Abstract
Sesamol is the main constituent of sesame seed oil and is obtained from Sesamum indicum. Oral squamous cell carcinoma (OSCC) is one of the most common neoplasms affecting the oral cavity. In this study, we investigated the cytotoxic potentials of sesamol on human oral squamous carcinoma (SCC-25) cells. Human oral squamous carcinoma cells were treated with different concentrations (62.5, 125, and 250 μM/mL) of sesamol for 24 h. Cytotoxicity was analyzed by 3- (4, 5- dimethylthiazol -2- yl) -2, 5-diphenyltetrazolium bromide (MTT) assay. Intracellular reactive oxygen species (ROS) expression was investigated by dichloro-dihydro-fluorescein diacetate assay. Apoptosis-related morphology was analyzed by acridine orange/ethidium bromide staining. Caspase-9 expression was analyzed by confocal microscopic double immunofluorescence staining. Mitochondrial apoptosis-related markers are analyzed using qPCR. Sesamol treatment caused a significant cytotoxic effect in OSCC cells. Sesamol-induced cytotoxic effect was associated with intracellular ROS generation. Sesamol treatments induced a significant increase in the early and late apoptotic cells. This treatment also induced caspase-9 expression in OSCC cells. Sesamol treatments caused downregulation of Harvey rat sarcoma viral oncogene homolog (HRAS) expression at protein and gene levels. Sesamol treatment modulates intrinsic apoptotic marker gene expression in OSCC cells. Overall results confirm the anti-cancer potential of sesamol and it seems to be a promising candidate for OSCC.
Introduction
Squamous cell carcinomas comprise at least 90% of all oral malignancies. 1 Globally, oral squamous cell carcinoma (OSCC) ranks eighth in the cancer incidence and third most common malignancy in South-central Asia with the epidemiologic variations between different geographic regions.2,3 Development of OSCC is associated with several etiological factors, namely, tobacco, alcohol, diet and nutrition, human papillomavirus infection, radiation, ethnicity, familial and genetic predisposition, oral thrush, immunosuppression, use of mouthwash, syphilis, dental factors, occupational risks, and mate.4,5 Current treatment modalities for OSCC include chemo/radiotherapy, radical ablative surgery, targeted therapies with epidermal growth factor receptors inhibitors, and cyclooxygenase-2 inhibitors, and photodynamic therapy have led to the major problems related to non-specific cell death.6,7 Despite the vast amount of research and the clinical advancement in oncology and surgery, OSCC causes significant mortality and morbidity worldwide, and the mortality rates remain unchanged. 8 The overall 5-year survival rate in treated head and neck squamous cell carcinomas patients remains about 50%. 9 Moreover, radiotherapy with or without chemotherapy to control OSCC progression is associated with radiation-induced toxicity and various side effects, including oral mucositis, osteoradionecrosis, xerostomia, and dysphagia. 10 Hence, there is an urgent need to identify the drug as a part of chemotherapy with minimal or no side effects in OSCC patients. Accordingly, natural products especially medicinal plant-derived compounds have contributed significantly to cancer chemotherapy and remain a crucial source of molecular and mechanistic diversity for anticancer drug discovery. 11
Sesamol (3,4-methylenedioxyphenol) is a main constituent of sesame seed oil and is obtained from Sesamum indicum.12–14 Sesamol also has antioxidant, antimicrobial, chemoprevention, and antimutagenic properties.15–17 In experimental studies, sesamol is shown to have promising beneficial effects against hepatotoxicity, pancreatitis, heart diseases, diabetes, and neurotoxicity.18–22 In vitro studies, cytotoxicity and apoptosis-inducing potentials of sesamol have been reported previously against lung adenocarcinoma, human colon cancer cells, human platelets, human hepatoma cells.23–26 In vivo, sesamol administration was shown to act as a chemosensitizer by activating death receptors in cancer models.15,27 These studies show that sesamol may have beneficial effects against various ailments, and also, it can serve as a cytotoxic agent and chemosensitizer. Therefore, it is reasonable to assume that sesamol could be cytotoxic to OSCC cells. Further, sesamol cytotoxic potential has not been studied against OSCC cell lines. Hence, in this study, we investigated the anticancer effect of sesamol against the human oral squamous cell carcinoma (SCC-25) cell line.
Materials and methods
Chemical and reagents
Sesamol (3, 4-methylenedioxyphenol) was purchased from Sigma Chemical Co. India. Dulbecco’s minimum essential low glucose medium (DMEM), dimethyl sulfoxide (DMSO), penicillin, streptomycin, trypsin-EDTA, 3- (4, 5- dimethylthiazol -2- yl) -2, 5-diphenyltetrazolium bromide (MTT), fetal bovine serum (FBS) was obtained from GIBCO BRL (Gaithersburg, MD). All other chemicals were of analytical grade.
Cell cultures and treatment
The human oral squamous cell carcinoma (SCC-25) cell line with the passage numbers 17-20 was used in this study. SCC-25 cells were cultured in 25 cm2 culture flasks using low glucose DMEM supplemented with FBS (10%) and penicillin (100 units/mL) and streptomycin (100 μg/mL). Cells were maintained in a standard humidified culture conditions with 5% CO2 at 37°C. After a couple of passages, cells were seeded for experiments. Upon reaching confluence, the cells were removed by trypsin application (0.25%) and were used for studies. All the experiments were performed with 70–80% confluence.
Dimethyl sulfoxide (0.1%) was used to dissolve sesamol (v/v). SCC-25 cells were plated at a density of 10,000 cells/cm2. After 24 h of seeding, the attached cells were fed with fresh expansion culture medium containing 62.5, 125, and 250 μM/mL concentrations of sesamol or the corresponding volumes of the vehicle. After 24 h of sesamol treatment, cells were detached using trypsin and were processed for following experiments. The cell morphology was investigated under an inverted microscope.
MTT assay
The cytotoxic potential of sesamol in SCC-25 cells was assessed by MTT assay. 28 Cells were seeded at a concentration of 5 × 104 cells/well in a 96-well plate. After 24 h of seeding, the existing medium was aspirated and cells were replaced with fresh expansion medium containing different concentrations (62.5, 125, and 250 μM/mL) of sesamol and incubated for 24 h. At the end of the treatment period, media was aspirated from the control and the sesamol-treated cells. Then 50 μL of freshly prepared MTT (0.5 mg/mL of PBS) was added to each well. Cell plate was kept in a shaker for 30 min and then incubated for 4 h at 37°C in a CO2 incubator. After incubation, MTT solution was aspirated and the colored crystal of formazan product was dissolved in DMSO. The purple-blue colored formazan product was quantified at 570 nm using an ELISA reader (BIORAD).
Dichloro-dihydro-fluorescein diacetate assay
The intracellular reactive oxygen species (ROS) generation was analyzed using a non-fluorescent probe, dichloro-dihydro-fluorescein diacetate (DCFH-DA) assay. 29 The expression of intracellular ROS was estimated in control and sesamol-treated SCC-25 cells. Briefly, 8 × 106 cells/mL was made up to 2 mL with normal PBS (pH 7.4). From this one ml was taken, to which 100 μL of DCFH-DA (10 μM) was added and it was incubated at 37°C for 30 min. The DCFH-DA–stained control and sesamol-treated cells were examined and imaged under a Nikon fluorescence inverted microscope. Fluorescent measurements were done by excitation and emission filters were set at 485 ± 10 nm and 530 ± 10 nm, respectively (Perkin Elmer Multimode reader).
Acridine Orange/Ethidium Bromide (AO/EB) staining and fluorescent microscopy
Ethidium bromide/acridine orange staining was performed by the method of Gohel et al. (1999). 30 SCC-25 cells were seeded in 48-well plates at a density of 1 × 104 cells/well. After reaching 70–80% confluent, the cells were treated with sesamol for 24 h. After 24 h, the existing culture medium was discarded from each well and cells were gently washed twice with PBS at room temperature. Then, cells were collected using trypsin and centrifuged. To the cell pellet, known volume of media was added. Then, 100 μL of cell suspension was mixed with 100 μL of dye mixture (1:1) and immediately investigated under a Nikon inverted fluorescence microscope (Ti series). The cells of each sample were counted at five different fields. The percentage of apoptotic cells was calculated by dividing total number of apoptotic cells and total number of cells counted.
Double immunofluorescence staining
Approximately, 1 × 104 of SCC-25 cells were grown on glass coverslips in 48-well plates. After 24 h post-plating, the sesamol treatment was done for 24 h. After 24 h, the cells were washed using D-PBS and fixed for 20 min with 4% formalin at room temperature. The fixed cells were washed, and endogenous peroxidase activity was inactivated after the incubation of cells with 3.3% hydrogen peroxide for 3 min. Then, cells were incubated for 10 min with Triton X-100 (1% in D-PBS) at room temperature for permeabilization. After washing twice with PBS, the cells were incubated for 1 h with blocking solution containing 1% BSA. Then, cells were incubated with the mouse monoclonal caspase-9 primary antibody (1:150) (Dako, Denmark) for 1 h at room temperature. After couple of washed with PBS, propidium iodide nuclear counterstaining was performed. Then, image was captured under a Nikon fluorescence inverted microscope.
Gene expression analysis of HRAS
After the experimental period, cells were fixed in trizol reagent and total RNA was isolated according to standard protocol. The RNA concentration was quantified using a nano drop (Wilmington, USA). After RNA quantification, 1 μg of RNA was used to synthesize cDNA by reverse transcriptase using M-MLV (Promega, Madison, WI) and oligo (dT) primers (Promega) according to the manufacturer’s protocol. Then, template cDNA (2 μL) was added to the reaction mixture containing 20 μL. RT-PCR (40 cycles) was done involving denaturation (95°C for 15 s), annealing (60°C for 20 s), and elongation (72°C for 20 s). The GAPDH and HRAS primer sequences were taken from literatures. Glyceraldehyde-3-phosphate dehydrogenase
Protein expression analysis of HRAS
After sesamol treatment for 24 h, the cell pellet was collected. The collected cells were washed with PBS and harvested in RIPA buffer (Sigma Aldrich, USA). The cell lysate was mixed with gel loading buffer (Thermo Scientific, USA) and heated for 5 min at 95oC. After protein quantification, 50 μg of protein from each sample was taken for sodium dodecyl sulfate-polyacrylamide gel (10%) electrophoresis. The electrophoresed gel was electro-transferred to polyvinylidene difluoride membranes. Once transfer done, the membranes were incubated for 2 h in blocking solution (5% skimmed milk powder) at room temperature. Then, the membranes are incubated over night at 4oC with the HRAS primary antibody (monoclonal, IgG1 (1:1000), BioRad, Chennai, India). Next day, the membranes were incubated with corresponding horseradish peroxidase-conjugated secondary antibodies (1:2000) at room temperature for 2 h. β-actin was used as an internal control for normalization. The membranes were then developed using the Pierce ECL plus western blotting substrate (Thermo Scientific, USA), followed by quantification using the NIH ImageJ analysis software.
Gene expression analysis by real-time polymerase chanin reaction (PCR)
The apoptosis-related marker gene expressions were analyzed by real-time PCR. Total RNA was extracted from control and sesamol-treated cells. The total RNA concentration was quantified using Nanodrop. The commercial cDNA conversion Kit (high cDNA, Applied Biosystem) was used for RNA (2 μg) cDNA conversion. The targeted genes such as Bax, Bcl-2, cytochrome c, apoptotic protease activating factor-1 (Apaf-1), and caspase-3 were amplified using gene-specific primer sequences. β-actin was used as an internal control. Real-time PCR reaction was performed in AbiprismIM 7700 PCR machine (Applied Biosystem). The real time PCR results were calculated by 2−∆∆CT method, and results are expressed as fold change.
Statistical analysis
Data were expressed as mean ± standard error of the mean and analyzed by One-way ANOVA following Dunnett’s test to determine the significant differences between groups. A p value <0.05 was considered to be significant.
Results
Sesamol treatments induced cytotoxicity in OSCC cells
In this study, we investigated the cytotoxic effect of sesamol on OSCC cell line by MTT assay. Initially, to find out the effective concentration range, we have treated OSCC with sesamol concentration ranges between 10 and 1000 μM (10, 50, 100, 200, 500, and 1000 μM/mL). Different concentrations of sesamol treatment for 24 h caused significant (p < 0.001) and dose-dependent increase in the cytotoxicity of OSCC cells (Figure 1(a)). In this preliminary investigation, the inhibitory concentration of sesamol was found at 250 μM/mL on OSCC cells, and hence, further analysis was carried out with concentrations lower than that of inhibitory concentration, that is, 62.5, 125, and 250 μM/mL. The control and sesamol-treated cells were displayed cancer cell morphology (Figure 1(b)). (a) Cytotoxic effect of sesamol in OSCC cells. Values are expressed as mean ± SEM (n = 3). *** p < 0.001 versus control OSCC cells. (b) Morphology of control and 24 h of sesamol treated OSCC cells. OSCC: oral squamous cell carcinoma.
Sesamol treatments induced intracellular ROS in OSCC cells
Furthermore, to determine the exact reason for cytotoxic effect, we analyzed the ROS inducing potential of sesamol on OSCC cells by DCFH-DA staining assay. Fluorescent microscopic images revealed that sesamol treatments for 24 h could cause ROS expression than control OSCC cells. The ROS-expressed cells were more prominent at 125 and 250 μM/mL of sesamol treatments (Figure 2(a)). There was a significant (p < 0.001) and dose-dependent increase in the percentage of fluorescent intensity was observed upon sesamol treatments (Figure 2(b)). Sesamol induced ROS generation in OSCC cells. (A) Green fluorescence indicates the ROS expressed OSCC cells. (B) Quantification of fluorescent intensity. Values are expressed as mean ± SEM (n = 3). ap < 0.001 versus control OSCC cells. OSCC: oral squamous cell carcinoma; ROS: reactive oxygen species.
Sesamol treatments induced morphological changes related to apoptosis in OSCC cells
To ascertain the consequences of ROS generation, we investigated OSCC cells' morphology after sesamol treatment. The apoptosis-related morphological changes and DNA damage was done by AO/EB staining. Sesamol treatments for 24 h caused significant alterations in the morphology of OSCC cells and there was no significant change detected in the control group. Early apoptotic cells were seen as marked yellow-green with AO nuclear staining in the low dose of sesamol treatment (62.5 μM) while late apoptotic cells localized with red nuclear EB staining were detected in all experimental groups except control (Figure 3(a)). The quantification of apoptotic cells after AO/EB staining further confirms the dose-dependent increase in the presence of apoptotic cells in experimental groups than that of control (Figure 3(b)). (a) Representative images of OSCC control and sesamol treated cells stained with ethidium bromide and acridine orange stains. White and blue arrow heads indicate early and late apoptotic cells respectively. (b) Quantification of percentage number of apoptotic cells stained using ethidium bromide-acridine orange stain. Values are expressed as mean ± SEM (n = 3). Apoptotic cells were individually calculated and the percentage of apoptotic cells relative to the total number of cells in each random field was also calculated and represented the average of three independent experiments ± SEM ***p < 0.001. OSCC: oral squamous cell carcinoma.
Sesamol treatments induced caspase-9 expression in OSCC cells
To determine whether caspase-9 became activated upon sesamol treatments, we investigated the caspase-9 expression by confocal dual immunofluorescence assay. Caspase-9 plays a significant role in the intrinsic (mitochondrial) pathway of apoptosis could be upregulated by sesamol treatment in OSCC cells. To demonstrate the involvement of the mitochondrial pathway, we analyzed caspase-9 expression. Double immunofluorescence staining demonstrated that in untreated cells, caspase-9 is not expressed. In contrast, in sesamol-treated cells, the two signals (green for caspase-9 and red for PI) were completely distinguished, indicating the expression of caspase-9 upon sesamol treatment. Propidium iodide was used as a nuclear counterstain and was expressed only in the nucleus (Figure 4). Caspase-9 expression by dual staining. Representative immunostaining images of control and sesamol-treated (125 μM) OSCC cells. Cells were treated with caspase-9 and propidum iodide nuclear staining. Merged image confirms the expression of caspase 9 (green) and nucleus stains only with PI (red). OSCC: oral squamous cell carcinoma; PI: propidium iodide.
Sesamol treatments increased HRAS expression at gene and protein levels in OSCC cells
HRAS expression is most frequently increased in OSCC patient’s tissue; therefore, we investigated the effect of sesamol on HRAS expression at protein and gene levels. Sesamol treatments for 24 h caused downregulation of HRAS at gene and protein level as compared to the control OSCC cells (Figure 5(a)). The densitometry analysis for protein expression also confirms significant decrease in HRAS expression (p < 0.001) after 24 h of sesamol treatments as compared to the control OSCC cells (Figure 5(b) and (c)). Expression of HRAS (a) HRAS gene expression. (b) HRAS protein expression. (c) Quantification of HRAS protein expression. Values are expressed as mean ± SEM (n = 3). Statistical analysis was performed by One way ANOVA followed by Tukey’s test. ap < 0.001 versus control OSCC treatments. OSCC: oral squamous cell carcinoma.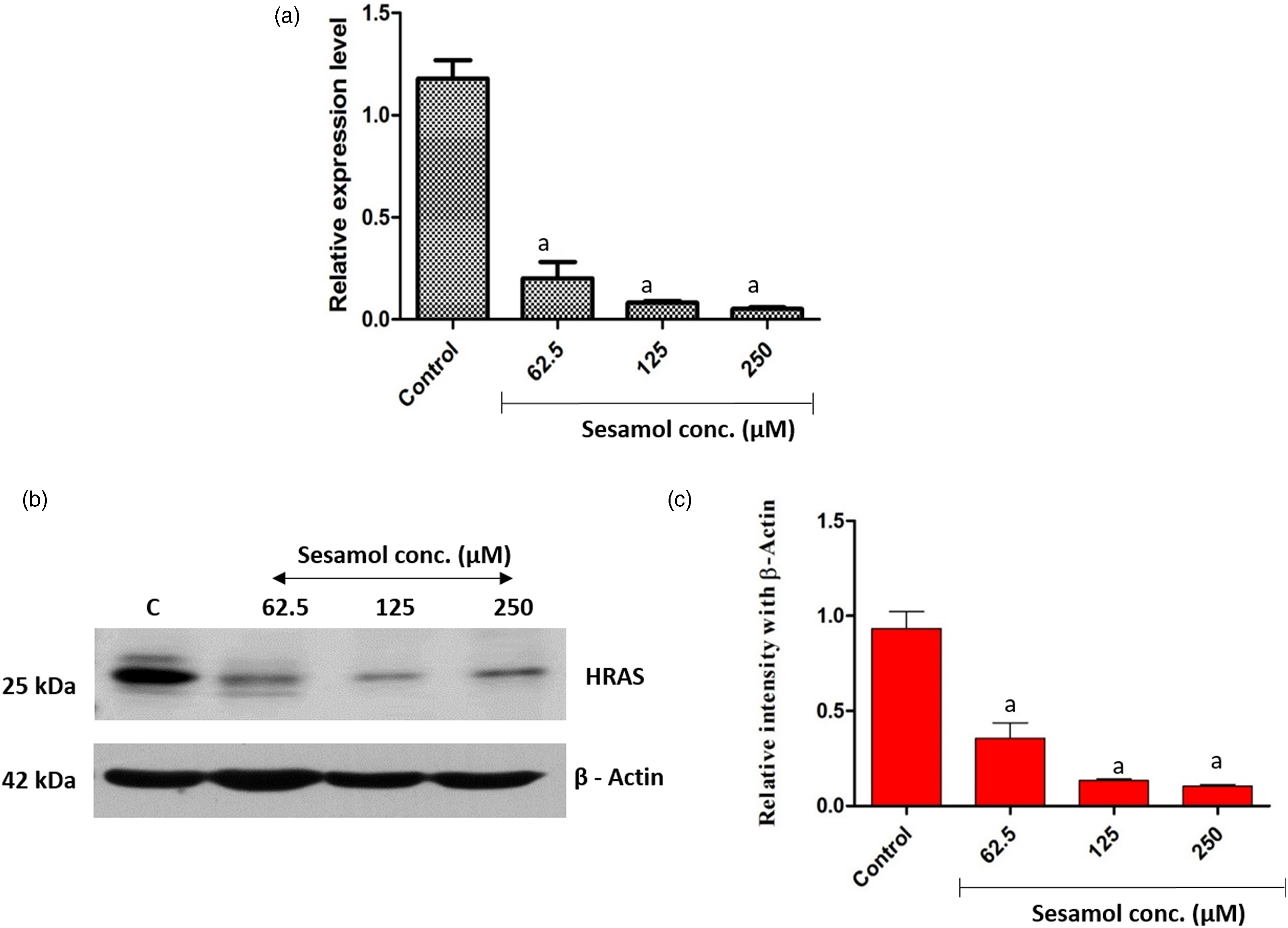
Sesamol treatments modulated intrinsic apoptosis-related marker gene expressions in OSCC cells
Further, to analyze whether sesamol-induced cytotoxicity was due to apoptosis induction, we investigated the apoptosis marker gene expressions in OSCC cells. Sesamol treatments caused upregulation of pro-apoptotic marker genes such as Bax (p < 0.01 vs 62.5 μM and p < 0.001 vs 125 and 250 μM), Bax to Bcl-2 ratio (p < 0.01 vs 62.5 μM and p < 0.001 vs 125 and 250 μM), cytochrome c (p < 0.01 vs 62.5 μM and p<0.001 vs 125 and 250 μM), Apaf-1 and caspase-3 (p < 0.01 vs 62.5 μM and p < 0.001 vs 125 and 250 μM). Sesamol treatment also caused downregulation of anti-apoptotic marker, that is, Bcl-2 in OSCC cells. However, at a low concentration (62.5 μM), sesamol did not modulate the bcl-2 and Apaf-1 expression (Figure 6). Intrinsic apoptosis pathways-related marker gene expression. Values are expressed as mean ± SEM (n = 3). Statistical analysis was performed by One way ANOVA followed by Tukey’s test. bP < 0.01 and aP < 0.001 are considered as significant. β-actin was used as internal control for normalization.
Discussion
Natural products represent a rich reservoir of potential phytoconstituents exhibiting cancer growth inhibition and anticancer properties. 31 Several plant-derived agents are currently successfully used in cancer treatment, such as vincristine and vinblastine and etoposide. More than 3000 plant species have been reported to treat cancer and about thirty plant-derived compounds have been isolated so far and have been tested in cancer clinical trials. 32 In this study, sesamol is one of the plant-derived compounds tested for its cytotoxic potential against OSCC cells.
Sesamol treatments caused significant cytotoxicity in OSCC cells. Previous studies have shown the cytotoxic potential of sesamol in lung adenocarcinoma, human hepatoma, and colon cancer cells.23–26 Therefore, it is clear that regardless of cell line sesamol could induce cytotoxicity. In this study, sesamol treatments also induced significant intracellular ROS expression, indicating the possibility of oxidative stress-inducing potentials of sesamol in OSCC cells. In previous studies, sesamol has been shown to generate intracellular ROS in cancer cells and the ROS inducing potential of sesamol has been implicated in cytotoxicity of cancer cells.24,26 The excessive intracellular ROS has been shown to induce cell membrane damage and cytotoxicity in a veriety of cancer cells, including OSCC cells.33–38 Taken together, the cytotoxic effect of sesamol observed in the present study could be due to the induction of intracellular ROS and their interaction with OSCC cells membrane.
Intracellular ROS generation has been shown to induce apoptosis in several cancer cell lines, including OSCC cells.39–45 Sesamol also induced apoptosis through intracellular ROS generation in various cancer cell lines other than OSCC cells.24,46,47 AO/EB stains are used to distinguish the early and late stage of apoptotic cells. 48 The dual staining assay confirms that sesmol treated OSCC cells are in early and late apoptotic phases. Early apoptotic cells were present in the experimental group treated with low concentrations of sesamol. While the cells were treated with maximum concentration of sesamol produced high fluorescence intensity for EB indicates late apoptosis and DNA damage. Moreover, a previous study has reported that sesamol could directly interact with DNA 49 and this could be the probable reason for the DNA damage induced by sesamol.
The RAS family proto-oncogenes, including HRAS KRAS, and NRAS, encode proteins responsible for regulating cell growth, differentiation, and survival of many cell types. The OSCC cells were shown to overexpress HRAS oncogene. 50 Clinically, HRAS gene overexpression was reported in OSCC patients and it was suggested to be useful as a prognostic marker and target for effective molecular therapy. 51 Sesamol treatments have been shown to downregulate the oncogene HRAS expression at gene and protein level in OSCC cells, indicating its anticancer potential. The downregulation of HRAS signaling may decrease the oncogenic signaling in OSCC cells.
Targeting mitochondria as a cancer therapeutic strategy is gained attention for researchers in recent years due to its essential role in cancer cell proliferation and apoptosis.52,53 Mitochondria are highly sensitive intracellular organelle, and the majority of herbal compounds target them to provoke cancer cell-specific death program.
54
Increased intracellular ROS has been shown to alter the mitochondrial homeostasis directly or by upregulation of bax and concomitant downregulation of Bcl-2.
44
In this study, sesamol treatments increased bax to bcl-2 ratio, which clearly indicate disturbance in bax/bcl-2 homeostasis. The ROS-induced alteration cause cytochrome c release from mitochondria to the cytosol. In the cytosol, cytochrome c form apoptosome along with apaf-1 and deoxy adenosine triphosphate. The apoptosome subsequently induce caspase-9 and -3 to trigger apoptosis.
45
Caspases are crucial mediators of apoptosis. Among them, caspase-9, and -3 are frequently activated in mitochondrial apoptosis,
55
and its activation is said to a critical determinant of genotoxic stress-induced apoptosis.
56
Detection of caspases in cells and tissues is an important method for apoptosis induced by a wide variety of apoptotic signals.
57
Previous studies also showed that sesamol treatment could trigger caspase expression, which indicates the proapoptotic potential of this compound.24,47 Caspase-3 is also required for apoptotic chromatin condensation and DNA fragmentation.
58
In light of the above report, this study suggests that sesamol-could induce caspase-3 expression in OSCC cells and this may be directly correlated with its apoptosis induction and genotoxic potential. Thus, sesamol induce mitochondrial toxicity and cytochrome c release via intracellular ROS accumulation and interference of bax/bcl-2 homeostasis. In the cytosol, cytochrome c forms apoptosome, which triggers the activation of caspases to induce apoptosis (Figure 7). Possible pro-apoptotic mechanism of sesamol in oral cancer cells. N: nucleus; M: mitochondria.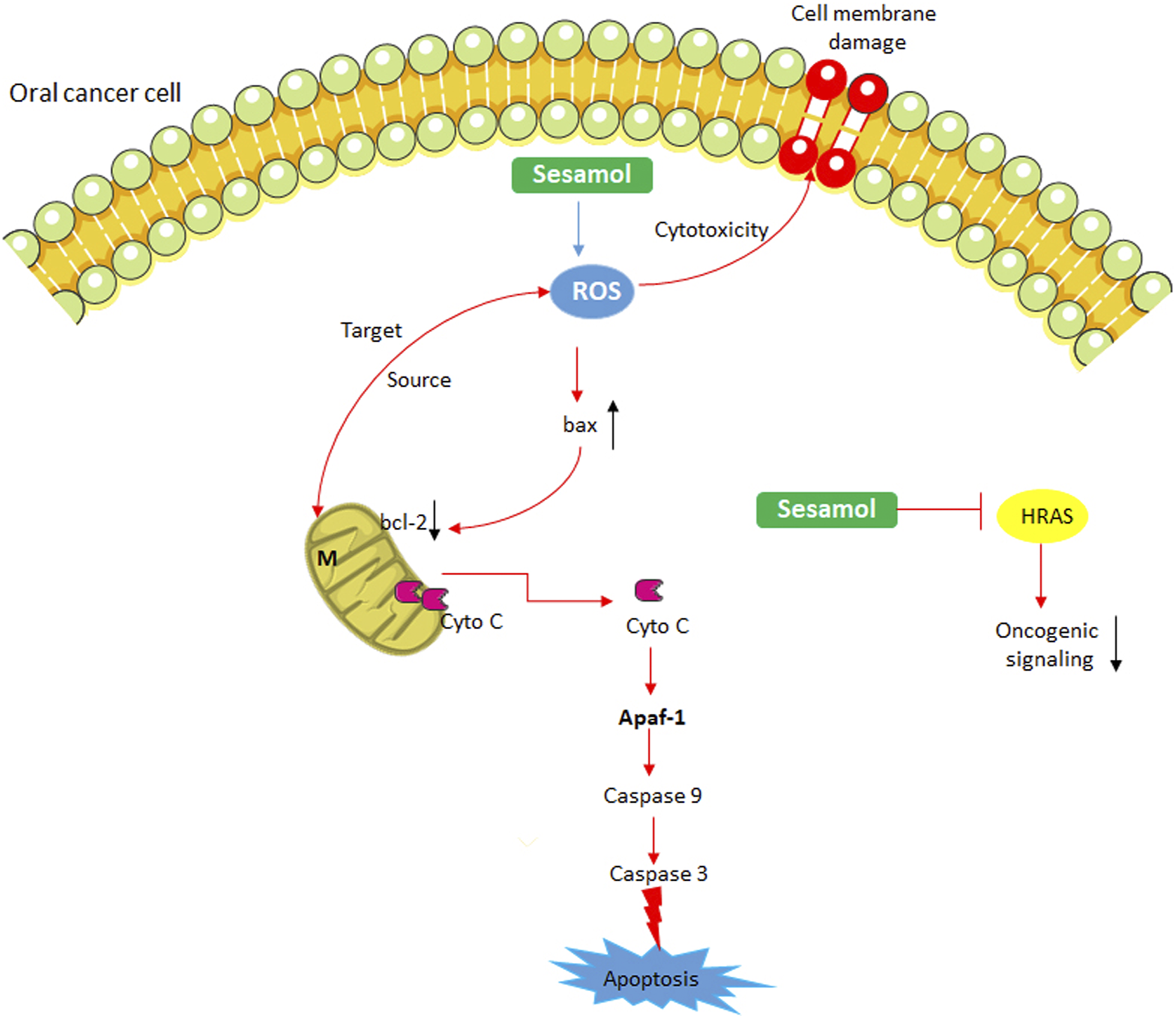
In conclusion, the results of this study suggest that sesamol has the potential to interact with OSCC cells DNA and thus impedes the process of cell proliferation. Sesamol accumulates the intracellular ROS and alters Bax and Bcl-2 homeostasis in OSCC cells and causes mitochondrial toxicity. Sesamol triggers apoptosis in OSCC cells by activation of caspases-9 and -3. These results support that sesamol can induce apoptosis in OSCC cells by ROS induction and it could be a promising candidate for OSCC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the researchers supporting project number (RSP-2021/165) king saud university Riyadh Saudi Arabia.
