Abstract
Glomerulonephritis (GN) is inflammation of glomeruli. The four major categories that cause human GN are mediated by immunoglobulin or complement or both, and they include (1) immune complex–mediated GN, (2) anti-glomerular basement membrane–mediated GN, (3) antineutrophil cytoplasmic autoantibody–mediated GN, and (4) complement factor 3 glomerulopathy mediated by complement dysregulation. Initiating processes include infection, autoimmunity, exogenous antigens, and neoplasia. Often there are predisposing and modulating genetic, epigenetic, and/or environmental factors. Animal models facilitated the recognition and elucidation of the pathogeneses of all four categories of GN, and they continue to be used in preclinical studies to identify and validate therapies for all four types of GN. Advanced diagnostic modalities (e.g., transmission electron microscopy and immunofluorescence) are helpful and sometimes required for the correct categorization of GN in humans and animals. This review provides historical background on the discovery of the different GN pathogeneses, describes some of the animal models used to discover and understand each GN pathogenic category, reviews the diagnostic classification of each category of GN, and compares human GN to spontaneous forms of nonhuman GN.
Introduction
In toxicologic pathology, the International Harmonization of Nomenclature and Diagnostic Criteria project provides diagnostic categories of proliferative and nonproliferative lesions of the urinary tract in rodents, of which glomerular lesions have 3 major patterns: glomerulonephritis (GN), glomerulopathy, and glomerulosclerosis (Frazier et al. 2012). The rationale for this classification system was to establish a simplified scheme based solely on histomorphology to be used by toxicologic pathologists. This would allow harmonization of diagnostic terms and facilitate coding using a computerized system (e.g., Standard for Exchange of Non-clinical Data). These accepted terms could also be easily understood by regulatory reviewers. Each pattern has a varying number of subcategories further defined by their histologic features, (e.g., mesangioproliferative, membranoproliferative, and hyaline). This is in comparison to human nephropathology, in which categories of glomerular disease are distinguished based on a wide variety of criteria including histology with a panel of histochemical stains, transmission electron microscopy (TEM), immunofluorescence (IF; Bonsib 2007), clinicopathologic data, and even response to treatment (L. Beck et al. 2013). Veterinary nephropathology has made similar advancements in the identification and characterization of glomerular injury mostly in small companion animals (R. E. Cianciolo et al. 2013).
For decades, both spontaneous and experimental animal models have served as tools to elucidate the pathogenesis of GN in humans. Understanding the history, discoveries, and limitations of these models is important for the appreciation of their continued relevance in glomerular disease research. This is true in both basic science research and toxicologic pathology, where these models are used to test the efficacy of novel therapies. Furthermore, with the development of new biotherapeutics, the glomerulus could be a site of off-target injury. Therefore, correct classification of the mechanism of injury can be crucial for assessing the safety profile of these treatments.
Put simply, GN is inflammation of the glomeruli. In humans, there are 4 major categories of GN: (1) immune complex–mediated GN, (2) anti-glomerular basement membrane (GBM)-mediated GN, (3) antineutrophil cytoplasmic autoantibody (ANCA)-associated GN, and (4) complement factor 3 (C3)-mediated glomerulopathy. Animal models were used in the discovery and characterization of these categories, and this review will provide the historical background for each and will describe the histologic, ultrastructural, and IF features of each disease phenotype.
One of the earliest steps in the identification of glomerular disease occurred in 1914 by Drs. Volhard and Fahr (1914) who discerned that injured glomeruli could have an increased number of nucleated cells within capillary lumens (endocapillary) or outside the glomerular tuft (extracapillary). At the time, it was theorized that native glomerular cells were undergoing mitosis, so the term “proliferative” was used. Although we now realize that much of the hypercellularity is due to recruitment of inflammatory cells, nephropathologists continue to use the term “proliferative.”
Animal models of GN have been used since the 1950s. For example, administration of rabbit antibodies against homogenized rat kidney tissue to other rats resulted in the development of GN (Germuth 1953; Mellors 1955; Dixon, Feldman, and Vazquez 1961). Comparisons to human GN led to the discovery that immunoglobulins (Igs) often played a crucial role in GN pathogenesis. Furthermore, the diagnostic tests that were used in the animal models of glomerular disease, IF and TEM, were also found to be useful in the diagnosis of spontaneous glomerular disease in humans. Specifically, applying an immunofluorescent antibody against human IgG onto fresh frozen human renal tissue might demonstrate a granular (or “lumpy bumpy”) pattern of staining, providing evidence for the presence of deposits of IgG along the glomerular capillary walls (Mellors 1955). Immunofluorescent antibodies for IgA and IgM were also developed for routine diagnostic use. Moreover, an immunofluorescent antibody against human C3 can also label the glomeruli in a similar fashion, indicating that the Igs were activating the complement system in that location. TEM in these cases revealed electron-dense deposits, which were determined to be immune complexes of antigen and antibodies within the glomerular tuft. The location of the electron-dense deposits could vary from subepithelial (between the podocyte and GBM), subendothelial (between the GBM and the endothelial cell), intramembranous (within the lamina densa of the GBM), mesangial, or a combination of the above. Subtypes of immune complex–mediated GN could be identified (e.g., IgA nephropathy, lupus GN, infection-associated GN), but the differences in their pathogeneses would not be elucidated for many decades.
In the meantime, another very aggressive type of glomerular injury was discovered. Notably, animal models were also key to the elucidation of this disease pathogenesis. Sheep were repeatedly injected with compounds composed of heterologous GBM material (derived from rabbits, rats, and humans) and Freund’s adjuvant, and they developed severe GN with damage to the capillary wall and/or Bowman’s capsule basement membrane (Steblay 1962). This type of damage results in the formation of a crescent, which is the accumulation of fibrin and cells, often neutrophils and macrophages, within Bowman’s space. The pattern of glomerular injury could also be induced by passively transferring serum antibodies from one treated sheep to a naive sheep (Steblay 1962). The histologic pattern resembled a type of aggressive crescentic GN in humans, and eventually, anti-GBM antibodies were also identified in humans. Interestingly, the IF pattern of anti-GBM disease is a strong linear staining of the GBM, as opposed to the granular pattern in immune complex–mediated GN (Sturgill and Westervelt 1965; Lerner, Glassock, and Dixon 1967). Ultrastructurally, electron-dense deposits are not present because each anti-GBM Ig binds directly to the GBM, without forming a discrete complex (Jennette and Nickeleit 2007). Of note, there are no spontaneous cases of anti-GBM-mediated GN in companion animals (R. Cianciolo and Mohr 2015).
The next large category of GN to be identified was ANCA-mediated GN, a disease caused by autoantibodies against neutrophil cytoplasmic components. Specifically, these autoantibodies target primary granule proteins including proteinase-3 and myeloperoxidase. When these autoantibodies bind to neutrophils, there is damage of small vessels. The glomerular and pulmonary capillaries are preferentially affected. Rupture of glomerular capillaries leads to a pattern of crescentic GN, which can be histologically similar to anti-GBM-mediated GN. In contrast to anti-GBM disease, the IF evaluation is essentially negative or “pauci-immune” because the autoantibodies are neither contained within immune complexes nor directly bound to the GBM (Stilmant et al. 1979; Falk and Jennette 1988). It also follows that TEM evaluation will not demonstrate electron-dense deposits in ANCA-mediated GN. Although dogs have been shown to spontaneously synthesize ANCAs (Mancho et al. 2010), a definitive case of spontaneous ANCA-mediated GN has not been reported in companion animals. Rarely, cynomolgus monkeys have had mildly positive ANCA serologic titers using the human assay (K. Frazier, Personal Communication, August 13, 2018). This species difference is not yet understood.
Relatively recently, the category of C3 glomerulopathy was described (Bomback and Appel 2012), again with animals playing a major role in the identification of this disease. Prior to the creation of this diagnostic category, C3 glomerulopathy was often lumped together with Ig-mediated GN, based on the positive nonlinear IF pattern and the presence of electron-dense deposits with TEM. Investigation of the complement system in related Norwegian pigs with a membranoproliferative pattern of glomerular injury identified mutations in complement regulator factor H. These mutations can lead to uncontrolled activation of the complement system and GN. The IF evaluation in these pigs demonstrated strong C3 labeling and little to no Ig labeling. Ultrastructurally, there were sausage-shaped electron-dense deposits along the lamina densa of the GBM (Hogasen and Jansen 1998; Jansen and Nordstoga 1992), similar to the deposits seen in dense deposit disease in humans, a disease which was previously considered a subtype of immune complex–mediated GN. Subsequently, genetic analysis of humans with dense deposit disease also identified mutations in inhibitors of the complement pathway (Bomback and Appel 2012). These types of mutations can lead to other types of complement-mediated glomerular injury such as direct endothelial damage, which is the underlying pathogenesis of thrombotic microangiopathy (Keir and Coward 2011). While C3-mediated glomerulopathy has many overlapping histologic features of immune complex–mediated GN, the correct diagnosis of this subtype of glomerular injury, via IF and TEM evaluation, is important for prognosis and treatment. Complement hyperactivity needs to be mitigated and monitored for possible recurrence, which may occur if there is activation of the complement system for any reason. Therapeutic antibodies against complement proteins are currently available, and others are in clinical trials (Rousset-Rouviere et al. 2014). Also, given the genetic basis for this category of GN, it could be a cause of familial GN.
In addition to the four main categories of GN, there are multiple histologic patterns of lesions within each category. Notably, the GN categories can transition from one pattern to another as the disease progresses, and the pattern is a main component of the diagnosis in human and veterinary nephropathology. For example, glomerular hypercellularity can be easily identified, but location of the hypercellularity (mesangial, endocapillary, extracapillary, or a combination) is always included in the description of the lesion. Presence of fibrinoid necrosis of the glomerular tuft and fibrinocellular crescents are markers for activity of the disease, whereas sclerosis (effacement of peripheral capillary loops by extracellular matrix) is a marker of chronicity and scarring. Some categories of GN are more likely to have certain pattern of lesions, such as crescent formation in anti-GBM disease.
Furthermore, the histologic pattern is also a major component of the subcategorization of GN, especially in immune complex–mediated GN. For example, membranous GN is mediated by immune complexes that are subepithelial, located between the podocyte and the GBM. This is a common cause of proteinuria in humans and companion animals. Early in the disease, the glomeruli might appear histologically normal, with evidence of immune complex deposition only identified through electron-dense subepithelial deposits on TEM and granular labeling of the capillary walls on IF. Over time, however, the GBM is remodeled by the synthesis of new GBM material from the podocytes. This eventually leads to a histologic pattern of thickened capillary walls. Special histochemical stains can be useful in the diagnosis. The Jones’s methenamine silver method can demonstrate the GBM remodeling (spikes and holes) in chronic cases (Nangaku and Couser 2005). Interestingly, even within this subcategory of membranous GN, there are two separate pathogeneses identified in humans. The most common type in humans is due to an autoantibody against the M-type phospholipase A2 receptor that is present on podocytes (L. H. Beck et al. 2009). Rarely, other podocyte antigens have been reported as causes of “primary” membranous GN, and serologic assays are used to determine whether a patient has an autoantibody against a podocyte antigen. These biomarkers are used to monitor for the recurrence of disease. The other type of membranous GN is secondary to an underlying disease that releases circulating antigens (e.g., neoplasia). If the antibodies that bind to the antigens have a low affinity, the antigens might pass though the GBM at a time when they are not bound to the antibody. They are trapped in the subepithelial space and then reassociate with the Igs that have also traversed the GBM, thereby reforming the immune complex (Iskandar and Jennette 1983). In addition to the subepithelial immune complexes, secondary membranous GN has immune complexes that are located in mesangial zones, whereas cases of primary membranous GN do not. Taken together, multiple features are integrated into the diagnostic algorithm for membranous GN including histologic pattern with special stains, TEM, IF, serologic assays, and presence of proteinuria (Figure 1).
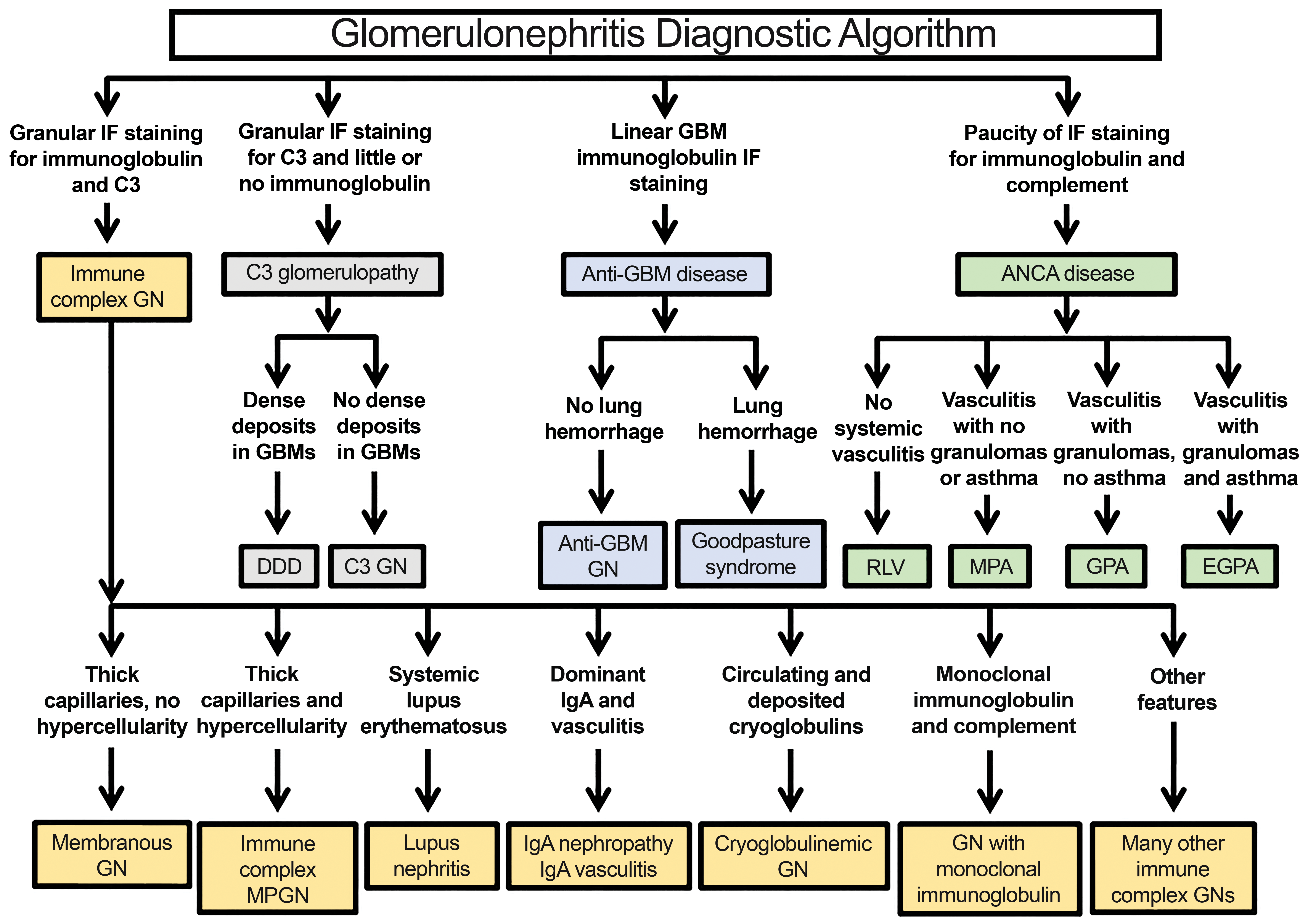
Diagnostic algorithm for glomerulonephritis in humans. GBM, Glomerular basement membrane; GN, Glomerulonephritis; ANCA, Anti-neutrophilc cytoplasmic antigen; DDD, Dense deposit disease; RLV, Renal limited vasculitis; MPA, Microscopic polyangiitis; GPA, Granulomatosis with polyangiitis; EGPA, Eosinophilic granulomatosis with polyangiitis; MPGN, Membranoproliferative GN.
Although proteinuric companion animals can demonstrate ultrastructural patterns of immune complex deposition that resemble both primary and secondary membranous GNs, a causative podocyte antigen has not yet been identified. It is important to note that the diagnosis of membranous GN in domestic animals relies on evidence of immune complex deposition in subepithelial space, no matter how thick the GBM is (R. E. Cianciolo et al. 2016; R. Cianciolo and Mohr 2015). As noted above, early in the disease, the glomerulus might appear completely normal because the complexes are small and only detectable with IF and TEM. This is in contrast to previously published criteria in veterinary pathology that relied on a thickened GBM for the diagnosis. If immune complexes are not identified along a thickened GBM in companion animal renal biopsy samples, then a more appropriate diagnosis is “glomerulopathy characterized by thickened GBM.” This will prevent the use of unnecessary immunosuppression in animals that do not have definitive evidence of immune complex deposition.
Lupus nephritis is another common subcategory of immune complex–mediated GN in humans and is due to the presence of autoantibodies against a wide array of cellular components. There are multiple histologic, IF, and TEM patterns of lupus GN, and a classification system based on the glomerular lesions is used to assess severity of injury (Markowitz and D’Agati 2007). This classification system also enables clinicians to monitor the progression of disease in patients with repeat biopsies, which is especially important in a disease known to present with periodic flares of active disease. Glomeruli can range from being histologically normal to hypercellular, they can have thickened GBM or GBM of normal thickness, and they can have crescents. The IF evaluation often identifies multiple types of Igs and complement components, and TEM demonstrates electron-dense deposits in many parts of the glomerular tuft (subendothelial, mesangial, intramembranous, and subepithelial) and even in the tubulointerstitium (Weening et al. 2004). Notably, there are numerous mouse models of lupus GN; however, none of them can completely recapitulate the heterogeneity of the lesions seen in human lupus. Even so, the mouse models have contributed significantly to our understanding of the basic pathologic mechanisms of this disease. They are used as preclinical models for drug development and proof of concept for novel therapeutics. The use of animal models in the investigation of lupus nephritis has been recently reviewed in detail (Richard and Gilkeson 2018). Although lupus GN has been reported in dogs (Osborne et al 1973), the diagnoses were questionable because they did not demonstrate positive labeling of multiple types of Igs in numerous locations throughout the glomerulus and tubulointerstitium.
Another common subcategory of immune complex–mediated GN is IgA nephropathy. This diagnosis relies on the detection of IgA-dominant or codominant deposits in glomeruli on IF. The electron-dense deposits are often present in mesangial zones with a few smaller scattered deposits along capillary loops. Glomeruli have mesangial hypercellularity with or without associated endocapillary hypercellularity. IgA nephropathy is a common cause of glomerular injury in humans, but it is exceedingly rare in nonprimate animals. Primates synthesize 2 types of IgA molecules (IgA1 and IgA2), whereas nonprimates only make IgA2. The pathogenesis of IgA nephropathy involves an abnormal glycosylation pattern of the IgA1 hinge region, exposing molecules that are normally hidden by galactose and sialic acids. This abnormal hinge region can be an antigen for IgG autoantibodies, which is why the glomeruli might have IgG codominance on IF evaluation (Wyatt and Julian 2013). Unfortunately, because IgA1 is not synthesized by mice, murine models of this disease have been difficult to create (Suzuki et al. 2014). Even so, transgenic mouse models have been created, and spontaneous IgA GN has been reported in laboratory marmosets (Schroeder et al. 1999). As previously stated, IgA GN is rare in companion animals, and this has been the experience of veterinary nephropathologists. Fewer than 1% renal biopsies from proteinuric animals have had IgA dominance or codominance on IF evaluation (unpublished data from the International Veterinary Renal Pathology Service July 7, 2018). Diagnosis of this subcategory of disease requires the advanced diagnostic modalities, and correct diagnosis allows clinicians to track disease progression and monitor for recurrence.
The last subcategory of immune complex–mediated GN to be discussed is immune complex–mediated proliferative or membranoproliferative GN. It is now recognized that there are multiple categories of disease that can result in the histologic pattern of proliferative or membranoproliferative disease. As stated above, nephropathologists use IF to distinguish between immune complex deposition and C3-mediated injury. If Igs are identified, then a search for underlying infection, neoplasia, or autoimmune disease is warranted. The deposits typically contain IgG and are located between the endothelial cell and GBM. Double contours of the GBM (“tram-tracking) is another distinguishing feature of membranoproliferative GN. Activation of the complement pathway at this subendothelial location attracts circulating leukocytes resulting in endocapillary hypercellularity. Over time, the endothelial cells synthesize new GBM resulting in the double-contoured capillary walls. The mesangial cells concurrently extend branches of cytoplasm out into the capillary wall, enhancing the appearance of double contours.
Notably, immune complex–mediated membranoproliferative GN is a common cause of proteinuria in companion animals (Schneider et al. 2013). In dogs, like in humans, it is often associated with an underlying infection. Similar to the scenario in membranous GN, the diagnosis of the immune complex–mediated membranoproliferative GN requires evidence of immune complex deposition. If immune complexes cannot be detected, then the preferred diagnosis is “glomerulopathy with membranoproliferative pattern.” One example of a nonimmune complex–mediated disease that can result in a membranoproliferative pattern in small animals is thrombotic microangiopathy, a disease in which endothelial cells are damaged directly (Holm et al. 2015). Correct diagnosis will avoid the use of immunosuppression in patients who do not have a proven pathogenesis of immune complex deposition, again highlighting the importance of ultrastructural evaluation of biopsy samples.
Subcategories of anti-GBM disease and ANCA-mediated GN also exist, with focus being mainly on which organs have evidence of vascular damage. In both diseases, pulmonary and glomerular capillaries can be injured, with pulmonary hemorrhage being life-threatening. Unfortunately, animal models often do not adequately represent all of the concomitant phenomenon seen in human patients with vasculitis of small renal and pulmonary vessels. Even so, IF evaluation allows nephropathologists to rapidly classify renal biopsies from human patients as having linear IgG staining or a pauci-immune pattern, so that physicians can quickly institute plasmapheresis to remove the autoantibodies.
Conclusion
In addition to the four main GN categories and subcategories, there are other types of glomerular injury which are not included because they are beyond the scope of this discussion. It should be emphasized, however, that many types of glomerular disease can result in overlapping histologic lesions, and some glomerular injuries might not demonstrate histologic lesions at all (e.g., early membranous GN). The advanced diagnostic techniques of IF and TEM are often essential in making the correct diagnosis. From the standpoint of a patient, the correct diagnosis will lead to treatment with the right medication (e.g., complement pathway inhibitor for patients with C3-associated glomerulopathy) and adequate monitoring in conditions known to recur (e.g., lupus GN). Also, relatives could be tested for types of GN that are known to be familial (e.g., IgA GN). From the research perspective, optimization of animal models will help investigators better understand complex multifactorial diseases such as lupus GN. These models can be used in drug development for proof of concept studies. Lastly, with the increasing use, and ongoing development, of novel biotherapeutics for a wide variety of diseases, correct identification of the mechanism of renal injury can be crucial in safety assessment decisions. If off-target glomerular damage is detected, then TEM on formalin-fixed tissue is often adequate to determine whether immune complexes were present. These data can guide toxicologic pathologists in future study designs and provide a basis for the decision to harvest fresh frozen tissue in the event that IF studies are warranted.
Footnotes
Author Contributions
Authors contributed to conception or design (RC, JJ); data acquisition, analysis, or interpretation (RC, JJ); drafting the manuscript (RC); and critically revising the manuscript (RC, JJ). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
