Abstract
The aim of this study was to evaluate the role of α-lipoic acid (α-LA) on oxidative damage and inflammation that occur in endothelium of aorta and heart while constant consumption of high-fructose corn syrup (HFCS). The rats were randomly divided into three groups with each group containing eight rats. The groups include HFCS, HFCS + α-LA treatment, and control. HFCS was given to the rats at a ratio of 30% of F30 corn syrup in drinking water for 10 weeks. α-LA treatment was given to the rats at a dose of 100 mg/kg/day orally for the last 6 weeks. At the end of the experiment, the rats were killed by cervical dislocation. The blood samples were collected for biochemical studies, and the aortic and cardiac tissues were collected for evaluation of oxidant–antioxidant system, tissue bath, and pathological examination. HFCS had increased the levels of malondialdehyde, creatine kinase MB, lactate dehydrogenase, and uric acid and showed significant structural changes in the heart of the rats by histopathology. Those changes were improved by α-LA treatment as it was found in this treatment group. Immunohistochemical expressions of tumor necrosis factor α and inducible nitric oxide synthase were increased in HFCS group, and these receptor levels were decreased by α-LA treatment. All the tissue bath studies supported these findings. Chronic consumption of HFCS caused several problems like cardiac and endothelial injury of aorta by hyperuricemia and induced oxidative stress and inflammation. α-LA treatment reduced uric acid levels, oxidative stress, and corrected vascular responses. α-LA can be added to cardiac drugs due to its cardiovascular protective effects against the cardiovascular diseases.
Introduction
High-fructose (HF) corn syrup (HFCS) has several industrial advantages over sugar, such as freshness, texture, color, not masking original taste, and, especially, low price. 1 –3 HFCS has been named as F30, F42, and F55 according to the ratio (30%, 40%, and 55%, respectively) of fructose content. HF consumption for a long time has been shown to cause metabolic syndromes including insulin resistance, hypertriglyceridemia, hyperuricemia, and endothelial dysfunction in rats. 4 The proportions of this syndrome increase in the Western world with an estimated prevalence of around 25% epidemically. 5,6 The mortality associated with the syndrome is likely related to the cardiovascular diseases. These metabolic disorders are major risk factors for the development of cardiovascular diseases and type II diabetes, 7,8 and there was a 2.5-fold increase in cardiovascular mortalities. 9
Chronic HFCS consumption increases hepatic production of uric acid. An elevated level of uric acid causes the development of hypertension by inducing oxidative stress and endothelial dysfunction. 10 –14 HFCS increases free oxygen radicals and causes decrease of nitric oxide (NO) production. Also NO synthase activity decreases in aorta of fructose-fed rat. 15 –18 Vascular endothelial injury induced by oxygen free radicals attenuates the production of NO and reduces NO synthase activity. 19,20 The endothelial damage occurs due to reduced NO levels, and vasodilation of blood vessels leads to a decrease in the response. Hypertension may occur when the reduction of the NO levels causes less vascular relaxation. 21,22
α-Lipoic acid (α-LA) is a strong antioxidant that has been shown to protect tissues against oxidative damage and inflammation. 23,24 There is a strong evidence of blocking oxidative damage and inflammation occurring in several diseases, especially diabetes mellitus by α-LA. 25,26 Studies about α-LA usage for endothelium and heart damages occurring in the cardiovascular diseases are limited. In this study the ameliorative effects of α-LA on oxidative damage and inflammation that occurred in endothelium of aorta and heart with the chronic consumption of HFCS.
Materials and methods
Experimental conditions
All experiments in this study were performed in accordance with the guidelines for animal research from the National Institutes of Health and were approved by the Committee on Animal Research at Suleyman Demirel University, Isparta, Turkey (03/10/2013-02).
Female Wistar albino rats weighing 250–300 g were placed in a controlled room temperature (21–22°C) maintained under 12-h light/12-h dark cycle and humidity (60 ± 5%). All the rats were fed with standard commercial chow diet (Korkuteli yem, Isparta, Turkey) ad libitum that composed of 88% dry material (mostly oat crust), 23% protein, 7% cellulose, and 8% ash, ash that does not dissolve in 2% hydrochloric acid, 1–1.8% calcium ion (Ca+2), 0.9% phosphate, 0.5–0.8% sodium, 1% sodium chloride (NaCl), 0.3% methionine, and 1% lysine.
Chemicals
HFCS (F30) was obtained from a local company (Toposmanoglu, Isparta, Turkey), which contains approximately 24% fructose and 28% dextrose in the syrup of 73% total solids. HFCS (F30) was applied into drinking water for 10 weeks during the experiment.
Thioctacid tablets (600 mg; MEDA Pharma, Turkey), a commercial form of α-LA, were used for the treatment.
To make a vascular contraction and relaxation during organ bath, phenylephrine hydrochloride (Sigma-Aldrich, Switzerland), acetylcholine chloride (Sigma-Aldrich, Switzerland), antihypertensive Ca channel blocker agent, diltiazem 25 mg ampoule (Mustafa Nevzat, Turkey) flacon, and ampoule forms were used.
Experimental design
Twenty-four rats were randomly divided into three groups (eight rats in each group).
The groups include group I: (control group), ad libitum; group II (HFCS group), 30% of F30 corn syrup in drinking water (10 weeks); and group III (α-LA treatment group), 30% of F30 corn syrup in drinking water + 100 mg/kg/day α-LA orally (HFCS, first 4 weeks) + HFCS + α-LA (last 6 weeks).
HFCS form F30 was prepared as 30% fructose syrup solution and given to the rats through drinking water for 10 weeks either in the presence or absence of α-LA. These concentrations of corn syrup were determined according to the sugar content of numerous soft drinks in which sugar ranges from 7% to 15%. 27 α-LA was given to rats by oral gavage for the last 6 weeks of the experiment and a single daily dose of 100 mg/kg orally. 28 Insulin sensitivity reduced at the end of the fourth week approximately during corn syrup consumption. 29
At the end of the experiment and 24 h after the last administration of α-LA, three rats were killed by cervical dislocation daily through 8 days because of our organ bath capacity. In this direction, the first three rats included in the study were killed first.
After the abdominal incision, blood samples were collected and aortic and cardiac tissues were quickly removed. Cardiac and aortic tissues were divided equally into two longitudinal sections. Abdominal aortas were put into Krebs–Henseleit solution and delivered to pharmacology laboratory for investigations of tissue bath analysis. One half of cardiac tissues were placed in formaldehyde solution for routine histopathological and immunohistochemical examination. Thoracic aortas and the other half of the cardiac tissues were homogenized and kept at −80°C for oxidant–antioxidant enzyme malondialdehyde (MDA) levels and catalase (CAT) activities. Blood samples were extracted to determine the serum levels of reatine kinase MB (CKMB), lactate dehydrogenase (LDH), and uric acid parameters.
Analysis
Biochemical analysis
For biochemical analysis, tissue samples from the heart and aorta were collected. Both tissues were homogenized in a motor-driven tissue homogenizer (IKA Ultra-Turrax T25 Basic; Labortechnic, Staufen, Germany) and sonicator (UW 2070 Bandelin Electronic, Germany) with phosphate buffer (pH 7.4). Unbroken cells, cell debris, and nuclei were sedimented by centrifugation at 10,000g for 10 min. The levels of protein and MDA and the activities of CAT were determined in the supernatants. The CAT activities were measured using the method described by Aebi 30 and expressed as kilo units per gram protein. Protein levels in the homogenate and supernatant were determined according to the method of Bradford. 31 The MDA levels in tissues were determined from the homogenate following the double heating method of Drapper and Hadley. 32 An autoanalyzer (Beckman Coulter AU 680, Pasadena, California, USA) was used to determine the CKMB and LDH activities and uric acid levels.
Aortic ring isolations and vascular response studies
Before killing, rats were not administered with any medical treatment or anesthetic agent. The anesthetic agent could not be used for this experiment because of the effect on vascular responses during organ bath investigations. 33 –35 After cervical dislocation, thoracotomy was applied to obtain thoracic aorta. Tissues of rats were quickly removed and put into cold Krebs–Henseleit solution. Krebs–Henseleit solution was prepared with following composition (in mM): NaCl, 119.0; potassium chloride, 4.7; monopotassium phosphate, 1.2; calcium chloride, 2.5; magnesium sulfate, 1.5; sodium bicarbonate, 25; and glucose, 11.0. The adhering perivascular fat and connective tissue were carefully removed and arterial segments were cut into about 2–3 mm rings. Caution was exercised to preserve the endothelial layer during the preparation of the aortic rings. The aortic rings were mounted in 25 ml organ baths containing the Krebs–Henseleit solution. The medium was maintained at 37°C and continuously gassed with 95% oxygen and 5% carbon dioxide. Tissues were allowed to equilibrate for 90 min under 1.0 g resting tension. Tension measurement was performed using isometric transducers (TRI201, Panlab SA, Spain), an amplifier (ML118/D Quad Bridge, ADInstruments, New Zealand), interface PowerLab/4SP (ML750, AD Instruments), and computerized system with ProtoWin version 1.0 software.
After 90-min washout period (with an interval of 15 min), the damage degree of endothelium and tunica media caused by corn syrup was determined by applying 10−6 mol/l acetylcholine (Ach) and 10−9–10−4 mol/l cumulative dose of diltiazem on 10−6 mol/l phenlyephrine (Phe)-precontracted aortic rings. After the first application of Ach and Phe or diltiazem and Phe, tissues were washed with Krebs–Hanseleit solution and again were left to 90-min washout period (with an interval of 15 min). At the end of this period, the same protocol was repeated.
After evaluation of the vascular damage (endothelial and tunica media) by applying 10−6 mol/l Ach, the response to Ca2+ channel blocker was determined by applying 10−9–10−4 mol/l cumulative dose of diltiazem on 10−6 mol/l Phe-precontracted aortic rings. After the first application of Ach and Phe, tissues were washed with Krebs–Hanseleit solution and again left to 90-min washout period (with an interval of 15 min). At the end of this period, the same protocol was repeated.
Histopathological and immunohistochemical analyses
One half of the cardiac tissue and thoracic aorta samples were collected during necropsy, fixed in 10% neutral-buffered formalin, and embedded in paraffin. Sections (5 µm) were stained with hematoxylin and eosin (H&E) for histopathological assessment, and the evaluations were performed on blinded samples. The histopathological changes were scored as (0) score (negative score): no structural damage; (1) score (one positive score): minimal damage; (2) score (two positive scores): moderate damage; and (3) score (three positive scores): severe damage. 36,37
The semiquantitative evaluation method was used to determine the immunohistochemical staining of inducible nitric oxide synthase (iNOS) and tumor necrosis factor α (TNF-α) concentrations in sections of cardiac and aortic tissue.
Selected cardiac tissue and thoracic aorta samples were stained to demonstrate TNF-α (anti-TNF-α antibody (ab66579), 1:100 dilution; Abcam, UK) and iNOS (anti-iNOS antibody (ab15323), 1:100 dilution; Abcam). The reaction was carried out using a streptavidin–biotin peroxidase technique according to the manufacturer’s instructions. The sections were incubated with the primary antibodies for a period of 60 min, and immunohistochemistry was carried out using biotinylated secondary antibody and streptavidin–alkaline phosphatase conjugate. Antigens were demonstrated using 3,3-diaminobenzidine as the chromogen. For specificity, the primary antibody was omitted in negative controls. To evaluate the percentage of immunopositive cells, 10 different areas were examined in each section and 100 cells counted under the 40× objective of Olympus CX21 light microscope and the Database Manual Cell Sens Life Science Imaging Software System (Olympus Corporation, Tokyo, Japan).
Statistical analysis
Variables were presented as frequencies, percentages, mean ± standard deviations, median, or minimum–maximum. The Kolmogorov–Smirnov test was used for determining normal distribution of continuous variables and Levene test for homogeneity of variance. Data characterized by a normal distribution were expressed as mean ± standard deviation. Parameters without such a distribution were expressed as median with range. The groups were compared using nonparametric Kruskal–Wallis test, followed by Mann–Whitney U test. Biochemical parameters were shown to fit the normal distribution and repeated measures analysis of variance. Post hoc least significant difference, Bonferroni, and Tukey’s tests were used to compare groups. The value of p < 0.05 was set as statistically significant.
Results
Biochemical analyses
Due to increased oxidative stress, MDA levels increased significantly in the HFCS group (p = 0.045), while α-LA treatment decreased these levels significantly (p = 0.019). CAT activities were decreased significantly in HFCS group (p = 0.003), while α-LA treatment increased significantly these activities (p = 0.003; Table 1). Biomarkers of cardiac muscle damage, CKMB and LDH levels, were increased in HFCS group but only CKMB levels were significant, and all of them were decreased significantly in α-LA treatment group. Similar to oxidative stress, the uric acid levels were increased in HFCS group and decreased significantly in α-LA treatment group (Table 2).
Oxidative stress markers of cardiac tissues.

HFCS: high-fructose corn syrup; α-LA: α lipoic acid; MDA: malondialdehyde; CAT: catalase.
a p < 0.05 vs control group.
b p < 0.05 vs HFCS.
Biochemical markers of blood samples.

HFCS: high-fructose corn syrup; α-LA: α lipoic acid; CKMB: creatine kinase MB; LDH: lactate dehydrogenase.
a p < 0.05 vs control group.
Cardiac histopathological and immunohistochemical analyses
There was no pathological condition observed in tissue samples of control group (Figure 1(a)). HFCS showed significant structural changes in heart of the rats. The degeneration of heart muscle cells (p = 0.000), mononuclear cell infiltration (p = 0.000) in the myocardial layer (p = 0.000), vascular congestion (p = 0.000), hemorrhagic areas (p = 0.001), and lipoidosis (p = 0.000) were increased (Figure 1(b) to (d)). In α-LA treatment group, the changes improved significantly due to the HFCS group. Cardiac muscle cells damage (p = 0.000), mononuclear cell infiltration (p = 0.001), vascular congestion (p = 0.001), and lipoidosis (p = 0.001) were decreased, and no bleeding of foci was found in this treatment group (Figure 1(e)).

Histopathological appearance of the heart. (a) Control: normal histological profile in heart; (b, c, and d) HFCS group: degeneration in heart muscle cell (black arrow), macrovesicular lipidosis (blue arrow), microvesicular lipidosis (red arrow), mononuclear cell infiltration (green arrow), vascular congestion (yellow arrow), and connective tissue (thick black tissue) were increased; and (e) HFCS and α-LA-given group: micro vesicular and macro vesicular lipoidosis were decreased and an improvement in heart muscle cells was observed. HFCS: high-fructose corn syrup; α-LA: α-lipoic acid.
In immunohistochemical staining, there was no TNF-α and iNOS staining found in control group (Figure 2(a)), and the expressions of TNF-α and iNOS were increased in HFCS group compared with the control group (p = 0.00) (Figure 2(b) to (e)). The HFCS group treated with α-LA showed a decreased expression of TNF-α and iNOS (p = 0.001; Figure 2(c) to (f)). These immunohistochemical findings were similar to histopathological findings.

Histopathological appearance of the heart. (a) Control: there was no staining, (iNOS immunostaining ×10); (b) HFCS group: particularly intense expressing around the vessel (black arrow; iNOS immunostaining ×10); (c) corn syrup and α-lipoic acid-given group: there was a little staining around vessel (iNOS immunostaining ×10); (d) control: there was no expression (TNF-α immunostaining ×10); (e) HFCS group: intense expression in the muscle cells (iNOS immunostaining ×10); and (f) corn syrup and α-LA-given group: there was a little expressing (TNF-α immunostaining, ×10). HFCS: high-fructose corn syrup; α-LA: α lipoic acid; iNOS: inducible nitric oxide synthase; TNF-α: tumor necrosis factor α.
Aortic histopathological and immunohistochemical analyses
No abnormality was observed in the aortic endothelium in all groups at gross examination (Figure 3(a)). HFCS group showed significant structural changes in aorta of the rats that were examined. The changes were the degeneration of muscle cells (p = 0.001) in the tunica media layer, mononuclear cell infiltration (p = 0.000), and an increase of fat cells (p = 0.000) in the tunica media layer as shown in Figure 3(b). In α-LA treatment group, the damage was markedly improved with a decrease in aortic muscle cells damage (p = 0.009), mononuclear cell infiltration (p = 0.004), and lipoidosis (p = 0.004; Figure 3(c)).

Histopathological appearance of the aorta. (a) Control group: normal histological profile in aorta; (b) HFCS group: mononuclear cell infiltration in the tunica media (black arrow) and an increases in lipoidosis (red arrow); (c) α-LA treatment group: mononuclear cell infiltration in the tunica media and adipose cells were decreased. HFCS: high-fructose corn syrup; α-LA: α lipoic acid; iNOS: inducible nitric oxide synthase.
Similar to histopathological findings, there was no TNF-α staining in control group (Figure 4(a)) but a weak iNOS expressing seen in the endothelial layer (Figure 4(d)). Immunohistochemical expressions of TNF-α and iNOS increased in HFCS group (p = 0.000; Figure 4(b) and (e)) and decreased with α-LA treatment (p = 0.000) (Figure 4(c) and (f)). As shown in Figure 4(e), only the intact part of endothelium was expressed with iNOS.

Histopathological appearance of the aorta. (a) Control: there was no staining, (TNF-α immunostaining, ×40); (b) HFCS group: intense expression at endothelial (green arrow) and adventitial tissue (red arrow) and small amounts of expressing in tunica media layer (thick arrow) were observed (TNF-α immunostaining, ×40); (c) corn syrup and α-LA-given group: there was a little expressing observed (TNF-α immunostaining, ×40); (d) control: a weak expressing seen in the endothelial layer (iNOS immunostaining, ×40); (e) HFCS group: there was a intense expressing in intact parts of endothelium (thin arrow) and surrounding adventitial vessels (thick arrow) was shown (iNOS immunostaining ×40); (e) corn syrup and α-LA-given group: there was a little expressing in endothelium observed (iNOS immunostaining, ×40). HFCS: high-fructose corn syrup; α-LA: α lipoic acid; iNOS: inducible nitric oxide synthase; TNF-α: tumor necrosis factor α; iNOS: inducible nitric oxide synthase.
Tissue bath analysis of thoracic aorta
To show the endothelial damage, Ach (10−6 mol/l) was used after phenylephrine-induced contractions. Phenylephrine caused repeated contractions in the isolated thoracic aorta of rats. Ach relaxation responses wer found to be 56.87 ± 3.63%, 53.70 ± 5.99%, 66.63 ± 2,36% in control, HFCS, and HFCS + α-LA group, respectively. The only responses that found in HFCS + α-LA group were statistically significant compared with the HFCS group (p = 0.008).
The maximum diltiazem response occurred with the HFCS was inhibited at a ratio of 59.55 ± 3.16 in the presence of diltiazem. The potency (half-maximal effective concentration) values to diltiazem in aortic rings from control, HFCS group and HFCS + α-LA group were 1.18 × 10−7, 1.23 × 10−8, and 1.04 × 10−9, respectively. Differences of inhibition of contraction responses by diltiazem between control, HFCS, and HFCS + α-LA were statistically significant. The dose range of 10−9 mol/l (p = 0.001), 10−8 mol/l (p = 0.008), and 10−7 mol/l (p = 0.007) were compared with the control group. The dose range of 10−9 mol/l (p = 0.010) and 10−7 mol/l (p = 0.022) were compared with the HFCS group.
These relaxations were corrected with α-LA in the dose range of 10−9 mol/l and 10−7 mol/l when compared with HFCS group. Except these findings, all the responses were increased in HFCS group and ameliorated by α-LA treatment; however, this change wasn’t significant (Figure 5).
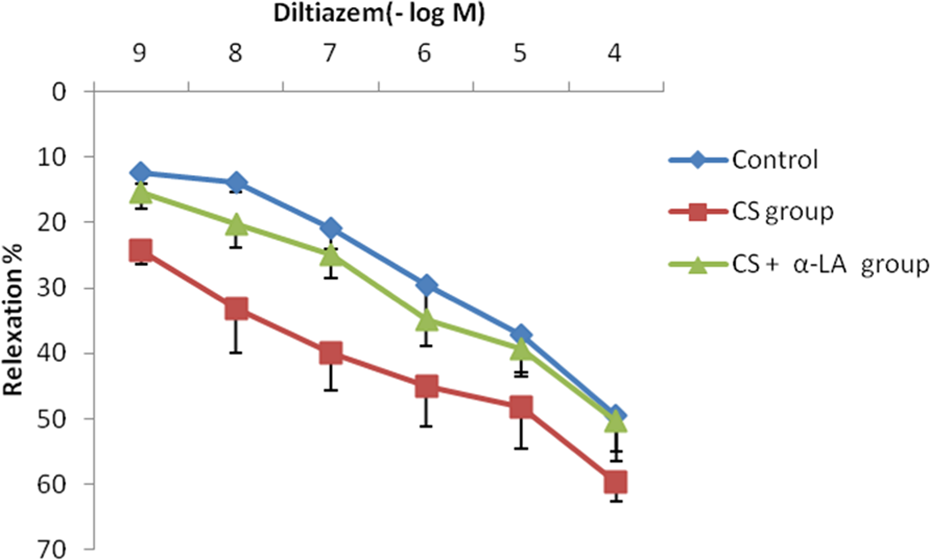
The concentration response curves for diltiazem in aortic rings. Values are expressed as means ± SEM (n = 6–8).
Discussion
HFCS consumption causes several metabolic problems like lipoidosis, insulin resistance, and hyperuricemia. 38 These abnormalities enhance cardiac and endothelial problems. 39 Akar et al. 40 studied the protective effect of resveratrol on HFCS-induced vascular dysfunction associated with metabolic disturbance in rats. They found that HFCS intake could be a potential risk factor in the development of cardiovascular and metabolic disorders by causing vasculopathy, and resveratrol had protective effects against the damage that occurred by HFCS consumption. Researchers found that uric acid-induced endothelial dysfunction was associated with mitochondrial alterations and decreased intracellular adenosine triphosphate. Therefore, uric acid-induced oxidative stress caused vascular damage. 41,42 Geetha et al. 43 studied cardiac damage induced by HF diet-fed rats and evaluated the protective effect of troxerutin. They found beneficial effects of troxerutin on HFCS-induced cardiac damage. In this study, firstly, uric acid levels that caused oxidative damage in the heart and endothelium were increased by HFCS, and these alterations were decreased by α-LA administration. The marker of oxidative stress MDA levels and antioxidant CAT activities supported this damage. Increased MDA levels and decreased CAT levels confirmed the oxidative damage in the heart. α-LA treatment protected the heart by decreasing the MDA levels and increasing the CAT activities. Sabharwal and May showed that α-LA had a protective effect against oxidative stress in cell culture of EA.hy926 endothelial cells that they supported the results of this study. 44 In another study, researchers evaluated the effects of α-LA on NOS in lung tissues. They shared that superoxide dismutase also increased with α-LA, inducible NOS and endothelial NOS increased in diabetic animals. As a result, α-LA can prevent some diabetic effects on the lungs and can also protect from vascular damages. 45 Similar to this condition, CKMB and LDH indicators of cardiac muscle damage were increased in HFCS group because of oxidative stress and lipoidosis. These markers were reduced by antioxidant α-LA strongly. Similar to these biochemical markers, histopathological findings induced by HFCS supported these endothelial and cardiac damages. For example, lipoidosis, the mononuclear cell infiltration, degeneration of heart muscle cells, vascular congestion, and hemorrhagic areas occurred in heart, and the lipoidosis, degeneration, and mononuclear cell infiltration of muscle cells in the tunica media layer of endothelium showed this oxidative damage. Antioxidant α-LA may effectively attenuate mitochondria-dependent cardiac apoptosis and suppress mitochondrial oxidative damage in diabetic cardiomyopathy. 46 Sakr applied high cholesterol and fructose diet and showed that multifocal vacuolar degeneration vascular congestion in cardiac blood vessel. 47 In another study, researchers showed abnormal myocardial architecture and enlarged interstitial space in the fructose-induced metabolic syndrome. 48 Kho et al. examined histological changes by staining with H&E in thoracic aorta in HF fad rats. They showed that endothelial layers of thoracic aorta were roughened, layers of the tunica intima media were increased, and the adipocytes were hypertrophy induced by HF diet compared with the control group. 49
The damages in heart and endothelium were markedly improved and reduced by α-LA treatment in this study. Cardiac muscle cells damage and lipoidosis were decreased, and no bleeding of foci was found in this treatment group. TNF-α and iNOS as inflammatory markers increased the inflammatory and oxidative processes. In immunohistochemical findings, both inflammatory markers, TNF-α and iNOS, were expressed with high intensity according to the damage occurred by HFCS. Also α-LA treatment improved these damages. Similar to these findings, there were lower relaxation observed at a dose of Ach in HFCS group because of the endothelial damage occurred in HFCS group, especially by inflammation and oxidative damage. The histopathological and immunohistochemical findings also supported this damage. The results of this study revealed unexpected responses to Ca2+ channel blockers in HFCS group were observed, α-LA treatment ameliorates this relaxation. There was more relaxation occurred by diltiazem in HFCS group at doses of 10−9, 10−8, and 10−7 mol/l, and these were statistically significant when compared with the control group and α-LA treatment reduced this damage. Oxidative damage and inflammation caused endothelial dysfunction and inhibition of NO synthesis leading to activated and enhanced expression of L-type Ca2+ channels. 50 When Ca2+ channel blockers were used at this condition, the relaxation responses were higher than in the other groups.
Conclusion
In conclusion, chronic consumption of HFCS caused several problems like cardiac and endothelial injury of aorta by hyperuricemia and induced oxidative stress and inflammation. Thromboembolic events and several organ damage could be triggered in this case as endothelial damage. α-LA treatment reduced uric acid levels, oxidative stress, and improved vascular responses. Therefore, α-LA can be used for the treatment of cardiovascular problems that induced by hyperuricemia like metabolic syndrome. It can be added to cardiac drugs due to its cardiovascular protective effects against oxidative stress and inflammation.
Footnotes
Acknowledgment
The authors would sincerely like to thank Dean of Faculty of Medicine, Professor Huseyin Yorgancigil for his technical support.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
The protocols for the research project and survey have been approved by a suitably constituted ethics committee of our institution and conform to the provisions of the Declaration of Helsinki (as revised in Edinburgh 2000). All experiments in this study were performed in accordance with the guidelines for animal research from the National Institutes of Health and were approved by the Committee on Animal Research at Suleyman Demirel University, Isparta, Turkey (03/10/2013-02).
