Abstract
The persistent inflammation and oxidative stress at the local site in ulcerative colitis reportedly extend to the testes via systemic circulation resulting in testicular dysfunction. The influence of 6-gingerol (6G), a phenolic compound isolated from Zingiber officinale, on colitis-mediated testicular dysfunction in mice was investigated in this study. Chronic ulcerative colitis was induced in mice using 2.5% dextran sulfate sodium (DSS) in drinking water for three cycles. Each cycle consisted of 7 consecutive days of exposure to DSS-treated water followed by 14 consecutive days of normal drinking water. 6G (100 mg/kg) or sulfasalazine (SZ; 100 mg/kg) was orally administered alone or in combination with DSS-treated water during the three cycles. SZ served as standard reference drug for colitis in this study. 6G significantly prevented the incidence of rectal bleeding, decrease in the body weight gain and colon mass index in DSS-exposed mice. 6G significantly prevented colitis-mediated decreases in luteinizing hormone, follicle-stimulating hormone and testosterone and decreases oxidative stress indices, pro-inflammatory cytokines and caspase-3 activity with concomitant augmentation of antioxidant enzymes activities, sperm characteristics, marker enzymes of testicular function and histoarchitecture in DSS-exposed mice. 6G exerted protective influence against ulcerative colitis-induced testicular damage via mechanisms involving its antioxidant and anti-inflammatory properties.
Introduction
Ulcerative colitis (UC) is a type of inflammatory bowel disease which is characterized by recurrent and long-lasting episodes of diarrhoea and inflammation of the gastrointestinal tract. 1 Several lines of evidence from human and experimental studies have indicated that the concomitant involvement of genetic predisposition, environmental factors, immune dysfunction and microbial agents plays a pivotal role in the development of the disease. 2,3 Moreover, the increase in the production of pro-inflammatory cytokines, activated lymphocytes and reactive oxygen species (ROS) are major contributory factors to the progression of the disease in the local inflamed tissue. 4,5
UC is well reported to disrupt the gut mucosa which allows the translocation of gut bacteria to other organs consequently leading to extra-intestinal manifestations including hepatobiliary disease, osteoporosis, amyloidosis and reproductive defect. 6 –8 Indeed, young patients with inflammatory bowel disease reportedly had delay in the onset and progression of puberty. 9,10 Experimental evidence from dextran sulphate sodium (DSS) model of UC in male rodent demonstrated delayed onset and progression of puberty which was associated with a significant reduction in the testes size, disruption in reproductive hormones namely testosterone, follicle-stimulating hormone (FSH) and luteinizing hormone (LH) as well as low sperm count. 11 –13 Recently, UC-induced testicular toxicity was reported to involve the migration of damaged inflammatory cells from the systemic circulation into the seminiferous tubules via testicular artery resulting in testicular damage. 13 The DSS-induced UC was associated with elevated inflammation, oxidative stress, cellular and DNA damage in the testes of mice. 13 Persistent inflammation and oxidative stress in the testes may induce reproductive deficits and impair fertility. 14
Contemporary therapeutic agents for inflammatory bowel disease including 5-amino salicylic acid, steroids and immunosuppressive and immune-regulatory agents have demonstrated limited therapeutic efficiency and long-term toxicity. 15 The incorporation of functional foods into therapeutic regimens for the disease has gained increasing attention due to their generally low toxicity profiles and high patient compliance. 16 In addition, the control of oxidative stress and inappropriate immune responses remains the target of many emerging treatments. 16,17
Zingiber officinale commonly known as ginger is a globally marketed flavouring agent and cooking spice with a long history of human health benefits. 18 It is commonly used in traditional oriental medicine as a remedy for various inflammatory disorders and gastrointestinal discomfort. 19,20 Moreover, ginger has been reported to exhibit androgenic activity, 21 ameliorate testicular dysfunction in male diabetic rats 22 and re-established systolic blood pressure and reproductive function in hypertensive male rats. 23 6-Gingerol (1-[4′-hydroxy-3′-methyoxyphenyl]-5-hydroxy-3-decanone (6G); Figure 1) is one of the principal bioactive components in ginger which is well documented to exhibit anti-inflammatory, anticancer and antioxidant properties. 24 Recent studies from our laboratory and others have demonstrated that 6G elicited anti-colitis effects in rodents through augmentation of endogenous antioxidant status, suppression of pro-inflammatory mediators and biomarkers of oxidative stress in mice. 25,26

Chemical structure of tested 6G. 6G: 6-gingerol.
However, there is dearth of information on the influence of 6G on colitis-associated testicular toxicity in experimental animals. Thus, based on the previously documented anti-colitis effects of 6G and its proven anti-inflammatory and antioxidant activities, the present study evaluated for the first time, the possible modulatory role of 6G in colitis-mediated reproductive dysfunction in male mice. SZ was used in this study as standard reference.
Materials and methods
Chemicals
DSS (molecular weight 37–40 kD) was procured from TdB Consultancy (Uppsala, Sweden). Reduced glutathione (GSH), epinephrine, hydrogen peroxide (H2O2), thiobarbituric acid, 5′,5′-dithio-bis-2-nitrobenzoic acid and 1-chloro-2,4-dinitrobenzene were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). All other reagents were of analytical grade and purchased from the British Drug House (Poole, Dorset, UK).
Isolation of 6G
Ginger plants comprising the rhizomes and the foliage were purchased from a local vendor in Bodija Market, Ibadan, Nigeria. The authentication of the plant samples was carried out by Mr Dunatus Esimekhuai at the Department of Botany, University of Ibadan, where a sample (voucher specimen number UIH-22390) was deposited in the herbarium. Subsequently, 6G was isolated from the ginger rhizomes and characterized according to the standardized method. 25 The purity of the isolated 6G is 91%.
Animal husbandry
Sixty sexually matured male BALB/c mice (6 weeks old, 21.4 ± 2.5 g) were obtained from the Faculty of Veterinary Medicine, University of Ibadan. The mice were housed in plastic cages and allowed to acclimatize for a week before starting the experiment. The mice were allowed free access to standard mice chow and drinking water and subjected to natural photoperiod of 12-h light:12-h dark cycle. All the animals received humane care according to the criteria specified in the ‘Guide for the Care and Use of Laboratory Animals’ prepared by the National Academy of Science and published by the National Institute of Health. The experimental procedures were carried out after approval by the University of Ibadan ethical committee. The ethical regulations have been followed according to the national and institutional guidelines for the protection of animal welfare during experiments. 27
Experimental design
The study consisted of six groups of ten mice each. The experimental design involved treatment of the mice for 63 consecutive days as follows: Group I: Control mice orally treated with corn oil at 2 mL/kg. Group II: Mice orally treated with 100 mg/kg of 6G alone. Group III: Mice orally treated with 100 mg/kg of SZ alone and served as standard reference. Group IV: Mice were exposed to three cycles of 2.5% DSS in drinking water. Group V: Mice were orally treated daily with 100 mg/kg of 6G during exposure to three cycles of 2.5% DSS in drinking water. Group VI: Mice were orally treated daily with 100 mg/kg of SZ during exposure to three cycles of 2.5% DSS in drinking water. Note: A cycle is defined as 7 days of exposure to 2.5% DSS in drinking water followed by 14 days of normal drinking water.
All the mice were sacrificed 24 h by cervical dislocation after the last treatment. The blood was collected from retro-orbital venous plexus using heparin-containing tubes. Plasma samples obtained by centrifugation of the blood samples at 3000 g for 10 min were subsequently used for the determination of the reproductive hormones concentrations using enzyme-linked immunosorbent assay (ELISA) strip reader (Robonik India Private Limited, Mumbai, India). The colons and testes were immediately excised, and the colons measured longitudinally after flushing with ice-cold phosphate-buffered saline. Subsequently, the testes and the colons were blotted with filter paper and weighed. The testes were then processed for biochemical determinations and histology.
Assessment of clinical manifestations of colitis
The clinical manifestations of colitis were evaluated according to the method described by Cooper et al., 28 using diarrhoea (as detected by perianal fur soiling), rectal bleeding and the colon mass index, calculated as the ratio of colon weight in milligrams to total body weight in grams, which was used to evaluate the severity of colonic inflammation.
Determination of oxidative stress indices
The testes samples were homogenized in eight volume of 50 mM Tris–HCl buffer (pH 7.4) containing 1.15% potassium chloride. Subsequently, the homogenate was centrifuged at 10,000 × g for 15 min at 4°C and the supernatant was used for the biochemical estimations. Protein concentration was assayed according to the method of Lowry et al., 29 using bovine serum albumin as standard. Superoxide dismutase (SOD) activity was assayed according to the method described by Misra and Fridovich. 30 Catalase (CAT) activity was assayed using H2O2 as a substrate according to the method described by Clairborne. 31 Glutathione peroxidase (GPx) activity was assayed according to the method described by Rotruck et al. 32 GSH was assayed according to the method described by Jollow et al. 33 H2O2 generation was assayed according to the method described by Wolff. 34 Lipid peroxidation was determined as malondialdehyde levels (MDA) according to the method described by Farombi et al. 35
Testicular concentrations of pro-inflammatory biomarkers
Nitric oxide (NO) level was assayed by measuring the testicular nitrites content, the stable end products of NO. Testicular nitrites content was obtained using a sodium nitrite curve as standard and expressed as micromolar of nitrites per milligram of protein according to the method described by Green et al. 36 Myeloperoxidase (MPO) activity was assayed according to the method described by Granell et al. 37 MPO activity was expressed as micromolar of H2O2 per minute per milligram of protein. The testicular concentrations of pro-inflammatory cytokines namely interleukin 1β (IL-1β) and tumour necrosis factor alpha (TNF-α) were assessed with the aid of DMM 9602 Microplate Reader (China) using commercially available ELISA kits (ABCAM PLC, UK).
Determination of activities of marker enzymes of testicular function
Marker enzymes of testicular function were determined in the testes supernatant. Lactate dehydrogenase-X (LDH-X) activity was determined according to the method described by Vassault, 38 which is based on the interconversion of pyruvate and lactate. Acid phosphatase (ACP) and alkaline phosphatase (ALP) activities were assayed according to established method, 39,40 which is based on the hydrolysis of p-nitrophenyl-phosphate in acid and alkaline medium, respectively.
Sperm progressive motility assay
The progressive motility of the sperm was assessed according to the method described by Zemjanis. 41 Briefly, epididymal sperm from the mice was obtained by cutting the cauda epididymis with surgical blades and released onto a sterile clean glass slide. Subsequently, the sperm was diluted with 2.9% sodium citrate dehydrate solution which had been pre-warmed to 37°C, mixed carefully and covered with a 24 × 24 mm coverslip. The sperm motility was observed under a phase contrast microscope in 10 microscopic fields at 200× magnification. Sperm motility was calculated by scoring the number of all progressive sperm, followed by the non-progressive and then the immotile sperm in the same field. The data were expressed as percentage of sperm progressive motility.
Epididymal sperm count
The epididymal sperm count was determined according to the method described in the World Health Organization manual. 42 Briefly, the sperm from the mice was obtained by mincing the caudal epididymis in normal saline and filtering through a nylon mesh. An aliquot of 5 µL of the sperm was mixed with 95 µL of diluent (0.35% formalin containing 5% NaHCO3 and 0.25% trypan blue). Subsequently, 10 µL of the diluted sperm was transferred the haemocytometer, allowed to sediment by standing for 5 min before counting using the improved Neubauer (Deep 1/10 m; LABART, Munich, Germany) chamber with a light microscope at 400×.
Assessment of sperm viability and morphological abnormalities
Sperm viability was determined by staining the sperm cells with 1% eosin and 5% nigrosine in 3% sodium citrate dehydrate solution according to Wells and Awa. 43 Morphological examination was carried out according to Wells and Awa 43 by placing a portion of the sperm suspension on a glass slide, smeared out with another slide and stained with a reagent containing 0.2 g eosin and 0.6 g fast green dissolved in distilled water and ethanol at a ratio of 2:1. A total of 400 sperm cells from each mouse were used for morphological examination.
Estimation of reproductive hormones
The circulatory concentrations of reproductive hormones namely FSH (RPN 2560; Amersham, UK), LH (RPN 2562; Amersham, UK) and testosterone (EIA-5179; DRG Diagnostics GmbH, Marburg, Germany) were assessed using commercial enzyme immunoassay kits according to the manufacturer’s protocols. The sensitivity of LH was 0.08 ng at 85% whereas FSH sensitivity was 0.06 ng at 96%. The sensitivity of the testosterone assay was 0.05 ng/mL with negligible cross-reactivity with other androgen derivatives such as androstenedione, methyl testosterone and 5α-dihydrotestosterone. The intra-assay coefficients of variations were 3.0% for LH, 3.7% for FSH and 3.6% for testosterone, respectively. All the samples were determined on the same day to avoid the inter-assay variation.
Determination of testicular caspase-3 activity
The testicular caspase-3 activity was determined with the aid of DNM 9602 Microplate Reader (China) using commercially available ELISA kits (Elabscience Biotechnology Company, Beijing, China) in accordance with the procedure described in the assay manual.
Histological examination
Representative testes specimens were fixed with Bouin’s solution and processed for histology according to standardized procedure. 44 Briefly, the tissues were dehydrated in an increasing concentration of alcohol, cleared twice in xylene with bench top tissue processor (Leica TP102; Illinios, USA) and embedded in paraffin using Leica EG1150H Illinios, USA. Subsequently, 4–5 μm sections of the embedded tissues were sectioned by a microtome and stained with haematoxylin and eosin. The slides were examined under a light microscope (Olympus CH; Olympus, Tokyo, Japan) and photomicrographs were taken with a Sony DSC-W 30 Cyber-shot (Sony, Tokyo, Japan) by pathologists who were blinded to control and treatment groups.
Statistical analysis
Statistical analyses were carried out using one-way analysis of variance to compare the various experimental groups followed by Bonferroni’s test to identify significantly different groups (SPSS for Windows, version 17). Values of p < 0.05 were considered significant.
Results
6G suppressed DSS-induced colitis and decrease in testes weight
The body weight gain, incidence of diarrhoea and bleeding, colon mass index and testes weight of control and experimental mice are presented in Table 1. Oral administration of DSS alone in mice resulted in a severe disease characterized by decrease in body weight gain and increase in the incidence of diarrhoea and rectal bleeding in the mice. Further, there was marked reduction in colon mass index, the absolute and relative testes weight (RTW) gain in mice exposed to DSS alone as compared to control. There were no treatment related effects of 6G or SZ administered alone when compared with the control. However, co-administration of 6G or SZ with DSS decreased the incidence of diarrhoea and rectal bleeding as well as restored the body weight gain, colon mass index, RTW and testes weight in as compared to mice that received DSS alone (p < 0.05).
Effects of 6G and SZ on body weight gain, diarrhoea, rectal bleeding, colon mass index and testes weight in DSS-treated mice.

6G: 100 mg/kg of 6-gingerol; SZ: 100 mg/kg of sulfasalazine; DSS: 2.5% dextran sulphate sodium in drinking water; BWG: body weight gain; CMI: colon mass index; TW: testes weight; RTW: relative testes weight.
aValues represent the mean ± SD of 10 mice.
bValues differ significantly from control (p < 0.05).
cValues differ significantly from 6G alone (p < 0.05).
dValues differ significantly from DSS group (p < 0.05).
eValues differ significantly from DSS + 6G group (p < 0.05).
6G inhibited testicular oxidative damage in DSS-exposed mice
To evaluate the status of antioxidant defence mechanisms in testes of mice during DSS-induced colitis, the antioxidant enzymes activities (i.e. SOD, CAT and GPx) and the levels of GSH, H2O2 and MDA were assessed in the testes homogenates. The influence of 6G on the testicular antioxidant status following induction of colitis by exposure to DSS is presented in Figures 2 and 3. The testicular activities of SOD, CAT and GPx were significantly decreased in the mice exposed to DSS alone when compared with the control. However, co-administration of 6G restored the activities of these enzymes and maintained their normalcy in DSS-exposed mice. Further, there was a significant reduction in the GSH level with significant increase in the levels of H2O2 and MDA in testes of mice exposed to DSS alone when compared with the control group. 6G co-treatment significantly increased GSH level and restored the H2O2 and MDA levels to normalcy in testes of DSS-treated mice. Administration of SZ alone significantly decreased the antioxidant enzymes activities, GSH level and increased the H2O2 and MDA levels in the testes of mice when compared with the control group. Moreover, the mice co-treated with SZ and DSS showed significant increase in GSH level and decreased H2O2 and MDA levels in testes of the treated mice as compared to DSS alone. However, co-administration of 6G was more effective than co-administration SZ in inhibiting testicular oxidative damage in DSS-exposed mice.

Effects of 6G on testicular antioxidant enzymes activities in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase.

Effects of 6G on testicular oxidative stress indices in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water; CAT: catalase; GPx: glutathione peroxidase.
6G suppressed inflammatory biomarkers in testes of DSS-exposed mice
To examine the influence of 6G on inflammatory biomarkers in testes of DSS-exposed mice, the levels of IL-1β, TNF-α, NO and MPO activity were measured in testicular tissues of the mice. The effects of treatments on the inflammatory mediators are depicted in Figure 4. Administration of DSS alone resulted in a significant elevation in the pro-inflammatory cytokines, namely, IL-1β and TNF-α, as well as in NO level and MPO activity when compared with the control group. However, co-treatment with 6G or SZ significantly inhibited the increases in these inflammatory biomarkers in testes of the DSS-treated mice. Administration of 6G or SZ alone did not affect the levels of these biomarkers of inflammation in the treated mice as compared to control. However, co-administration of 6G was more effective than SZ in inhibiting testicular IL-1β level and MPO activity in DSS-exposed mice.
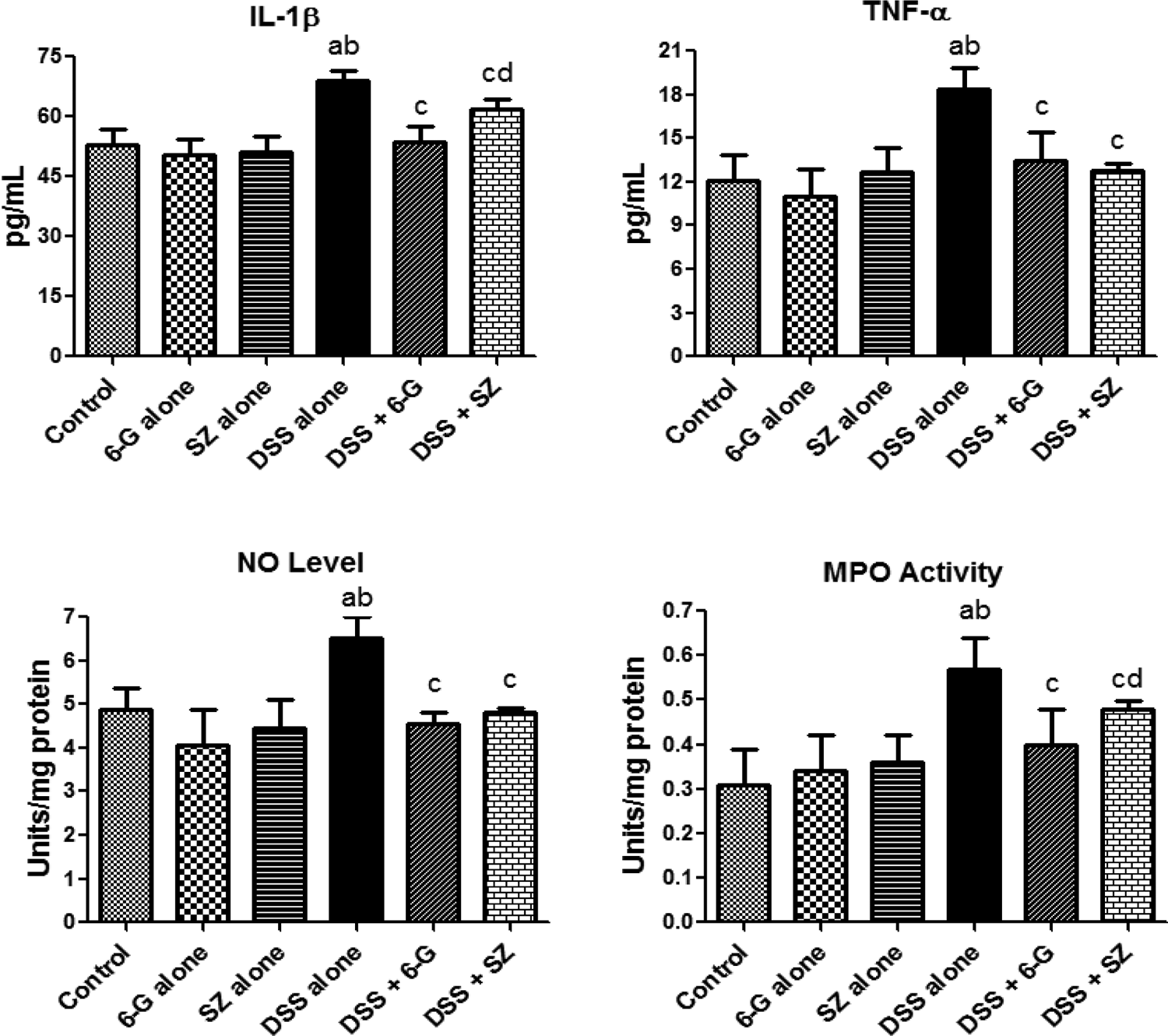
Effects of 6G on testicular inflammatory biomarkers in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water; IL-1β: interleukin 1β; TNF-α: tumour necrosis factor alpha; NO: nitric oxide; MPO: myeloperoxidase.
6G ameliorates colitis-induced decreases in sperm functional characteristics
To investigate the influence of 6G and SZ administration on sperm functional characteristics which are vital for reproductive success, the assessment of sperm count, motility, viability and morphology was carried out in control and experimental mice. The influence of 6G or SZ administration alone and their respective co-administration on sperm characteristics in normal and DSS-exposed mice is presented in Figure 5. The results indicated a significant (p < 0.05) decrease in sperm count and motility in mice exposed to SZ alone and DSS alone whereas there were no treatment-related effects in mice treated with 6G alone when compared with the control. Co-administration of 6G or SZ with DSS significantly increased the sperm count and motility when compared with DSS-alone group. However, co-administration of 6G was more effective than SZ in boosting the sperm count and motility in DSS-alone-treated mice. Moreover, although the sperm viability remained unaffected in all the treatment groups, there were significant increase in the sperm abnormalities, consisting majorly head and mid-pieces abnormalities in DSS-exposed mice as compared to all other treated groups. Co-administration of 6G was more effective than SZ in decreasing the sperm abnormalities significantly to near normal in DSS-exposed mice.

Effects of 6-gingerol on sperm count, motility, viability and morphology in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water.
6G recovers colitis-induced deficits in circulatory concentrations of reproductive hormones
To delineate the mechanisms of chemoprevention of 6G against colitis-mediated testicular dysfunction, the levels of circulatory concentrations of reproductive hormones, namely, LH, FSH and testosterone, were measured in plasma of the experimental mice. The influence of 6G or SZ co-administration on circulatory concentrations of pituitary and testicular hormones in normal and DSS-exposed mice is depicted in Figure 6. There were no significant differences in the circulatory concentrations of LH, FSH and testosterone between the control and mice treated with either 6G alone or SZ alone. However, plasma concentrations of LH, FSH and testosterone were significantly (p < 0.05) decreased in mice exposed to DSS alone when compared with the control. Co-administration of 6G or SZ significantly reversed the reduction of these hormones by increasing their concentrations to near control in DSS-exposed mice.

Effects of 6G on circulatory concentrations of reproductive hormones in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water; FSH: follicle-stimulating hormone; LH: luteinizing hormone.
6G prevents colitis-induced inhibition of marker enzymes of testicular function
The influence of 6G on marker enzymes of testicular function was investigated to further delineate its protective mechanisms against colitis-mediated impairment in spermatogenesis in the treated mice. Figure 7 shows the influence of 6G administration on the marker enzymes of testicular function in DSS-exposed mice. The testicular activity of LDH was increased significantly whereas ACP and ALP activities were significantly decreased in mice exposed to DSS alone when compared with control. However, co-administration of 6G was more effective than SZ in preventing these alterations and restoring the activities of these enzymes toward normalcy in DSS-exposed mice.

Effects of 6G on marker enzymes of testicular function in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water; LDH-X: lactate dehydrogenase-X; ACP: acid phosphatase; ALP: alkaline phosphatase.
6G inhibits colitis-induced activation of caspase-3 activity in testes of mice
Further investigation into the involvement of caspase-3 activity (an executioner caspase in apoptosis) in the testis samples was conducted. The influence of 6G and SZ co-administration on the testicular caspase-3 activity in DSS-exposed mice is depicted in Figure 8. There were no significant differences in the testicular caspase-3 activity between the control and mice treated with either 6G alone or SZ alone. Mice exposed to DSS alone showed significant increase in caspase-3 activity when compared with control. However, co-administration of 6G was more effective than SZ in decreasing caspase-3 activity significantly to near normal in DSS-exposed mice.
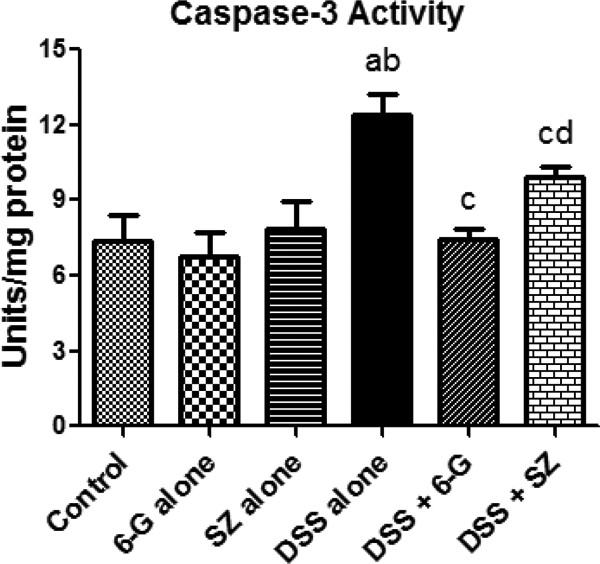
Effects of 6G on testicular caspase-3 activity in DSS-treated mice. Each bar represents mean ± SD of 10 mice. (a) Values differ significantly from control (p < 0.05). (b) Values differ significantly from 6G alone (p < 0.05). (c) Values differ significantly from DSS-alone-treated group (p < 0.05). (d) Values differ significantly from DSS + 6G co-treated group (p < 0.05). 6G, 100 mg/kg of 6-gingerol; SZ, 100 mg/kg of sulfasalazine; DSS, 2.5% dextran sulphate sodium in drinking water.
Histological observations
Figure 9 shows the representative photomicrographs of testes sections of control and experimental mice. The testes from control and 6G-alone-treated mice showed seminiferous tubules with normal architecture. Testicular sections from mice administered SZ alone showed seminiferous tubules with marked spermatogenic epithelium degeneration (green arrow). The DSS-alone exposed testes showed seminiferous tubules containing masses of sloughed spermatogenetic cells in their lumina (blue arrow). Testicular sections from mice co-administered with 6G and DSS showed closely packed seminiferous tubules with normal architecture. Mice co-administered with SZ and DSS showed seminiferous tubules with mild destruction of the spermatogenic epithelium.
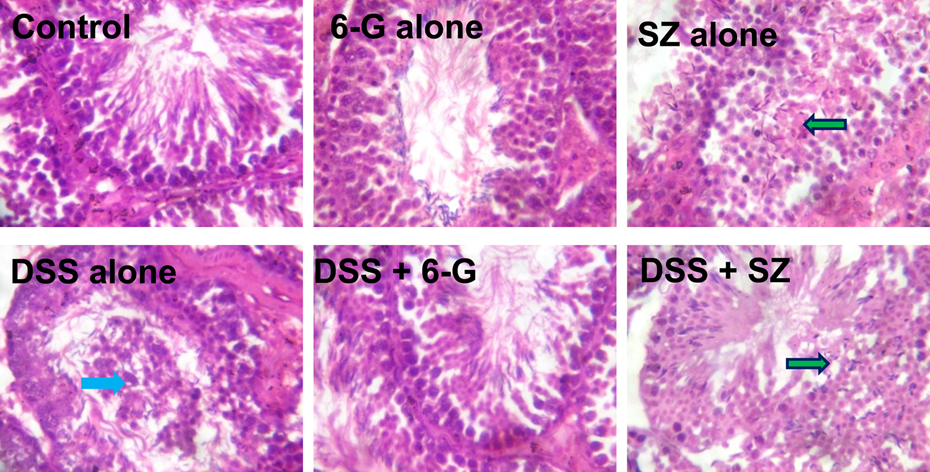
Photomicrographs of the testes of control and experimental mice. Testes from control and 6G-alone-treated mice showing normal architecture. Testes from SZ-alone-treated mice showing seminiferous tubules with marked spermatogenic epithelium degeneration (green arrow). DSS-alone-treated mice showing seminiferous tubules with masses of sloughed epithelial cells in their lumina (blue arrow). Testes of mice from DSS plus 6G showing closely packed seminiferous tubules with normal architecture. The seminiferous epithelium of mice co-treated with DSS and SZ (green arrow) showing mild destruction of the spermatogenic epithelium. Original magnification: ×400, H&E. 6G: 6-gingerol; SZ: sulfasalazine; DSS: dextran sulphate sodium; H&E: haematoxylin and eosin.
Discussion
The pharmacological activity of 6G administration in the colitis-mediated testicular toxicity in BALB/c mice was characterized in the present study. The chemical colitogen DSS is a useful tool for understanding the pathways and the possible targets for the treatment and management of colitis owing to its ability to mimic the clinical features of human UC. 25,45,46 In the present study, chronic exposure of mice to DSS alone resulted in diarrhoea, rectal bleeding and decreased body weight gain with a significant decrease in colon mass index when compared with control and other treatment groups. However, co-administration of 6G or SZ significantly ameliorated DSS-induced colitis evidenced by prevention of diarrhoea, rectal bleeding and decrease in body weight gain and mass index in the DSS-exposed mice. The present observations corroborate earlier findings on the anti-colitis effects of 6G. 25,26
Testicular weight is an important marker of male reproductive toxicity whereas the RTW is an established indicator of degenerative changes including swelling, atrophy or hypertrophy. 47 The decrease in the absolute and RTWs observed in mice exposed to DSS alone and SZ alone indicates testicular atrophy mediated by DSS-induced colitis and SZ, respectively. Our observation on the reduction in testes weight is in contrast to previous observations in acute testicular effect of SZ administration in Sprague Dawley rats for 14 days. 48 This contradiction may be due to differences in chronic treatment (i.e. 63 days) and animal model (i.e. mice) used in the present study. Moreover, administration of 6G alone did not affect the testes of the mice during this investigation. Co-administration of SZ alone did not change the decrease in testes weight in the DSS-exposed mice whereas 6G co-administration modulated it. The increased in both absolute and RTW in mice co-administered with 6G revealed a restoration of normal reproductive health possibly due to inhibition of the tissue degeneration in chronically DSS-alone exposed mice.
In the present study, the amelioration of colitis-mediated testicular oxidative damage in mice co-treated with 6G was evidenced by significant decrease in the levels of H2O2 and MDA in testes of DSS-exposed mice. The involvement of oxidative stress in colitis-mediated testicular damage in mice has been previously reported. 13 Moreover, the decrease in the activities of SOD, CAT and GPx with concomitant reduction in GSH level indicates suppression of the antioxidant defence mechanisms in the testes of DSS-exposed mice. The decrease in testicular SOD activity in mice exposed to DSS alone could lead to accumulation of ROS, which subsequently inactivated CAT and caused accumulation of H2O2 in testes of the treated mice. The reduction in GSH level suggests its overutilization in the detoxification of free radicals and may decrease GPx activity, which consumes GSH as a substrate during the removal of H2O2 and lipid hydroperoxides to prevent oxidative damage in the testes. 49 It is worthy of note that 6G co-administration protected against GSH diminution, inhibition of CAT and GPx activities as well as the H2O2 elevation in testes of the DSS-treated mice, thus indicating that the protective mechanism of 6G in testes of mice with colitis involves its antioxidant action.
Exposure of mice to DSS caused inflammatory reaction with elevated levels of TNF-α, IL-1β, NO and MPO activity in testicular tissue. Elevated TNF-α level is well known to up-regulate the inducible NO synthase, which subsequently increases NO production. 50 Similarly, high levels of NO exerts toxicological effects due to its ability to react with superoxide anion to generate peroxynitrite radical which causes further cellular damage. The significant decrease in the levels of TNF-α, IL-1β and NO in testes of DSS-exposed mice during 6G co-administration indicates its anti-inflammatory activity in testicular tissue. Besides, MPO is a well-known index of inflammation damage. 51 Persistent testicular inflammation could activate MPO activity, an enzyme which utilizes H2O2 to generate hypochlorite and ROS which eventually may result in testes damage. Thus, the increase in the testicular MPO activity in DSS-exposed mice in the present study indicates testicular inflammation damage in the mice. Our data corroborate the increase in the testicular MPO activity previously reported in mice with UC. 13 The apparent reduction in MPO activity in mice co-treated with 6G indicates the efficacy of 6G in suppressing inflammatory infiltrates and MPO activity in the testes of DSS-exposed mice.
The present investigation demonstrated significant decrease in sperm count and sperm motility with significant increase in sperm abnormalities in DSS-exposed mice. These findings suggest that the observed testicular oxidative damage was sufficient to impair sperm quality and quantity in mice with colitis. The alteration in the regulation of the hypothalamus-pituitary-testicular axis was also obvious in the present study. During spermatogenesis, LH stimulates Leydig cell to synthesize and secrete testosterone while both LH and FSH cooperate to enhance spermatogenic cell survival by suppressing pro-apoptotic signals. 52 Thus, the significant decrease in the sperm functional parameters in the present study may be associated with the decrease in circulatory concentrations of testosterone in the DSS-alone-treated mice. Consistent with the previous investigation, 48 the present data showed that administration of SZ alone significantly decreased sperm count and sperm motility of the treated mice. The lack of treatment-related effects of SZ on the investigated hormones may indicate direct toxic effect of SZ on the testes of the mice. However, evaluation of the spermiogram and circulatory hormones concentrations in mice co-administered with 6G or SZ revealed that 6G improved LH, FSH and testosterone levels and restored sperm parameters in DSS-exposed mice more than the reference drug, SZ.
The involvement of LDH, ACP and ALP in energy metabolism and stabilization of the testes is vital for spermatogenesis and sperm maturation. 53 LDH is a testicular germ cell marker enzyme, which plays an important role in the lactate metabolism of spermatogenic cells. 54 Lactate is produced by the Sertoli cells but it is utilized primarily by the germ cells for adenosine triphosphate production during spermatogenesis. 55 Moreover, lactate has been reported to effectively inhibit apoptosis of testicular germ cells in a dose-dependent manner. 56 Thus, the increase in testicular LDH in mice treated with DSS alone and SZ alone may indicate LDH protective mechanism against germ cell death in the treated mice. ALP is a biomarker for primordial germ cells and its high activity in spermatogonium is related to mitosis and transfer of glucose to spermatogenic cells. 53 Thus, the decrease in the activities of ACP and ALP in DSS-alone-treated mice may reflect impairment of mitosis and glucose utilization in germ cells. However, the restoration of LDH, ACP and ALP activities in mice co-treated with 6G indicates the protection of the testicular germ cells from colitis-mediated degeneration, as evidenced histologically by the significant amelioration of seminiferous tubule degeneration.
Elevated ROS production and oxidative changes have been implicated in apoptosis of numerous cell types. 57,58 Caspase-3 is a key executioner protease, which is responsible for the cleavage of cellular proteins that produce morphologic features of apoptosis. 59,60 Interestingly, the present investigation showed that chronic exposure to DSS alone increased caspase-3 activity in testes of mice with colitis, thus suggesting induction of apoptotic cell death. The apparent decrease in testicular caspase-3 activity in mice co-treated with 6G indicates the efficacy of 6G in suppressing caspase-3 activity in the testes of DSS-exposed mice. Furthermore, co-administration of 6G was more effective than SZ in suppressing caspase-3 activity in testes of DSS-exposed mice. These findings suggest that 6G, a naturally occurring compound, may be beneficial than SZ in ameliorating testicular toxicity especially when administered for a long time. Thus, 6G, an important phytochemical with antioxidant activity, showed a higher efficacy in suppressing testicular apoptotic cell death when compared with SZ.
In conclusion, the findings from the present study highlight the chemoprotective mechanisms of 6G in colitis-induced testicular toxicity in mice (Figure 10). 6G efficiently ameliorated experimental colitis-mediated testicular damage via mechanisms involving inhibition of inflammation, caspase-3 activity and oxidative and nitrosative stress along with augmentation of sperm functional characteristics. Thus, 6G may be a safe and potent drug candidate for the re-establishment of reproductive health in humans afflicted with UC.

Proposed pathway delineating the cellular and molecular mechanisms of 6G chemoprotection against colitis-mediated testicular toxicity in mice. 6G: 6-gingerol; SZ:sulfasalazine; DSS: dextran sulphate sodium; SOD: superoxide dismutase activity; CAT: catalase; GPx: glutathione peroxidase; GSH: reduced glutathione; H2O2: hydrogen peroxide; LPO: lipid peroxidation; IL-1β: interleukin; TNF-α: tumour necrosis factor alpha; NO: nitric oxide; MPO: myeloperoxidase; FSH: follicle-stimulating hormone; LH: luteinizing hormone; LDH: lactate dehydrogenase-X; TSN: testosterone; ACP: acid phosphatase; ALP: alkaline phosphatase; CASP3: caspase-3.
Footnotes
Acknowledgements
The technical assistance of Dr Ganiyat K. Oloyede and Mr C.C. Nwabueze of the Department of Chemistry, University of Ibadan, is appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Multidisciplinary Research Grants under the Staff Training and Research Capacity Building Programme of the John D. and Catherine T. MacArthur Foundation Grant (USA) endowment from the University of Ibadan, Nigeria, awarded to EOF.
