Abstract
We examined the possible molecular mechanisms underlying the cytotoxicity and anticarcinogenic potential of Mentha leaf extracts (petroleum ether, benzene, chloroform, ethyl acetate, methanol, and water extracts) on 6 human cancer (HeLa, MCF-7, Jurkat, T24, HT-29, MIAPaCa-2) and normal (IMR-90, HEK-293) cell lines. Of all the extracts tested, chloroform and ethyl acetate extracts of M piperita showed significant dose- and time-dependent anticarcinogenic activity leading to G1 cell cycle arrest and mitochondrial-mediated apoptosis, perturbation of oxidative balance, upregulation of Bax gene, elevated expression of p53 and p21 in the treated cells, acquisition of senescence phenotype, while inducing pro-inflammatory cytokines response. Our results provide the first evidence of direct anticarcinogenic activity of Mentha leaf extracts. Further, bioassay-directed isolation of the active constituents might provide basis for mechanistic and translational studies for designing novel anticancer drugs to be used alone or as adjuvant for prevention of tumor progression and/or treatment of human malignancies.
Introduction
Cancer is the one of the leading causes of death in the world. According to World Health Organization’s recent estimate, the cumulative death toll due to cancer will be 12 million by 2030. 1 Plants have been a prime source of highly effective conventional drugs for the treatment of many forms of cancer and more than 60% of overall agents used for anticancer therapy are derived from either plants or marine and micro organisms. 2 Because of serious side effects associated with chemotherapies and radiotherapies, a surge to seek alternative and complimentary medicaments from natural sources has been recently observed. 3
Epidemiological studies have consistently suggested an inverse association between cancer risk and intake of fruits and vegetables. These health benefits have been linked to the additive and synergistic combination of biologically-active secondary metabolites such as alkaloids, flavonoids, terpenoids, essential oils, and glycosides present in fruits and vegetables that exert anticarcinogenic activity. 4 –6 Evidence from laboratory investigations have revealed the anticancer properties of a multitude of medicinal herbs, fruit, and vegetable extracts that are mediated through different mechanisms including altered carcinogen metabolism, induction of DNA repair systems, immune activation, and suppression of cell cycle progression/induction of apoptosis. 7 –10 The advantages of using such plant-derived agents are their relatively very low toxicity and availability in a consumable form. 11
Mentha piperita Linn. or peppermint (Family—Lamiaceae) has a long history of safe use, both in medicinal preparations and as a flavoring agent. Peppermint is used as a remedy for common cold, inflammatory processes of the mouth, pharynx, sinuses, liver, gallbladder, and bowel, as well as gastrointestinal tract ailments such as nausea, vomiting, diarrhea, cramps, flatulence, and dyspepsia. 12,13 A number of studies using oil and aqueous extract of M piperita leaves have demonstrated radioprotective and chemopreventive activity. However, a majority of these effects was attributed to protective activity against radiation-induced alterations in hematological constituents of peripheral blood and phosphatase activities in serum, lipid peroxidation. 14,15 Several herbal products derived from Lamiaceae family have shown potential anticarcinogenic properties. 16 –18 Despite these reports, direct cytotoxic potential of this important herb has never been evaluated. Assessment of the anticarcinogenic effects of this widely used dietary supplement is important not only to address the precise molecular mechanisms involved in its anticancer property, but also to gain critical insights for developing therapeutic entities from this amenable and vital natural source.
To determine the molecular mechanisms of cytotoxic and anticarcinogenic activity of Mentha leaves, six extracts were prepared in sequence of polarity, viz. petroleum ether, benzene, chloroform, ethyl acetate, methanol, and water extracts using a standard protocol. 19 –21 Studies were conducted on 6 different cancer and 2 normal cell cultures of human origin, cervical adenocarcinoma (HeLa), breast adenocarcinoma (MCF-7), T-cell lymphoblast (Jurkat), urinary bladder carcinoma (T24), colon adenocarcinoma (HT-29) and pancreatic adenocarcinoma (MIAPaCa-2), normal lung fibroblast (IMR-90), and kidney epithelial (HEK-293) to address the specific cytotoxic and anticarcinogenic activity of the extracts toward tumor cell types of different lineage. For cytotoxic evaluation, apoptotic index and internucleosomal DNA fragmentation was studied using all extracts. With extracts that showed greater cytotoxic potential, anticarcinogenicity was assessed by mitochondrial membrane potential assay, estimation of active caspase-3, DNA cell cycle analysis, p53 and p21 western blot, quantification of Bax gene expression, induction of cellular senescence, reactive oxygen species, status of antioxidant defense enzyme glutathione reductase, and pro-inflammatory cytokine response.
Materials and Methods
Reagents
To quantify cytotoxicity, Annexin-V Fluos and Apoptotic DNA ladder kit from Roche Applied Sciences, Mannheim, Germany, was used. For evaluation of anticarcinogenicity, incidence of mitochondrial-mediated apoptosis was studied by MitoScreen JC-1 kit and Active caspase-3 PE kit from BD Biosciences, San Diego, California. Determination of ROS was performed by CM-H2DCFDA from Molecular Probes, Invitrogen Co, Carlsbad, California. Quantification of glutathione reductase (GR) activity was performed by using GR ELISA assay kit from Trevigen Inc, Gaithersburg, Maryland. Analysis of secreted cytokine levels was performed using Human inflammation multiplex cytometric bead array (CBA) assay kit from BD Biosciences. DNA cell cycle analysis was investigated using BD Cycle TEST PLUS DNA Reagent Kit, BD Biosciences. Expression of p53 and p21 through western blot was performed using antibody obtained from Santa Cruz Biotechnology Inc. (Santa Cruz, California). For relative gene expression analysis by quantitative real-time PCR, RNA isolation was done using Trizol reagent (Invitrogen Co) and Transcriptor One-step RT-PCR kit with SYBR green dye was used from the kit obtained from Roche Applied Sciences, Mannheim, Germany. Cellular senescence assay was performed using senescence-associated β-gal (SA-β-gal) staining kit procured from Millipore (Billerica, Massachusetts). Plant extracts were prepared using high pure grade solvents (99.98%) procured from Merck Ltd, Mumbai, India.
Cell Culture
Six different human cancer cell lines, HeLa (cervical adenocarcinoma), MCF-7 (breast adenocarcinoma), Jurkat (T-cell lymphoblast like cell line), T24 (urinary bladder carcinoma), HT-29 (colon adenocarcinoma), and MIAPaCa-2 (pancreatic adenocarcinoma) along with 2 normal human cell lines IMR-90 (lung fibroblast) and HEK-293 (kidney epithelial) were obtained from ATCC (Manassas, Virginia) and seeded at the density of 2 × 105 cells/60 mm culture dish (BD Discovery Labware, Bedford, Massachusetts) as per ATCC’s catalogue instructions. All cell lines used in the investigation were positive for wild type p53 status. Cells were incubated at 37°C in a humidified atmosphere of 95% air and 5% CO2. After optimum confluence, cells were treated with the drugs. At the onset of the experiments, the cells were at an exponential and asynchronous phase of growth. Untreated cells were considered as controls.
Study Design
Studies (n = 3) were conducted in 2 sections: dose-dependent and time-course kinetics. Dose-dependent response of plant extracts on different human cancer and normal cell cultures were conducted with concentration 1 μg/μL at 1×, 10×, and 100×, whereas time course experiments were performed with constant concentration of 1× and 10× at time intervals ranging from 1 to 48 hours.
Extract Preparation
M piperita leaves were locally procured, shade dried, and coarsely powdered. Extraction was performed using hot-soxhalation protocol. 19 –21 The powdered material (100 g) was soxholated using 500 mL of petroleum ether, benzene, chloroform, ethyl acetate, methanol, and water, each carried out separately. The extracts were filtered through double layer of 100 µm nylon wire mesh and concentrated under reduced pressure. The dried residue was free of solvents and weighed approximately 2 g for each extract. For investigations, the dried residue was dissolved in phosphate-buffered saline (pH 7.0) and studies were conducted with concentration 1×, 10×, and 100×, whereas time course experiments were performed with constant concentration of 1× and 10× at time intervals ranging from 1 to 48 hours. The extracts were not further characterized.
Evaluation of Cytotoxicity
Annexin-V-FITC/PI assay
Measurement of apoptotic index was performed using Annexin-V-FITC/PI assay. A sum total of 10 000 events were acquired in HI mode and stained cells were subjected to analysis by Cell-Quest software (BD-IS, San Jose, California) using BD FACS Calibur flow cytometer. 22
Analysis of internucleosomal DNA fragmentation
To determine internucleosomal DNA fragmentation (ladder), cells following treatment were washed twice in PBS, resuspended in 200 μL PBS; DNA was isolated as previously described. 23 DNA was eluted in prewarmed elution buffer by centrifugation at 8000 rpm for 1 minute. The samples were dissolved in 15 μL of loading buffer and were subjected to electrophoresis in 1% agarose gel.
Assessment of Anticarcinogenic Potential
Detection of mitochondrial membrane potential
Mitochondrial membrane potential of the cells was detected by staining cells with JC-1 (5, 5’, 6, 6’-tetrachloro-1,1’,3,3’-tetraethylbenzimidazolcarbocyanineiodide) through flow cytometry. The gate was applied in the FSC/SSC dot plot to restrict the analysis to cells only. The ratio of FL1/FL2 cell-associated fluorescence was evaluated for 10 000 events recorded in HI mode. 23
Estimation of active caspase-3
The activity of caspase-3 was measured by washing the cells with cold 1 × PBS and then resuspending in BD cytofix/cytoperm solution at a concentration of 1 × 106 cells/mL followed by incubation of 20 minutes on ice. The cells were harvested, washed, followed by incubation with antibody for 30 minutes at room temperature. The cells were washed and analyzed by flow cytometry using FL2 channel. 23
Analysis of DNA cell cycle arrest
Cellular DNA content was determined by flow cytometric analysis of propidium iodide (PI)-labeled cells. Sum total of 30 000 events were acquired in LO mode and PI stained cells were subjected to flow cytometric analysis. Histogram displays were overlaid with graphical representations of the modeled G0/G1, S, and G2/M populations and data were expressed as percentage of cells for any given phase of the cell cycle. 24,25
Cell cycle regulation through western blot analysis
Briefly, cells were rinsed twice in PBS and lysed in the buffer (10% SDS, 1 mol/L Tris pH-7.6, 5 mmol/L EDTA). The obtained cell lysates were centrifuged at 12 000 rpm for 10 minutes at 4°C, and the supernatant was collected. Protein concentrations were determined from the supernatant by Bradford assay. An amount of 100 µg protein was analyzed through 10% SDS-PAGE and transferred onto nitrocellulose membrane in 25 mmol/L Tris, 194 mmol/L glycine, and 20% methanol at 4°C with semidryer transfer unit. Analysis of p53 and p21 proteins was performed as reported previously. Quantitation of the p53 and p21 signals was done by using Quanti One software, Bio-Rad, Philadelphia, Pennsylvania. 26
Relative gene expression analysis by quantitative real-time PCR
For gene expression analysis, total RNA, from treated and control groups of cells at 6, 12, and 24 hours posttreatment with constant concentration of 10×, was isolated using Trizol reagent and was quantified spectrophotometrically. Purity and integrity of RNA was verified by ethidium bromide staining following resolution in 1% agarose gels. Transcriptor One-step RT-PCR with SYBR green dye was performed as described elsewhere. 26 Primers for Bax was synthesized from TIB MOLBIOL Universal Probe Library, GmbH·Eresburgstrasse, Berlin, Germany. Primers for Bax gene were as follows: forward sequence 5′ GGGTGGTTGCCCTTTTCTACT 3′ and reverse sequence 3’CCCGGAGGAAGTCCAGTGTC5′, with corresponding housekeeping gene sequence (GAPDH) being forward 5′GTATTGGGCGCCTGGTCACC3′ and reverse 3′ CGCTCCTGGAAGATGGTGATGG 5′.
Cellular senescence assay
Cellular senescence assay was performed through senescence-associated β-gal staining (SA-β-gal) as described in our previous studies, and stained cells were examined under a phase contrast microscope. 27 Persistence of DNA damage in senescent cells was determined through γ-H2AX foci formation. 28
Assay for intracellular ROS
A fresh stock solution of CM-H2DCFDA (5 mmol/L) was prepared in DMSO and diluted to a final concentration of 1 μmol/L in 1× PBS. The cells were washed with 1× PBS followed by incubation with 50 μL of working solution of fluorochrome marker CM-H2DCFDA (final working concentration adjusted to 2.5 μg/50 μL) for 2 hours. The cells were harvested, washed in PBS, and cell-associated fluorescence was measured by flow cytometry in FL1 channel. 27
Estimation of GR activity
Levels of the antioxidant defense system enzyme, GR, was measured at 340 nm in accordance with our previous studies through ELISA. 23
Inflammatory cytokines
Supernatants collected from cultures were subjected to measuring inflammatory response by determining levels of cytokines, interleukin (IL)-6, IL-12p70, IL-1β, tumor necrosis factor (TNF), IFN-γ, and IL-8, and the assay was performed as per the manufacturer’s instructions. Data acquisition and analysis were carried out on a flow cytometric platform using BD CBA software. 29
Statistical Analysis
Statistical analysis was performed by using analysis of variance (ANOVA). The Statistical Package for Social Sciences (SPSS) software package (SPSS Inc., Chicago, Illinois) was used to perform statistical analysis and a P value of ≤.001 was considered to be significant.
Results
Evaluation of Cytotoxicity
Apoptotic index
Exposure of phosphatidyl serine (PS) on the cell surface is a conserved feature of apoptosis and annexin-V is a Ca2+-dependent phospholipid-binding protein with high affinity for PS. This protein is used as a sensitive probe for PS exposure upon the outer leaflet of the cell membrane and suitable for detection of apoptotic cells. The number of apoptotic cells was incremental with increase in dose of Mentha extracts (Figure 1 ). However, among all the extracts tested, chloroform and ethyl acetate extracts of Mentha leaves showed significant higher apoptotic index following 6 hours treatment in a dose-dependent manner. All 6 cell lines tested in this study stained positive for annexin-V binding, showing statistically identical apoptosis following treatment with both chloroform and ethyl acetate extracts of Mentha leafs and no differential cytotoxicity was observed (Figure 1). As compared to cancer cell lines, no significant changes were observed in IMR-90- and HEK-293-treated cells (data not shown).
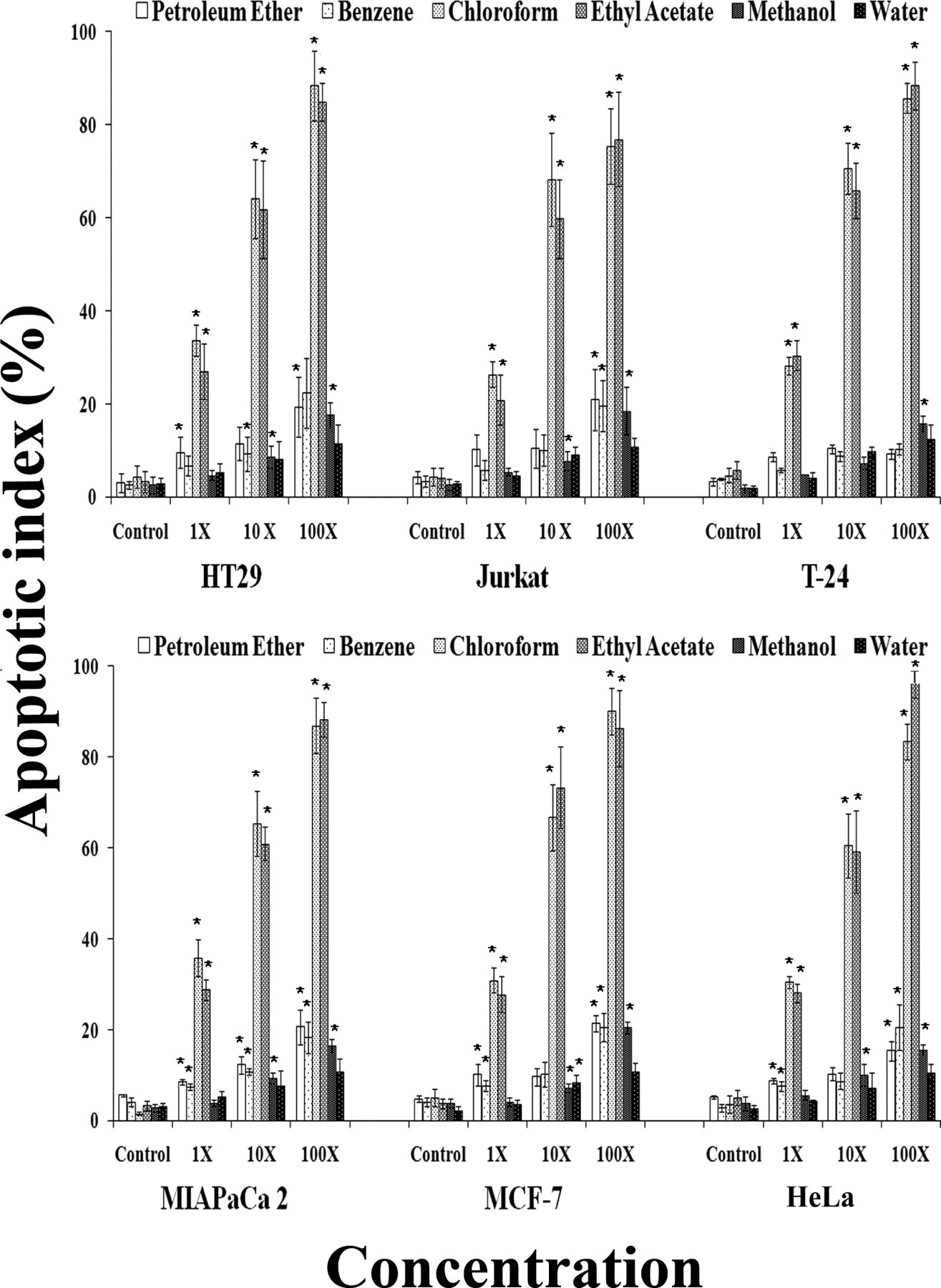
Induction of apoptosis by Mentha piperita leaf extracts in human cell lines. Histogram depicting the apoptotic index in cultured human cancer cell lines after treatment with Mentha extracts at 1× (1 µg/µL), 10× (10 µg/µL), and 100× (100 µg/µL) concentration at 6 hours. The data shown is representative of mean of 3 independent experiments. *P ≤ .001.
DNA fragmentation
Analysis of DNA fragmentation in the form of a ladder detected on agarose gel electrophoresis was observed uniformly in all cell lines treated with chloroform and ethyl acetate extract of Mentha leaves.
Assessment of Anticarcinogenic Potential
Mitochondrial membrane depolarization
Effect of Mentha leaf extracts on mitochondrial transmembrane potential (Δψ) was evaluated by membrane-permeable lipophilic cationic fluorochrome JC-1 (5, 5′, 6, 6′-tetrachloro-1, 1′, 3, 3′-tetraethylbenzimidazolcarbocyanine iodide). JC-1 penetrates into cells and its fluorescence is a reflection of Δψ. This assay showed a trend similar to that observed in annexin-V/PI assay, which was dose-dependent. Upon treatment, chloroform and ethyl acetate extracts showed depolarization of the mitochondria (loss of Δψ) and highest activity with chloroform extract was observed in T24 cells (58.3 ± 4.11) and ethyl acetate-treated HeLa cells (64.2 ± 8.57) at 6 hours (Figure 2a ).
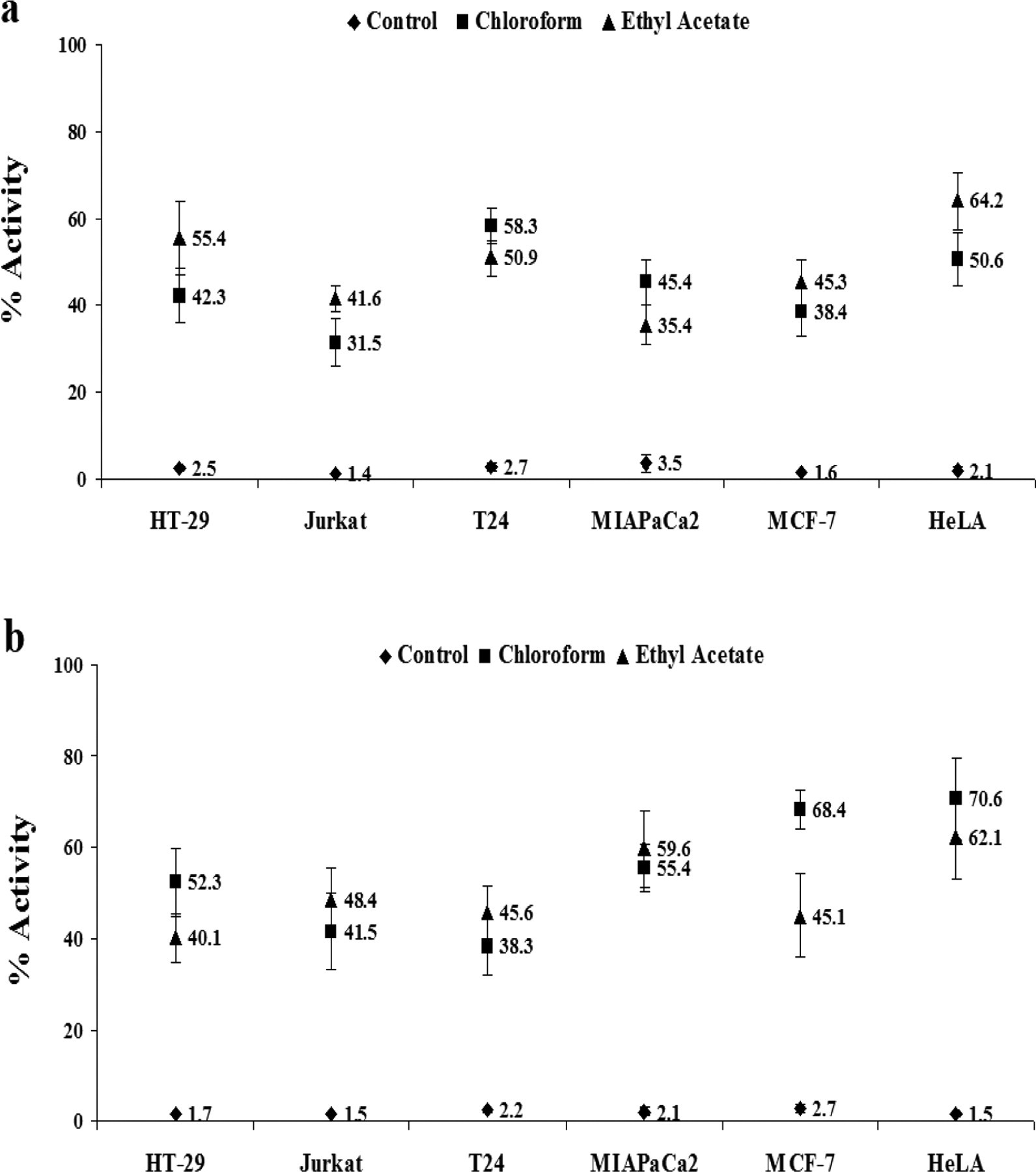
Effect of Mentha piperita leaf extract on mitochondrial-mediated apoptotic pathway. Chloroform and ethyl acetate extracts of Mentha leaves at 10× concentration induced significant depolarization of mitochondria membrane potential (a); activation of caspse-3 (b) in human cancer cell lines after 6 hours of treatment.
Activation of caspase-3
Caspase-3-mediated proteolysis is a critical element of the apoptotic process mediated through mitochondria. Active caspase-3 comprises a heterodimer of 17- and 112-kDa subunits, which in turn are derived from a 32-kDa proenzyme, a marker for cells undergoing apoptosis. Active caspase-3 proteolytically cleaves and activates other caspases and relevant targets in the cytoplasm. Maximum activation of caspase-3 was observed in HeLa cells following 6 hours treatment with fixed concentration (10×) of chloroform and ethyl acetate extracts (Figure 2b).
Cell cycle arrest
The profile of the DNA content of the chloroform and ethyl acetate extract-treated cells was obtained using flow cytometric analysis to measure the fluorescence of PI-DNA binding and the stage at which these extracts induced growth inhibition. Figure 3 depicts exposure to extracts resulted in progressive and sustained accumulation of cells in the G1 phase at 1 hour and 3 hours. Furthermore, a sub-G1 peak in treated cells indicative of inception of apoptosis at 6 and 12 hours after treatment was noticed (Figure 3).
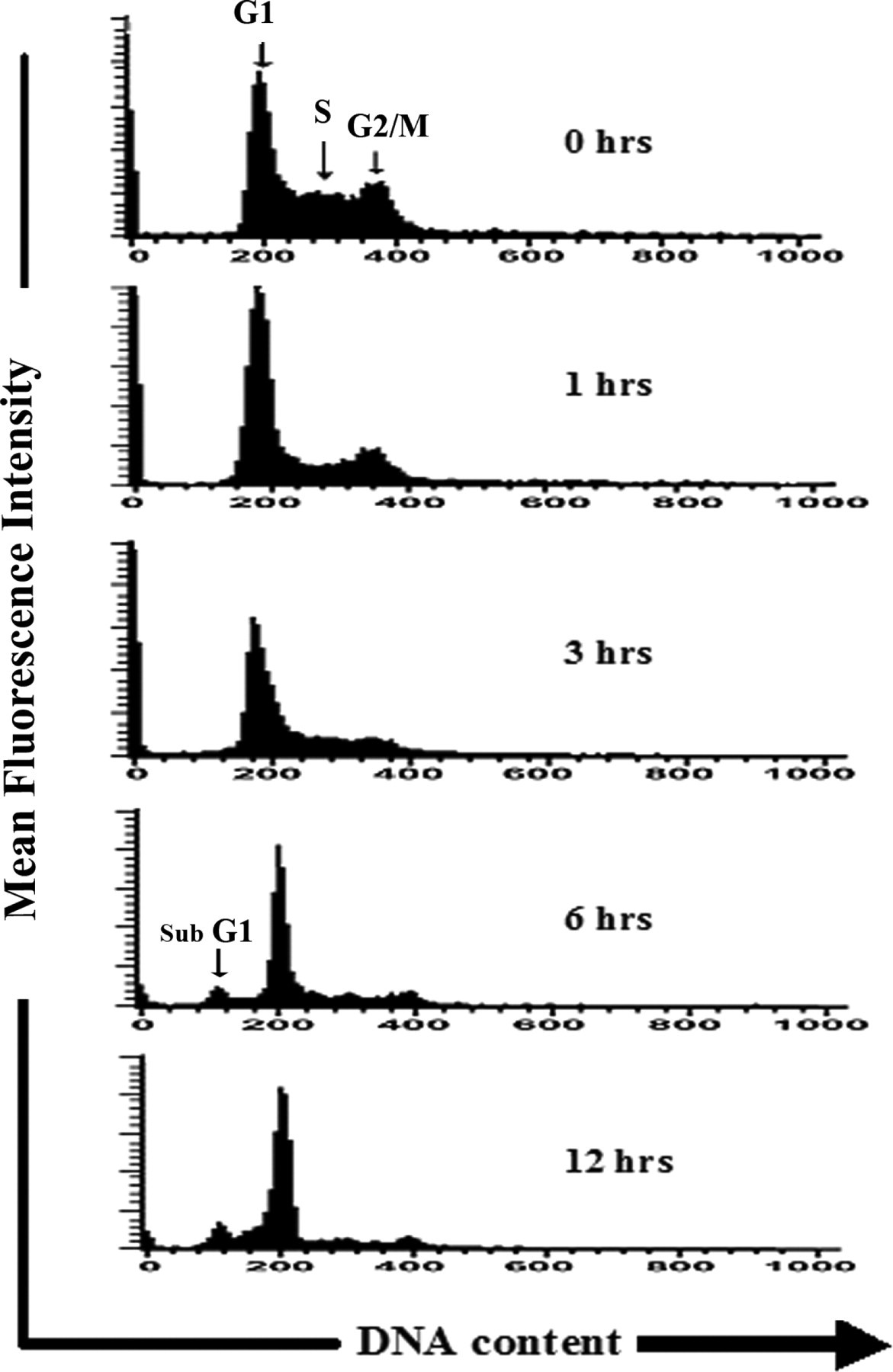
DNA cell cycle analysis through flow cytometry in MIAPaCa-2 cells. Cells were monitored at 1, 3, 6, and 12 hours following treatment with 10× chloroform extract of Mentha piperita.
Cell cycle regulation
In order to examine the molecular mechanisms and underlying changes in cell cycle patterns, we investigated the effects of cell cycle regulatory proteins. Western blot analysis revealed upregulation of p53 and p21 proteins along the time course in chloroform and ethylacetate extracts-treated cells up to 24 hours of observation period (Figure 4 ).
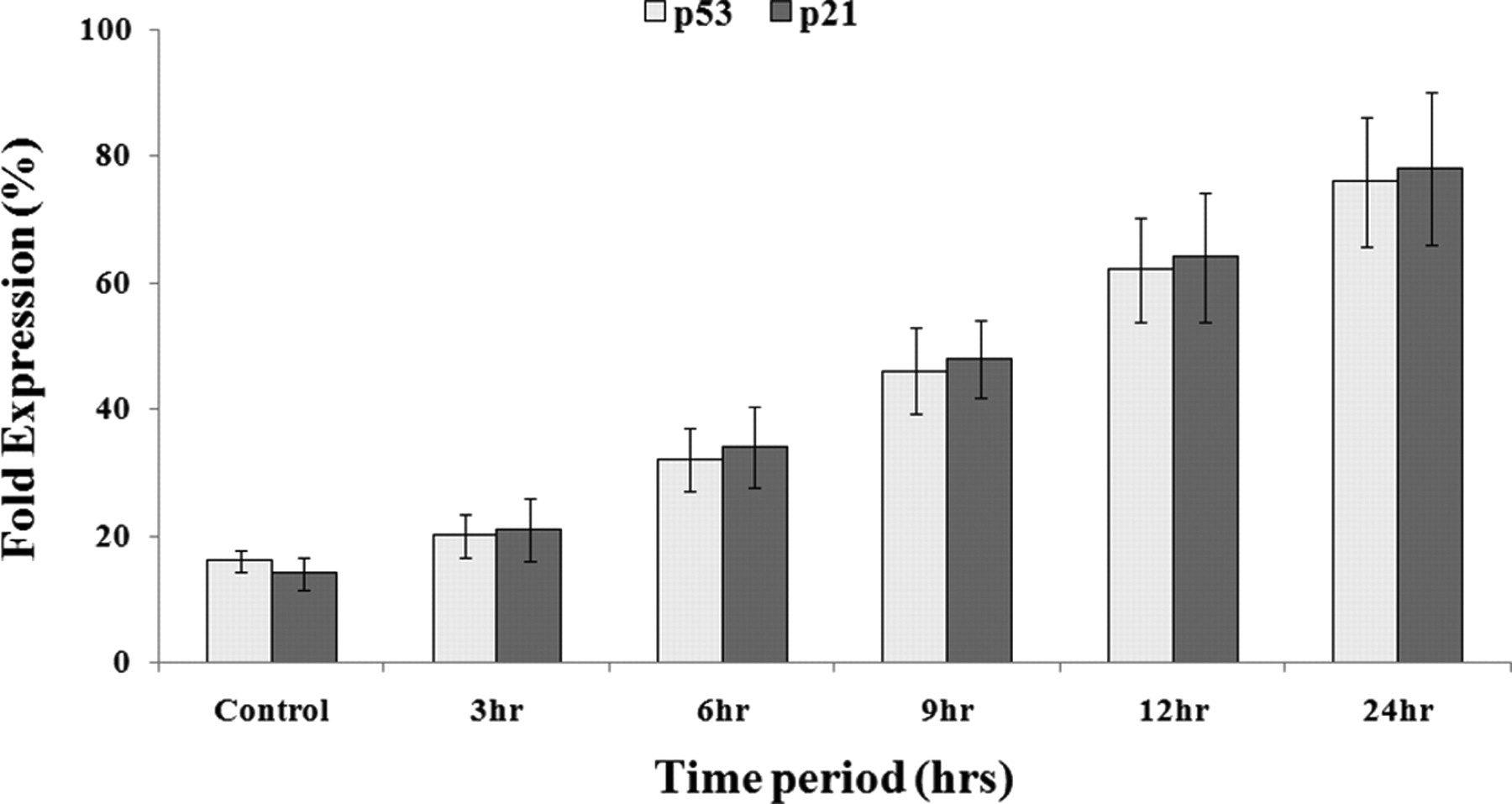
Status of cell cycle regulatory proteins in HeLa cells. (A) Western blot analysis of p53 and p21proteins in control and treated cells after exposure to 10× ethyl acetate extract of Mentha piperita. observed at 1 to 24 hours.
Bax gene expression
Quantification of pro-apoptotic gene, Bax, displayed escalation in its expression along the time course. Of all extracts tested, the maximum impact was shown by ethyl acetate and chloroform extracts at 6 hours and 24 hours, with a 9.7- and 10.1-fold, respectively, increase at 10× concentration (Table 1 ).
Relative Expression of Bax Gene in MCF-7 Cells After Treatment With 10× Concentration of Mentha Leaf Extracts at Different Time Course
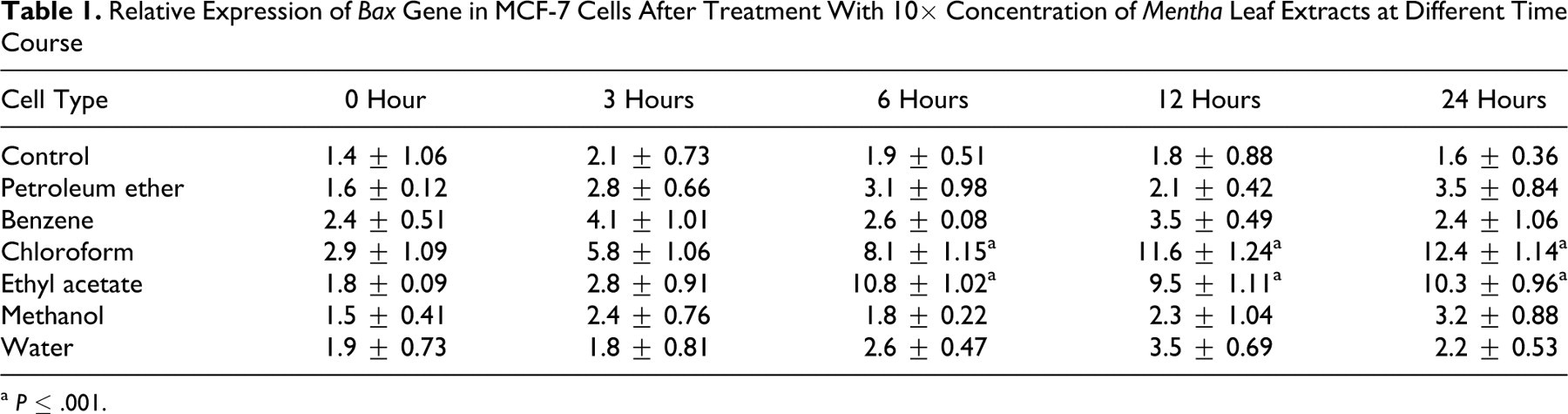
a P ≤ .001.
Senescence
Cellular senescence assay performed in all the treated cells demonstrated significant morphological changes after 12 and 24 hours treatment with 1× concentration. Induction of senescence after 48 hours of treatment through perceptible increase in cell size, along with acquisition of distinct flat morphology and γH2AX foci, was evident in the treated cells with incremental SA-β-gal staining pattern (Figure 5 ).
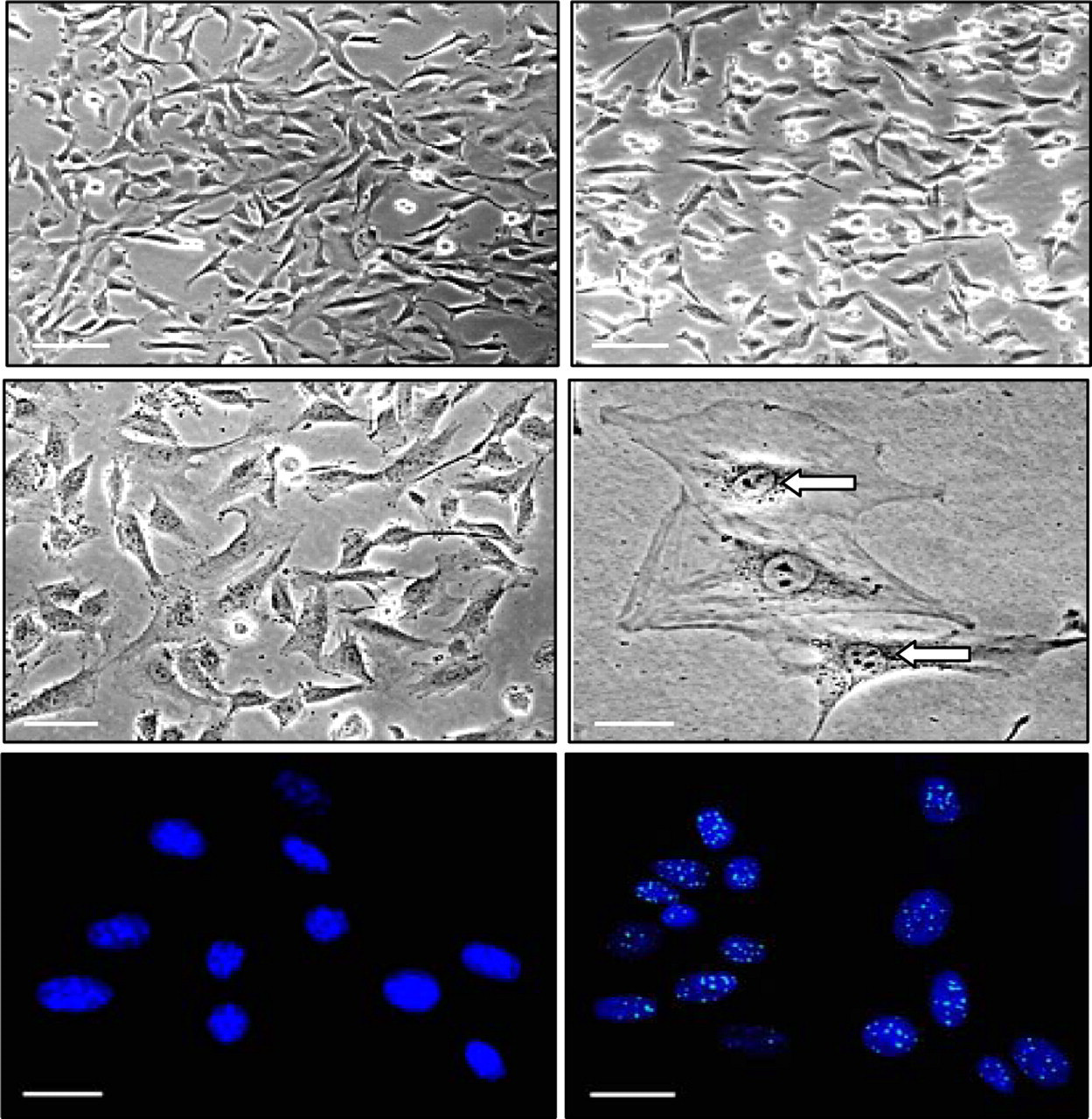
Morphological changes and senescence induction by Mentha piperita extracts. Representative phase contrast microphotographs (×200) of T24 cells prior to (upper left panel) and after treatment with 1× chloroform extract of Mentha piperita at 12 hours (upper right panel), 24 hours (middle left panel), and after 48 hours (middle right panel). Treated cells showing persistence of γH2Ax foci at 48 hours (lower right panel) with respect to control cells (lower left panel).
Oxidative stress
The production of intracellular ROS was measured by DCFH oxidation in cells treated with chloroform and ethyl acetate extracts of Mentha leaves with dosage of 10× at 6 hours. A significant increase in the H2O2 generation, as indicated by DCF fluorescence, was recorded in all cell types studied in comparison to respective controls. Notably, the ethyl acetate extract exerted its maximum effect in generation of ROS in MIAPaCa-2 cells with values being 41.6 ± 8.26, while chloroform extract demonstrated highest effect in MCF-7 cells with 28.4 ± 3.41 (Figure 6 ).
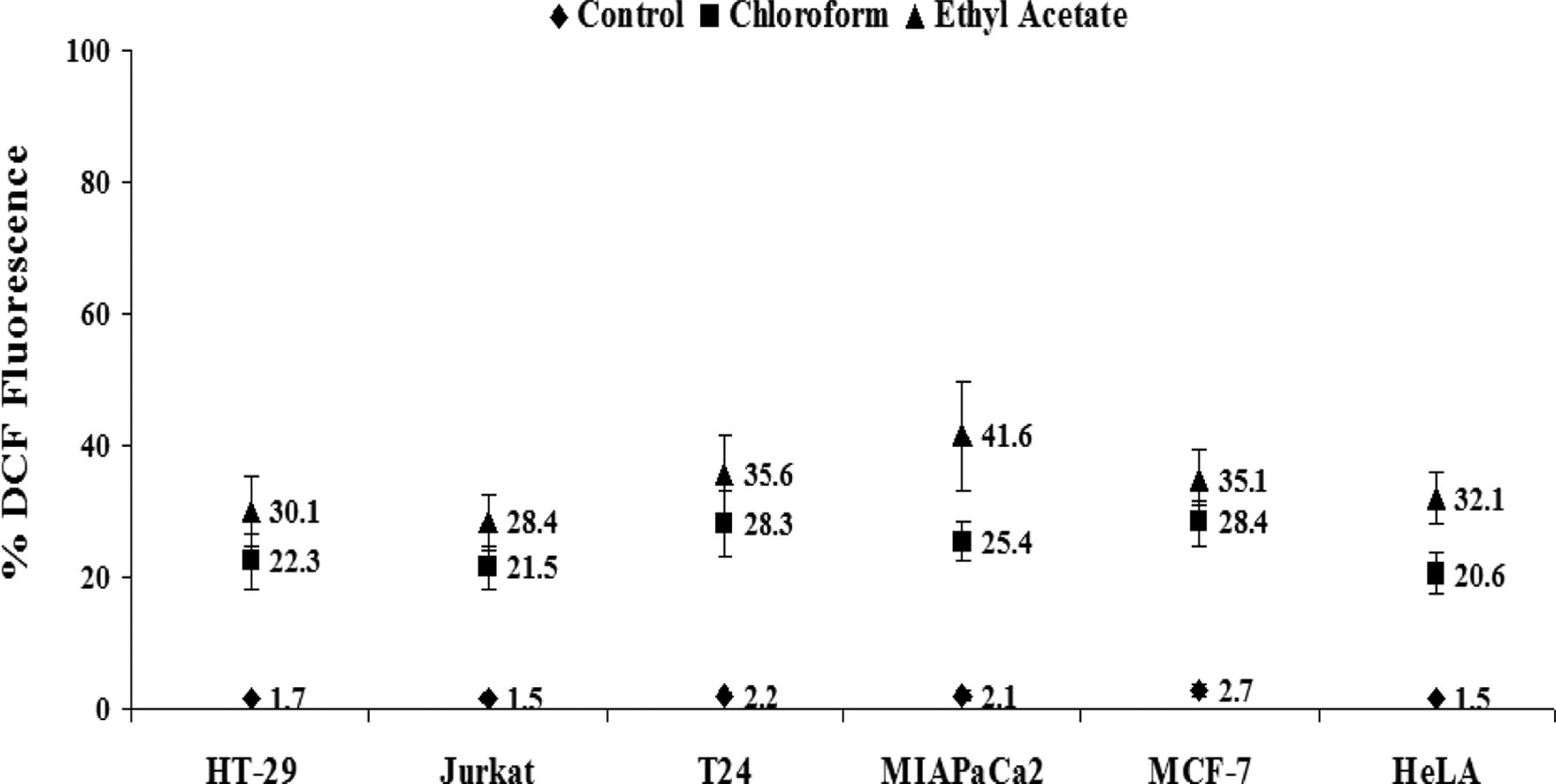
Mentha piperita extracts induces ROS generation. Generation of ROS [CM-H2DCFDA] in different human cancer cell lines after treatment with chloroform and ethyl acetate extracts of Mentha leaves at 10× concentration for 6 hours.
Depletion of GR activity
In order to determine the status of antioxidant defense system in treated cells, we measured the activity of antioxidant enzyme glutathione reductase. A significant decline in GR activity was recorded in all cell lines treated with 10× concentrations of chloroform and ethyl acetate extract. Both ethyl acetate and chloroform extracts caused the maximum inhibition in the GR activity relatively in MIAPaCa-2 and T24 cells with respect to controls (Figure 7 ).
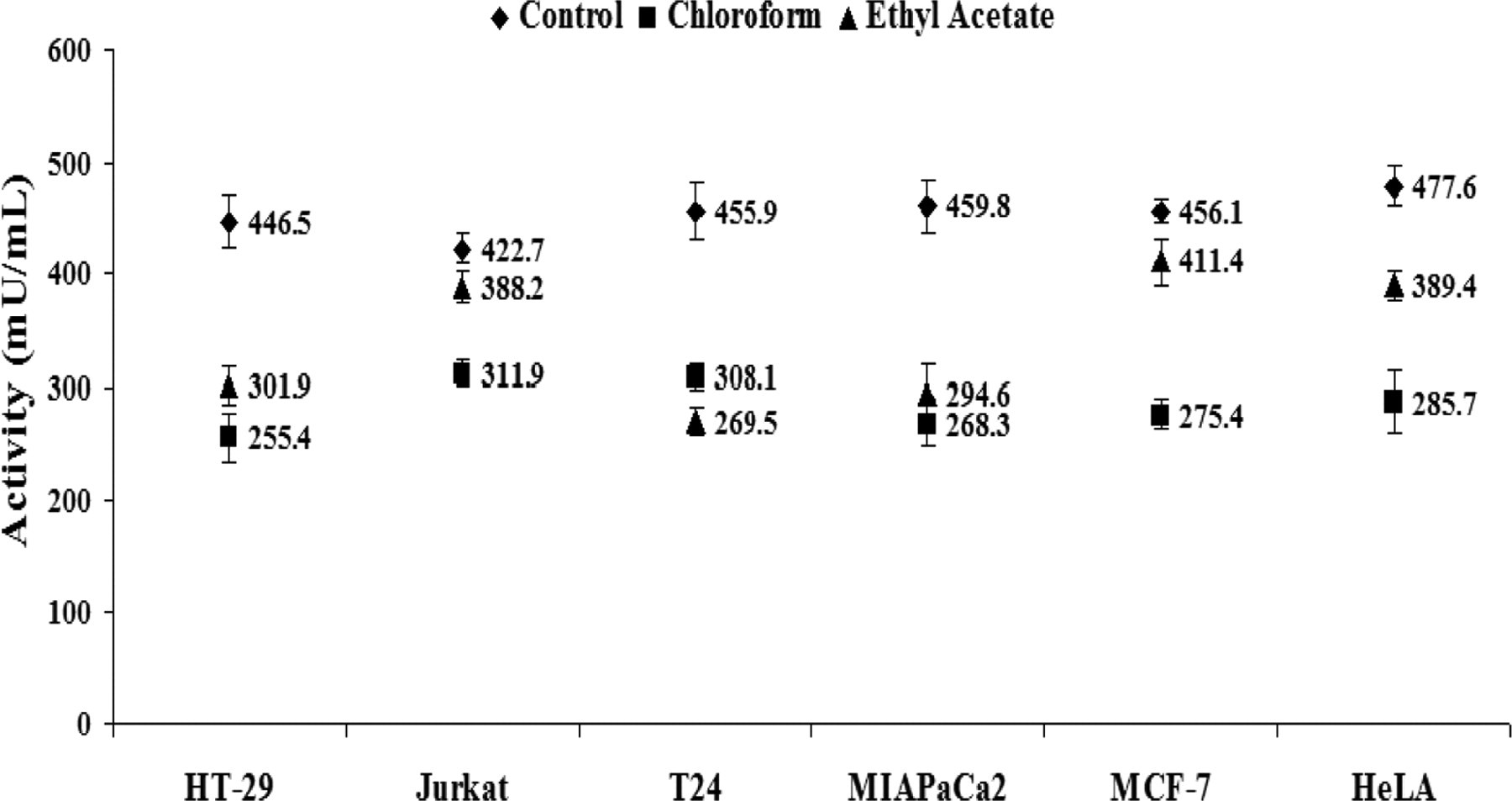
Depletion of antioxidant defense by Mentha piperita extracts. Graph showing depletion in the activity of antioxidant defense states enzyme glutathione reductase (GR) in human cancer cell lines after 6 hours treatment of chloroform and ethyl acetate extracts.
Inflammation
Multiplex CBA assay for human inflammatory cytokines displayed an increase in levels of pro-inflammatory cytokines, IL-6, IL-12p70, IL-1β, TNF, IFN-γ, and IL-8 in culture supernatant of all the cell lines tested with chloroform and ethyl acetate extracts (10×) of Mentha leaves. An increase by nearly 2-fold in IL-12p70 and IL-6 and nearly 3-fold in IL-8 and IFN-γ levels was observed. Interassay and intraassay variability was negated since the assay involved independent antigen-antibody interaction for each cytokine. Representative data for increase in levels of cytokines secreted in culture supernatant at 6 hours following exposure to chloroform and ethyl acetate extract (10×) has been shown (Figure 8 ).
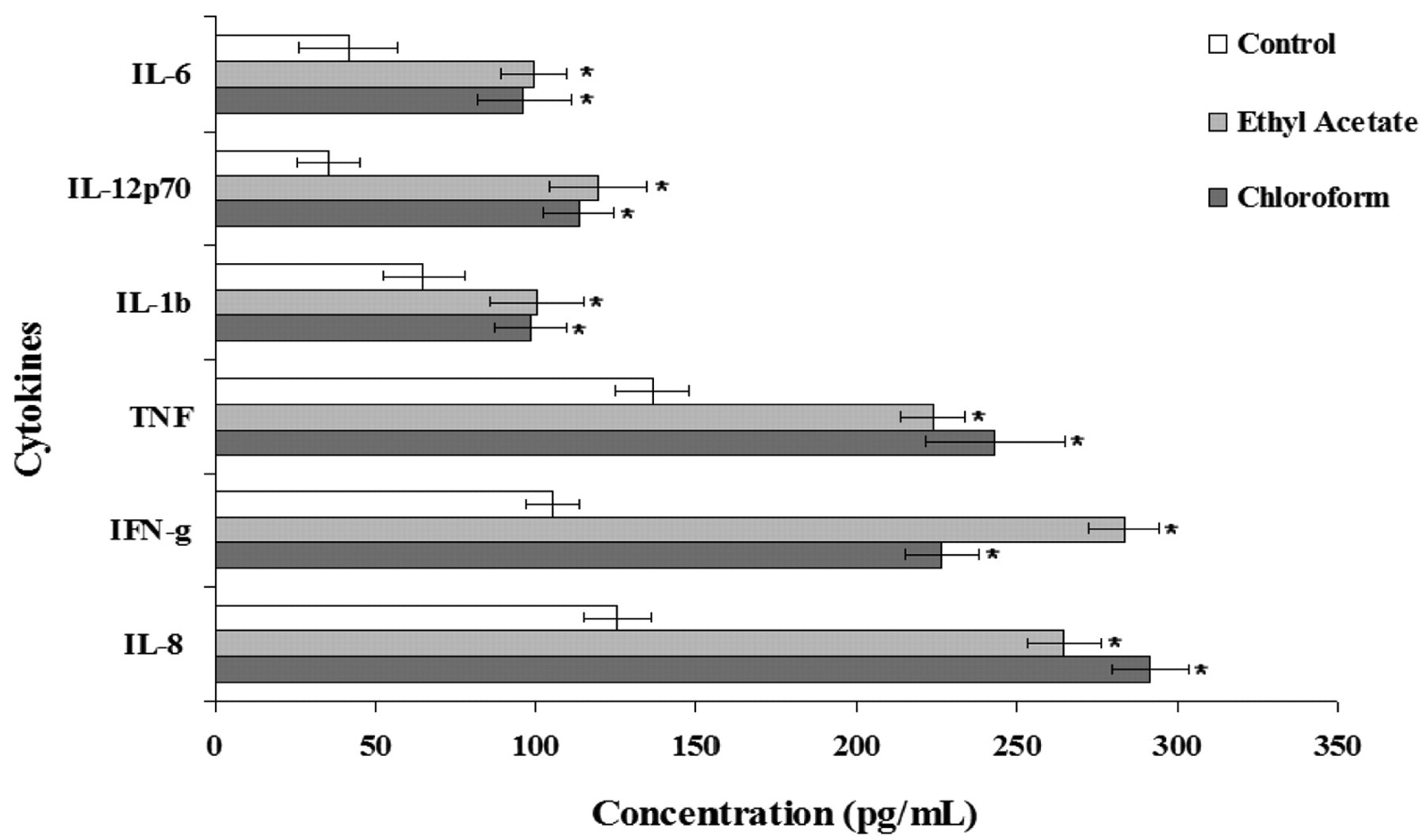
Inflammatory cytokine response in Jurkat cells. Effect of chloroform and ethyl acetate extracts (10× concentrations) of Mentha piperita leaves on proinflammatory cytokine response evaluated in cell culture supernatant. Values are mean ± SE from 3 independent experiments conducted in duplicate. *P ≤ .001.
Discussion
Phytochemicals have been used for treating various human diseases since time immemorial. The use of complementary and alternative medicine such as herbal extracts is becoming increasingly popular among patients with cancer. 30 Typically, herbal medicines emphasize the use of whole extracts from a plant mix or from complex formulations. 31 Anticancer agents may alter regulation of the cell cycle machinery, resulting in cellular arrest at different phases of the cell cycle and, thereby, reducing the growth and proliferation of, and even inducing apoptosis in, cancerous cells. The present research documents the effect of Mentha extract treatment in vitro in a cell culture model involving human cancer and normal cell lines. Our results show that chloroform and ethyl acetate extracts of M piperita leaves can act as a potential natural anticancer agent inducing cellular apoptosis through mitochondria mediated pathway, inhibition of proliferation, perturbation of oxidative balance, upregulation of Bax gene, elevated expression of p53 and p21 in the treated HT-29, Jurkat, T24, MCF-7, MIAPaCa-2, and HeLa cells with significant arrest in G1 phase, while at the same time scaling up proinflammatory cytokines (IL-8, IFN-γ, TNF, IL-1β, IL-12p70, and IL-6) levels.
Apoptosis has been well characterized by a variety of hallmark events, including rapid reduction in cellular volume, chromatin condensation, and internucleosomal DNA cleavage. 32 Induction of apoptosis is suggested to be one of the major modes of action of chemotherapeutic anticancer drugs on malignant cells. 33,34 Decreased cell viability in the tumorogenic cells has been accounted by previous studies for the action of medicinal herbs and inducing apoptotic activity through an early redistribution of plasma membrane phosphatidylserine. 35,36 In the present investigation of all the extracts examined, significant cytotoxic activity was shown by chloroform and ethyl acetate extracts at 1×, 10×, and 100× concentrations uniformly in all the cancer cell types studied (Figure 1). No significant apoptotic index was recorded in normal human cell lines, IMR-90 and HEK-293, on treatment with Mentha leaf extracts suggesting their nontoxic nature. Selective inhibition in proliferation of cancer cells by chloroform and ethyl acetate extracts might be attributed to the active constituents such as terpenoids and alkaloids reported to be present in similar organic extracts of different Mentha species. 37,38 Apoptotic DNA ladder pattern observed in cells treated with chloroform and ethyl acetate extracts also showed a similar trend. Depolarized Δψ with simultaneous activation of caspase-3 further corroborate the stronger anticarcinogenic potential of chloroform and ethyl acetate extracts of M piperita through mitochondrial-mediated pathway (Figure 2a and b).
Regardless of peripheral mediators, the behavior of a cell is ultimately dictated by its genetic profile. Thus, investigating changes in gene expression profiles as a result of herbal treatment for cancer may help define the underlying mechanisms of action and validate their efficacy as anticancer herbs. In many human cancers, the proapoptotic proteins, like Bax, have reduced expression. 39 Therefore, search of new cytotoxic agents that can increase Bax expression or restore the ability of tumor cells to undergo apoptosis are imperative. Our data demonstrate upregulation of proapoptotic gene Bax following treatment with chloroform and ethyl acetate extracts of Mentha leaves. Both the extracts displayed similar kind of expression of Bax genes in the treated cells (Table 1).
Although the exact mechanism for activating the expression and function of Bax is not fully understood, p53 molecule is thought to play a major role in this process. 40 Incidentally, extracts from Lamiaceae family have shown their potential as anticancer agents by virtue of activation of cell death signals through inhibition of growth and a G1 phase arrest of the cell cycle along with p53 and p21 upregulation in cancerous cells. 16 –18 Our present study followed suit with arrest of cells in G1 phase as early as 1 and 3 hours (Figure 3) and increased expression of p21 and p53 in cells treated with the chloroform and ethyl acetate extract of Mentha leaves (Figure 4).
One approach to control progression of cancers is through growth inhibition by which the disease can be prevented, slowed down, or substantially reversed. To accomplish this, attempts have been made by administration of one or more naturally occurring or synthetic agents. 41 Studies in cancerous cells have shown that bioactive extracts of plant inhibit cell proliferation through alterations in cell cycling, energy metabolism proteins, and ultimately the cancer cell environment. 42,43 Concomitantly, in the present study, with the increase in the duration of exposure, there was a reduction in cell growth with apparent morphological changes in extracts treated HT-29, Jurkat, T24, MIAPaCa-2, MCF-7, and HeLa cells. Their growth was noticeable in a more disperse manner with progressive accumulation of β-gal stain after attaining senescence at 48 hours (Figure 5).
Cancer therapies and cancer progression can increase oxidative stress that might account for drug-induced toxicity in patients with cancer. As a consequence, therapeutic selectivity becomes the most important consideration in cancer therapy to preferentially kill malignant cells while minimizing harmful effects to normal cells, which in turn, depends on our understanding of the biological differences between cancer and normal cells. Malignant cells in general are more active than normal cells in the production of free radicals such as O2
Based on the outcome of our current findings, Mentha leaf extracts merit investigation as antitumor agents for treatment of human cancers. Although chloroform and ethyl acetate extracts demonstrated a universal effect on various human tumor cell lines, however, further studies to characterize the bioactive constituents in these extracts would be most important not only to extend the understanding of the molecular framework of anticarcinogenic activity but also to provide basis for mechanistic and translational studies for designing novel anticancer drugs to be used alone or as an adjuvant for prevention of tumor progression and/or treatment of human malignancies.
Footnotes
Acknowledgment
Authors thankfully acknowledge Mr Naveen Kumar Khare for providing necessary technical assistance.
The author(s) declared a potential conflict of interest as follows: This article is not an official FDA or NCI guidance or policy statement, and no official endorsement by the FDA or NCI is intended.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This research was partially supported by the Grant-in-Aid received from Department of Science & Technology, Govt. of India, New Delhi, to NP.
