Abstract
Exposure to the fungicide carbendazim (CBZ) has been associated with hepatorenal dysfunction. Quercetin, a naturally occurring polyphenolic phytochemical, reportedly possesses beneficial health effects. However, there is paucity of scientific information on the impact of quercetin on CBZ-induced hepatorenal damage. The present study investigated the protective mechanism of quercetin in CBZ-induced hepatic and renal damage in rats. The treatment groups consisted of control, CBZ alone (50 mg/kg), quercetin alone (20 mg/kg), and rats co-treated with CBZ and quercetin for 14 consecutive days. Quercetin co-treatment significantly (p < 0.05) abated CBZ-induced increase in biomarkers of hepatorenal damage when compared to CBZ alone. Also, quercetin abrogated CBZ-mediated decrease in antioxidant status as well as the increase in reactive oxygen and nitrogen species and lipid peroxidation in the treated rats. Furthermore, quercetin significantly suppressed CBZ-mediated increase in interleukin-1β, tumor necrosis factor alpha, and caspase-3 activity in the liver and kidney of the rats. Histopathological examination demonstrated that the severity of CBZ-induced hepatic and renal injury was ameliorated in rats co-treated with quercetin. Taken together, quercetin-mediated hepatorenal protection in CBZ-treated rats involves antioxidant, anti-inflammatory, and antiapoptotic mechanisms.
Introduction
Carbendazim (CBZ, methyl N-(1H-benzimidazol-2-yl) carbamate) is a broad-spectrum fungicide which is extensively used in agriculture worldwide for the control of fungal infections, foot rot, and damping-off diseases. 1 Moreover, CBZ is often applied as a worm control agent to protect amenity turf, namely, golf courses and tennis courts. 1 According to the European Union (EU) regulation number 528/2012, CBZ is commonly applied as a biocide for the preservation of product types, including film, leather, rubber, fiber, polymerized materials, and facades of buildings. 2,3 The extensive application of CBZ is a major global concern because it is often released during rainfall and subsequently transported to locations where it poses threat to the environment, animal, and human health. CBZ is a persistent environmental contaminant because its benzimidazolic ring is difficult to break and its degradation is slow. 4 Previous studies from several countries have reported that CBZ is frequently detected in soil and water. 5,6
Humans can be exposed to CBZ occupationally and environmentally. The main routes of exposure to CBZ include inhalation and consumption of contaminated water, fruits, and vegetables. 7 Several experimental studies have demonstrated that CBZ adversely affects the reproductive system and disrupts endocrine function in animals. 8,9 Recent studies have also shown that CBZ induces renal and hepatic toxicity 10,11 which can be associated with gut microbiota dysbiosis and hepatic lipid metabolism disorder. 12,13 Mechanistically, CBZ-mediated toxicity has been associated with the induction of microtubule disturbances during cell divisions, oxidative stress, and apoptosis in rodents and zebrafish. 8,9 Therefore, an appropriate intervention strategy to abate the CBZ-induced toxicity would involve the suppression of oxidative stress and apoptosis using natural antioxidants.
Quercetin (3,5,7,3′,4′-pentahydroxyflavone) is a naturally occurring polyphenolic flavonoid. It is widely found in many fruits, including grapes, cherries, apples, and cranberries as well as in vegetables, namely, peppers, onion, and asparagus. 14 The estimated daily quercetin intakes reportedly range from 3 mg to 40 mg in Western diets, whereas “high-end consumers” of fruits and vegetables were reported to consume about 250 mg of quercetin/day. 15 Specifically, an average of 6–18 mg of quercetin/day has been estimated to be consumed in the United States, China, and Europe. 16 Moreover, purified quercetin is commercially available in the EU and United States as capsules or aqueous extract administered sublingually. 17 In Canada, quercetin is used in Natural Health Products as an antioxidant or in Herbal Medicine Products as a capillary/blood vessel protectant. 15 The intended quercetin intake through dietary supplements (1 g/day) is often largely greater than real dietary intake levels of quercetin. 18 Several studies have demonstrated that quercetin produces ample health beneficial effects which are linked to its free radicals scavenging ability, suppression of oxidative stress and inflammation, as well as its anticancer activity in vitro and in vivo. 15 Nevertheless, there is a paucity of scientific information on the role of quercetin in CBZ-mediated hepatorenal toxicity.
Therefore, the present study evaluated the possible chemoprotective effects of quercetin on CBZ-induced hepatorenal dysfunction by assessing antioxidant enzymes activities, oxidative stress indices, pro-inflammatory cytokines levels, caspase-3 activity, as well as the histological lesions in liver and kidneys of experimental animals.
Materials and methods
Chemicals
CBZ (97% pure), quercetin (≥95% pure), 5′,5′-dithiobis-2-nitrobenzoic acid, thiobarbituric acid, 1-chloro-2,4-dinitrobenzene, epinephrine, hydrogen peroxide (H2O2), and glutathione (GSH) were obtained from Sigma Chemical Co. (St Louis, Missouri, USA). Enzyme-linked immunosorbent assay (ELISA) kits for the evaluation of interleukin-1β (IL-1β), tumor necrosis factor alpha (TNF-α), and caspase 3 activity were procured from Elabscience Biotechnology Company, Beijing, China.
Care of animal
Forty adult male Wistar rats (9 weeks old, 180 ± 5 g) obtained from the Experimental Animal Unit, Faculty of Veterinary Medicine, University of Ibadan, were used for this investigation. The animals were housed in plastic cages situated in a well-ventilated vivarium, provided rat chows and water ad libitum. The rats were subjected to a natural photoperiod of 12-h light/dark and adequately cared for according to the conditions specified in the “Guide for the Care and Use of Laboratory Animals” as promulgated by the National Institute of Health. In addition, the study was executed following authorization by the University of Ibadan Ethical Committee and in accordance with the US NAS guidelines.
Experimental design
Following a week of acclimatization, the experimental animals were randomized into four (4) groups of 10 rats each and were orally treated for 14 consecutive days as follows.
Control group: The rats were administered with 2 ml/kg of corn oil.
CBZ alone group: The rats were administered with 50 mg/kg of CBZ alone.
Qt alone group: The rats were administered with 20 mg/kg of quercetin alone.
CBZ and Qt group: The rats were coadministered with CBZ (50 mg/kg) and quercetin (20 mg/kg).
The doses of CBZ and quercetin used in the present investigation were selected from our preliminary studies and earlier published studies. 10,19 Blood obtained from the retro-orbital venous plexus was transferred into plain tubes before the animals were killed by cervical dislocation on day 15. Thereafter, serum samples were prepared by centrifuging the clotted blood at 3000 g for 10 min. The serum samples were then kept frozen at −20°C until needed for the liver and kidney function analysis.
Evaluation of liver and kidney function biomarkers
Serum analysis of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), creatinine, and urea levels were done using available commercial kits from Randox Laboratories Limited (UK).
Evaluation of hepatic and renal antioxidant status
The liver and kidney samples of the rats were homogenized in 50-mM Tris–hydrogen chloride buffer (pH 7.4) followed by centrifugation at 12,000 × g for 15 min at 4°C to obtain the supernatant which was used for the biochemical analyses. Protein concentration of the liver and kidney was assayed according to the method described by Bradford, 20 lipid peroxidation (LPO) was assessed in line with the established method, 21 GSH levels were assayed according to the method described by Jollow et al., 22 superoxide dismutase (SOD) activity was assayed in line with the method described by Misra and Fridovich, 23 catalase (CAT) activity was assayed according to the method described by Clairborne, 24 and glutathione peroxidase (GPx) activity was assayed according to the established method, 25 whereas glutathione-S-transferase (GST) activity was assayed in line with the method of Habig et al. 26
Quantification of reactive oxygen and nitrogen species level
The level of reactive oxygen and nitrogen species (RONS) in the liver and kidney of experimental rats was quantified using an established protocol which was based on the RONS-dependent oxidation of 2′,7′-dichlorodihydrofluorescin diacetate (DCFH-DA) to Dichlorofluorescein (DCF). 27 Succinctly, the reaction mixture contained 10 µl of the sample, 150 µl of 0.1-M potassium phosphate buffer (pH 7.4), 35 µl of distilled water, and 5 µl of DCFH-DA (200 µM, final concentration 5 µM). The fluorescence emission of DCF resulting from DCFH-DA oxidation was analyzed at 488-nm excitation and 525-nm emission wavelengths for 10 min at 30 s intervals using a SpectraMax plate reader (Molecular Devices, San Jose, California, USA). The rate of DCF formation was expressed in percentage of control group.
Measurement of pro-inflammatory biomarkers and caspase 3 activity
Myeloperoxidase (MPO) activity in the liver and kidney of experimental animals was evaluated according to the method described by Granell et al., 28 whereas the level of nitric oxide (NO) was evaluated using Griess reagent according to established protocol. 29 Succinctly, the reaction mixture comprised of equal volume of sample and Griess reagent was incubated for 15 min before the absorbance was determined at 540 nm. Subsequently, the level of NO was extrapolated from the standard curve and expressed as units per milligram of protein. Furthermore, IL-1β and TNF-α concentrations as well as caspase-3 activity were assessed using ELISA Kits (Elabscience Biotechnology Company, Beijing, China) with the aid of a SpectraMax plate reader (Molecular Devices).
Histological analysis of liver and kidney
Liver and kidney samples were fixed in 10% phosphate buffered formalin for 3 days followed by careful dehydration procedures. Subsequently, the tissues were embedded in paraffin before making sections of 4–5 µm using a microtome and staining with hematoxylin and eosin. Histology of the liver and kidney samples was viewed using a light microscope and the histopathological lesions scored by pathologists who were blinded to the treatment groups.
Statistical analysis
Results were analyzed using the one-way analysis of variance and post hoc Bonferroni’s test with the aid of GRAPHPAD PRISM 5 (GraphPad Software, La Jolla, California, USA) to ascertain significant differences in the treatment groups. p Values less than 0.05 were considered to be significant.
Results
Quercetin decreases biomarkers of hepatic and renal toxicity in CBZ-treated rats
Assessment of serum levels of creatinine and urea was done to confirm renal damage, whereas serum ALT, AST, ALP, and LDH levels were evaluated to establish hepatic damage in the experimental animals. The results presented in Table 1 indicated that exposure to 50 mg/kg of CBZ for 14 consecutive days significantly (p < 0.05) elevated all the serum biomarkers of hepatic and renal damage in the rats, whereas quercetin at 20 mg/kg elicited no treatment-related effect on the biomarkers when compared with the control rats. Conversely, co-treatment with 20 mg/kg of quercetin significantly diminished the serum levels of creatinine, urea, blood urea nitrogen (BUN)/creatinine ratio, ALT, AST, ALP, and LDH in CBZ-exposed rats when compared with CBZ alone group, although not to the levels of either the control or quercetin alone.
Quercetin modulates hepatic and renal functional indices in CBZ-treated rats.
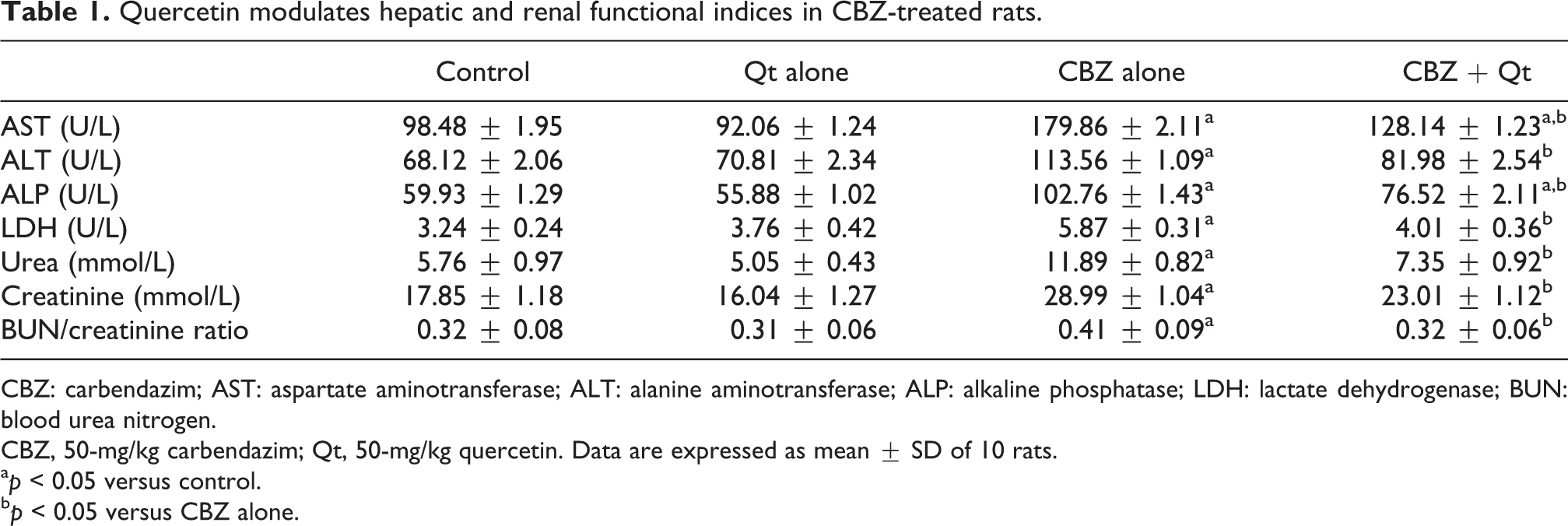
CBZ: carbendazim; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase; BUN: blood urea nitrogen.
CBZ, 50-mg/kg carbendazim; Qt, 50-mg/kg quercetin. Data are expressed as mean ± SD of 10 rats.
a p < 0.05 versus control.
b p < 0.05 versus CBZ alone.
Quercetin inhibits hepatic and renal oxidative damage in CBZ-treated rats
Table 2 represents the influence of quercetin co-treatment on antioxidant enzymes activities in the kidney and liver of CBZ-treated rats. Results indicated that administration of CBZ alone significantly (p < 0.05) decreased the activities of SOD, CAT, GST, and GPx in the kidney and liver of the treated rats. However, co-treatment with quercetin abrogated the decrease in these antioxidant enzyme activities and restored their hepatic and renal levels to near normal. Moreover, Figure 1 shows that exposure to CBZ alone significantly decreased GSH level and increased RONS and LPO levels in the kidney and liver of the treated rats, whereas co-treatment with quercetin markedly mitigated against increased renal and hepatic oxidative damage evidenced by a marked elevation in GSH level with concomitant reduction in RONS and LPO levels when compared with CBZ alone group.
Quercetin improves hepatic and renal antioxidant enzyme activities in CBZ-treated rats.
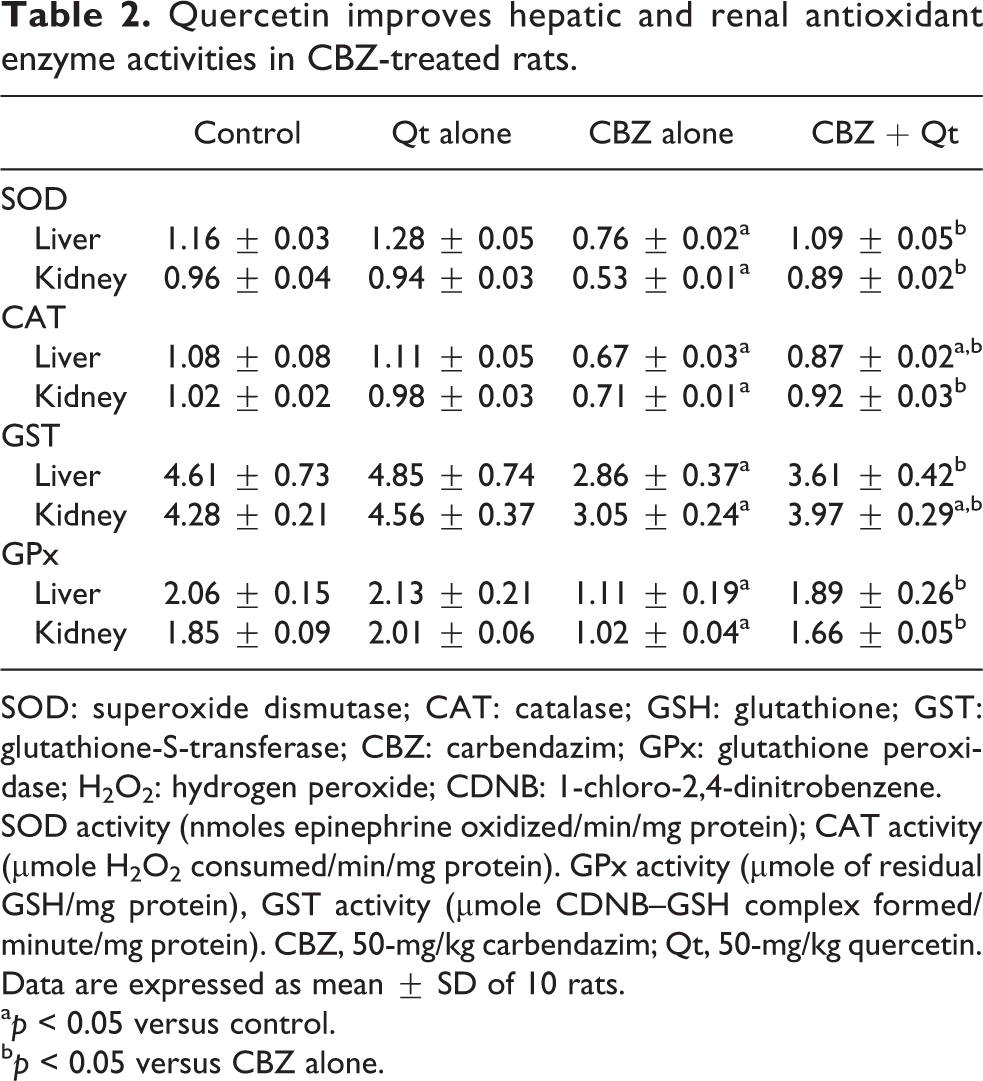
SOD: superoxide dismutase; CAT: catalase; GSH: glutathione; GST: glutathione-S-transferase; CBZ: carbendazim; GPx: glutathione peroxidase; H2O2: hydrogen peroxide; CDNB: 1-chloro-2,4-dinitrobenzene.
SOD activity (nmoles epinephrine oxidized/min/mg protein); CAT activity (µmole H2O2 consumed/min/mg protein). GPx activity (µmole of residual GSH/mg protein), GST activity (µmole CDNB–GSH complex formed/minute/mg protein). CBZ, 50-mg/kg carbendazim; Qt, 50-mg/kg quercetin. Data are expressed as mean ± SD of 10 rats.
a p < 0.05 versus control.
b p < 0.05 versus CBZ alone.
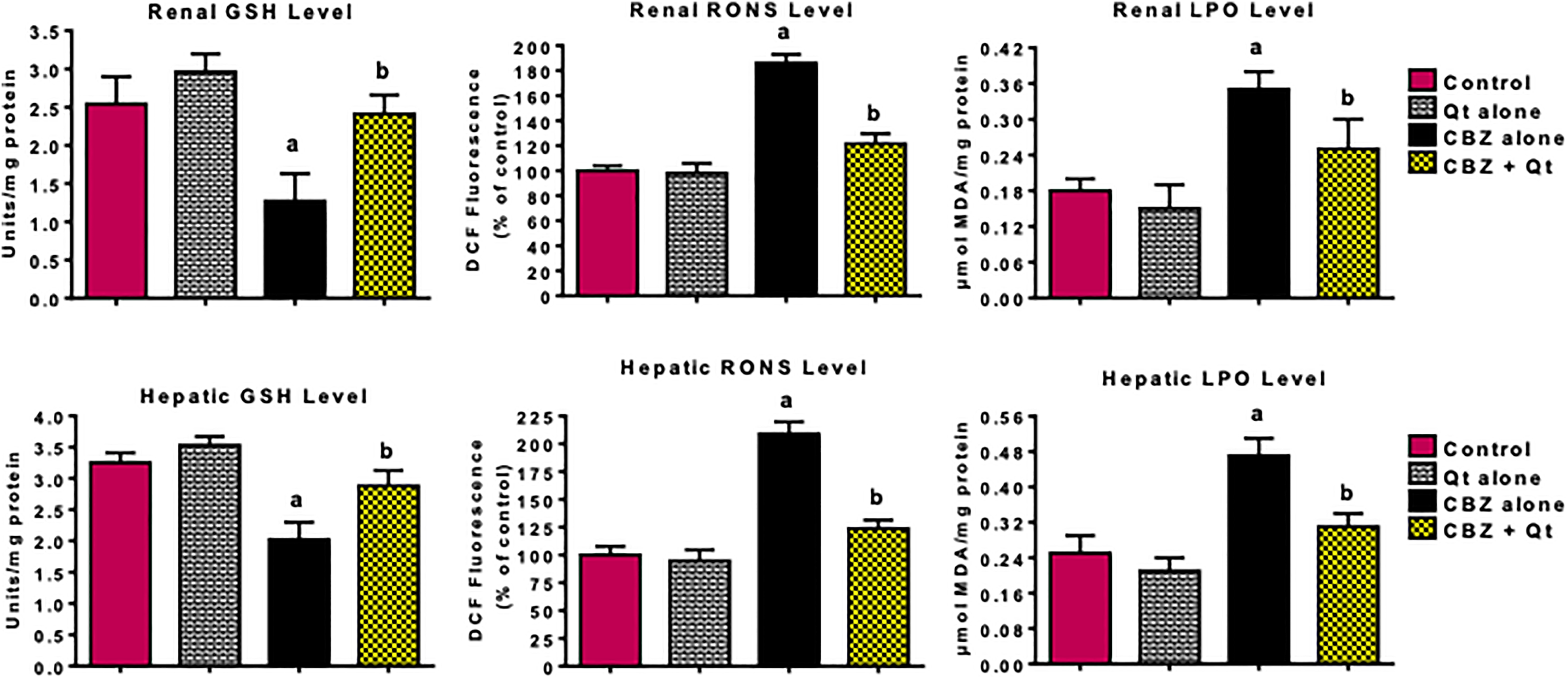
Influence of quercetin on hepatic and renal levels of GSH, RONS, and LPO in CBZ-treated rats. CBZ, 50-mg/kg carbendazim; Qt, 20-mg/kg quercetin. Each bar represents mean ± SD of 10 rats. a p < 0.05 versus control; b p < 0.05 versus CBZ alone. GSH: glutathione; RONS: reactive oxygen and nitrogen species; LPO: lipid peroxidation; CBZ: carbendazim.
Quercetin inhibits biomarkers of inflammation and apoptosis in CBZ-treated rats
The results on the influence of CBZ and quercetin on biomarkers of inflammation and apoptosis are depicted in Figures 2 and 3. In comparison with the control, administration of CBZ for 14 consecutive days to rats markedly increased the hepatic and renal MPO activity as well as levels of NO, TNF-α, and IL-1β in the treated rats. In addition, administration of CBZ alone to rats caused a significant increase in the hepatic and renal caspase-3 activity when compared with control. However, co-treatment with quercetin abated CBZ-mediated increase in these indices of inflammation and apoptosis in the liver and kidney of the treated rats when compared with the CBZ alone group.
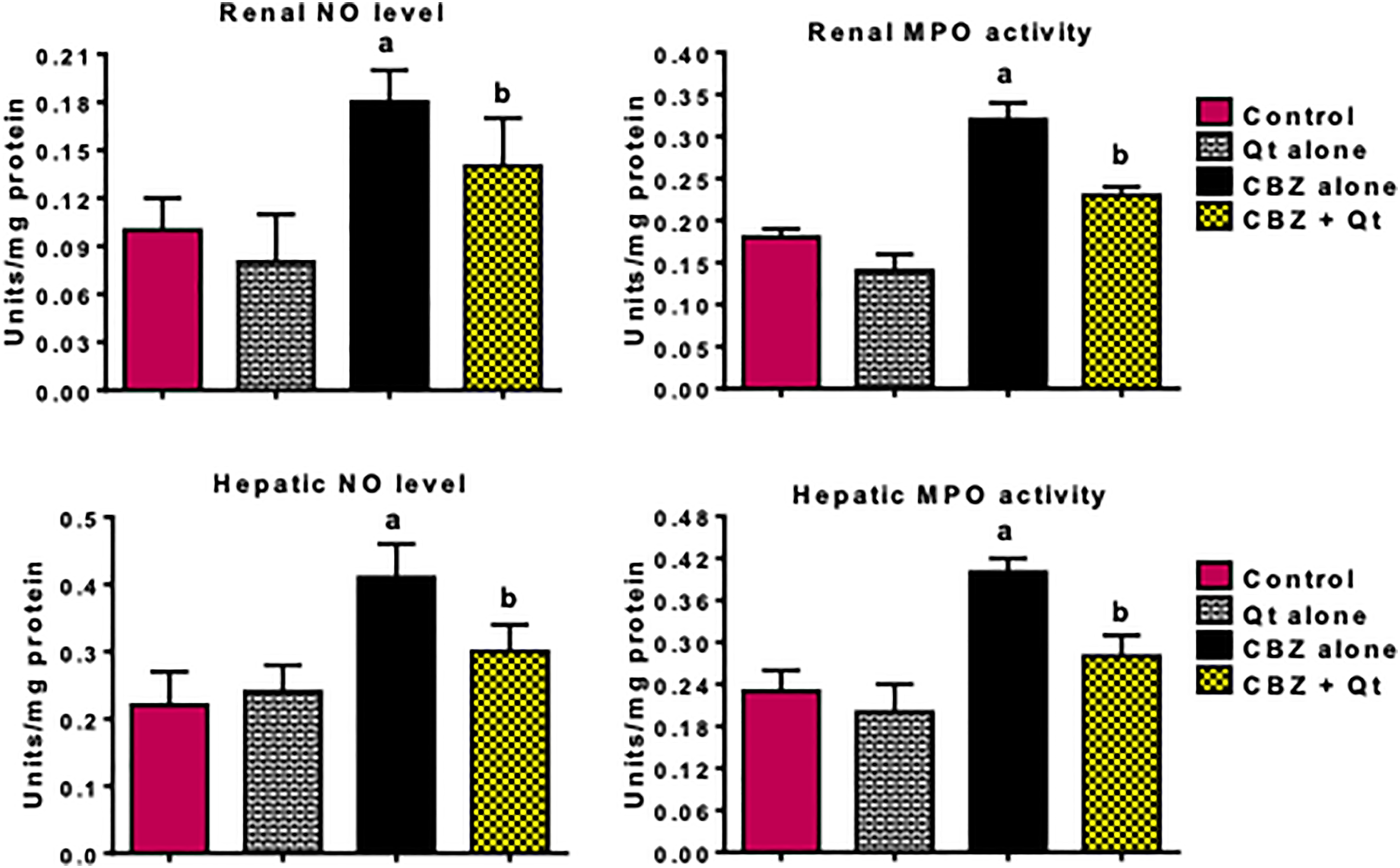
Influence of quercetin on hepatic and renal level of NO and MPO activity in CBZ-treated rats. CBZ, 50-mg/kg carbendazim; Qt, 20-mg/kg quercetin. Each bar represents mean ± SD of 10 rats. a p < 0.05 versus control; b p < 0.05 versus CBZ alone. NO: nitric oxide; CBZ: carbendazim; MPO: myeloperoxidase.
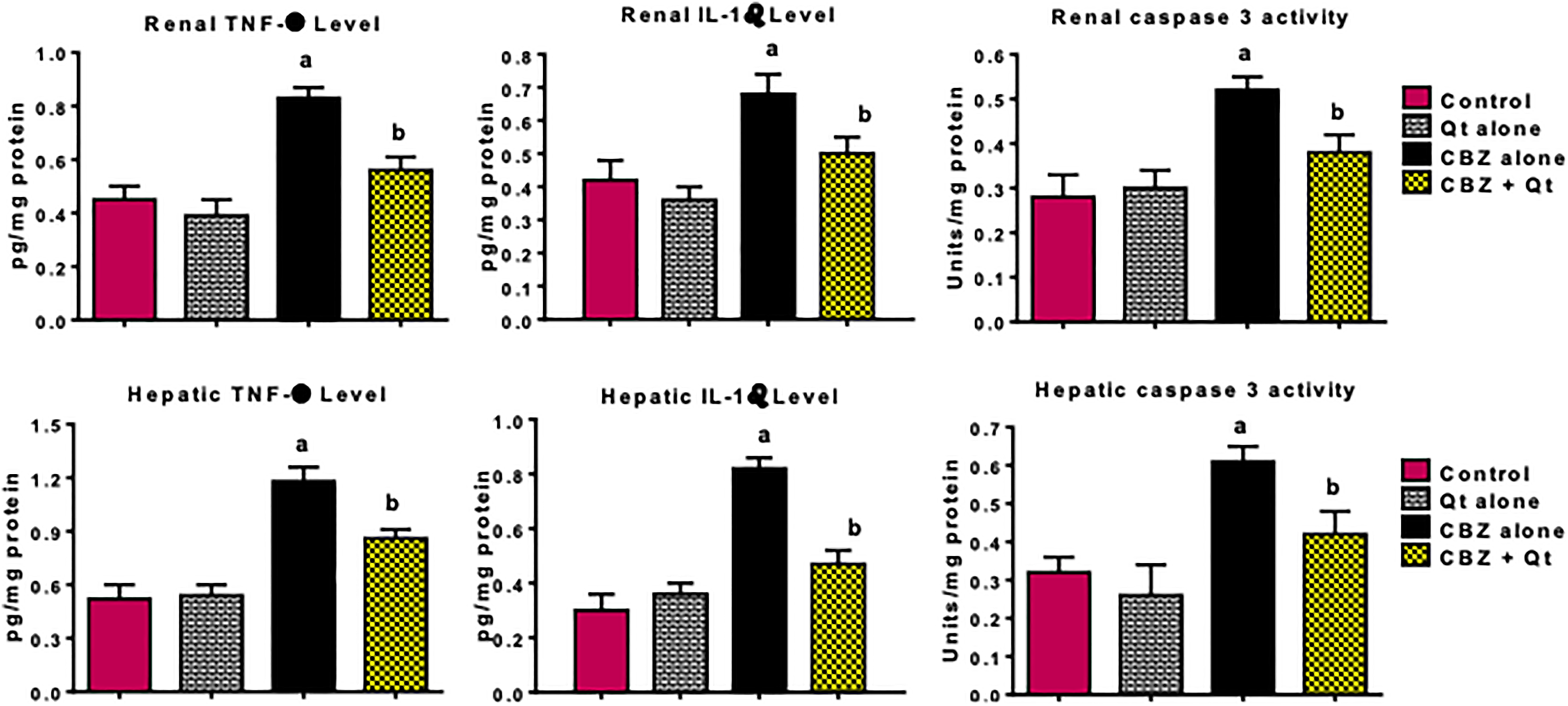
Influence of quercetin on hepatic and renal levels of IL-1β, TNF-α, and caspase-3 activity in CBZ-treated rats. CBZ, 50-mg/kg carbendazim; Qt, 20-mg/kg quercetin. Each bar represents mean ± SD of 10 rats. a p < 0.05 versus control; b p < 0.05 versus CBZ alone. IL-1β: interleukin-1β; TNF-α: tumor necrosis factor alpha; CBZ: carbendazim.
Quercetin abates hepatorenal lesions in CBZ-exposed rats
The representative photomicrographs showing the influence of quercetin on CBZ-induced histological damage in the kidney and liver of experimental rats are presented in Figure 4. The kidney and liver of control and rats treated with quercetin alone appeared normal with well-preserved morphology. However, the kidney of rats exposed to CBZ alone showed focal area of necrosis and the presence of inflammatory cells, whereas the liver showed severe disseminated congestion and infiltration of inflammatory cells. However, the kidney and liver of rats co-treated with quercetin appeared somewhat similar to control but with mild disseminated congestion.
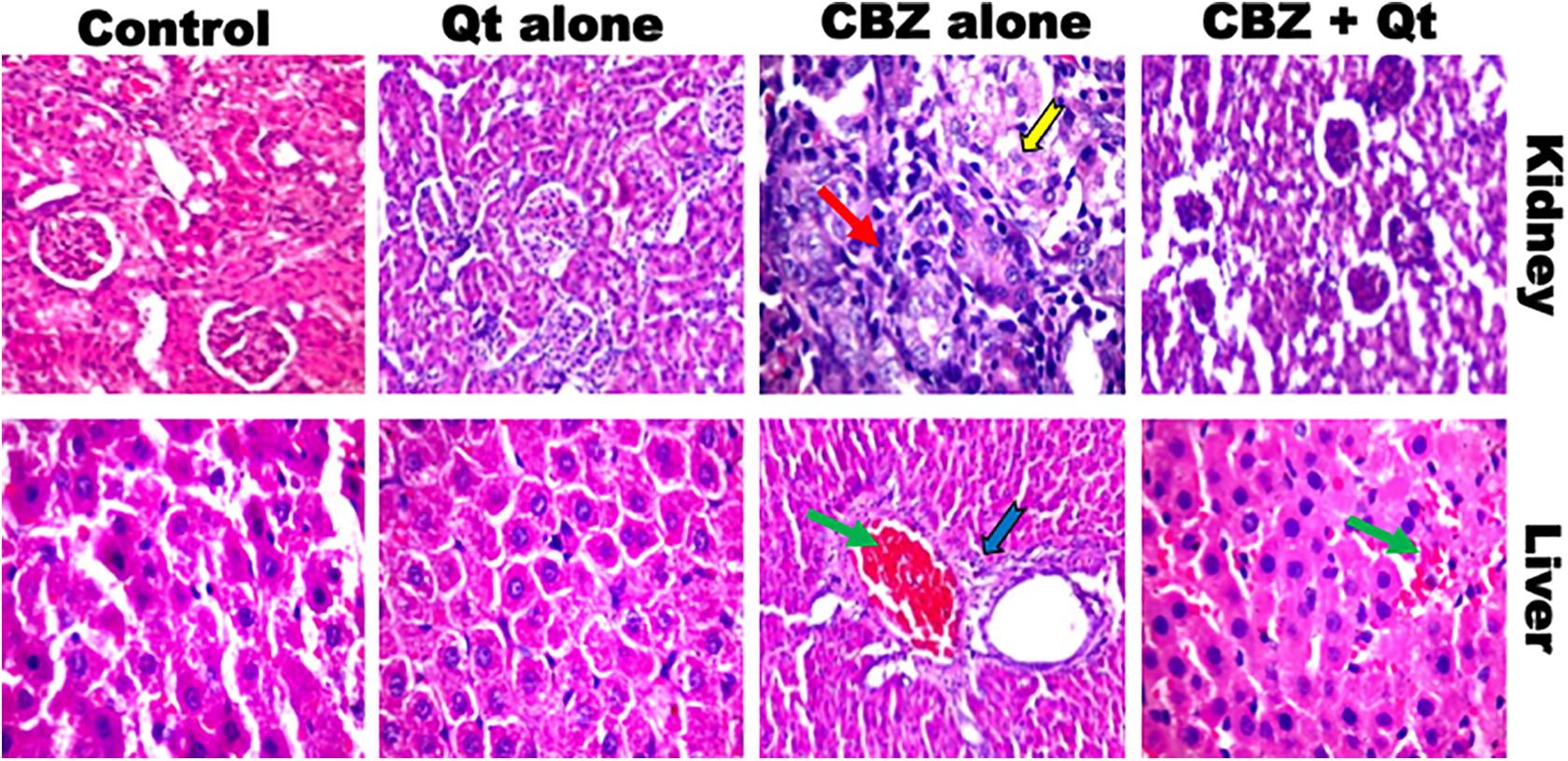
Representative photomicrographs of liver and kidney. Liver and kidney of control rats showing normal morphology. Kidney of rats treated with CBZ alone showing focal area of necrosis (yellow notched arrow) and presence of inflammatory cells (red arrow), whereas the liver showed severe disseminated congestion (green arrow) and infiltration of inflammatory cells (green arrow). However, the kidney and liver of rats co-treated with quercetin showing mild disseminated congestion (green arrow). CBZ: carbendazim.
Discussion
The present study demonstrated, for the first time, the beneficial effects of dietary quercetin on hepatic and renal toxicity resulting from exposure to the fungicide CBZ in rats. The liver integrity in the experimental rats was assessed by analyzing standard biochemical indices namely the serum levels of aminotransferases (AST and ALT), ALP and LDH along with microscopic examination of the liver. It is well known that aminotransferases are confined in periportal hepatocytes where they play a pivotal role in transamination reactions during amino acid metabolism but their serum activities increase following cellular membrane damage and leakage. 30,31
In general, a less than twofold increase in the serum biomarkers of liver and renal toxicity is interpreted as moderate elevation. 32,33 Thus, the moderate elevation in the serum ALT and AST observed in rats that were exposed to CBZ alone in the present study signifies hepatic damage which may be related to disrupted membrane permeability. Moreover, ALP is an index enzyme for the hepatobiliary system integrity and bile flow into the small intestine, whereas LDH is an established biomarker for an early stage of acute liver injury. 34,35 The elevation in serum LDH and ALP activities in the present investigation connotes hepatic injury and cholestatic effect following CBZ exposure. However, the marked diminution of these indices of hepatic damage in rats coadministered with quercetin evidenced the hepatoprotective effects of quercetin on CBZ-induced hepatic injury in rats.
Furthermore, the present study clearly demonstrated that CBZ treatment elicited marked decrease in renal function as evidenced by moderate elevation in serum levels of creatinine, urea, and BUN/creatinine ratio. Our findings on CBZ-mediated renal and hepatic injury are in agreement with earlier studies. 36,37 Biochemically, an elevated serum urea level is associated with decreased reabsorption at the renal epithelium, whereas elevation in the serum creatinine connotes impairment in the kidney function especially in the glomerular filtration rate. 38 However, coadministration of quercetin markedly decreased the serum levels of these indices of renal toxicity in rats, thus connoting the renoprotective effects of quercetin on CBZ-induced renal damage in rats. Moreover, the histopathological findings evidenced that oral exposure to CBZ demonstrated detrimental effects to both liver and kidney of exposed rats, hence substantiating the biochemical data. However, coadministration with quercetin effectively protected against CBZ-mediated hepatorenal damage in the treated rats.
In an attempt to delimit the protective mechanism of action of quercetin in the liver and kidney, the influence of quercetin administration on the hepatic and renal antioxidant defense system, biomarkers of inflammation, and caspase 3 activity was investigated. Endogenous antioxidant enzymes, namely SOD, CAT, GPx, and GST, are accountable for the detoxification of noxious free radicals and their activities are used to evaluate cellular antioxidant status and oxidative stress level. 39 In mechanistic term, metalloenzyme SOD accelerates the conversion of endogenous cytotoxic superoxide radicals to H2O2, whereas CAT converts the harmful peroxide radicals into water and oxygen. 8 Moreover, the biochemical conjugation of electrophilic oxidants with GSH to form water-soluble products which are easily excreted from the system is catalyzed by GST, 40,41 whereas GPx is responsible for the detoxification of H2O2 and lipid hydroperoxides. Thus, the marked reduction in the SOD, CAT, GST, and GPx activities as well as GSH level in CBZ-treated rats simply reflects injurious predominance of Reactive Oxygen Species (ROS). Our observations on CBZ-mediated decrease in hepatic and renal antioxidant enzyme activities are in agreement with previous studies. 37 However, the significant increase in these antioxidant enzyme activities in rats co-treated with quercetin demonstrates the chemoprotective role of quercetin in assuaging oxidative stress via enhancement of antioxidant defense system.
Moreover, the increase in the RONS and LPO levels observed in rats exposed to CBZ alone revealed excessive ROS production and cellular oxidative damage, whereas elevated levels of NO, MPO, and TNF-α connote involvement of aggravated inflammatory response in CBZ-mediated hepatorenal injury in the rats. The marked reduction in the RONS, LPO, NO, MPO, and TNF-α levels in rats co-treated with quercetin connotes an anti-lipid peroxidative and anti-inflammatory mechanisms of quercetin in the protection against CBZ-induced tissue injury. Moreover, in the present study, the rats that were exposed to CBZ alone demonstrated a significant increase in hepatic and renal caspase-3 activity. Caspase-3 is an aspartate-specific cysteine protease and is a well-known downstream key apoptotic initiator. 38,42 Thus, elevation in the caspase-3 activity signifies induction of apoptotic cell death in the liver and kidney of CBZ-treated rats. However, co-treatment with quercetin markedly suppressed the increase in caspase-3 activity, thus signifying the antiapoptotic properties of quercetin.
In conclusion, the protective influence of quercetin on CBZ-induced hepatorenal damage in the present investigation is related to improvement in endogenous antioxidant defense systems, inhibition of LPO, and RONS generation with concomitant suppression of inflammation and caspase-3 activities. Hence, the extrapolation of the present animal study to human indicates that quercetin may have potential beneficial health effects in fungicide CBZ-exposed individuals.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
