations of YKL-40. thiazolyl blue tetrazolium bromide (MTT) assay was used to examine the cell proliferation. Annexin V-fluorescein isothiocyanate isomer (FITC)/propidium iodide staining and scratch assay were performed to test the cell apoptosis and migration. The concentrations of transforming growth factor-β1 (TGF-β1), Smad3, Smad7, alpha-smooth muscle actin (α-SMA), interleukin-4 (IL-4), IL-6, and IL-8 in the cell culture supernatant were detected by enzyme-linked immunosorbent assay. The messenger RNA and protein levels of YKL-40, TGF-β1, Smad3, Smad7, and α-SMA were detected by reverse transcription polymerase chain reaction and Western blot. BEAS-2B cells cultured with different concentrations of YKL-40 showed significantly higher cell proliferation and migration and inflammatory cytokines compared with that of control group, while the cell apoptosis was significantly lower than that of control group (p < 0.05). In addition, BEAS-2B cells cultured with YKL-40 had increased TGF-β1, Smad3, Smad7, and α-SMA levels in the supernatant, compared with that of BEAS-2B cells cultured alone (p < 0.05). Furthermore, LY364947, as TGF-β1/Smads signaling pathway inhibitor, decreased cell proliferation and migration ability and enhanced cell apoptosis of BEAS-2B cells compared with control group (p < 0.05). However, YKL-40 administration reversed the effect of LY364947 on the biological behavior of BEAS-2B cells. YKL-40 could affect the biological behaviors of BEAS-2B cells, which might be related to the TGF-β1/Smads pathway.
Introduction
Allergic asthma is a chronic respiratory disease with airway hyperresponsiveness and mucus hypersecretion. 1,2 The airway epithelium cells, as the first barrier to the pathogens, are important for the initiation for development of airway inflammation. 3,4 There are several different human bronchial epithelial cell culture models to evaluate the biological behavior and function of airway epithelium cells in vitro. 5 Thus, it is critical to understand the function and the correlation of the epithelium relevant to asthma, which might provide new insight for allergic asthma treatment.
YKL-40, a chitinase-like glycoprotein, has been reported to be associated with severe asthma, lung function impairment. 6 In asthma, the expression of YKL-40 was high in serum of patients with irreversible airway obstruction or with severe exacerbations. 7 In these patients, four clusters of YKL-40 were identified, and C3, C4 showed activation of non-type 2 inflammatory pathways. 7 Another study also showed that YKL-40 was significantly elevated in serum of patients with asthma and chronic obstructive pulmonary disease compared to healthy controls. 8 However, little is known regarding the functional role of YKL-40 in human bronchial epithelial cell proliferation and molecular mechanisms.
Transforming growth factor-β (TGF-β) plays its biological function by activating the downstream Smad signaling pathway. As the most abundant isoform, TGF-β1 can be infiltrated inflammatory cells. 9 TGF-β1 extensively stimulates the phosphorylation of Smad2 and Smad3, and then activated Smad2 and Smad3 bond with Smad4 to form oligomeric complexes. 10 TGF-β1/Smads pathway plays many essential roles in organ fibrosis, which is associated with a gradual destruction of normal tissue architecture and function. 11 Previous studies revealed that TGF-β1/Smads pathway was activated in the airways of children with asthma, and the TGF-β1 levels were correlated with the number of sputum eosinophils and neutrophils. 12 In airway remodeling mice, TGF-β1, Smads, and αSMA expression are increased to reduce airway inflammatory response. 13 More and more evidences have shown that YKL-40 is involved in asthma development. YKL-40 concentration was increased in asthmatic serum and correlated with asthma severity. YKL-40 was reported to induce interleukin-8 (IL-8) expression from bronchial epithelium and regulate smooth muscle proliferation and migration via mitogen-activated protein kinase and nuclear factor kappa B pathway. 14 Another group showed that YKL-40 induced the release of IL-8 via protein kinase C and signal-regulated kinase pathways. 15 However, whether YKL-40 directly functions on bronchial epithelium cells and the signaling pathways involved are still illustrated.
Materials and methods
Cell culture and treatment
According to previous reports, 16,17 human bronchial epithelial cell line (BEAS-2B) was purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and cultured in Roswell Park Memorial Institute (RPMI)-1640 medium (Gibco, Rockville, Maryland, USA) supplemented with 10% fetal bovine serum (Sigma-Aldrich, St. Louis, Missouri, USA) at 37°C in a 5% CO2 incubator (Thermo Fisher Scientific, Waltham, Massachusetts, USA). Cells were treated as shown in Table 1. According to previous study, we used 10 mmol/L LY364947 to inhibitor TGF-β1. 18 In the study, the YKL-40 was obtained from eBioscience (San Diego, California, USA).
The experimental groups.

MTT assay
1 × 104 cells were seeded in a 96-well flat-bottomed plate (100 μL per well with 6 duplicate wells). Cells were incubated with 20 μL of 5 mg/mL MTT (R&D, Minneapolis, MN, USA) for 4 h, and then the supernatant was carefully discarded. dimethyl sulfoxide (DMSO) (200 μL) (Solarbio, Minneapolis, MN, USA) was added to each well, and the mixture was evenly blown. The absorbance (OD value) at 490 nm was measured by a microplate reader, and the proliferation of cells in each group was evaluated.
Apoptosis analysis
Apoptosis rates were assessed using the Annexin V-FITC/Propidium Iodide (PI) Apoptosis Detection Kit (eBioscience). Briefly, different treated cells were washed twice with precooled phosphate-buffered saline (PBS) at 4°C. 1× 106 cells were resuspended in 250 μL of 1× binding buffer and incubated with 5 μL of FITC-labeled Annexin-V and 10 μL of PI solution at a concentration of 20 μg/mL for 10 min at room temperature in the dark. Apoptosis rates were analyzed in a flow cytometer (Beckman Coulter, Brea, California, USA).
Wound-healing assay
3 × 105 cells/well were seeded in a six-well plate and cultured until the cells reached 90% confluence. Artificial wounds were prepared by scarping using a sterile10 μL pipette tip. The streaked cells were washed with PBS to remove cell debris. Cell migration was observed under an inverted microscope (Olympus, Japan) and photographed at 0 h and 24 h, respectively. The distance between the two edges of a wound was calculated using Image J software (NIH).
Enzyme-linked immunosorbent assay
In order to measure the contents of TGF-β1, Smad3, Smad7, α-SMA, IL-4, IL-8, and IL-6 secreted by BEAS-2B cells after different concentrations of YKL-40 treatment, cell supernatants were collected and centrifuged. Fifty microliters of each sample were applied to an enzyme-linked immunosorbent assay 96-well plate to detect different proteins using following antibodies: TGF-β1 (ab100647; Abcam, UK), Smad3 (ab186038; Abcam), Smad7 (Bs-0566p, Bioss, Guangzhou, China), α-SMA (YM-S1139; Yuanmu Biological Tech, China), IL-4 (PD4050; R&D systems), IL-8 (PD8000C; R&D systems), IL-6 (PD6050; R&D systems).
Reverse transcription polymerase chain reaction
Total RNA was isolated from cell lysate using TRIzol (Invitrogen, Carlsbad, California, USA). Two micrograms of total RNA were subjected to reverse transcription, and the synthesized cDNA was used for real-time quantitative polymerase chain reaction with SYBR Green I Supermix (Takara, Dalian, China) according to the manufacturer’s protocol. The results were analyzed using 2ΔΔCT. All experiments were repeated three times. The primer sequences of TGF-β1, Smad3, Smad7, and α-SMA are as follows. TGFβ1 Forward: 5′-CTAATGGTGGAAACCCACAACG-3′; TGFβ1 Reverse: 5′-TATCGCCAGGAATTGTTGCTG-3′; Smad3 Forward: 5′-CCAGTGCTACCTCCAGTGTT-3′; Smad3 Reverse: 5′-CTCGGTGTCGCTAGTTTCTC-3′; Smad7 Forward: 5′-GGCTTTCAGATTCCCAACTTC-3′; Smad7 Reverse: 5′-CGCCATCCACTTCCCTTGT-3′; α-SMA Forward: 5′-GTTTCTCGCACGTCTCCTCT-3′; α-SMA Reverse: 5′-CAGGCAGTTCGTAGCTCTTC-3′.
Western blot
Cells were collected, and the total protein was extracted from cell lysate using radio immunoprecipitation assay (RIPA) lysis buffer. Protein concentration was measured by the bicinchoninic acid (BCA) Protein Assay Kit (Pierce, Waltham, Massachusetts, USA). Equal amounts of samples (40 μg) were loaded, separated by dodecyl sulfate, sodium salt -polyacrylamide gel electrophoresis (SDS-PAGE) gels (Bio-Rad, Hercules, California, USA), transferred to a polyvinylidene fluoride (PVDF) membrane (Millipore, Burlington, Massachusetts, USA). The membranes were blocked at room temperature for 1 h with 5% nonfat milk in tris-buffered saline and tween-20 (TBST) buffer, and the following primary antibodies were added. Mouse anti-TGF-β1 antibody (ab92486, 1:1000; Abcam), rabbit anti-Smad3 antibody (ab40854, 1:1000; Abcam), rabbit anti-Smad7 antibody (ab90086, 1:1000; Abcam), mouse anti-α-SMA antibody (A5228, 1:1000; Sigma), mouse anti-β-actin antibody (ab8226, 1:1000; Abcam). After overnight incubation at 4°C, the membrane was washed by TBST and further incubated with the secondary anti-goat anti-rabbit IgG-horseradish peroxidase (HRP) (1:2000, ab6721; Abcam) for 2 h at room temperature. enhanced chemiluminescence (ECL) method was used for detection, and gray scale scanning and quantification were performed by Image J software (NIH). Protein expression levels were normalized to β-actin.
Statistical analysis
The data were analyzed by SPSS 19.0 statistical software. Results were expressed as mean ± standard deviation. Data analysis between multiple groups was based on one-way analysis of variance (ANOVA), subsequent analysis using the least significant difference test. Before ANOVA was performed, Levene test was used to analyze homogeneity of variance. A value of p <0.05 indicated a significant difference.
Results
YKL-40 promotes the proliferation and inflammatory cytokines of human bronchial epithelial cell line BEAS-2B
As shown in Figure 1(a), after different concentration of YKL-40 treatment, the cell proliferation of BEAS-2B cells was enhanced compared with that of control group (p < 0.05). Cells treated with 0.1 μg/mL YKL-40 had the most significant promotion of cell growth among YKL-40 treatment groups. Meanwhile, we also observed that 0.1 μg/mL YKL-40 treatment had a time-dependent effect on cell proliferation with highest cell proliferation at 48 h (p < 0.05). The data also showed there was no toxicity of YKL-40 in BEAS-2B cells (Figure 1(b)). Further, the secretion of IL-4, IL-8, and IL-6 was obviously increased in BEAS-2B cells after different concentration of YKL-40 administration when contrasted to control group (p < 0.05) (Figure 1(b) to (d)). The levels of IL-4, IL-8, and IL-6 were highest when YKL-40 concentration was 0.1 μg/mL. Similarly, the production of IL-4, IL-8, and IL-6 was analyzed at different time points with 0.1 μg/mL YKL-40 treatment. The data showed the levels of IL-4, IL-8, and IL-6 increased with prolonged time.

YKL-40 treatment promotes the proliferation and inflammatory cytokines of BEAS-2B cells. (a) BEAS-2B cells were treated with different concentrations of YKL-40 (0, 0.01, 0.1, 1, and 10 μg/mL). (b) After 6, 12, 24, 48, and 72 h, the survival rate of BEAS-2B cells was analyzed by MTT assay. (c to e) The expression of IL-4, IL-8, and IL-6 was analyzed by ELISA after treatment of different concentration of YKL-40 (0, 0.01, 0.1, 1, and 10 μg/mL), different time point (0, 6, 12, 24, 48 h) with YKL-40 stimulation. Values are mean ± standard deviation, n = 6. *p < 0.05 versus control group; # p < 0.05 versus 0.01 μg/mL YKL-40 group; ^ p < 0.05 versus 0.1 μg/mL YKL-40 group; % p < 0.05 versus 1 μg/mL YKL-40 group. # p < 0.05 versus 6 h; ^ p < 0.05 versus 12 h; % p < 0.05 versus 24 h. ELISA: enzyme-linked immunosorbent assay; IL: interleukin.
YKL-40 inhibits cell apoptosis of BEAS-2B cells
To explore the influence of YKL-40 on cell apoptosis, BEAS-2B cells were cultured with different concentration of YKL-40 (Figure 2(a)). Compared with control group, YKL-40 treatment significantly reduced cell apoptosis rate (p < 0.05). The inhibitory effect was most obvious with 0.1 μg/mL YKL-40 concentration. We also stimulated BEAS-2B cells with 0.1 μg/mL YKL-40 and analyzed the cell apoptosis rate at different time points (Figure 2(b)). Compared with control group, YKL-40 treatment significantly inhibited BEAS-2B cell apoptosis. Interestingly, YKL-40 inhibited the apoptosis of BEAS-2B in a time-dependent manner.

YKL-40 inhibits apoptosis in BEAS-2B cells. (a) The apoptosis of BEAS-2B cells was analyzed by flow cytometry after treatment with different concentrations of YKL-40 for 48 h. *p < 0.05 versus control group; # p < 0.05 versus 0.01 μg/mL YKL-40 group; ^ p < 0.05 versus 0.1 μg/mL YKL-40 group; % p < 0.05 versus 1 μg/mL YKL-40 group. (b) The apoptosis of cells cultured alone (control) or with 0.1 μg/mL YKL-40 for 6 h, 12 h, 24 h, or 48 h, respectively. *p < 0.05 versus control group; # p < 0.05 versus 6 h; ^ p < 0.05 versus 12 h; % p < 0.05 versus 24 h. Values are mean ± standard deviation, n = 6.
YKL-40 promotes migration of BEAS-2B cells
As shown in Figure 3(a), compared with control group, different concentration of YKL-40 treatment significantly enhanced BEAS-2B cells migration rate (p < 0.05). BEAS-2B cells treated with 0.1 μg/mL YKL-4 showed the highest migration rate among groups (Figure 3(a)). We also stimulated BEAS-2B cells with 0.1 μg/mL YKL-40 and analyzed the cell migration rate at different time points (Figure 3(b)). YKL-40 treatment significantly increased BEAS-2B cell migration at different time points compared with that of control group (p < 0.05).

YKL-40 promotes migration of BEAS-2B cells. (a) Effect of different concentrations of YKL-40 treatment on the migration ability of BEAS-2B cells. *p < 0.05 versus control group; # p < 0.05 versus 0.01 μg/mL YKL-40 group; ^ p < 0.05 versus 0.1 μg/mL YKL-40 group; % p < 0.05 versus 1 μg/mL YKL-40 group. (b) Effect of different treatment time of 0.1 μg/mL YKL-40 on the migration ability of BEAS-2B cells. *p < 0.05 versus control group; # p < 0.05 versus 6 h; ^ p < 0.05 versus 12 h; % p < 0.05 versus 24 h. Values are mean ± standard deviation, n = 6.
YKL-40 induces expression of TGF-β1, Smad3, Smad7, and α-SMA in TGF-β1/Smads signaling pathway
As shown in Figure 4(a), compared with control group, the expression of TGF-β1, Smad3, Smad7, and α-SMA was significantly increased after stimulation with different concentrations of YKL-40, with the highest levels of expression when cells were stimulated by 0.1 μg/mL YKL-40. In Figure 4(b), 0.1 μg/mL YKL-40 treatment significantly increased TGF-β1, Smad3, Smad7, and α-SMA expression in BEAS-2B cell at different time points compared with that of control group (p < 0.05). Consistently, the messenger RNA (mRNA) levels and protein levels of TGF-β1, Smad3, Smad7, and α-SMA in cultured BEAS-2B cells with YKL-40 stimulation were significantly enhanced compared with those of control group (p < 0.05), while 0.1 μg/mL YKL-40 stimulation induced highest mRNA levels and protein levels in BEAS-2B cells (Figures 5(a) and 6(a)). Furthermore, we also investigated the expression of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cells cultured with 0.1 μg/mL YKL-40 for different times (Figures 5(b) and 6(b)). The results suggested that YKL-40 could induce TGF-β1, Smad3, Smad7, and α-SMA expression in the TGF-β1/Smads pathway in a time-dependent manner in BEAS-2B cells.

ELISA detection of TGF-β1, Smad3, Smad7, and α-SMA expression in cell supernatants. (a) Effects of different concentrations of YKL-40 stimulation on the expression of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cell culture supernatant. *p < 0.05 versus control group; # p < 0.05 versus 0.01 μg/mL YKL-40 group; ^ p < 0.05 versus 0.1 μg/mL YKL-40 group; % p < 0.05 versus 1 μg/mL YKL-40 group. (b) BEAS-2B cells were treated with 0.1 μg/mL YKL-40 for different time on the expression of TGF-β1, Smad3, Smad7, and α-SMA expression in the supernatant. *p < 0.05 versus control group; # p < 0.05 versus 6 h; ^ p < 0.05 versus 12 h; % p < 0.05 versus 24 h. Values are mean ± standard deviation, n = 6. ELISA: enzyme-linked immunosorbent assay; TGF-β1: transforming growth factor-β1.

RT-PCR detection of TGF-β1, Smad3, Smad7, and α-SMA mRNA expression in BEAS-2B cells. (a) Effects of different concentrations of YKL-40 stimulation on the mRNA levels of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cells. *p < 0.05 versus control group; # p < 0.05 versus 0.01 μg/mL YKL-40 group; ^ p < 0.05 versus 0.1 μg/mL YKL-40 group; % p < 0.05 versus 1 μg/mL YKL-40 group. (b) BEAS-2B cells were treated with 0.1 μg/mL YKL-40 for different time and the mRNA levels of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cells were examined by RT-PCR. *p < 0.05 versus control group; # p < 0.05 versus 6 h; ^ p < 0.05 versus 12 h; % p < 0.05 versus 24 h. Values are mean ± standard deviation, n = 6. TGF-β1: transforming growth factor-β1; mRNA: messenger RNA; RT-PCR: reverse transcription polymerase chain reaction.
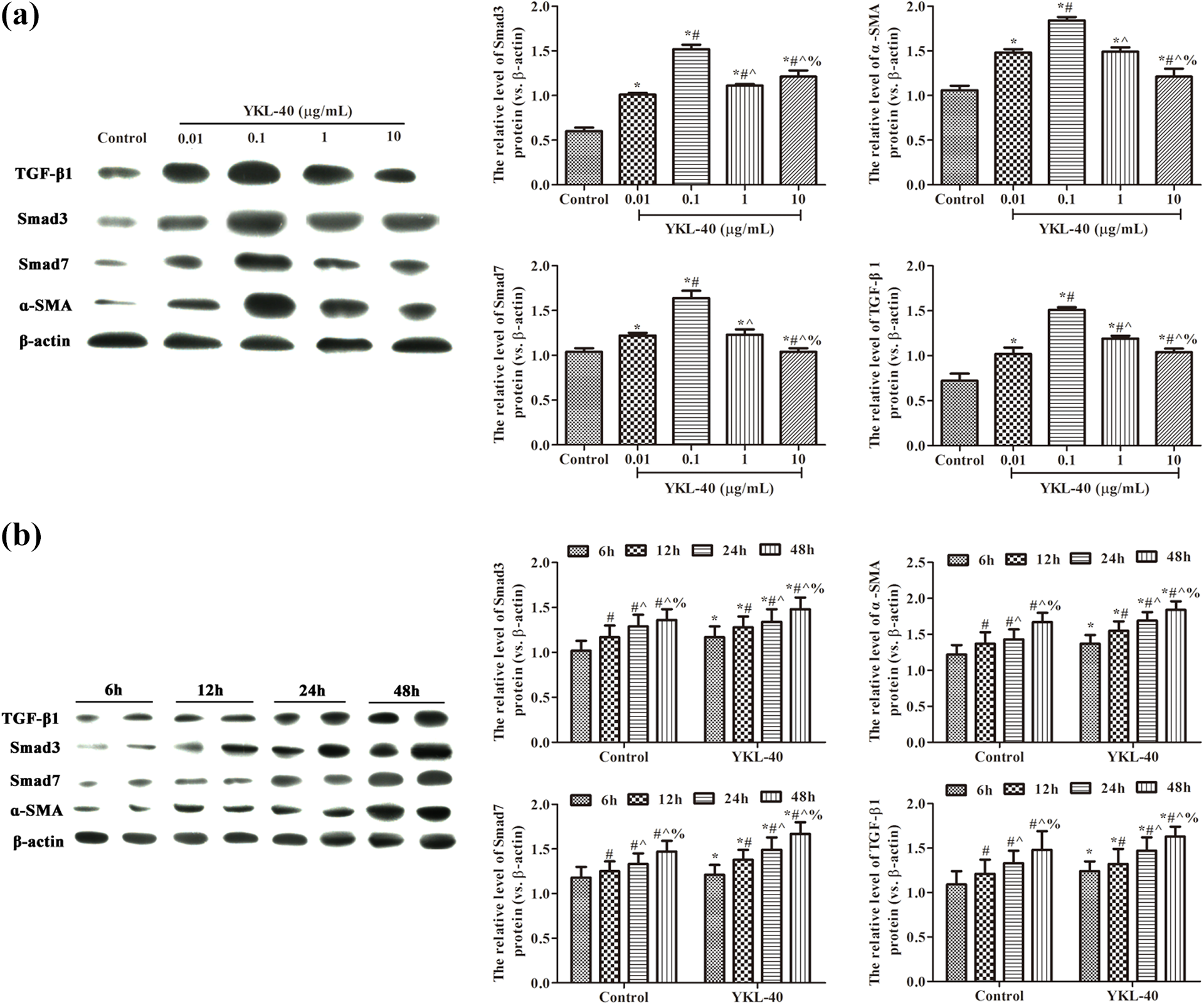
TGF-β1, Smad3, Smad7, and α-SMA protein expression in BEAS-2B cells analyzed by Western blot. (a) Effects of different concentrations of YKL-40 stimulation on the protein expression of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cells. *p < 0.05 versus control group; # p < 0.05 versus 0.01 μg/mL YKL-40 group; ^ p < 0.05 versus 0.1 μg/mL YKL-40 group; % p < 0.05 versus 1 μg/mL YKL-40 group. (b) BEAS-2B cells were treated with 0.1 μg/mL YKL-40 for different time and the protein expression of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cells were examined by Western blot. *p < 0.05 versus control group; # p < 0.05 versus 6 h; ^ p < 0.05 versus 12 h; % p < 0.05 versus 24 h. β-actin was used as an internal reference. Values are mean ± standard deviation, n = 6. TGF-β1: transforming growth factor-β1.
YKL-40 promotes cell proliferation/migration and inhibits apoptosis through the TGF-β1/Smads signaling pathway
To clarify whether YKL-40 functions directly on TGF-β1/Smads signaling pathway, we used 0.1 μg/mL YKL-40 and TGF-β1 inhibitor LY364947 to further study. As shown in Figure 7(a), compared with control group, the proliferation of BEAS-2B cells treated with LY364947 was dramatically inhibited (p < 0.05). However, BEAS-2B cells treated with LY364947 combined with YKL-40 had mild decrease of cell proliferation. Consistently, compared with control group, LY364947 treatment increased cell apoptosis and inhibited cell migration ability (Figure 7(b) to (d)) significantly (p < 0.05). YKL-40 treatment could reverse the function effect of LY364947 treatment on BEAS-2B cells, with less cell apoptosis and higher cell migration rate. The effect of LY364947 on IL-8, IL-4, and IL-6 was observed (Figure 7(e)), the results showed the production of IL-8, IL-4, and IL-6 was obviously suppressed when contrasted to control group (p < 0.05). YKL-40 treatment reversed the function of LY364947.

YKL-40 promotes cell proliferation/migration and inhibits apoptosis through the TGF-β1/Smads signaling pathway. BEAS-2B cells were culture alone, treated with 10 mol/L LY364947, or treated with 10 mmol/L LY364947 and 0.1 μg/mL YKL-40 for 48 h. (a) Cell proliferation was tested with MTT assay. (b and c) Flow cytometry to examine the cell apoptosis in different groups. (d) Scratch test to detect the cell migration. (e) ELISA analysis to test IL-8, IL-4, and IL-6. Values are mean ± standard deviation, n = 6. *p < 0.05. TGF-β1: transforming growth factor-β1; ELISA: enzyme-linked immunosorbent assay; IL: interleukin.
We also investigated the TGF-β1/Smads pathway-associated proteins in BEAS-2B cells, as shown in Figure 8. Compared with control group, BEAS-2B cells treated with LY364947 had decreased expression levels of TGF-β1, Smad3, Smad7, and α-SMA in cell culture supernatant (Figure 8(a)), mRNA levels (Figure 8(b)), and protein levels (Figure 8(c)) in BEAS-2B cells, and YKL-40 could reverse the inhibitory function of LY364947 on TGF-β1/Smads signaling pathway.
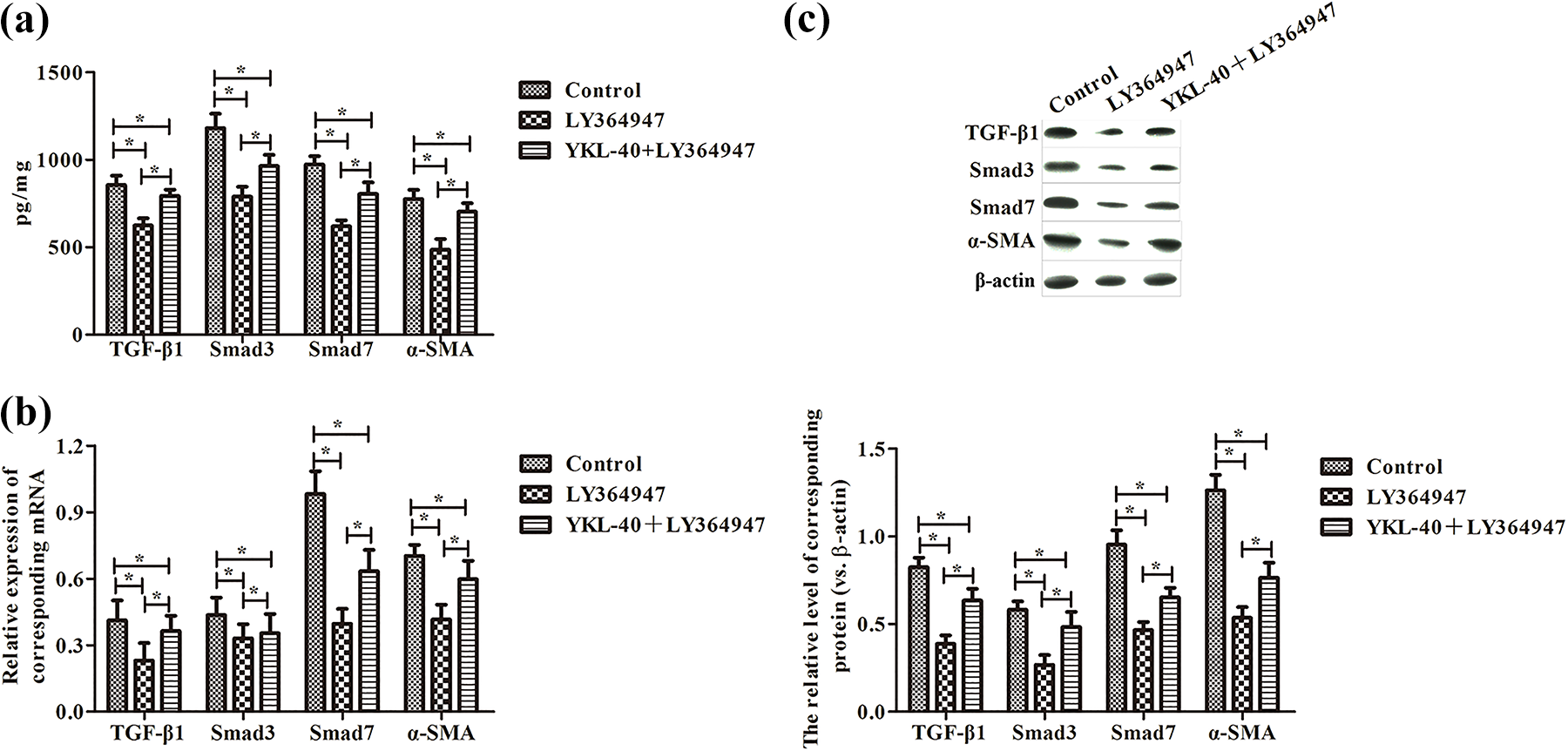
The TGF-β1, Smad3, Smad7, and α-SMA expression in BEAS-2B cells. BEAS-2B cells were culture alone, treated with 10 mmol/L LY364947, or treated with 10 mmol/L LY364947 and 0.1 μg/mL YKL-40 for 48 h. (a) ELISA detection of TGF-β1, Smad3, Smad7, and α-SMA expression in cell supernatants. (b) RT-PCR detection of TGF-β1, Smad3, Smad7, and α-SMA mRNA expression in BEAS-2B cells. (c) Western blot analysis of TGF-β1, Smad3, Smad7, and α-SMA protein expression in BEAS-2B cells. Values are mean ± standard deviation, n = 6. *p < 0.05. TGF-β1: transforming growth factor-β1; ELISA: enzyme-linked immunosorbent assay; RT-PCR: reverse transcription polymerase chain reaction; mRNA: messenger RNA.
Discussion
In this study, we found that YKL-40 promoted the proliferation, migration, and inflammatory cytokines of human bronchial epithelial cell line BEAS-2B, while inhibited cell apoptosis. In addition, we demonstrated that YKL-40 treatment could enhance the expression of TGF-β1, Smad3, Smad7, and α-SMA in BEAS-2B cells (shown in Figure 9). Furthermore, YKL-40 stimulation reversed the inhibition of TGF-β1/Smads signaling pathway caused by inhibitor LY364947. Our findings suggest that YKL-40 could be used for targeting TGF-β1/Smads signaling pathway in allergic asthma treatment.

Concept map.
Serum YKL-40 can be used as a useful biomarker for asthma. 19,20 YKL-40 is secreted by bronchial macrophages and airway epithelial cells in asthma patients and allergen-sensitized mice. 6,21 There are several studies focusing on the role of YKL-40 in asthma, in which YKL-40 promoted bronchial smooth muscle cell proliferation and migration in a PAR-2-dependent manner. 11 In our study, consistent with previous studies, YKL-40 treatment regulated the biological behavior of human bronchial epithelial cells, such as cell proliferation, migration, and cell apoptosis.
Inflammatory cytokines play an important role in allergic asthma, which have the potential to induce respiratory disease in humans. It was found that the release of many proinflammatory cytokines (such as IL-4, IL-8, IL-6) increased in asthmatic patients. 22 Several studies showed inhibition pro-inflammatory cytokines production could control allergic asthma and ameliorate airway condition in an allergic asthma model, which mechanism is associated with TGF-β1/Smads pathway. 13 Interestingly, in this study, YKL-40 treatment increased the secretion of IL-4, IL-8, and IL-6 in BEAS-2B cells. These results were consistent with Väänänen et al. who reported that YKL-40 concentration correlated positively with IL-6 and IL-7 levels. 23
In order to clarify whether YLK-40 is involved in TGF-β1/Smads pathway in bronchial epithelial cells, the TGF-β1/Smads signaling inhibitor LY364947 18 was used in this study. Our results showed LY364947 treatment decreased cell proliferation and migration of BEAS-2B cells, co-stimulation with LY364947 and YKL-40 showed improved cell proliferation and migration. These data suggested that YKL-40 regulated biological behavior of BEAS-2B cells by TGF-β1/Smads signaling pathway.
Although our study clearly demonstrated that YKL-40 regulated the biological behavior of BEAS-2B cell by targeting TGF-β1/Smads signaling pathway, more cell lines need to be tested. Due to the complicated signaling pathway network, further experiments focused on the role of YKL-40 in animal asthma models are highly demanded to fully explore the mechanisms.
Conclusion
In summary, we demonstrated that suitable concentration of YKL-40 treatment could promote proliferation of BEAS-2B cells, which mechanism was possible by activating TGF-β1/Smads pathway. These results provided a new insight for allergic asthma treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The National Science Foundation for Young Scientists of China (81701587).
