Abstract
High fructose corn syrup (HFCS) has been shown to cause cardiovascular toxicity via oxidative stress and inflammation. The aim of this study is to demonstrate the protective effects of melatonin (MLT) against HFCS-induced endothelial and cardiac dysfunction via oxidative stress and inflammation. Thirty-two Sprague Dawley male rats were distributed into three groups as control, HFCS, and HFCS + MLT. HFCS form F55 was prepared as 20% fructose syrup solution and given to the rats through drinking water for 10 weeks, and MLT administrated 10 mg/kg/day orally for last 6 weeks in addition to F55. After decapitation, blood and half of the heart samples were collected for biochemical analysis and other half of the tissues for histopathological and immunohistochemical analysis. Aspartate transaminase, creatine kinase MB, lactate dehydrogenase, total oxidant status and oxidative stress index, and caspase-3 levels increased and total antioxidant status levels decreased significantly in HFCS group. MLT treatment reversed all these parameters. Histopathologically, hyperemia, endothelial cell damage and increased levels of angiogenin, C-reactive protein, inducible nitric oxide synthase, myeloperoxidase and decreased sirtuin-1 (SIRT-1) expressions were observed in HFCS group. MLT ameliorated all these changes. MLT has an anti-inflammatory, antioxidant, antiapoptotic effects on HFCS-induced cardiovascular toxicity through enhancing the expression of SIRT-1.
Introduction
Over the last 30 years, high fructose corn syrup (HFCS) has been widely used in many commercial products, because it extends the shelf life of foods, does not disturb its normal taste, and has a low price. 1,2 Currently, the daily intake of fructose in children and adults is estimated to be 54.7 g in United Nations. 3 Different forms of HFCS (F30, F42, F55) contain 30%, 40%, and 55% fructose, respectively, and loads in body approximately 60–70 mg by daily consumption. 4 Therefore, it affects human body negatively by the wide range of products, and studies should be carried out to eliminate harmful effects, mainly as metabolic syndromes. 5,6 HFCS-55 is the most commonly used among these sweeteners, and 20% HFCS intake causes more serious metabolic consequences than other forms. 7,8 Metabolic syndromes such as hypertension, obesity, and insulin resistance have been shown to developed in HFCS-fed rats. 9 –11
It is known that high fructose consumption increases the incidence of many metabolic disorders as obesity and hypertension via oxidative stress, inflammation, and apoptosis. 12,13 Inflammation, which has a key role in the pathophysiology of metabolic disorders, may increase the risk of cardiovascular diseases as well. 6,14 Inflammation and oxidative stress have a relationship and they induce each other’s by various mechanisms; increasing level of damage by hyperglycemic activity triggers the release of pro-inflammatory cytokines, increases oxidative stress and apoptosis, with the decrease of antioxidant enzymes. 15 Utilization for detoxification or reduced production of antioxidant enzymes contribute the excessive damages. 16
Pro-inflammatory factors promote the formation of metabolic and vascular insulin resistance, and thus increasing the risk of development of cardiovascular diseases. 17 Fructose intake reduces endothelial nitric oxide synthase (eNOS) activity and leads to decrease the level of endothelial nitric oxide; this decrement also triggers insulin resistance and hypertension. 18 Besides, it causes myocardial damage by these mechanisms and elevates cardiac damage markers such as creatine kinase MB (CKMB) and lactate dehydrogenase (LDH). 19 –21
Sirtuin-1 (SIRT-1), acetylating protein, induces eNOS levels, enhances antioxidant enzyme synthesis, and ameliorates the increased levels of pro-inflammatory cytokines to contribute the anti-inflammatory and antioxidant response of tissues such as the heart and endothelium. 22 –27
Chronic HFCS consumption increases oxidative stress, inflammation, and endothelial dysfunction via raising some parameters such as angiogenin (ANG), inducible nitric oxide synthase (iNOS), an acute phase reactant, C-reactive protein (CRP), myeloperoxidase (MPO), and decreasing SIRT-1. These markers have an important role in metabolic health of many tissues including heart and endothelium. 28 –30
Besides these indicators, the caspase-3 (Cas-3), a member of the cysteine-aspartic acid protease, plays a central role in the execution phase of cell apoptosis; some stressing factors as oxidative stress and inflammation are known to trigger intracellular apoptotic signaling. 31 As a response to these factors, extrinsic and intrinsic pathways activate Cas-3 to initiate apoptotic process in apoptotic cells which interacts with Cas-8, 9, and 12. 32
Melatonin (MLT), an M1/M2 receptors analog, has a strong antioxidant and anti-inflammatory activity which act to all subcellular structures due to lipophilic properties in vascular and cardiac tissues. 33 It decreases inflammation, mitochondria-triggered oxidative stress, and blood pressure in obesity and hypertension by anti-inflammatory and antioxidant properties. 34,35
The aim of this study is to show the protective effects of MLT on HFCS-induced endothelial and cardiac dysfunction via oxidative stress and inflammation.
Materials and methods
Animals
This experimental study was approved by Mehmet Akif Ersoy University Animal Experiments Local Ethics Committee (Ethic no: 241/30.11.2016). Thirty-two male, 12–16 months aged, weighing 150–250 g Sprague Dawley rats were maintained at a constant ambient temperature of 21–23°C, with a humidity of 55–60%, 12 h of light and 12 h of darkness. Feed (Standard Rat Feed, Feed My Korkuteli, Turkey) and fluids were given ad libitum. The stools and the fluids they got were changed daily.
Experimental conditions
The rats were distributed into three groups (12 rats in each group). Groups were as follows.
1. Control group: 1 ml 0.9% sodium chloride solution orally for last 6 weeks.
2. HFCS group: HFCS form F55 was prepared as 20% fructose syrup solution and given to the rats through drinking water for 10 weeks.
3. HFCS + MLT group: 20% of F55 corn syrup in drinking water for first 4 weeks, then 10 mg/kg/day MLT orally for 6 weeks in addition to F55. 36
The concentration of corn syrup was chosen according to a study about HFCS administration and investigating the sugar content in beverages. 37 As the treatment, MLT was preferred to use from fourth week to the end of the study because insulin resistance was shown to develop at the end of the fourth week approximately by corn syrup consumption in rats, previously. 38
HFCS (F55: 56% fructose and 37% glucose) was obtained from a local company (Toposmanoglu, Isparta, Turkey), and MLT were obtained from Sigma-Aldrich Chemical Inc. (St Louis, Missouri, USA). All reagents except phosphate buffers were prepared on the day of experiment and used fresh. During the 10-week trial period, six animals died. Animals were decapitated under anesthesia with 10% ketamine (Alfamin, Alfasan IBV) and 2% xylazine (Alfazin, Alfasan IBV), administered intraperitoneally. Blood samples were collected for biochemical analysis. Half of the heart tissues were placed into Eppendorf tubes and stored at −20°C until assayed for biochemical analysis and another half of heart tissues and endothelial tissues were collected during the necropsy and fixed in 10% buffered formaldehyde solution for histopathological and immunohistochemical analysis.
Biochemical analysis
Measurement of biochemical cardiac markers in blood
An autoanalyzer (Beckman Coulter AU680, Brea, California, USA) is used to determine the activities of CKMB, LDH activities, and aspartate transaminase (AST) levels in the supernatants of the blood samples.
Measurement of the total antioxidant status
Total antioxidant status (TAS) was measured using a novel automated colorimetric measurement method developed by Erel. In this method, antioxidants in the sample reduce dark blue-green colored 2,2′-azino-bis (3-ethylbenzthiazoline-6-sulphonic acid) (ABTS) radical to colorless reduced ABTS form. The change of absorbance at 660 nm is related with total antioxidant level of the sample. This method determines the antioxidant effect of the sample against the potent free radical reactions initiated by the produced hydroxyl radical. The results are expressed as millimolar Trolox equivalent per liter. 39
Measurement of total oxidant status and oxidative stress index
Total oxidant status (TOS) was measured using a novel automated colorimetric method described by Erel. In this method, oxidants present in the sample oxidize the ferrous ioneo-dianisidine complex to the ferric ion. The oxidation reactions are enhanced by glycerol molecules, which are abundantly present in the reaction medium. The ferric ion forms a colored complex with xylenol orange in an acidic medium. The color intensity, which can be measured spectrophotometrically, is related to the total amount of oxidant molecules present in the sample. The assay is calibrated with hydrogen peroxide, and the results are expressed in terms of micromolar hydrogen peroxide equivalent per liter (µmol hydrogen peroxide equiv./L) 40 Oxidative stress index (OSI) was calculated using the formula OSI = TOS/TAS. 41
Western blot analysis
Heart tissues were dissected, homogenized, and lysed in ice-cold Radioimmunoprecipitation assay buffer (RIPA) buffer (1× PBS, 1% nonidet P-40, 0.5% sodium deoxycholate, and 0.1% SDS, pH 8.0) containing a protease inhibitor. Following centrifugation at 14,000 r/min for 10 min at 4 °C insoluble materials were removed. Protein concentrations were determined by the Bicinchoninic acid assay (BCA) protein assay kit (Thermo Fisher Scientific, Pittsburgh, Pennsylvania, USA). Typically, 40-μg of total protein were used for immunoblotting (IB). Samples were denatured in 4x SDS-PAGE loading buffer (200-mM Tris-hydrochloride pH:6.8, 8% SDS, 0.4% bromophenol blue, 40% glycerol, 4% 14.7 β-mercaptoethanol, 10% 0.5-M EDTA) at 95°C for 5 min and were separated on hand cast polyacrylamide gels. Following electrophoresis, proteins were transferred to the Polyvinylidene Fluoride Membrane (PVDF) membrane (Millipore). After that typic IB steps (blocking, incubating with primary and secondary antibodies, respectively), specific proteins were visualized using Pierce ECL Western blotting substrate kit (Thermo Fisher Scientific) by ChemiDocTM Imaging System (Bio-RAD, California). Polyclonal anti-Cas-3 (9662); monoclonal anti-NF-kß (D14E12) and anti-p-NF-kß (Ser536) (93H1) were from Cell Signaling Technology, Danvers, MA; anti-ß-actin (A5316) was from Sigma Aldrich (St. Louis, Missouri) and Horseradish peroxidase (HRP)-conjugated anti-mouse or anti-rabbit Immunoglobulin G (IgG) was purchased from Pierce. Cleaved Cas-3 levels were analyzed using the densitometric analysis of three independent samples using Image J software (NIH, Bethesda, MD) (http://imagej.nih.gov/ij/).
Histopathological and immunohistochemical analysis
Histopathological examinations
Heart and aorta samples were collected and fixed in 10% neutral formalin solution during the necropsy. After routine tissue processing (Leica ASP300S, Wetzlar, Germany), they were embedded in paraffin and sectioned to 5-μm thickness (Leica RM2155 rotary microtome-Leica Microsystems, Wetzlar, Germany), stained with hematoxylin-eosin (HE) and examined under light microscope.
Immunohistochemical examinations
Samples were then immunostained with AGN (Anti-Angiogenin antibody (14017.7) (ab10600)), CRP (Anti-C Reactive Protein antibody—Aminoterminal end (ab65842)), iNOS (Anti-iNOS antibody (ab15323)), MPO (Anti-Myeloperoxidase antibody (ab9535)), and SIRT-1 (Anti-SIRT-1 antibody (E104) ab32441)) according to the manufacturer’s instructions using a routine streptavidin–biotin peroxidase technique. All primary antibodies were used in 1/100 dilution. Expose mouse and rabbit specific HRP/DAB detection IHC kit (ab80436) was used as secondary antibody. All kits were purchased from Abcam, UK. Positive controls were used for each antibody. Primary antibodies were not applied to the negative controls for immunohistochemistry.
For immunohistochemical procedure after deparaffinization and rehydration, hydrogen peroxide solution applied on the tissues. Then, the sections were incubated with the protein block for 20 min, and primary antibodies for 60 min, and immunohistochemistry was carried out using biotinylated secondary antibody and streptavidin-alkaline phosphatase conjugate 20 min for each one. EXPOSE Mouse and Rabbit Specific HRP/DAB Detection IHC kit (ab80436) were used as secondary antibody and diaminobenzidine (DAB) as the chromogen. For negative controls, primary antiserum step was omitted. All examinations were performed by a pathologist who was unaware the groups. All of the slides were analyzed for immunopositivity, and a semiquantitative analysis was conducted. Samples were analyzed by examining five different sections in each sample, which were then scored from 0 to 3 according to the intensity of staining (0: absence of staining, 1: slight; 2: medium, and 3: marked). For evaluation, 10 different fields for every section at a magnification under 40× objective for all groups. Results obtained from the image analyzer were subjected to statistical analysis. Morphometric analyses were performed using the Database Manual Cell Sens Life Science Imaging Software System (Olympus Co., Tokyo, Japan).
Statistical analyses
For biochemical and immunohistochemical parameters; one-way analysis of variance test was used to determine significant differences between the groups. To determine differences between the groups and compare the groups in immunohistochemical analyses, the Bonferroni–Dunn and Least Significant Differences (LSD) multiple comparison methods were used. Calculations were made using the SPSS 18.0 program (SPSS Inc., Chicago, Illinois, USA). Differences were considered as significant for p < 0.05. Descriptive statistics were given as mean ± standard deviation for each group.
Results
Biochemical analysis
Cardiac damage markers in blood
AST, CKMB, and LDH levels significantly increased in HFCS group compared with control group and these levels significantly decreased in MLT treatment group compared with HFCS group (p < 0.001 for all; Figure 1).

Biochemical analyses of cardiac markers levels in blood. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA test (Bonferroni test). ANOVA: analysis of variance; HFCS: high fructose corn syrup; MLT: melatonin. ***p < 0.001, HFCS versus control; †††p < 0.001, HFCS + MLT versus HFCS group.
Oxidative stress markers in cardiac tissue
There was a statistically significant difference in TAS, TOS, and OSI (p < 0.001 for all). TAS levels were decreased in HFCS and HFCS + MLT groups significantly compared with control group (p < 0.001 and p = 0.001, respectively). MLT treatment significantly increased TAS levels compared with HFCS group (p = 0.045). TOS and OSİ levels were significantly increased in HFCS group compared with control group (p < 0.001 for both) and significantly decreased in HFCS + MLT group compared with HFCS group (p < 0.001 and p = 0.001, respectively; Figure 2).

Oxidative stress markers of heart tissues. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA test (post hoc Least Significant Differences (LSD) test). ANOVA: analysis of variance; HFCS: high fructose corn syrup; MLT: melatonin. ***p < 0.05, HFCS versus control; †p < 0.05, †††p < 0.001, HFCS + MLT versus HFCS.
Western blot results of cardiac tissues
Fragmented Cas-3 (17 kDa) is one of the key mediators of the apoptotic process. Our IB results showed that cleaved Cas-3 levels observed in HFCS group decreased statistically significantly after MLT administration (Figure 3).
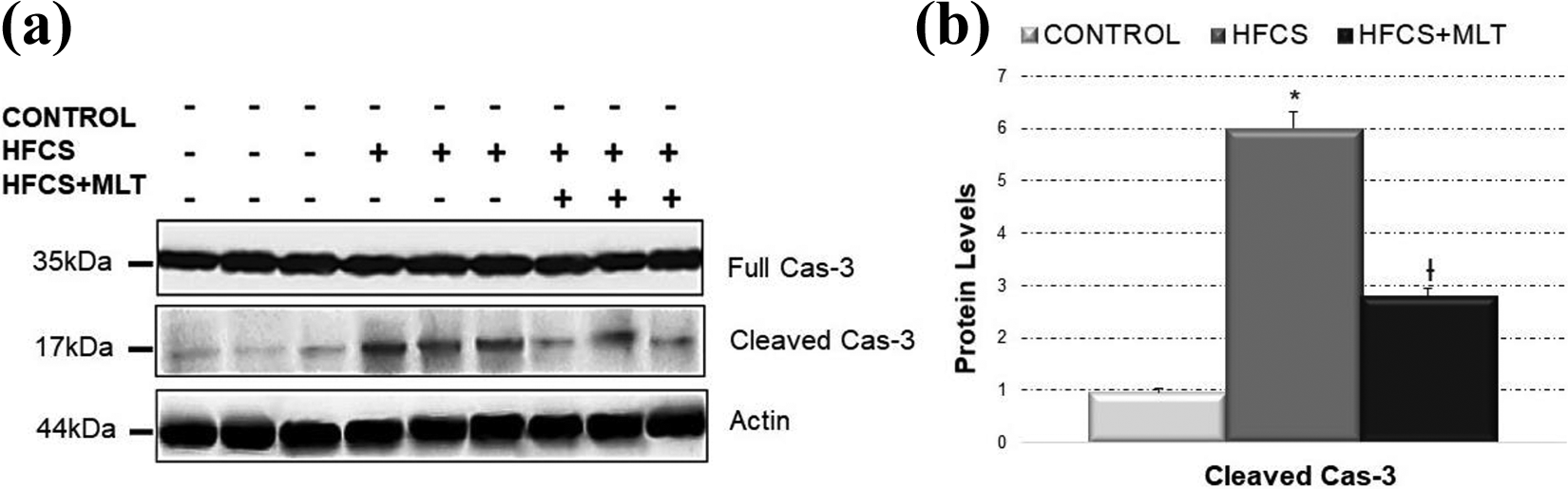
The protein expression levels of Cas-3 in cardiac tissues. (a) No changes in full Cas-3 expressions in any groups. (b) Cleaved Cas-3 expression was increased in HFCS and decreased in HFCS + MLT group. Cas-3: caspase-3; HFCS: high fructose corn syrup; MLT: melatonin. *p < 0.05, HFCS versus control; †p < 0.05, HFCS versus HFCS + MLT.
Histopathological and immunohistochemical results
Histopathology results
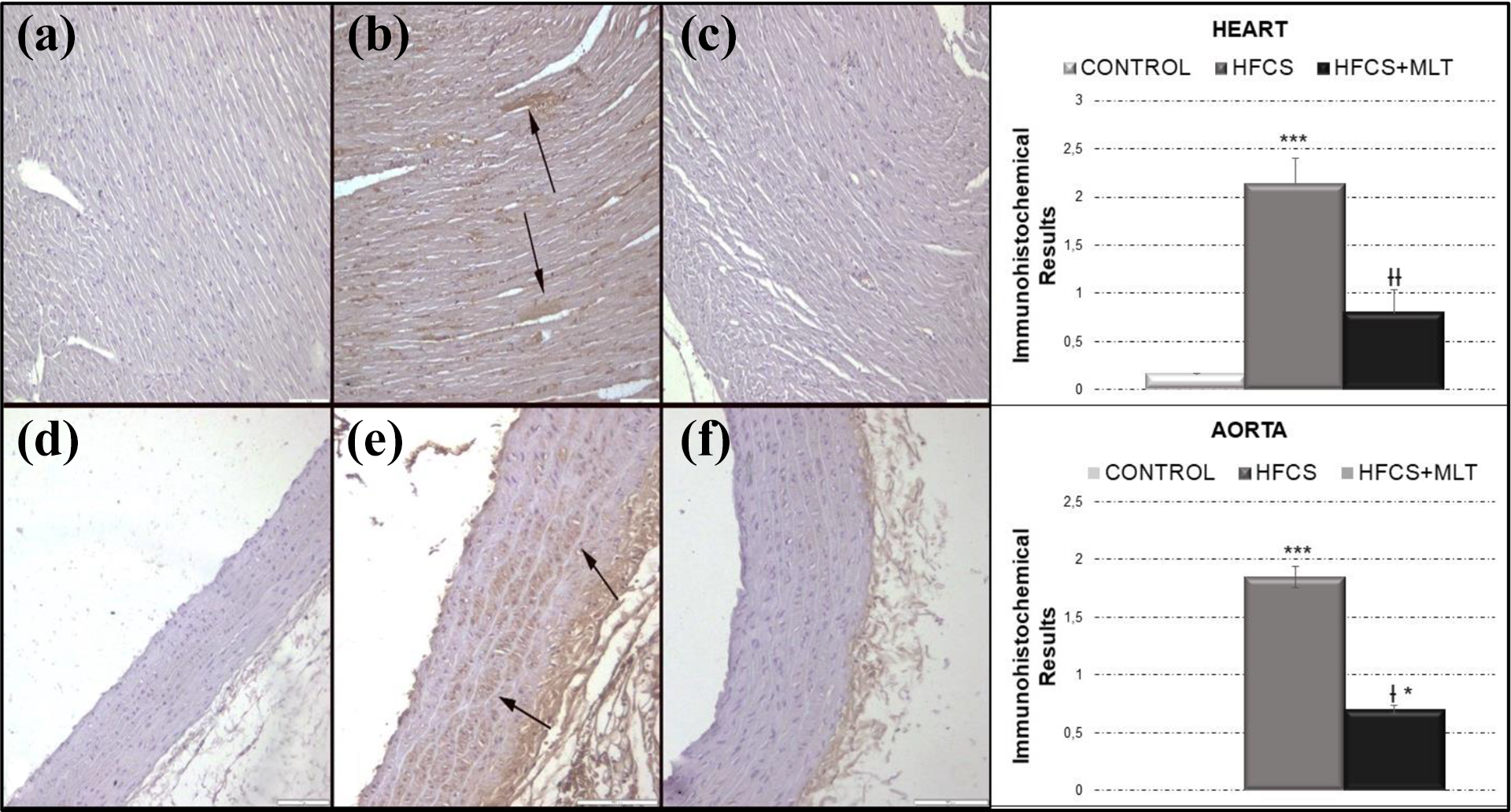
Hyperemia was the common histopathological finding in myocardium in HFCS group. No other pathological findings were observed in hearts in any groups. There were no marked histopathological changes noticed in aortas in any group except rare endothelial cell damage in HFCS group in some rats (Figure 4).

Histopathological appearance of the hearts and aortas. (a) Normal histological architecture in control group, (b) hyperemic vessels (arrows) in HFCS group, and (c) normal histology in HFCS + MLT group. HE, bar = 100μm. Microscopical appearance of the aortas between the groups. (d), (e) and (f) Normal histology in aortas in control, HFCS, and HFCS + MLT groups, respectively. HE, Bars = 100 μm. HFCS: high fructose corn syrup; MLT: melatonin; HE: hematoxylin-eosin.
Immunohistochemical results
AGN expression increased in both hearts and aortas in HFCS group (Figure 5). CRP also markedly expressed in myocardial cells and smooth muscle cells in medial layer of aortas in HFCS group (Figure 6). Although increase in iNOS expression was observed in myocardial cells in HFCS group, there was no expression in any group in aortas (Figure 7). Marked MPO expressions were noticed in hearts and aortas of HFCS group (Figure 8). SIRT-1 moderately expressed in myocardial cells of the heart and endothelial cells of aortas in control group while decreased in HFCS group. MLT treatment decreased the expression of all markers, except SIRT-1 (Figure 9). Statistical analysis of the results belonging to the expression of the markers was shown in Figures 5 to 9.

ANG expressions and immunohistochemical results in hearts and aortas. (a) No expression in control group, (b) marked increase in immunoreaction (arrows) in HFCS group, (c) negative expression in HFCS + MLT group, (d) no expression in control group, (e) increase expression in some smooth muscle cells (arrows) in HFCS group, and (f) slight expression in HFCS + MLT group. Streptavidin biotin peroxidase method, bars = 100 μm. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA Bonferroni test. ANG: angiogenin; ANOVA: analysis of variance; HFCS: high fructose corn syrup; MLT: melatonin. ***p < 0.001 compared with control; †p < 0.05, †††p < 0.001 compared with HFCS group.

CRP expressions and immunohistochemical results in hearts and aortas. (a) Negative expression in control group, (b) marked increase in myocardial cells (arrows) in HFCS group, (c) no expression in HFCS + MLT group, (d) negative expression in control group, (e) marked increase in smooth muscle cells (arrows) in HFCS group, and (f) no expression in HFCS + MLT group. Streptavidin biotin peroxidase method, bars = 100 μm. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA Bonferroni test. CRP: C-reactive protein; ANOVA: analysis of variance; HFCS: high fructose corn syrup; MLT: melatonin. *p < 0.05, ***p < 0.001 compared with control; †p < 0.05, ††p < 0.01 compared with HFCS group.

iNOS expressions and immunohistochemical results in hearts and aortas. (a) No immunoreaction in control group, (b) marked increase in myocardial cells (arrows) in HFCS group, (c) slight expression in HFCS + MLT group, and (d), (e), and (f) no expression in control, HFCS, and HFCS + MLT groups, respectively. Streptavidin biotin peroxidase method, bars = 100 μm. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA Bonferroni test. iNOS: inducible nitric oxide synthase; ANOVA: analysis of variance; HFCS: high fructose corn syrup; MLT: melatonin. ***p < 0.001 compared with control; ††p < 0.01 compared with HFCS group.
MPO expressions and immunohistochemical results in hearts and aortas. (a) Negative expression in control group, (b) increased expression in myocardial cells (arrows) in HFCS group, and (c) no expression in HFCS + MLT group. Myeloperoxidase expression in aortas in groups. (d) Negative expression in control group, (e) increased expression in smooth muscle cells (arrows) in HFCS group, and (f) slight expression in HFCS + MLT group. Streptavidin biotin peroxidase method, bars = 100 μm. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA Bonferroni test. MPO: myeloperoxidase; HFCS: high fructose corn syrup; MLT: melatonin; ANOVA: analysis of variance. *p < 0.05, ***p < 0.001 compared with control; †p < 0.05, ††p < 0.01 compared with HFCS group.

SIRT-1 expressions and immunohistochemical results in hearts and aortas. (a) Marked expression in control group (arrows), (b) decreased expression in myocardial cells (arrows) in HFCS group, (c) increased expression in HFCS + MLT group, (d) numerous positive endothelial cells in control group (arrows), (e) negative expression in HFCS group, and (f) marked expression in endothelial cells (arrows) in HFCS + MLT group. Streptavidin biotin peroxidase method, bars = 100 μm. Values are presented as means ± SD. The relationships between groups and results of biochemical markers are assessed by one-way ANOVA Bonferroni test. SIRT: sirtuin-1; ANOVA: analysis of variance; HFCS: high fructose corn syrup; MLT: melatonin. *p < 0.05, **p < 0.01, ***p < 0.001 compared with control; ††p < 0.01, †††p < 0.001 compared with HFCS group.
Discussion
Chronic consumption of corn syrup has been claimed as the responsible factor underlying the etiopathogenesis of many chronic diseases as diabetes mellitus, metabolic syndrome, hypertension, and so on. 42 Corn syrup induced inflammation and oxidative stress in blood triggering cardiac and endothelial damage by enhancing pro-inflammatory cytokine synthesis, leading to hyperuricemia and production of reactive oxygen species (ROS). 1
Heart and endothelial tissues are one of the main targets of corn syrup due to vascular structure. Enhancing the level of oxidative stress and inflammation in blood is the leading cause of tissue damage, which increase cumulatively in the case of chronic exposure. 43 Releasing of markers from heart like AST, CKMB, and LDH as a reflection of injury in blood accompanies to these changes. 44 In the current study, chronic consumption of HFCS increased abovementioned cardiac indicators, and MLT treatment ameliorated the changes
Besides the changed biochemical parameters in blood, changes were observed in tissues, too. Observed alterations in TAS, TOS, and OSI indexes addressed the oxidative stress in heart tissue which resulted the release of indicators mentioned earlier. As shown in a previous study, HFCS increases the level of several oxidative stress parameters and antioxidant enzymes as malondialdehyde and catalase in heart and endothelium. 4 Similar changes in oxidative stress markers through TAS and TOS levels were observed in this study. In many articles regarding HFCS and cardiac tissues, there is no satisfactory level of paper about the relationship between TAS/TOS levels and HFCS. As a potent antioxidant, MLT reversed the oxidative parameters of heart in previous studies. 45,46 As expected, MLT treatment ameliorated the oxidative changes in our study, too.
Production of ROS and methylglyoxal formation by HFCS lead to inflammatory changes through the mitochondrial and endoplasmic reticulum damages in heart and endothelial tissues. 25 Inflammatory changes induced by oxidative stress lead to activation of human antioxidant enzyme system as SIRT-1 and triggered catalase and superoxide dismutase enzymes. 47 In our previous study, we found that aspirin and vitamin C enhanced the decreasing levels of SIRT-1 and decreased the elevated levels of Cas-3 to protect the cardiovascular tissues against HFCS-induced oxidative stress, inflammation, and apoptosis. 1,48 Similarly, in current study, evaluating changes in protein levels with IB, we found that increased levels of cleaved Cas-3 in the HFCS group decreased significantly after MLT administration. Increased level of SIRT-1 plays a major role in defense system against to imbalanced homeostasis by blocking NF-kß transcription that promotes cytokine synthesis. 49
Similar to the literature, SIRT-1 levels decreased in HFCS group and MLT treatment reversed these parameters in heart tissue of this study. Increased SIRT-1 levels by MLT acted in two ways; first, it induced antioxidant enzyme synthesis and decreased oxidative stress, and second, it caused NF-kß downregulation that resulted in decrement of cytokine synthesis as iNOS, CRP, and MPO to ameliorate inflammation and apoptosis in current study, and MLT treatment decreased the levels of CRP, iNOS, and MPO, which reflect the severity of inflammation.
ANG, a potent inducer of blood vessel formation, takes places in inflammation by inducing angiogenesis. 50 HFCS increased ANG levels and causes hyperemia as shown in histopathological and immunohistochemical figures. Decreasing levels of ANG expression by MLT reflect that it also reduces the revascularity of inflammation in addition to the aforementioned anti-inflammatory effects.
In conclusion, MLT has an anti-inflammatory, antioxidant, and antiapoptotic effects on HFCS-induced cardiovascular toxicity through enhancing the expression of SIRT-1. This expression blocked NF-kß activation that leads to decrease the production of cytokines synthesis from nucleus to negatively effect on inflammation and apoptosis and enhances antioxidant enzyme levels to decrease oxidative stress. In the light of all findings, governments should either reduce the quota of this corn syrup or add preservatives such as MLT in addition to such foods.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
