Abstract
The efficacy of methotrexate (MTX) as an anticancer drug is limited by some adverse effects including hepatic and renal toxicities. The present study investigated the possible protective effect of protocatechuic acid (PCA), a phenolic phytochemical widely present in several edible vegetables and fruits, on hepatorenal toxicity associated with MTX treatment in rats. Male Wistar rats were randomly assigned to five groups (n = 10), namely control, MTX alone (20 mg/kg), PCA alone (50 mg/kg), and rats that were coadministered MTX and PCA at 25 and 50 mg/kg. The MTX was administered as a single intraperitoneal dose on the first day, whereas PCA treatment lasted 7 days. Results indicated that PCA significantly (p < 0.05) abrogated MTX-mediated elevation in indices of hepatorenal toxicity. Furthermore, PCA protected against MTX-induced decreases in glutathione level and antioxidant enzyme activities as well as the increase in reactive oxygen and nitrogen species and lipid peroxidation in the liver and kidney of the treated rats. Administration of PCA markedly abated MTX-induced increases in interleukin-1β, tumor necrosis factor alpha, and caspase 3 activity in the rats. The biochemical data on the hepatorenal protective effects of PCA were well supported by the histological data. Collectively, PCA protected against MTX-induced hepatorenal toxicity via antioxidant, anti-inflammatory, and antiapoptotic mechanisms.
Introduction
Chemotherapy is a major method in the management of several cancer types. Methotrexate (MTX) is a folate antagonist and cytotoxic chemotherapeutic drug commonly used in the management of malignancies specifically lymphoblastic leukemia, osteosarcoma, and tumors of the head and neck. 1,2 Unfortunately, the efficacy and usefulness of most chemotherapeutic drugs are associated with side effects due to their detrimental effects on both cancerous and normal cells. Specifically, the clinical use of MTX reportedly elicits hematological disorder, myelosuppression, hepatotoxicity, 3 –6 and nephrotoxicity. 7,8 Administration of MTX is associated with renal toxicity because more than 90% of the drug is excreted primarily the kidney. 9 Indeed, acute renal failure and precipitation of MTX and/or its metabolites are often detected in acidic urine from patients. 10 Although the precise mechanisms by which MTX elicits hepatic and renal toxicities are not fully known, induction of oxidative stress, pro-inflammatory cytokines release, and apoptosis have been reported. 11,12 Hence, therapeutic agents capable of regulating oxidative stress, inflammation, and apoptosis without decreasing MTX therapeutic efficiency is essential to abrogate these clinical problems associated with MTX administration.
Protocatechuic acid (PCA, 3,4-dihydroxybenzoic acid) is a simple phenolic compound widely found in many edible nuts, vegetables, and fruits. 13,14 Moreover, PCA has been shown to be the main stable metabolite of phytochemical anthocyanins. 15,16 Epidemiological and experimental data revealed that PCA possesses several beneficial health effects which have been linked to multiple biochemical mechanisms. Previous studies have demonstrated that PCA exhibits antioxidative, anti-inflammatory, and antiapoptotic effects in chemically induced cellular damage in rodents, 17 –19 whereas its antiproliferative and proapoptotic effects, as well as its inhibitory effects on the metastatic capacity of tumor cells, have been demonstrated in vitro and in vivo. 20 –22 PCA reportedly inhibited LPS-stimulated tumor necrosis factor-alpha (TNF-α) and interleukin-1β (IL-1β) secretion in RAW264.7 cells. 23
Up till now, there is no scientific report in the literature on the influence of PCA on hepatorenal dysfunction associated with MTX administration in rats. The present study examined, for the first time, the effectiveness of PCA as a chemoprotective agent against MTX-induced hepatorenal injury in adult Wistar rats. To achieve this, we evaluated oxidative stress biomarkers, antioxidant enzyme activities, and levels of inflammatory mediators along with histopathological examination of the liver and kidney to further gain mechanistic understanding of how PCA probably modulates hepatorenal dysfunction associated with administration of anticancer drug MTX in rats.
Materials and methods
Chemicals
MTX was procured from the Morningside Healthcare Limited, Leicester, UK. PCA (≥97% pure), epinephrine, thiobarbituric acid, glutathione (GSH), hydrogen peroxide (H2O2), 1-chloro-2,4-dinitrobenzene, and 5′, 5 ′-dithiobis-2-nitrobenzoic acid were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). Enzyme-linked immunosorbent assay (ELISA) kits for the assessment of IL-1β, TNF-α, and caspase 3 (CASP3) activities were purchased from E-labscience Biotechnology Company, Beijing, China.
Care of animal
Fifty sexually matured male Wistar rats (8 weeks old, 168 ± 7 g) obtained from the Experimental Animal Unit, Faculty of Veterinary Medicine, University of Ibadan, Ibadan, Nigeria, were used for the present study. The animals were kept in plastic cages situated in a well-ventilated house and provided rat chows and water ad libitum. The rats were subjected to natural photoperiod of 12-h light:12-h dark and adequately cared for according to the conditions indicated in the “Guide for the Care and Use of Laboratory Animals,” as published by the National Institute of Health. Besides, the study was performed following approval by the University of Ibadan Ethical Committee and in accordance with the US NAS guidelines.
Experimental design
Subsequent to 1 week of acclimatization, the experimental animals were randomly assigned to five groups of 10 rats each and were treated as follows: Control: The rats orally received 2 ml/kg of normal saline.
The doses of MTX and PCA used in the present study were chosen from our pilot studies and earlier published studies. 10,19 Specifically, 20 mg/kg of MTX was chosen from earlier studies and confirmed to cause hepatorenal toxicity within 7 days of a single intraperitoneal injection to rats, whereas 25 and 50 mg/kg PCA were the highest effective doses among the various doses (5, 25, 50, 100, and 200 mg/kg) investigated in our preliminary studies (data not shown). Blood samples were obtained from retro-orbital venous plexus using plain tubes before the animals were sacrificed by cervical dislocation on day 8. Subsequently, serum samples were prepared by centrifuging the clotted blood at 3000 g for 10 min. The serum samples were then kept frozen at −20°C until needed for the liver and kidney function analysis.
Biomarkers of hepatic and renal toxicity assessment
Serum assessment of aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), alkaline phosphatase (ALP), urea, and creatinine levels were performed using available commercial kits from Randox Laboratories Limited (UK).
Assessment of hepatic and renal oxidative stress levels
The liver and kidney samples of the experimental rats were homogenized in 50 mM Tris–HCl buffer (pH 7.4). Subsequently, the homogenate was centrifuged at 12,000 × g for 15 min at 4°C to obtain the supernatant, which was used for the biochemical analyses. Hepatic and renal protein concentration was evaluated according to the method described by Bradford. 24 Lipid peroxidation (LPO) was evaluated according to the method described by Farombi et al. 25 Superoxide dismutase (SOD) activity was evaluated according to the method described by Misra and Fridovich, 26 whereas catalase (CAT) activity was evaluated according to the method described by Claiborne. 27 GSH level was evaluated according to the method described by Jollow et al., 28 glutathione-S-transferase (GST) activity was evaluated according to the method described by Habig et al., 25,29 whereas glutathione peroxidase (GPx) activity was evaluated according to the method described by Rotruck et al. 30
Quantification of RONS level
The level of hepatic and renal reactive oxygen and nitrogen species (RONS) in the experimental rats was analyzed using established protocol, which is based on the RONS-dependent oxidation of 2′,7 ′-dichlorodihydrofluorescin diacetate (DCFH-DA) to 2, 7-dichlorofluorescein (DCF). 31 Briefly, the reaction mixture consisted of 150 µL of 0.1 M potassium phosphate buffer (pH 7.4), 10 µL of the sample, 35 µL of distilled water, and 5 µL of DCFH-DA (200 µM and final concentration 5 µM). The fluorescence emission of DCF due to DCFH-DA oxidation was analyzed at 488 nm excitation and 525 nm emission wavelengths for 10 min at 30 s intervals using a SpectraMax plate reader (Molecular Devices, San Jose, California, USA). The rate of DCF formation was then expressed in percentage of control group.
Assessment of biomarkers of inflammation and CASP3 activity
The level of hepatic and renal nitric oxide (NO) was assayed according to the method described by Green et al. 32 Briefly, the reaction mixture consisting of equal volume of sample and Griess reagent was incubated for 15 min and the absorbance was measured at 540 nm. The level of NO in the sample was subsequently quantified by extrapolation using the standard curve. Myeloperoxidase (MPO) activity was assayed according to established protocol. 33 In addition, hepatic and renal IL-1β and TNF-α levels, as well as CASP3 activity, were assayed using ELISA kits (Elabscience Biotechnology Company, Beijing, China) with the aid of a SpectraMax plate reader (Molecular Devices).
Histopathological examination
Samples of the liver and kidney were fixed using 10% phosphate-buffered formalin for 3 days followed by cautious dehydration procedures. The tissues were then embedded in paraffin before cutting into sections of 4–5 µm using a microtome. The slides were then stained with hematoxylin and eosin and coded before examination under a light microscope (Leica DM 500, Germany). The histopathological lesions were scored and the photomicrographs captured by means of a digital camera (Leica ICC50 E, Germany) by pathologists.
Statistical analysis
Data were analyzed using one-way analysis of variance to compare the various experimental groups followed by post hoc Bonferroni’s test to ascertain significantly different groups using GRAPHPAD PRISM 5 software (version 4; GraphPad Software, La Jolla, California, USA). Values of p < 0.05 were considered significant.
Results
PCA ameliorates hepatic and renal toxicities in MTX-treated rats
To investigate the influence of PCA on the hepatic and renal injury following MTX administration in rats, the assessment of serum levels of creatinine and urea was done to ascertain renal damage, whereas serum ALT, AST, ALP, and LDH levels were evaluated to ascertain hepatic damage. Figure 1 depicts the influence of PCA on the serum biomarkers of hepatic and renal toxicities in MTX-treated rats. Single intraperitoneal administration of 20 mg/kg of MTX significantly (p < 0.05) elevated all the serum biomarkers of hepatic and renal injury in the rats when compared with the control rats. However, administration of PCA at 25 and 50 mg/kg to MTX-treated rats evidently decreased the serum creatinine, urea, ALT, AST, ALP, and LDH levels when compared with rats treated with MTX alone. Administration of PCA alone at 50 mg/kg for seven consecutive days occasioned no treatment-related effect on these biomarkers.

Effect of PCA on hepatic and renal functional indices in MTX-treated rats. MTX, 20 mg/kg MTX; PCA 1, 25 mg/kg PCA; and PCA 2, 50 mg/kg PCA. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and #p < 0.05 versus MTX alone. PCA: protocatechuic acid; MTX: methotrexate; SD: standard deviation.
PCA attenuates MTX-induced hepatic and renal oxidative damage in rats
Subsequently, the influence of PCA on the oxidative stress indices in MTX-treated rats was evaluated. Figures 2 –4 depict the influence of PCA administration on antioxidant enzymes activities and biomarkers of oxidative stress in the kidney and liver of MTX-treated rats. Results showed that with exception of renal CAT and hepatic GPx activities, a single intraperitoneal injection of MTX alone significantly (p < 0.05) decreased the activities of SOD, CAT, GST, and GPx in the investigated tissues. However, administration of PCA at 25 and 50 mg/kg significantly abated the diminution in the activities of these antioxidant enzymes and reestablished their hepatic and renal levels to near control in MTX-treated rats. Besides, exposure to MTX alone markedly decreased GSH level but elevated RONS and LPO levels in the kidney and liver of the treated rats when compared with MTX alone group. However, administration of PCA evidently protected against renal and hepatic oxidative damage evidenced by significant increase in GSH level with concomitant decrease in RONS and LPO levels when compared with rats treated with MTX alone.

Effect of PCA on SOD and CAT activities in liver and kidney of MTX-treated rats. MTX, 20 mg/kg MTX; PCA 1, 25 mg/kg PCA; and PCA 2, 50 mg/kg PCA. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control; #p < 0.05 versus MTX alone; and @p < 0.05 versus MTX + PCA 1. PCA: protocatechuic acid; SOD: superoxide dismutase; CAT: catalase; MTX: methotrexate; SD: standard deviation.

Effect of PCA on GSH-dependent enzyme activities and GSH level in liver and kidney of MTX-treated rats. MTX, 20 mg/kg MTX; PCA 1, 25 mg/kg PCA; and PCA 2, 50 mg/kg PCA. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control; #p < 0.05 versus MTX alone; and @p < 0.05 versus MTX + PCA 1. PCA: protocatechuic acid; GSH: glutathione; MTX: methotrexate; SD: standard deviation.

Effect of PCA on RONS and LPO levels in liver and kidney of MTX-treated rats. MTX, 20 mg/kg MTX; PCA 1, 25 mg/kg PCA; and PCA 2, 50 mg/kg PCA. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control; #p < 0.05 versus MTX alone; and @p < 0.05 versus MTX + PCA 1. PCA: protocatechuic acid; RONS: reactive oxygen and nitrogen species; LPO: lipid peroxidation; MTX: methotrexate; SD: standard deviation.
PCA suppresses MTX-mediated increase in biomarkers of inflammation and apoptosis in rats
Additionally, the influence of PCA on the biomarkers of inflammation and apoptosis was assessed in the liver and kidney of MTX-treated rats. Figures 5 and 6 depict the impact of PCA on biomarkers of inflammation and apoptosis in MTX-treated rats. Administration of MTX significantly increased the hepatic and renal MPO activities as well as levels of NO, TNF-α, and IL-1β in the treated rats when compared with the control. However, administration of abrogated MTX-induced increase in these biomarkers of inflammation in the liver and kidney of the treated rats when compared with rats administered MTX alone. Moreover, MTX treatment alone caused a significant increase in the CASP3 activity, an executioner apoptotic protease, in the liver and kidney of the treated rats when compared with control. Conversely, administration of PCA markedly abrogated MTX-mediated increase in CASP3 activity in the liver and kidney of the treated rats when compared with rats administered MTX alone. Administration of PCA alone at 50 mg/kg for seven consecutive days caused no treatment-related effect on biomarkers of inflammation and CASP3 activity.
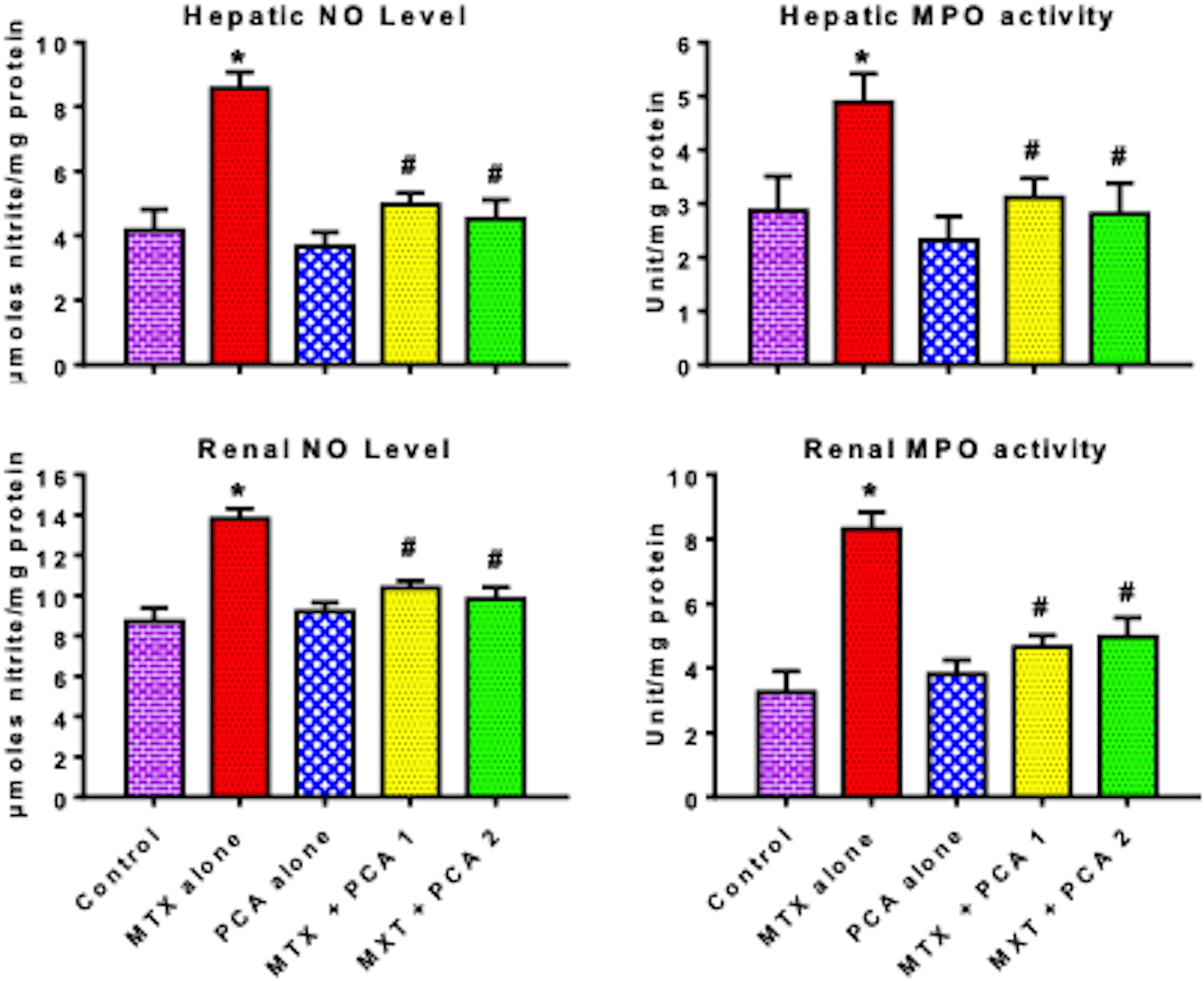
Effect of PCA on NO level and MPO activity in liver and kidney of MTX-treated rats. MTX, 20 mg/kg MTX; PCA 1, 25 mg/kg PCA; and PCA 2, 50 mg/kg PCA. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control and #p < 0.05 versus MTX alone. PCA: protocatechuic acid; NO: nitric oxide; MPO: myeloperoxidase; MTX: methotrexate; SD: standard deviation.

Effect of PCA on TNF-α, IL-1β, and caspase-3 activity in liver and kidney of MTX-treated rats. MTX, 20 mg/kg MTX; PCA 1, 25 mg/kg PCA; and PCA 2, 50 mg/kg PCA. Each bar represents mean ± SD of 10 rats. *p < 0.05 versus control; #p < 0.05 versus MTX alone; and @p < 0.05 versus MTX + PCA 1. PCA: protocatechuic acid; TNF-α; tumor necrosis factor alpha; IL-1β: interleukin-1β; CASP3: caspase-3; MTX: methotrexate; SD: standard deviation.
PCA abrogated MTX-induced hepatorenal lesions in rats
The representative photomicrographs depicting the influence of PCA on MTX-induced histological damage in kidney and liver of experimental rats are shown in Figures 7 and 8. The kidney and liver of control and rats treated with PCA alone appeared normal with well-preserved morphology. Administration of MTX alone caused disseminated congestion, infiltration of inflammatory cells, and glomerular hypercellularity, whereas the liver showed marked congestion and focal periportal infiltration by inflammatory cells. However, rats treated with PCA and MTX had the liver and kidney histological architecture preserved and similar to control.

Representative photomicrographs of the kidney. The kidney of control and PCA alone rats exhibited normal morphology. Kidney of rats treated with MTX alone showing disseminated congestion (black arrow), infiltration of inflammatory cells (yellow arrow), and glomerular hypercellularity (blue arrow). The kidney of rats coadministered MTX and PCA showing a well-preserved histological architecture similar to control. Magnification is ×100 except for MTX alone (2) which is ×400. PCA: protocatechuic acid; MTX: methotrexate.

Representative photomicrographs of liver. The liver of control and PCA alone rats showed normal morphology. Liver of rats treated with MTX alone showing marked congestion (black arrow) and focal periportal infiltration by inflammatory cells (yellow arrow). The liver of rats treated with MTX and PCA appears somewhat similar to control. Magnification is ×100 except for MTX alone (2) which is ×400. PCA: protocatechuic acid; MTX: methotrexate.
Discussion
Owing to the significance of drug-induced hepatorenal toxicity in clinical medicine, there is an increasing interest in the pursuit of basic knowledge about the toxicity particularly on the nature, mechanisms, and measures to minimize the incidence of this unpleasant side effect by researchers. 34 The assessment of the enzyme activities in the tissues and body fluids is essential in the diagnosis and investigation of diseases and cellular injury. 35,36 Indeed, tissue enzymes are recognized as sensitive indicators of cellular damage by xenobiotics prior to confirmation by histological analysis.
Serum marker enzymes of cellular injury were assessed in the present investigation based on their specific cellular locations. The hepatobiliary enzymes, namely AST, ALT, LDH, and ALP, reside largely in the liver under normal conditions. However, during hepatocyte necrosis or membrane injury, the enzymes are often released into the circulation. 35 Specifically, aminotransferases (AST and ALT) are situated in periportal hepatocytes where they participate in transamination reactions during amino acid metabolism but their serum activities are known to be elevated subsequent to cellular membrane damage and leakage. 37,38 Hence, the significant increase in the serum aminotransferases following administration of MTX alone is indicative of disrupted membrane permeability associated with hepatic damage in the treated rats. However, the marked reduction in the serum AST and ALT levels following PCA administration to MTX-treated rats indicates hepatoprotection against MTX-induced liver damage by PCA in rats.
Moreover, an ectoenzyme ALP associates with the plasma membrane. ALP is commonly used to assess the integrity of the plasma membrane because any alteration in its activity is related to the disruption of external boundary of the cells in the tissue. ALP is a recognized sensitive marker for cholestasis. 39 LDH is located in the cytosolic portion of the cell, which is in close proximity to the plasma membrane. Thus, slight modification in the structure of plasma membrane will consequently result in the leakage of the enzyme into the extracellular fluid. The marked elevation in the serum ALP and LDH activities following administration of MTX alone to rats in the present investigation attests to altered plasma membrane permeability with the consequence of their leakage from the hepatic cells into the serum. However, the serum ALP and LDH levels were significantly decreased following PCA administration to MTX-treated rats. This observation further reveals the hepatoprotective role of PCA in MTX-treated rats.
Moreover, the present study revealed that MTX administration was associated with renal dysfunction as evidenced by marked increase in serum urea and creatinine levels. In clinical biochemistry, an increase in serum urea level is indicative of reduced reabsorption at the renal epithelium, while an increase in the serum creatinine signifies impairment in the renal function especially in the glomerular filtration rate. 40 However, administration of PCA significantly decreased the serum levels of urea and creatinine in the treated rats, thus indicating the renoprotection against MTX-induced renal toxicity by PCA in rats. Also, the histopathological observations confirmed that MTX administration elicited obvious damaging effect to both liver and kidney of in rats, hence corroborating the biochemical results. Administration of PCA efficiently abated MTX-induced hepatorenal toxicity in the treated rats.
To elucidate the hepatorenal protective mechanism of PCA in the rats, the role of PCA administration on the hepatic and renal antioxidant defense systems, biomarkers of oxidative stress, inflammation, and apoptosis were explored. Antioxidant defense enzymes comprise SOD which is responsible for the conversion of noxious super oxide anion to H2O2, while CAT protects the cells against accumulation of toxic H2O2 by converting it to water and oxygen. Moreover, GPx is responsible for the removal of peroxyl radicals and maintenance of functional integrity of the cell membranes, whereas GST plays a pivotal role in the detoxification of electrophilic compounds. 41 In the present study, administration of MTX alone occasioned significant decreases in the activities of SOD, CAT, GST, and GPx in rats, thus specifying an inhibition of their antioxidant protective role. However, the marked increase in the activities of these antioxidant enzymes following administration of PCA to MTX-treated rats indicates the chemoprotective role of PCA in suppressing cellular damage possibly through augmentation of antioxidant defense mechanism.
An uncontrolled intracellular generation of free radicals is an important factor in tissue oxidative damage. Moreover, GSH is a ubiquitous and versatile antioxidant well-known to protect the cell via free-radical scavenging activity, restoration of the damaged molecules by donating hydrogen, reduction of peroxides, and preservation of protein thiols in the reduced state. 42 The significant decrease in hepatic and renal GSH levels in rats treated with MTX alone connotes its cellular depletion, which is related to an overutilization during detoxification of the RONS generated by MTX treatment. Administration of MTX alone markedly decreased GSH level, whereas hepatic GPx activity was not significantly decreased. The reason for this observation is not presently known. However, a possible explanation may be that the MTX exposure period is short and therefore not sufficient to inhibit GPx activity. However, the increase in GSH level following administration of PCA to MTX-treated rats indicates that the PCA elicited hepatorenal protection possibly due to enhancement of GSH availability essential for the cells to mitigate free radicals generated by MTX.
Further, LPO is a degenerative pathway of membrane components, which has been related to increased free radical production in the cell. 43 The present study demonstrated that administration of MTX to rats triggered elevation in RONS and LPO levels in the liver and kidney. The increase in RONS and LPO connotes that MTX administration elicited a destructive effect on the membrane functional state, which is related to the diminution in the antioxidant defense mechanisms capable of abrogating oxidative stress in the rats. However, the marked decrease in the RONS and LPO levels following administration of PCA signifies an antioxidative and anti-lipid peroxidative mechanism of PCA in the protection against MTX-induced tissue damage.
Nitric oxide is well reported to play a central role in inflammation and has also been implicated in cellular and molecular signaling under normal physiological conditions. 44,45 Indeed, elevated NO level reportedly increases nitration of proteins at tyrosine and, consequently, induces protein dysfunction or alterations in signal transduction pathways. 46 Besides, MPO activity has been associated with induction of oxidative stress and inflammation, 47 whereas TNF-α is well-known to be a “master regulator” of cytokine production during an inflammatory response. 48 Hence, the significant elevation in the MPO activity as well as levels of NO, IL-1β, and TNF-α in MTX-treated rats obviously signifies induction of inflammation in the treated rats. However, PCA exerted a protective effect against MTX-induced hepatorenal injury via mechanisms of action involving the reduction of MTX-mediated increases in MPO activity and levels of NO, TNF-α, and IL-1β. CASP3 is an aspartate-specific cysteine protease, which is well-known for its executioner role in apoptotic pathway. 49,50 Administration of MTX alone significantly increased hepatic and renal CASP3 activities in the present study, thus connoting an induction of apoptotic cell death in the liver and kidney of MTX-treated rats. However, the marked decrease in CASP3 activity following PCA administration signifies the antiapoptotic properties of PCA in the treated rats.
The beneficial hepatorenal effects of PCA observed in the present study are related to the structure–activity relationship of this phytochemical. Mechanistically, the existence of hydroxyl groups is essential for hydrogen atom donation activity, which reduces peroxyl radicals before their damaging interaction with the cell membranes and other cellular components. 51
Conclusively, PCA abrogated MTX-mediated hepatorenal damage via mechanisms involving enhancement of endogenous antioxidant defense, inhibition of LPO and RONS production along with suppression of inflammation and CASP3 activities. Hence, PCA may be a prospective chemoprotective supplement for cancer patients undergoing MTX chemotherapy. Nevertheless, PCA administration to humans requires some clinical trials with different doses and durations prior to its usage in cancer therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
