Abstract
The susceptibility of the kidneys to fluoride toxicity can largely be attributed to its anatomy and function. As the filtrate moves along the complex tubular structure of each nephron, it is concentrated in the proximal and distal tubules and collecting duct. It has been frequently observed that the children suffering from renal impairments also have some symptoms of dental and skeletal fluorosis. The findings suggest that fluoride somehow interferes with renal anatomy and physiology, which may lead to renal pathogenesis. The aim of this study was to evaluate the fluoride-associated nephrotoxicity. A total of 156 patients with childhood nephrotic syndrome were screened and it was observed that 32 of them had significantly high levels (p ≤ 0.05) of fluoride in urine (4.01 ± 1.83 ppm) and serum (0.1 ± 0.013 ppm). On the basis of urinary fluoride concentration, patients were divided into two groups, namely group 1 (G-1) (n = 32) containing normal urine fluoride (0.61 ± 0.17 ppm) and group 2 (G-2) (n = 32) having high urine fluoride concentration (4.01 ± 1.83 ppm). Age-matched healthy subjects (n = 33) having normal levels of urinary fluoride (0.56 ± 0.15 ppm) were included in the study as control (group 0 (G-0)). Kidney biopsies were taken from G-1 and G-2 only, who were subjected to ultrastructural (transmission electron microscopy) and apoptotic (terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end labeling) analysis. Various subcellular ultrastructural changes including nuclear disintegration, chromosome condensation, cytoplasmic ground substance lysis, and endoplasmic reticulum blebbing were observed. Increased levels of apoptosis were observed in high fluoride group (G-2) compared to normal fluoride group (G-1). Various degrees of fluoride-associated damages to the architecture of tubular epithelia, such as cell swelling and lysis, cytoplasmic vacuolation, nuclear condensation, apoptosis, and necrosis, were observed.
Keywords
Introduction
In India 21 out of 35 states are known to be endemic for fluorosis due to high fluoride contamination in underground drinking water. 1,2 Apart from the dental and skeletal fluorosis, exposure to toxic levels of fluoride is known to affect vital organs including the kidneys. 3 The kidneys are the main target organ of mammalian fluoride intake and renal toxicity occurs after acute and chronic fluoride intoxication. 4 –6 Numerous structural and functional changes have been noted in the kidneys of experimental animals receiving increased amounts of fluoride under different conditions. 7 In humans, few reports pertaining to kidney involvement in endemic fluorosis areas are available. Impaired renal functions in fluoride-exposed industrial workers have been reported. 8 Fluoride-induced renal tubular injury in different animal models has been observed. 9 In addition, changes including tubular degeneration, inflammation, fibrosis, parenchymatous nephritis, cloudy swellings, and dilatation of tubules have also been reported in experimental animals. 10 Chronic fluoride toxicity at a level of 14 mg F/kg bw/day and higher has been claimed to result in renal lesions in experimental models. 11 Fluoride-induced pathological changes were reported in the proximal, distal, and collecting tubules in experimental models. 12 Fatty degeneration and focal calcification in the tubules of the bovine kidneys are known. 13 In vitro exposure to fluoride at high concentrations induces profound cellular reactions, such as chromosome aberrations, inhibition of cell cycle progression, 14 stimulation of cytokine and arachidonic acid release, 15 and induction of apoptosis. 16,17 Fluoride may inhibit protein synthesis, activate superoxide production, and induce other changes in cellular metabolism. 18 The initial mechanisms involved in the fluoride-induced pathological changes are not fully understood. However, fluoride is known to activate different cellular signal transducing systems and has been accordingly used as an experimental tool in mechanistic studies and metabolism.
Apoptosis is a pathway of cell death, and excessive apoptosis leads to various kinds of organ injury. 19,20 Fluoride causes various renal histological changes such as necrosis of glomeruli and tubules, atrophic glomeruli and glomerular capsule, and tubular dilatation; moreover, severe tubular leakage appeared in animals, given high concentrations of fluoride in drinking water and food. 21 Tubular damage can result from ischemic or nephrotoxic injury to the kidney 22 Poor oxygenation leads to induction of a variety of secondary factors that promote the development of tubular injury, including the intracellular accumulation of calcium, generation of reactive oxygen species, depletion of adenosine triphosphate, and apoptosis. 23
Various types of fluoride-induced renal injuries are reported in different animal studies but the same are not confirmed in human. 24 To evaluate the fluoride related renal injury and ultrastructural changes in human, kidney biopsy is essential. However kidney biopsy cannot be advised in fluorosis patients just for studying fluoride-induced nephrotoxicity in fluorosis patients. Therefore, an indirect approach was adopted, and kidney biopsy tissues of nephrotic syndrome minimal change disease (NS-MCD) patients, who are routinely advised kidney biopsy for clinical diagnosis, coming from fluorosis endemic areas and having high levels of fluoride in their serum and urine were considered for the study. The present study was therefore designed to investigate ultrastructural changes in kidney tubules due to fluoride toxicity in human subjects to understand the pathogenesis involved.
Methodology
Subject selection
With an aim to evaluate the fluoride concentration in urine and serum and its impact on renal tubule, confirmed cases of childhood NS-MCD were selected from the pediatrics out patient department, All India Institute of Medical Sciences, New Delhi, India. The study was done from June 2012 to January 2015. Childhood nephrotic syndrome patients (NSP; n = 156) were screened for fluoride concentration in urine and serum. On the basis of fluoride concentration in urine and serum, NSP were divided into two groups: NSP with high urinary fluoride and NSP with normal urinary fluoride. All the recruited subjects were between 4 years and 12 years of age and grouped as follows: group 0 (G-0): healthy individuals (n = 32) with normal level of urinary fluoride (up to 1 ppm) were recruited as control group; group 1 (G-1): NSP with normal level of urinary fluoride (up to 1 ppm); and group 2 (G-2): NSP with increased level of urinary fluoride (above 1 ppm). After getting ethical permission from the Institutional Human Ethics Committee (vide certificate number IESC/T-56/03.02.2012, dated February 2, 2012), sample collections were started. Before the sample collection, the details of the research plan were explained to parents/guardians of the subjects. Spot urine, serum, renal biopsy (only for G-1 and G-2), and clinical history were taken from the recruited subjects. Urine samples (10 ml) were collected on the spot in sterile plastic vial and 5 ml blood samples were collected aseptically as per standard protocol.
Renal biopsy
The subjects with proteinuria (>1 g/l or PCR > 100 mg/mmol), with two normal sized, non-obstructed kidneys and no obvious cause, were suggested for renal biopsy. Microscopic and ultrastructural examination of the kidney tissues was required to confirm the diagnosis of MCD. Required blood parameters were investigated before the biopsy to rule out infection or any blood clotting (BT/CT) prior to the renal hydronephrosis, preexisting arteriovenous fistula in the kidney, and cystic kidney disease; small and shrunken kidneys were also ruled out. Informed consent was taken from the parents/guardians prior to the procedure. Ultrasound-guided renal biopsies were performed by the nephrologist and/or the interventional radiologist, and kidney tissues (core of kidney tissue, less than 1 mm in diameter and up to 1 cm in length) were taken by biopsy gun.
Fluoride estimation
Urinary and serum levels of fluoride were measured by potentiometric method using fluoride selective ion electrode (Orion, Thermo Scientific, Massachusetts, USA). Serum, urine, or water samples were diluted and measured in total ionic strength adjustment buffer to maintain constant ionic strength and to remove certain interferences. The pH of the urine samples was also adjusted using KCl or NaOH according to the pH of urine to avoid ionic interference.
Transmission electron microscopy
A part of the biopsy tissue was subjected to ultrastructural studies with the help of transition electron microscopy (TEM) to evaluate fluoride-associated ultrastructural changes in the kidney tissues. Kidney tissues having both medullary and cortical parts were taken for ultrastructural analysis. For TEM, a part of kidney tissue was fixed in paraformaldehyde and processed as follows:
(1) Fixation: Fixed in 2.5% glutaraldehyde + 2% paraformaldehyde, made in 0.1 M phosphate buffer (pH 7.4), for 12–24 h at 4°C, then washed thoroughly 4–5 times in 0.1 M PB. (2) Post-fixation: The tissues were fixed again in 1% osmium tetraoxide for 2 h at 4°C, then washed thoroughly 4–5 times in 0.1 M PB. (3) Dehydration: The tissues were dehydrated through various acetone dilutions at 4°C. 30% (30 min) > 50% (30 min) > 70% (15 min × 2) > 90°C (30 min) > 95°C (30 min) > dry acetone (15 min × 2) > dry acetone (30 min at room temperature (RT) × 2). (4) Clearing: The tissues were immersed in toluene for 30 min (× 2). (5) Infiltration: LR White (acrylic resin) and toluene were used for infiltration and embedding in different concentrations 1:3 (2 h at RT) > 2:2 (2–3 h at RT) > 3:1 (2 h at RT under vacuum) > pure resin (2 h at RT) > pure resin (2 h at 50°C). (6) Embedding and (7) Polymerization: The tissues were embedded in pure embedding medium (resin) using mold or BEEM capsules (Ted Pella, California, USA) and then polymerized at 50°C for 12–24 h in a dry heat bath, after this, temperature was raised to 60°C for continuous polymerization for 48–72 h. (8) Light Microscopy: The prepared blocks were cut in 1 μm thickness serially using glass knives to locate the desired part of the tissue under light microscope. The sections for light microscopy were stained with 1% toluidine blue (prepared in 1% borax). (9) Ultrathin sectioning: Ultrathin sections of 60–90 nm were cut from the located desired part of the tissue and mounted on 400 mesh-nickel grids. (10) Staining: Tissues were stained with uranyl acetate (10–15 min), washed with 50% ethanol and double distilled water, and the meshes were placed on drops of lead citrate (5–10 min), and were washed with 0.02 M sodium hydroxide and double distilled water. After staining, the meshes were dried and stored in grid boxes. (11) Viewing: The stained and dried copper grids with the ultrathin sections were viewed under Tecnai G2 200 kV, TEM (FEI, Oregon, USA) in the Electron Microscope Facility, department of Anatomy, AIIMS, New Delhi, India.
Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
Terminal deoxynucleotidyl transferase deoxyuridine triphosphate (dUTP) nick end labeling (TUNEL) assay was performed using in situ cell death detection kit, Fluorescein (Roche, Bavaria, Germany), according to the manufacturer’s instructions. The sections were counter stained with DAPI (4’,6-diamidino-2-phenylindole) (Molecular Probes, Oregon, USA) (in 0.1 M PB). and mounted with VECTASHIELD mounting medium (Vector Labs, Burlingame, CA, USA). The apoptotic cells in the renal tubule cells were identified on the basis of the presence of Fluorescein at 595 nm and DAPI at 460 nm in their nuclei, under Nikon epifluorescence microscope (Nikon Instruments, Japan). High-resolution high-power (20× objective) digital photomicrographs (10–15 non-overlapping photomicrographs/section) of kidney tissue sections were quantitatively analyzed for the apoptotic nuclei, with the help of image analysis software Image Pro-Plus 6.0 (Media Cybernetics, Rockville, USA).
Statistical analysis
The data obtained were presented as the mean and SD (Mean ± SD). Statistical comparison across the groups was done by using a one-way analysis of variance (ANOVA) or Student’s t test using Excel analysis ToolPak add-on for MS Office 2010. The significance between pairs of variable means was analyzed using Fisher’s least significant difference test at 5% level of significance. A difference at p < 0.05 was considered statistically significant.
Results
Fluoride concentrations were measured in urine and serum samples of all the recruited 156 childhood NSP. It was observed that (32 out of 156) 20.38% of NSP had significantly (p = 0.01) high urinary (4.01 ± 1.83 ppm) and serum (0.1 ± 0.013 ppm) fluoride concentration compared with G-0 (urine, 0.56 ± 0.15 ppm; serum, 0.07 ± 11 ppm). However, rest of the NSP had normal urinary (0.61 ± 0.17 ppm) and serum (0.07 ± 0.012 ppm) fluoride concentrations. These findings showed significant (p ≤ 0.05) variation (Figure 1) in urinary and serum fluoride concentration within the NSP. On the basis of body fluid fluoride concentration, NSP can be clearly classified into two groups: one with high fluoride concentration (G-2) and the other with low/normal fluoride concentration (G-1).

Graphical representation of (a) urinary fluoride concentration (y-axis) in different groups (from left to right G-0 to G-2). The concentration of urinary fluoride was significantly high in G-2 as compared to G-0 and G-1 (p = 0.001). (b) The concentration of serum fluoride was significantly high in G-2 as compared to G-0 and G-1 (p = 0.001). G-0: healthy subjects having normal levels of urinary fluoride; G-1: normal urine fluoride containing subjects; G-2: high fluoride containing subjects.
Ultrastructural changes
TEM images showed accumulation of multiple dark spherical microparticles within the tubular basement membranes (Figure 2(b)) and basement membrane disintegration (Figure 2(c)) in G-2, in which subjects had significantly increased urinary and serum fluoride concentrations. Glycogen lysis, rarefactions of cytoplasmic ground substances, hypervacuolation, and chromosome condensation were observed frequently (Figure 2(d) and (e)) in the renal tubule of G-2 while the same was less frequent in G-1. The increased levels of nuclear swelling, chromatin disintegration, and other signs of apoptosis were observed in G-2 as compared to G-1. The pyknotic changes in the cells of the renal tubules of G-2 observed but it was only occasional. Proliferation and moderate grade of apical blebbing of the endoplasmic reticulum of epithelial cells of renal tubule was observed (Figure 2(f)). An increased number of lysosomes was observed in the cells of proximal convoluted tubule (Figure 2 (e)), including occasional fusion of lysosomes forming irregular shaped dense bodies within the cytoplasm (Figure 2(d), (e) and (f)).

Electron micrographs of 70-nm ultrathin renal tubular tissue sections of NS-MCD. (a) Patients with normal levels of urinary and serum fluoride showed normal appearance of TBM in G-1. (b) TBM, along with intra-TBM electron dense deposits (ED-rectangle) in G-2. (c) Abnormal appearance due to TBM disintegration (circle) and gross proteolysis (rectangle) in G-2. (d) Nuclear membrane disintegration with occasional cytoplasmic ground substance lysis in G-2. (e) Renal tubular cells showing hypervacuolation (V), chromatin condensation and cytoplasmic glycogenolysis (eclipse), and rarefaction of cytoplasmic ground substances in G-2. (f) Showing ER blebbing (*), along with hyper-vacuolation. All these features indicate subcellular damages in renal tubular cell, which may lead to apoptosis due to fluoride toxicity in G-2. Scale bar: 0.5 μm. NS-MCD: nephrotic syndrome minimal change disease; TBM: tubular basement membrane; ER: endoplasmic reticulum; G-2: high fluoride containing subjects.
Apoptotic status
Apoptosis is a pathway of cell death, and altered levels of apoptosis leads to various kinds of tissue/organ injuries. In the present study, it was observed a significant level of apoptosis (22%) in renal tubular region (p = 0.001) in the patients of G-2, as compared to NSP (7%) of G-1 (Figure 3); a few animal model-based studies have reported that fluoride induces kidney lesions through apoptosis and our findings also reveal the same in human subjects.
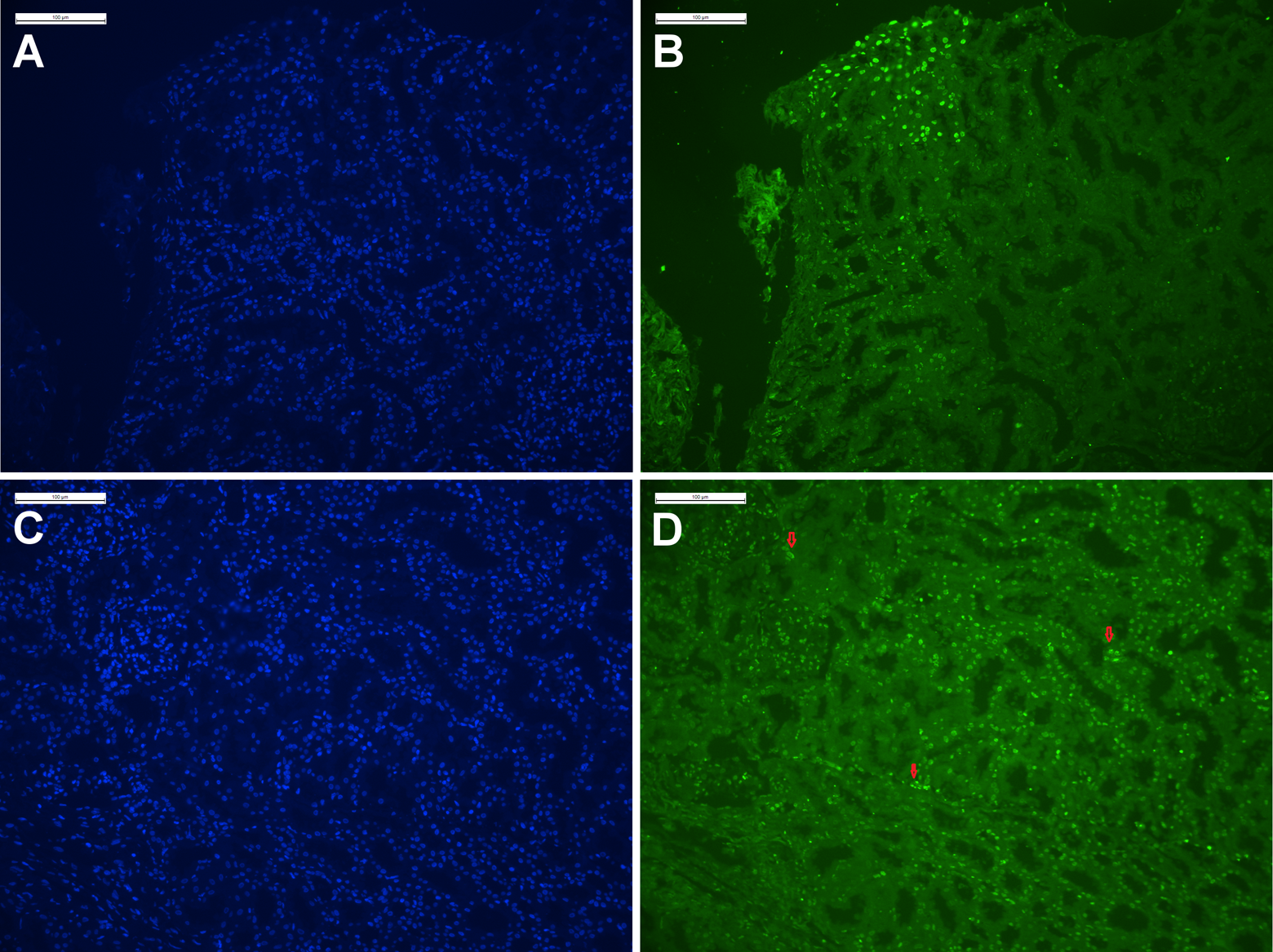
(a) and (c) Representative images of DAPI nuclear staining of paraffin embedded renal biopsy tissue from G-1 (a) and G-2 (c). Blue intense signals showing cells nuclei. (b) and (d) TUNEL-representative images of Fluorescein-labeled apoptotic nuclei. Hyper intense green fluorescent spots in the renal biopsy tissues showing only apoptotic cell nucleus in G-1 (b) and G-2 (d). In G-1 on an average 7% nuclei were TUNEL positive (apoptotic) while, about 93% were negative (normal non-apoptotic), while on an average 22% of nuclei were TUNEL positive (apoptotic) in the G-2. TUNEL positivity was calculated using image analysis software Image Pro-Plus 6.0 (Media Cybernetics, USA). G-1: normal urine fluoride containing group; G-2: high fluoride containing group; TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling.
Discussion
In the present study, NSP with MCD were included and the reason behind choosing MCD was to assess the fluoride-induced ultrastructural changes in renal tubule. In MCD, only glomerular part of the kidney gets impaired but the tubular part remains unaffected. Another advantage of the nephrotic syndrome MCD was the presence of variations in the body fluid fluoride concentration. In addition, almost all NSP with MCD are advised renal biopsy which, being an invasive procedure, cannot be routinely performed on fluorosis patients. The ultrastructural and apoptotic findings indicated various types of injuries to the tubular epithelia which include cellular edema, lysis, cytoplasmic hypervacuolation, nuclear condensation, apoptosis, and necrosis.
Kidney impairment has been demonstrated in fluoride-exposed animals mainly due to enhanced lipid peroxidation and decreased activities of antioxidant enzymes. 6 Lipid peroxidation may be one of the reasons for fluoride-induced tubular damage. As it is evident from the report of previous animal studies, fluoride toxicity causes pathological changes in the renal tubules. 12 In the present study, fluoride-associated renal tubular injury is observed, which includes various ultrastructural changes and increased level of apoptosis in G-2 (Figures 3, 4 & 5). In animal models, its effects on glomerular function are less severe, whereas proximal tubular injury is more evident. 25 Likewise the findings of the present study on human subjects are in agreement with the previous animal model-based experimental studies. Polyuria is one of the symptoms in fluorosis patients which indicates early renal impairment and the other changes in the renal tissue in G-2 seen in this study, which further strengthens the previous findings. The electron microscope findings include accumulation of multiple dark spherical microparticles within the tubular basement membranes. These multiple aggregates of microparticles may be cellular debris or electron dense deposits within the renal tubular basement membrane most likely derived from damaged and degenerated mitochondria in some proximal tubules. Glycogen lysis and rarefactions of cytoplasmic ground substances were frequently observed in the renal tubule of high fluoride containing group (Figure 2(b) to (f)). Many types of nuclear changes in epithelial cells of tubules were also observed, which include nuclear swelling, chromatin disintegration, and other signs of apoptosis (Figure 2(e)).

Graphical representation of TUNEL-positive (apoptotic) and TUNEL-negative (non-apoptotic) nuclei: (a) about 7% of renal tubular cells were positive for apoptosis in G-1, having normal levels of urinary and serum fluoride; (b) about 22% of renal tubular cells were apoptosis positive in G-2, having high concentrations of urinary and serum fluoride. The differences were statistically significant among G-1 and G-2. TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling; G-1: normal urine fluoride containing subjects; G-2: high fluoride containing subjects.
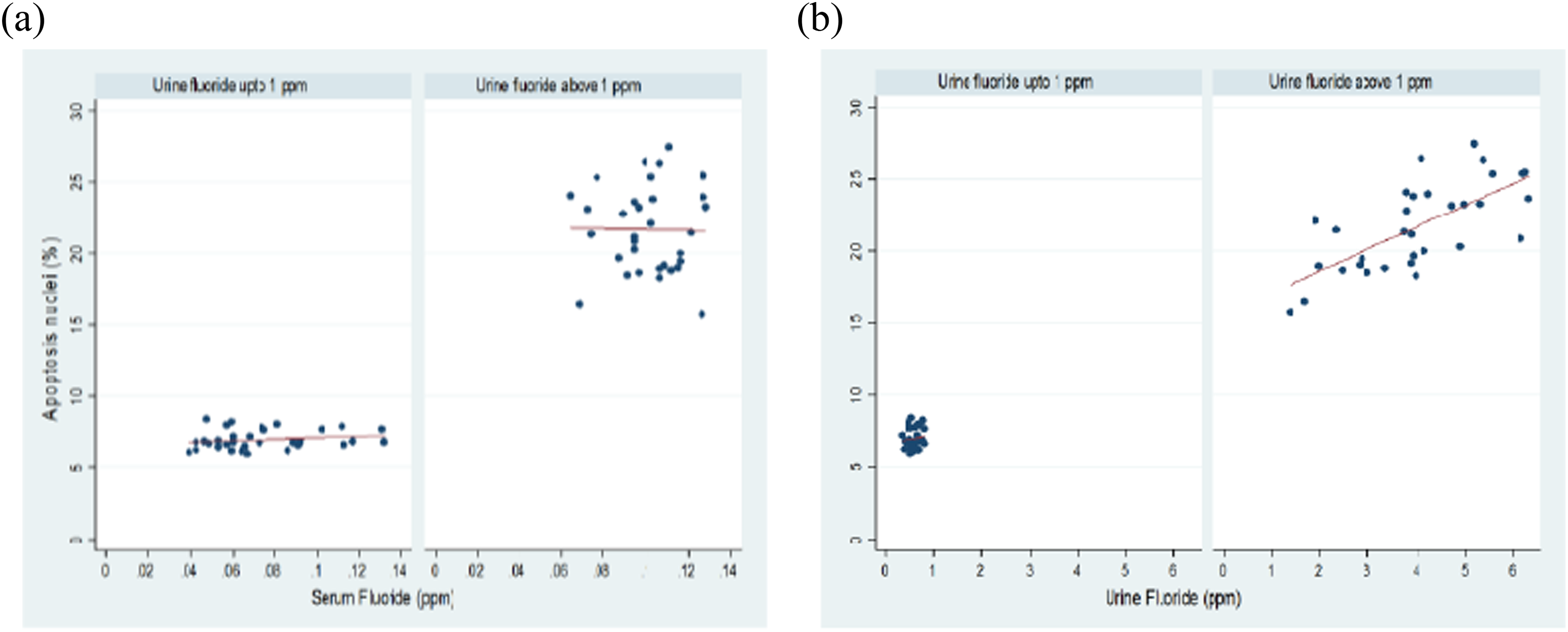
Graphical representation of correlation between apoptosis with respect to (a) serum fluoride and (b) urine fluoride among G-1 and G-2 (p = 0.001). G-1: normal urine fluoride containing subjects; G-2: high fluoride containing subjects.
Furthermore preexisting renal impairment may increase the toxic manifestations of chronic fluoride toxicity due to diminished urinary fluoride clearance rate (due to reduced glomerular filtration rate [GFR]). 26 It is possible that fluoride causes damage to renal tissues even after low levels of fluoride ingestion in the patients with compromised renal function. It may happen due to compromised renal filtration (reduced GFR) rate and hence retention of excess fluoride in the body. It was observed during patient workup for fluorosis diagnosis that a good number of patients with chronic fluoride toxicity also have some symptoms of renal impairment, irrespective of age and sex. These findings in human subjects are in agreement with previous fluoride-related nephrotoxicity reports concerning animal models like rabbits, rats, and cows. The findings of the present study provide preliminary evidence about adverse effects of fluoride toxicity on renal ultrastructure.
Conclusion
The ultrastructural and apoptotic findings indicate various degrees of fluoride-induced damage to the architecture of tubular epithelia, such as cell swelling and lysis, cytoplasmic vacuolation, nuclear condensation, apoptosis, and necrosis. The cumulative effects of fluoride-induced cellular and subcellular changes most likely activate apoptosis, which leads to cell death and renal injury.
Further studies are required in large population to establish and confirm the fluoride-associated renal impairment in adults. Urinary proteomics should also be undertaken in fluorosis patients, which will be helpful in understanding the mechanism of fluoride-induced renal impairment as well as in finding a reliable urinary marker of fluoride-induced tubular injury.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
