Abstract
Chalcones are naturally occurring compounds exhibiting biological activity through multiple mechanisms. Flavokawain B is one of chalcones found in kava plant. In our studies, we focused on the anticancer activity of flavokawain B in colorectal cancer cells LoVo and its resistant to doxorubicin subline—LoVo/Dx. Strong cytotoxic activity of flavokawain B and its ability to inhibit the proliferation in both cell lines was detected. These effects accompanied with induction cell cycle arrest in G2/M phase and the presence of SubG1 fraction. Flavokawain B at low concentration led to increase of caspase-3 activity. The chalcone-induced apoptosis was also confirmed by DNA fragmentation. In our work, the conversion of flavokawain B to corresponding flavanone—5,7-dimetoxyflavanone—was shown to be more extensive in cancer than in non-cancer cells. We found that the cyclization of the chalcone was related to the significant decrease in the cytotoxicity. Cell proliferation and cell cycle progression were not impaired significantly in the studied cancer cells incubated with 5,7-dimethoxyflavanone. We did not observe apoptosis in the cells incubated with flavanone. The results from biological studies agreed with the theoretical activity that emerges from structural parameters.
Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed malignancy and the fourth leading cause of cancer death in the world. Distribution of CRC increases in countries with a high or very high human development index. It is known that behavioral risk factors, such as a sedentary lifestyle or diet, account for a large proportion of cases. 1 The effectiveness of chemotherapeutic approaches in CRC is limited. The major obstacle in successful therapy is the multidrug resistance which can be primary phenomenon or can develop in CRC cells during treatment. 2 Despite unsatisfactory therapies of CRC and the number of adverse side effects caused by the presently used drugs, the emphasis has been put on the use of natural products that can be candidates for single use or for use in combinations with other substances.
Kava (Piper methysticum G. Foster, Piperaceae), a plant grown in the Pacific Islands, has been used for the preparation of solvent extracts (ethanol and/or acetone) that might treat some ailments, that is, mild and moderate anxiety, stress, insomnia, or restlessness. Kava extracts are commercially available as dietary supplements in Western countries and may be beneficial in the treatment of anxiety, insomnia, and muscular problems. 3 Biological activities of kava are related to the presence of specific chemical components in the plant: for example, chalcones, dihydrolactones, 5,7-dimethoxyflavanone, and cinnamic acid bornyl ester. It has been suggested that these compounds acting alone or in combination with other components of kava can be responsible for pharmacological action of the plant. 4
Chalcones that are open chain flavonoids are produced by vegetables, fruits, and some other edible plants. Because of their wide biological activity, chalcones are an interesting group of natural compounds. Chalcones are compounds derived from flavonoids. Their chemical structure consists of two aromatic rings linked by a three-carbon α,β-unsaturated carbonyl system. Plant-derived chalcones and also their synthetic analogues have been characterized as biologically active molecules. They possess anticancer, antimicrobial, antimalarial, and antioxidant activities. In addition, it has been indicated the potential of chalcones as anti-HIV, anti-inflammatory, and anti-protozoa molecules. 5 The structure-dependent anticancer activity seems to be one of the most crucial properties of chalcones, and their inhibitory potential against numerous molecular targets have been characterized. 6 Important field in the studies on anticancer activity of chalcones is their ability to induce apoptosis. It is well-known that different dysfunctions in this process, such as deficiency in caspase activation, contribute to pathogenesis of CRC but also to the development of resistance to treatment. 7,8 Regarding the apoptosis-based trends in CRC therapies, chalcones are promising candidates for further research.
Chalcones identified in kava plant are known as flavokawains: flavokawain A, flavokawain B, and flavokawain C. They possess a similar structure with some small differences in their respective side chains. Here, we focused on the flavokawain B, because according to previous research, this molecule is more effective in the treatment of cancer cells. 9 Our studies have been performed in a model of CRC cells LoVo. However, to further assess therapeutic potential of the studied compounds, we used also LoVo/Dx cells, resistant to doxorubicin subline of LoVo. We found that flavokawain B is cyclized to 5,7-dimethoxyflavanone by CRC cells LoVo and their resistant to doxorubicin subline LoVo/Dx. We studied and compared the cytotoxicity of both compounds, their influence on proliferation, and cell cycle distribution but also their ability to induce apoptosis in these cell lines. Our findings from experimental section of our study were compared and discussed in relation to the theoretical parameters obtained using SPARTAN’16 calculation package.
Materials and methods
Compounds
Flavokawain B (2′-hydroxy-4′,6′-dimethoxychalcone) was synthesized by Cleisen-Schmidt condensation of 2′-hydroxy-4′,6′-dimethoxyacetofenone (1) and benzaldehyde (2) following the method by Yadav et al. (Figure 1). 10 The reaction was carried out for 2 h in the presence of sodium hydroxide as the catalyst and methanol (MeOH) as a solvent.

The scheme of synthesis of flavokawain B (3) and 5,7-dimethoxyflavanone (4).
5,7-Dimethoxyflavanone (4) used in the experiments on the colorectal cell lines LoVo and LoVo/Dx was obtained from 2′-hydroxy-4′,6′-dimetoxychalcone (3) according to the previously described method. 11,12 Then, 5 g of the substrate was dissolved in ethanol (75 ml) with the addition of sodium acetate (8 g), and the reaction mixture was refluxed for 48 h. Crystallization from ethanol afforded pure flavanone (4). 5,7-Dimethoxyflavanone (4) was used as a standard for HPLC analysis. The chemicals used were purchased from Sigma-Aldrich (Poland).
For studies, both compounds were dissolved in dimethyl sulfoxide (DMSO; ROTH, Poland) to 30-mM stock solution.
Cell lines
The human colorectal adenocarcinoma cell line LoVo and its doxorubicin–resistant subline LoVo/Dx 13 –15 used in these studies were obtained from the Institute of Immunology and Experimental Therapy (Polish Academy of Science, Wrocław, Poland). Both cell lines were grown in F12 medium (Cytogen, Poland) containing 10% fetal bovine serum (Gibco, Poland), 1% antibiotic antimycotic solution (Sigma, Poland), and 1% glutamine (Prolabo, RPA). Madin-Darby canine kidney (MDCK) cells were used as a non-cancer model of mammalian cell line. These cells were grown in Dulbecco’s modified Eagle’s medium (DMEM; Cytogen) containing 10% fetal bovine serum (Gibco), 1% penicillin–streptomycin (Sigma), and 1% glutamine (Prolabo). All the cells used in the research were cultured at 37°C in humidified 5% carbon dioxide atmosphere and passaged twice a week. The cultured cells were gently dislodged from the flask surface using nonenzymatic cell dissociation solution (Sigma Aldrich, Poland). After each passage of LoVo/Dx cells, doxorubicin (Sigma Aldrich) at the concentration of 100 ng/ml was added to the medium for the LoVo/Dx cells to develop resistance to this drug.
The density of cells for each experiment was determined with EVE Automatic Cell Counter (NanoEnTek, Poland). Absorbance measurement was performed using spectrophotometer Tecan Infinite M 200.
Biotransformation of flavokawain B in cancer cells
For biotransformation, cells (1.5 × 105) were incubated with flavokawain B (3) at the concentration of 10 µM for 24 h. Contents of the flasks were extracted with ethyl acetate (2 × 2 ml); the organic layer was separated, dried over anhydrous magnesium sulfate, and filtered into a test tube; the solvent was evaporated off. Control cultivation with no substrate was also performed. The second control was flavokawain B incubated in F12 or DMEM medium only, without the cells. The residue was analyzed by high-performance liquid chromatography (HPLC). The products of biotransformation were not isolated after the experiment. The flavanone was identified in comparison with the standard obtained in chemical synthesis. The pH of the cell culture medium at the beginning and end of the experiments was the same for the controls and the flavokawain B experiment. Maintaining the pH was important to exclude spontaneous isomerization resulting from pH changes.
HPLC analysis
The HPLC analyses were performed with UHPLC Thermo Scientific (Poland) Ultimate 3000 or Waters 2690 instrument equipped with a Waters 996 (Milford, MA, USA) photodiode array detector, using an Agilent column Eclipse XDB-C18 (4.6 × 250 mm2, 5 µm, Santa Clara, California, USA) and C18 pre-column. The separation conditions were as follows (Figure 2): gradient elution, using 80% acetonitrile in 4.5% formic acid solution (eluent A) and 4.5% formic acid (eluent B); flow: 1 ml/min; detection wavelength 280 nm; program: 0–7 min, 10% A 90% B; 7–10 min, 50% A 50% B; 10–13 min, 60% A 40% B; 13–15 min, 70% A 30% B; 15–20 min 80% A 20% B; 20–30 min 90% A 10% B; 30–40 min, 100% A; (Figure 3): gradient elution, using 0.1% formic acid (eluent A) and 0.1% formic acid in acetonitrile (eluent B); flow: 1 ml/min; program: 0–12 min, 60% A 40% B; 12–17 min, 3% A 97% B; 17–21 min, 60% A 40% B; the enantiomeric excess values were determined using a Chiralpak AD-H HPLC column with diameters 4.6 × 250 mm2 (Diacel, Bratislava, Slovakia), with hexane:isopropanol (9:1) as the eluent (isocratic resolution).

HPLC analysis performed after incubation of flavokawain B with LoVo (a) and LoVo/Dx (b) using UHPLC Thermo Scientific Ultimate 3000 instrument. Measurements were carried out after 24 h of incubation. Presented control analysis was performed after 24 h of incubation of flavokawain B with F12 growth medium only. Measurements were carried out using Waters 2690 instrument (c). The experiments were repeated three times (n = 3). HPLC: high-performance liquid chromatography.

HPLC analysis performed after incubation of flavokawain B with MDCK cells (a) using UHPLC Thermo Scientific Ultimate 3000 instrument. Measurements were carried out after 24 h of incubation. Presented control analysis (b) was performed after 24 h of incubation of flavokawain B with DMEM medium only. The experiments were repeated three times. MDCK: Madin-Darby canine kidney; HPLC: high-performance liquid chromatography.
Quantitative analyses of the mixtures were performed by means of HPLC. Calibration curves for quantitative analyses were prepared using isolated and purified chemical synthesis products as standards.
Thin layer chromatography
The course of the chemical synthesis and the transformation was monitored by thin layer chromatography (Silica gel 60, Kieselguhr F254 plates, Merck, Darmstadt, Germany). Chromatograms were developed using the following developing systems: hexane:ethyl acetate (7:3), dichloromethane:ethyl acetate (1:1), and toluene:diethyl ether (4:1).
Spectroscopic methods
The nuclear magnetic resonance spectra were recorded with a DRX 600-MHz Bruker spectrometer (Bruker, Billerica, Massachusetts, USA). Mass spectra were obtained using high-resolution electrospray ionization (ESI+-MS) (Waters LCT Premier XE mass spectrometer, Milford, Massachusetts, USA). The optical rotation was measured by a Jasco P-2000-Na automatic polarimeter (ABL&E-JASCO, Kraków, Poland).
Sulforhodamine assay
Cytotoxic activity of flavokawain B and 5,7-dimethoxyflavanone in LoVo, LoVo/Dx, and MDCK cells was investigated using the sulforhodamine B (SRB) assay according to the procedure described previously. 16 Cells were incubated with compounds at concentrations: 5, 10, 25, 50, or 100 µM for 48 h.
To check the influence of the compounds on the resistance to doxorubicin in LoVo/Dx cells, the constant concentration of flavokawain B or 5,7-dimethoxyflavanone (5 µM) was used. In these experiments, doxorubicin at concentrations 0, 2.5, 5, 10, 15, and 25 µM was applied. Survival rate was presented as the percentage of survival cells in relation to the control cells that were incubated without any compound. The experiments were repeated three times. Cytotoxicity of DMSO against LoVo and LoVo/Dx cells was found to be negligible (below 0.5%).
Protein isolation
Proteins were isolated according to the procedures described previously. 16 The cells were washed with phosphate-buffered saline (PBS) and suspended in E buffer (50-mM Tris hydrochloride (HCl), 5-mM magnesium chloride, 50-mM sodium chloride (NaCl), and 250-mM sucrose). Then, the sonication on ice was performed. The mixture was centrifuged, and E buffer was added to the precipitate containing nuclear fraction. After next centrifugation, supernatant was removed and lysis buffer (50-mM Tris HCl, 250-mM NaCl, 0.1% NP-40, 0.5-mM phenylmethanesulfonyl fluoride (PMSF), and protease inhibitor cocktail) was added to the pellet. The mixtures were mixed and centrifuged. The Bradford method was applied to estimate the concentration of protein in the supernatant containing nuclear proteins. 17
Western blotting and immunodetection
Equal amounts of proteins were loaded onto the gels. The isolates were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis for 1.5 h at 100 V. Then, the proteins were transferred to a polyvinylidene difluoride membrane using a wet blotting method. To assess cellular proliferation, anti-proliferating cell nuclear antigen (PCNA) monoclonal Antibody (dilution 1:2000, Thermo Scientific) was used. Also, beta-actin monoclonal antibody (dilution 1:5000, Thermo Scientific) was applied for determining the expression of internal control. The incubations with the primary and secondary antibodies were performed as described previously. 16 To enable detection, Pierce Rabbit Anti-Mouse iGG (horseradish peroxidase (HRP) conjugated) secondary antibody (dilution 1:1000, Thermo Scientific) was used. 3-Amino-9-ethylcarbazole was applied as an HRP substrate. Reaction was carried out in the presence of 30% hydrogen peroxide and terminated by washing with water. The experiments were repeated three times.
Flow cytometry measurements
The influence of flavokawain and 5,7-dimethoxyflavanone on the cell cycle progression in LoVo and LoVo/Dx was estimated using flow cytometry method. The cells (800,000/flask) were seeded and allowed to attach for 24 h. Then, flavokawain B or 5,7-dimethoxyflavanone at final concentration of 10 and 50 µM, respectively, was added to the bottles. After 48 h of incubation, cells were scraped and washed with PBS (2×). Then, the cells were suspended in cold 70% ethanol and incubated on ice 2 h. In the next step, mixture was centrifuged (25,000 r/min/5 min/4°), the cells were resuspended in PBS, and RNaze (10 mg/ml) was added. After 2 h of incubation at 37°C, the cells were stained with propidium iodide (50 µg/ml) for 30 min in dark. Flow cytometric analysis was immediately performed in the Laboratory of Flow Cytometry (Faculty of Veterinary Medicine, Wrocław University of Environmental and Life Sciences, Wrocław, Poland) using an FACS Calibur (BD, Biosciences, San Jose, USA; cell cycle profiles were analyzed with BD CellQuest Pro 3.lf. software.
Caspase 3 activity
The activity of caspase-3 in the CRC cells incubated with flavokawain B and 5,7-dimethoxyflavanone was estimated using caspase 3 colorimetric assay kit (GenScript, Netherlands) The assay was based on the hydrolysis of a substrate DEVD-p-nitroaniline (pNA) by caspase 3 that led to the release of the pNA moiety. pNA can be detected using spectrophotometric methods. The cells (800,000/flask) were seeded and allowed to attach for 24 h. Then, flavokawain B or 5,7-dimethoxyflavanone (at the final concentration of 10, 20, 50, or 100 µM) was added to the bottles. After 48 h of incubation, cells were scraped and washed with PBS. The procedure was carried out according to the producer’s protocol as was previously described. 16 Relative caspase activity was introduced as ODprobe/ODcontrol ratio, where ODprobe and ODcontrol were the absorbance of pNA detected after incubation with the compound and with DMSO in medium, respectively.
DNA fragmentation
The cells (800,000/flask) were incubated with flavokawain B or 5,7-dimethoxyflavanone and then prepared for further proceedings in the same way as described in caspase 3 activity section. After 48 h of incubation, cells were centrifuged and washed with PBS. In order to isolate genomic DNA from the cells, the procedure previously described with minor modifications was applied. 16 The electrophoresis of obtained DNA was carried out for 1 h at 100 V in 1.5% agarose gel stained with Simply Safe (EURX). The separated fragments were visualized using the Gel Documented System KODAK MI v4.0.0.
Molecular calculations
All quantum calculations were performed using the SPARTAN ’16 calculation package. 18 Geometry of the molecule, in gaseous phase, of each investigated compound was optimized using the density functional theory (DFT) method with ωB97X-D exchange correlation potential (includes empirical corrections for dispersive interactions) in connection with 6-311+G** basic set. The optimized geometry of the given molecule was confirmed to be the real minima, by frequency analysis (no imaginary frequencies). 19 The solvent effect on geometry and value of quantum-mechanical parameters was performed using the conductor-like polarizable continuum model (CPCM) method for water. 20
Statistical analysis
The values of measured parameters (cell survival (%), the relative level of PCNA (%), and ODprobe/ODcontrol) were presented as the means ± standard deviation from three independent experiments. For data obtained in SRB, Western blotting, and caspase 3 activity research, the statistical significance was determined by Student’s t test (0.05 as threshold value) with the use of Statistica 10 software. The parameters estimated for probe with the compound have been compared with the parameters obtained for control without the compound.
Results
Identification of compounds
2′-Hydroxy-4′,6′-dimethoxychalcone (flavokawain B)
C17H16O4; Rt 24.2 min (HPLC); purity 98% (HPLC); proton nuclear magnetic resonance (1H NMR) (deuterated chloroform (CDCl3)) δ: 3.83 (3H, s, 4′-OCH3), 3.92 (3H, s, 6′-OCH3), 5.97 (1H, s, H-5′), 6.11 (1H, s, H-3′), 7.40 (3H, m, H-3, H-4, H-5), 7.60 (2H, d, J = 7.4 Hz, H-2, H-6), 7.79 (1H, d, J = 15.6 Hz, H-β), 7.91 (1H, d, J = 15.6 Hz, H-α), 14.29 (1H, s, 2′-OH); carbon 13 nuclear magnetic resonance (13C NMR; CDCl3) δ: 55.7 (4′-OCH3), 56.0 (6′-OCH3), 91.4 (C-5′), 93.9 (C-3′),106.5 (C-1′), 127.7 (C-α), 128.5 (C-6, C-2), 129.0 (C-3, C-5), 130.2 (C-4), 135.7 (C-1), 142.4 (C-β), 162.6 (C-4′), 166.4 (C-6′), 168.5 (C-2′), and 192.8 (–C=O). HRESI-MS [M+H+] (calculated/found) (m/z 285.1219/285.1215) (for details, see Supplementary materials).
5,7-Dimethoxyflavanone
C17H16O4; Rt 16.6 min (HPLC); purity 99% (HPLC);
Biotransformation of flavokawain B in the presence of human colon cancer cells
The product was determined in biotransformation for a 24-h incubation of LoVo and LoVo/Dx cells with substrate—flavokawain B. The compound detected by HPLC analysis at retention time of ∼16.6 min was identified as 5,7-dimethoxyflavanone. The remaining unreacted substrate was detected at retention time of ∼24.2 min (Figure 2(a) and (b)). In both cases, flavokawain B was transformed almost entirely to racemic 5,7-dimethoxyflavanone, and moreover, we detected high level of this conversion in biotransformation for a 24-h incubation (Figure 2(a) and (b)). Quantitative analyses indicated that the masses of flavokawain B and the flavanone obtained in biotransformation by LoVo were 2.23 × 10−3 mg and 9.17 × 10−3 mg, respectively (the ratio of the flavanone to the chalcone was 4.11). In the case of LoVo/Dx, the ratio of the flavanone to the chalcone was 6.72 (the masses of flavokawain B and the flavanone were 1.30 × 10−3 mg and 8.73 × 10−3 mg, respectively). 5,7-Dimethoxyflavanone was not observed in the controls contained only flavokawain B in medium without cells or only cells without flavokawain B. The chromatogram obtained for the control probe containing the chalcone in F12 medium without cells was presented in Figure 2(c). An unreacted flavokawain B (Rt = 24.5) and high peaks derived from components of the medium were detected. However, we did not notice any signal at retention time ∼16.6 (derived from 5,7-dimethoxyflavanone) what excluded spontaneous cyclization of the chalcone.
The conversion of flavokawain B to 5,7-dimethoxyflavanone has been also observed in the presence of non-cancer MDCK cells (Rt for the flavanone and for the chalcone was ∼8.4 min and ∼12.9 min, respectively). However, the conversion rate did not exceed 53% (Figure 3(a)). The obtained masses of flavokawain B and 5,7-dimethoxyflavanone were 6.20 × 10−3 mg and 7.39 × 10−3 mg, respectively. The calculated ratio of the product to the rest of the substrate of cyclization in MDCK cells (=1.19) was much lower than the ratios determined in experiments on colon cancer cells. We did not observe spontaneous cyclization of the chalcone in medium only (Figure 3(b)).
Cytotoxicity measurements
Studied chalcone, flavokawain B, exerted cytotoxic effect on the LoVo and LoVo/Dx cell lines. Strong inhibition of cells growth (p < 0.05) was observed for the all the concentrations used. The IC50 parameter was 18 µM in LoVo and 11 µM in LoVo/Dx cells (Figure 4).
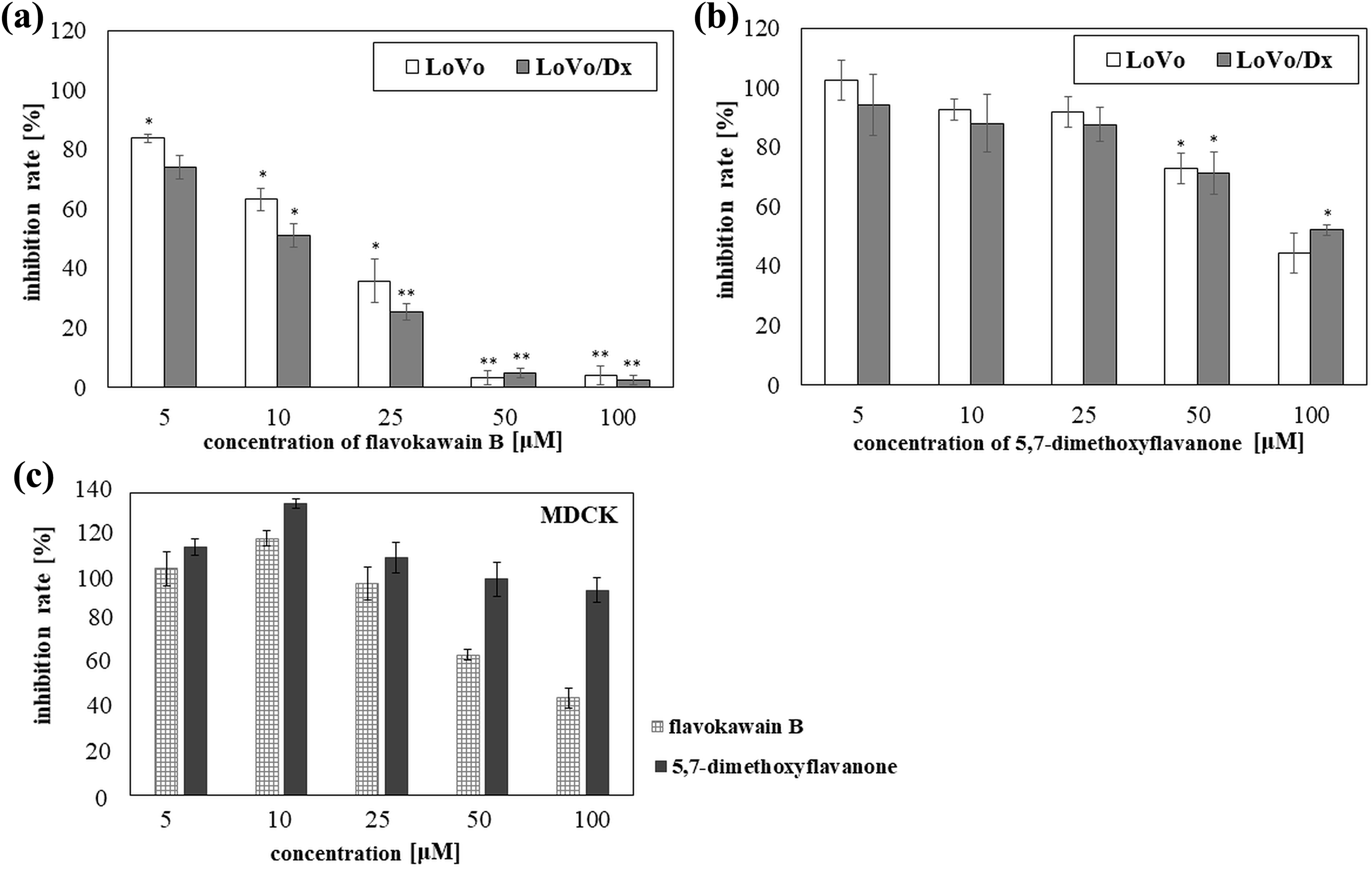
Cytotoxic effects of chalcone–flavokawain B (a) and corresponding flavanone–5,7-dimethoxyflavanone (b) in the cancer cells—LoVo, LoVo/Dx—and in the non-cancer cells—MDCK (c). The results were mean ± SD from three independent experiments. The results obtained at the concentration (5, 10, 25, 50, or 100 μM) were compared with the results obtained for control (without the compound). The statistical significance was determined obtained for the concentration using Student’s t test (*p < 0.05; **p < 0.001). MDCK: Madin-Darby canine kidney; SD: standard deviation.
We did not observe any differences between the cytotoxic activity of flavokawain B in LoVo and the cytotoxic activity of this compound in the Lovo/Dx cell line. A similar observation was made for of 5,7-dimethoxyflavanone. However, unlike flavokawain B, concentrations of 5,7-dimethoxyflavanone from 5 µM to 25 µM had a negligible effect on the growth of the cancer cell lines. Statistically significant cytotoxicity of this flavone (p < 0.05) was noticed in LoVo as well as LoVo/Dx cells only in high concentrations: 50 and 100 µM. Nevertheless, the cytotoxic activity of the studied flavanone was much lower than that of flavokawain B (p < 0.05). The IC50 parameter observed in LoVo was 88 µM. In the case of LoVo/Dx line, no IC50 could be established. (Figure 4(a) and (b)). Also, we performed cytotoxic measurements on the non-cancer MDCK cells. The observed activity of studied compounds in non-cancer cells was lower than their activity in cancer cells. It was shown that the studied compounds applied at low concentrations (5–10 µM in the case of flavokawain B and 5–25 µM in the case of corresponding flavanone) can slightly stimulate cell growth. The chalcone used at the higher concentrations (50–100 µM) reduced cell survival over 40%, whereas the same concentrations of 5,7-dimethoxyflavanone had a negligible effect on the growth of MDCK cells (Figure 4(c)).
Also, we checked whether flavokawain B and 5,7-dimethoxyflavanone can modulate the activity of doxorubicin in studied model of colon cancer cells. In our previous studies, we showed that the LoVo cells are more sensitive to this anticancer drug as compared to its subline—LoVo/Dx. 21 We found that none of the studied compounds has any influence on the cytotoxic activity of doxorubicin (Figure 5).

The influence of flavokawain B (flavB) and 5,7-dimethoxyflavanone (5,7-flav) on the cytotoxic activity of doxorubicin in resistant to this drug cancer cells LoVo/Dx. The results were mean ± SD from three independent experiments. The results obtained at the concentration of doxorubicin (2.5, 5, 10, 15 or 25 μM) were compared with the results obtained for control without this drug (only with flavB or 5,7-flav). The statistical significance was determined obtained for the concentration using Student’s t test (*p < 0.05; **p < 0.001). SD: standard deviation.
Effect of compounds on cancer cells LoVo and LoVo/Dx proliferation
Cellular proliferation was evaluated through the measurements of PCNA level. Expression of this protein is increased in rapidly proliferating cells. The effects were recorded using the Western blotting and immunodetection methods. We found that in both studied cancer cells incubated with flavokawain B the level of PCNA was much lower than cells incubated in the absence of this compound. The effect of 5,7-dimethoxyflavanone was not as marked as that of flavokawain B. We observed only slightly lower level of PCNA in LoVo and LoVo/Dx treated with the flavanone (Figure 6).
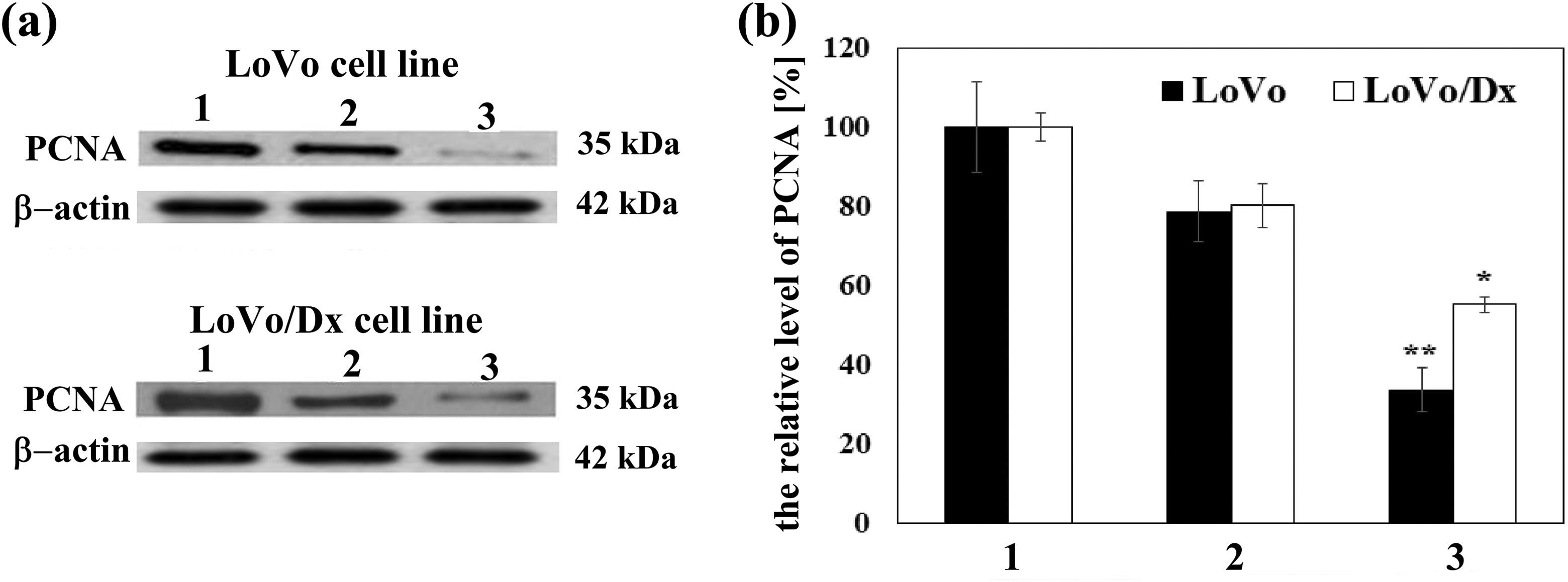
The level of PCNA protein in the presence of 5,7-dimethoxyflavanone (50 µM) and flavokawain B (10 µM). Control PCNA (1) derived from untreated cells. (a) Western blot analysis of PCNA level; β-actin was used as a reference protein. (b) The levels of PCNA were quantified using optical density measurements and normalized to the control without studied compounds (100%). Presented results were mean ± SD from three independent experiments. The statistical significance was determined obtained for the concentration using Student’s t test (*p < 0.05; **p < 0.001). SD: standard deviation.
Cell cycle analysis
In our work, we studied the impact of flavokawain B and its corresponding flavanone on the LoVo and LoVo/Dx cell cycle. We found that the antiproliferative effect of the chalcone was mediated by arresting cells in the G2/M phase. The population of treated cells in this phase was higher in both LoVo and LoVo/Dx cell line than the control (p < 0.05). The accumulation of cells in G2/M phase was accompanied by a decrease in the population of cells in G0/G1 phase. Our analysis showed also a significant subG1 cell population suggesting the late-stage apoptosis triggered by flavokawain B. Cyclization of the chalcone resulted in decreased ability to impair both LoVo and LoVo/Dx cell cycle. The percentage of cells accumulated in G2/M and in SubG1 phase in the presence of 5,7-dimethoxyflavanone was lower than in the presence of flavokawain B (Figure 7).

The analysis of LoVo/Dx (a) and LoVo (b) cell cycle in the presence of flavokawain B and 5,7-dimethoxyflavanone. The cellular distributions (percentage) in different phases of the cell cycle were determined after 48 h treatment with the chalcone at the concentration of 10 µM or with the flavanone at the concentration of 50 µM. The results were mean ± SD from three independent experiments. The statistical significance was determined using Student’s t test (*p < 0.05). SD: standard deviation.
Induction of apoptosis
We studied the ability of flavokawain B and its product of cyclization to induce apoptosis in LoVo and LoVo/Dx cells. For this purpose, the impact of studied compounds on the activity of caspase 3 was checked. Caspase 3 is a member of the cysteine-dependent proteases family participating in programmed cell death. This enzyme, known as one of the executive caspases, is responsible for the breakdown of the nucleus and other cellular compartments. 22 To investigate the influence of flavokawain B on caspase 3 activity, the concentrations of 10 µM and 20 µM were used. Both concentrations increased significantly the activity of this enzyme for both cell lines compared to the control (p < 0.05 and p < 0.001 for 10 and 20 µM flavokawain B, respectively). However, the effect observed in the resistant to doxorubicin LoVo/Dx cells was not as substantial as for LoVo. Additionally, statistically significant differences between caspase 3 activity in flavokawain B-treated LoVo and in LoVo/Dx cells were observed (Figure 8(a)). The apoptosis induced by flavokawain B was also confirmed by the detection of DNA bands observed after electrophoresis of genomic DNA obtained from the cells exposed to this compound (Figure 8(b)).

Investigation of apoptosis in LoVo and LoVo/Dx cells incubated with 5,7-dimethoxyflavanone or flavokawain B. (a) The activities of caspase-3 in studied cancer cells incubated with the compounds (ODprobe) for 48 h were normalized to the control derived from non-treated cells (ODcontrol). The results were mean ± SD from three independent experiments. The statistical significance was determined using Student’s t test (*p < 0.05; **p < 0.001); (b) Genomic DNA isolated from LoVo and LoVo/Dx cells incubated with flavokawain B (20 µM) or 5,7-dimethoxyflavanone (100 µM) for 48 h. DNA was separated by agarose gel (1.5%) electrophoresis. SD: standard deviation.
We did not observe changes in the activity of caspase 3 in LoVo and LoVo/Dx cells incubated with 5,7-dimethoxyflavanone at concentrations of 10 and 20 µM (data not shown). Due to the low cytotoxicity of this compound observed for the cell lines studied, it was decided to use higher concentrations of this compound. However, even at these concentrations, no noticeable change in caspase 3 activity was noted when compared to the control (the non-treated cells; Figure 8(a)). As we expected, this flavonoid did not cause DNA fragmentation (Figure 8(b)).
Molecular calculations
Theoretical chemical reactivity of investigated compounds can be estimated on the basis of frontier (HOMO, LUMO) molecular orbitals energy (E). Based on the density functional theory and the Koopmans’ theory of the frontier orbitals, energies are given by:
where I and A are the ionization potential and electron affinity, respectively. ΔE parameter, known as energy gap, is a difference between values of E HOMO and E LUMO. The theoretical chemical reactivity of chemical compounds was estimated using several descriptors, such as chemical hardness (η), chemical potential (μ), and electrophilicity index (ω). The values of the descriptors are closely related to energy of frontier orbitals, according to the following equations: 23 –25
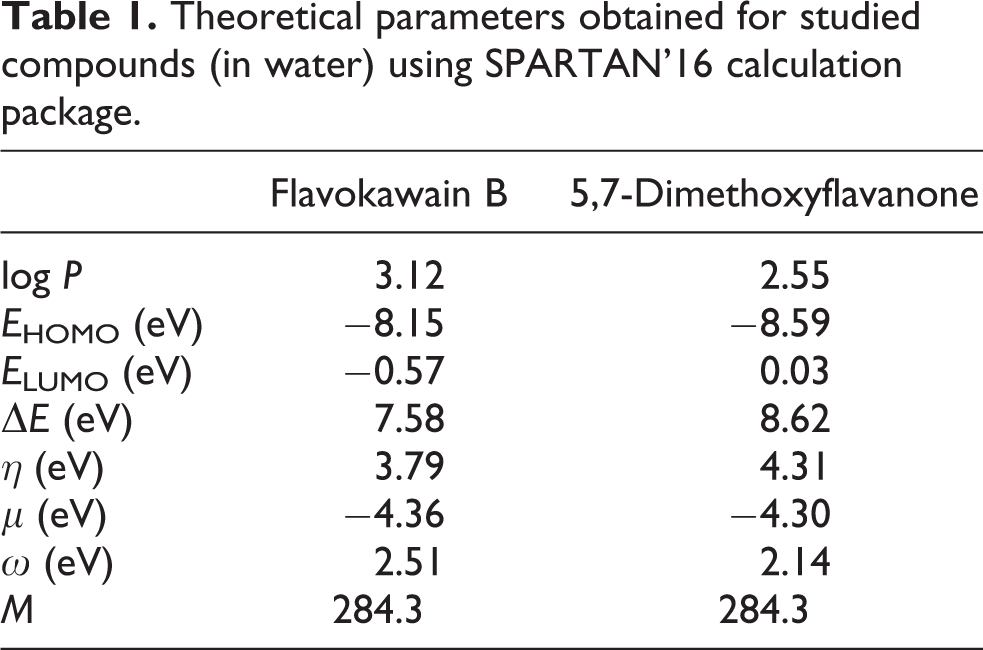
The obtained values of orbital energy HOMO and LUMO orbitals as well as calculated values of quantum chemical descriptors are given in Table 1. It was observed significantly lower values of chemical hardness (η) and energy gap (ΔE) for flavokawain B as compared to 5,7-dimetoxyflavanone. In parallel, the structure of flavokawain B was characterized by much higher values of log P and of electrophilicity index (ω) comparing to the studied flavanone. It was not any essential difference between chemical potential values between these compounds.
Theoretical parameters obtained for studied compounds (in water) using SPARTAN’16 calculation package.
Discussion
Flavokawain B, a chalcone isolated from kava plant, has been extensively studied due to its interesting pharmacological properties. 26 –28 In our studies, flavokawain B was synthesized by the Claisen–Schmidt condensation method. This method is very useful to obtain chemicals that are hard to reach from their natural sources or derivatives of compounds possessing specific groups in the structure. Also, other researchers have used the Claisen–Schmidt condensation method for the synthesis of compounds interesting from the point of view of their expected biological activity. 9,29
Due to the high incidence of colon cancer and the development of multidrug resistance followed by the low effectiveness of chemotherapy, we focused our research on the model of CRC cells LoVo and their resistant to doxorubicin subline LoVo/Dx. Our work discovered that CRC cells LoVo and cells resistant to doxorubicin of LoVo/Dx cell line catalyze a conversion of flavokawain B to the corresponding flavanone, 5,7-dimetoxyflavanone. The intracellular cyclization of chalcone has been found as one of the reactions in isoflavonoid biosynthesis pathway catalyzed by the enzyme chalcone isomerase in plants. However, in humans, chalcone isomerase does not exist. Previous studies proved that spontaneous isomerization of chalcone to flavanone may occur in a solution (without plant or human cells) and the process depends on pH of the environment in which the reaction takes place. 30 –32 For example, Mol et al. in their studies on self-cyclization of 2′,4,4′,6′-tetrahydroxychalcone reported that this process is dramatically reduced at pH ≤ 6.5 and in the presence of high concentrations of serum albumin. 30 Also, Simmler et al. in their research on isoliquiritigenin found that this chalcone can undergo isomerization to liquiritigenin flavanone in the medium (DMEM/F12 or RPMI 1640) with or without breast cancer cells MCF7. 32 In our studies performed using F12 or DMEM medium, we did not observe isomerization of flavokawain B to 5,7-dimethoxyflavanone in the absence of the studied cancer cells. Also, the reaction of cyclization of the chalcone was not a result of decrease in pH—we did not observe any changes in the value of pH in the medium during experiments. We found that the isomerization of the chalcone can occur in a model of non-cancer cells MDCK. However, the extent of cyclization was much greater in studied colon cancer cells, in particular—LoVo/Dx cells. These effects indicated that cancer cells could provide more optimal conditions for this process. In the next step, it was investigated whether cyclization of chalcone resulted in the change of biological activity of compound. In our further experiments, it was used 5,7-dimetoxyflavanone obtained by chemical synthesis.
Strong cytotoxic activity of flavokawain B was observed. Growth inhibition with IC50 value <20 µM was obtained in the case of both colon cancer cell lines, sensitive one and resistant to doxorubicin. The cytotoxic activity of flavokawain B has been also assessed in other colon cancer cells HCT-116 33 and HT-29. 34 Presented here, LoVo cell survival for 48-h incubation with flavokawain B was slightly higher than HCT-116 cell survival observed by Kuo et al. However, the cytotoxicity of the chalcone in HCT-116 cells was comparable to its cytotoxicity in LoVo/Dx shown in our work. The cytotoxic activity of flavokawain B in HT-29 was evaluated for 72-h incubation with the compound. 34 Nonetheless, the cytotoxicity of the chalcone used in the concentrations of 50–10 µM was lower than the cytotoxicity in LoVo and LoVo/Dx cells. An activity of flavokawain B as a growth inhibitor was also found in breast, 35 liver, 36 bone, 37 oral, 38 and prostate cancer cells. 39 In contrast to flavokawain B, 5,7-dimethoxyflavanone was characterized by low cytotoxicity against LoVo and LoVo/Dx cells. Only at high concentrations, this compound cause more than 20% growth inhibition. HPLC analysis confirmed the presence of both flavokawain B and 5,7-dimethoxyflavanone in the cells. However, cytotoxicity measurements indicated that the chalcone is much more active that the flavanone. This fact allowed us to suppose that most of the observed cytotoxic activity is due to flavokawain B.
Cyclization of the chalcone led to a significant reduction of the IC50 parameter observed in LoVo and LoVo/Dx. Similar effects have been observed also by other authors. Ketabforoosh et al. found that chalcones with a 3,4-dimethoxyphenyl moiety in their structure and their derivatives with an additional chloride atom or methoxy group at carbon C-5 are much more potent inhibitors of cell growth than corresponding flavanones as observed in three different cancer cell lines—neuroblastoma cell line SK-N-MC and breast cancer cell lines: MDA-MB-231 and MCF-7. 40 In contrast, the attachment of a pyrrolidinylmethoxy group at position C-7 of a flavanone structure increased the cytotoxicity of flavanones when compared to their parent chalcone.
The results of our research indicated that both flavokawain B and 5,7-dimethoxyflavanone did not influence the resistance of LoVo/Dx cells to anticancer drug—doxorubicin. Thus, these compounds can’t be used as chemosensitizing agents.
The results of our research on cytotoxicity of the two studied compounds in LoVo and LoVo/Dx cells correspond well to the results of other studies on the impact on cancer cell proliferation. Flavokawain B strongly decreased the level of PCNA, which is a standard marker used to assess the influence on DNA replication. 41 In addition, flow cytometry measurements showed the accumulation of cells incubated with flavokawain B in G2/M phase of cell cycle. In contrast, the impact of 5,7-dimethoxyflavanone on cancer cell proliferation and cell cycle distribution was much weaker. Our studies were in agreement with previous research exhibiting flavokawain B-inducted G2/M cell-cycle arrest in the CRC cell line HCT116 33 and also in osteosarcoma, 37 oral, 38 and squamous. 42 Moreover, it has been shown that anticancer activity of flavokawain B in case of several cancer cell lines is associated with the induction of apoptosis via both the extrinsic 43,44 and intrinsic pathways. 37,42,45 Cell cycle progression analysis performed in our studies revealed a significant subG1 population of LoVo and LoVo/Dx cells treated with flavokawain B suggesting later stage of apoptosis. Further studies confirmed the ability of this chalcone to induce apoptosis in studied cells. Flavokawain B used even at low concentrations significantly increased activity of caspase 3 activity. The induction of apoptosis was confirmed also by identification of the characteristic “ladder” in the electrophoresis of DNA isolated from cancer cells treated with flavokawain B. Our results indicated the involvement of both the extrinsic and intrinsic pathways of apoptosis in studied cells cultured with flavokawain B. Our findings correspond to the results of Malami et al. who observed, that is, an increased level of caspase 3 and DNA fragmentation in another colon cancer cells HT-29 incubated with this chalcone. 34 In contrast to flavokawain B, 5,7-dimethoxyflavanone did not display proapoptotic activity in the studied LoVo and LoVo/Dx cells. In the presence of this flavanone, a slight increase in caspase 3 activity was noticed but at very high concentrations of the compound. These changes were not statistically significant and could be a result of other cellular processes such as proliferation, migration, and repopulation of cancer cells. 46 Moreover, no DNA fragmentation after incubation with 5,7-dimethoxyflavanone was observed. Our results indicated a low anticancer activity of this compound in LoVo and LoVo/Dx cells. On the other hand, Li et al. showed that flavone with methoxy groups at position C-5 and C-7 (5,7-dimethoxyflavone) is an effective inhibitor of liver cancer cell proliferation and inductor of apoptosis. 47 Low effectiveness of 5,7-dimethoxyflavanone as compared to 5,7-dimethoxyflavone observed in cancer cells might be a consequence of a lack of a double bond between carbons C-2 and C-3 in flavanone.
Also, we checked whether the differences in the anticancer activity of flavokawain B and 5,7-dimethoxyflavanone in cell-based assays correspond to the differences in the theoretical activity assessed using the molecular descriptors calculated for these compounds by theoretical methods. The lipophilicity is an important property governing permeation of a compound across the cell membrane. This parameter can be used in the studies on the interactions of drugs and other chemical compounds with the lipids or cell membranes. The lipophilicity is described as the logarithm of partition coefficient between n-octanol and water (log P). 48 We found that log P of flavokawain B is higher than log P of 5,7-dimethoxyflavanone what might be responsible for its stronger ability to interact with the biological membranes.
The chemical hardness (η) describes the resistance of molecule to exchange electrons with the environment. This descriptor, together with ΔE parameter (known as energy gap), is associated with the stability and the reactivity of a molecular system. The higher parameters ΔE and η indicate higher stability and less reactivity of a molecule. 23 In parallel, the electrophilicity index (ω) measures the energy stabilization when molecule accepts an additional electron. This parameter depends on the chemical potential (μ) that expresses the tendency of a molecule to acquire an additional electron density, and the chemical hardness (η) that describes its resistance to exchange electrons with the environment. The higher value of ω indicates better electrophilic properties of a molecule and its higher reactivity. 25 Taking into account the values of molecular descriptors obtained for studied compounds, flavokawain B should be a less stable and more reactive compound as compared to 5,7-dimethoxyflavanone. This theoretical assumption corresponded well with the results from cell-based assays what suggested a significant impact of chemical structure of flavokawain B on the anticancer activity as compared to the corresponded flavanone.
Due to the high incidence and limited treatment of colon cancer, the studies on the new potential therapeutics with an ability to induce apoptosis or increase the activity of drugs applied in the therapy are desirable. Thus, our work was undertaken to check the effects of flavokawain B and its corresponding flavanone on colon cancer cells. We found that flavokawain B can’t modify the resistance of doxorubicin in resistant to this drug colon cancer cells. However, this compound is an effective growth inhibitor and apoptosis inducer in the studied cancer cells. The activity of this chalcone can be limited by its conversion to low active 5,7-dimethoxyflavanone. In our opinion, further experiments should be based on the relationship between the time-dependent anticancer activity of flavokawain B in colon cancer cells and the degree of its cyclization in the presence of these cells.
Supplemental material
Supplementary_Data - Cyclization of flavokawain B reduces its activity against human colon cancer cells
Supplementary_Data for Cyclization of flavokawain B reduces its activity against human colon cancer cells by A Palko-Łabuz, E Kostrzewa-Susłow, T Janeczko, K Środa-Pomianek, A Poła, A Uryga and K Michalak in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The costs of experiments were supported by the Polish Ministry of Science and Higher Education [Research Grant for Young Scientist no STM.A050.17.051].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


