Abstract
Background
Studies have shown oxidative stress and apoptosis are the main pathogenic mechanisms of renal ischemia/reperfusion (IR) injury (IRI). Genistein, a polyphenolic non-steroidal compound, has been extensively explored in oxidative stress, inflammation and apoptosis. Our research aims to reveal the potential role of genistein on renal IRI and its potential molecular mechanism both in vivo and in vitro.
Methods
In vivo experiments, mice were pretreated with or without genistein. Renal pathological changes and function, cell proliferation, oxidative stress and apoptosis were measured. In vitro experiments, overexpression of ADORA2A and knockout of ADORA2A cells were constructed. Cells proliferation, oxidative stress and apoptosis were analyzed.
Results
Our results in vivo showed that the renal damage induced by IR was ameliorated by genistein pretreatment. Moreover, ADORA2A was activated by genistein, along with inhibition of oxidative stress and apoptosis. The results in vitro showed that genistein pretreatment and ADORA2A overexpression reversed the increase of apoptosis and oxidative stress in NRK-52E cells induced by H/R, while the knockdown of ADORA2A partially weakened this reversal from genistein treatment.
Conclusions
Our results demonstrated that genistein have a protective effect against renal IRI by inhibiting oxidative stress and apoptosis via activating ADORA2A, presenting its potential use for the treatment of renal IRI.
Introduction
Renal ischemia-reperfusion (IR) injury (IRI), a common cause of acute kidney injury (AKI), commonly occurs following hypotension, severe infection, partial nephrectomy and renal transplantation. The IRI mainly comes from an initial ischemia and followed by reperfusion, which elicits proximal tubular damage including epithelial cells inflammatory response and oxidative stress generation. 1 These complex cellular events eventually lead to renal cells damage or even death. Owing to the complicated pathophysiological process, renal IRI is associated with delayed graft function and high mortality. Therefore, it is critical to explore molecular mechanism from various ways and find useful targets for clinical treatment.
The purine nucleoside adenosine is an important active substance produced in almost all cells. 2 Adenosine participates in the regulation of many physiological and pathological processes by four adenosine receptors, A1, A2A, A2B and A3. 3 Evidence has grown in recent years that adenosine A2A receptor (ADORA2A) is associated with fibrosis in various organs, including cardiac and renal4–6 ADORA2A receptor activation seems to be attributed to cyclic adenosine monophosphate (cAMP)- and PKA-dependent pathways, leading to reduced inflammation and oxidative stress, as well as endothelial cell injury and death. 6 It has been demonstrated that selective stimulation of the adenosine A2A receptor partly reversed the IR related injury in different organs including the liver, lung and cerebrum.7–9 Therefore, ADORA2A is expected to be an effective target for the prevention and treatment of renal IRI.
Genistein (4,5,7-trihydroxyisoflavone), a polyphenolic non-steroidal compound, is mostly isolated from leguminous plants. Genistein plays various effective pharmacological actions, such as antioxidation, anticancer, antibacterial and antiviral.10–12 Studies have shown that genistein has a protective effect on cerebral IRI, and the mechanism may be involved in scavenging reactive oxygen species free radicals, preventing the generation of lipid peroxidation, delaying the apoptosis of nerve cells by regulating the ratio of bcl-2/bax, and inhibiting the activation of caspase.13,14 In recent years, it has been reported that genistein have a protective effect of genistein on renal ischemia-reperfusion injury in rats. 15 However, its mechanism is still not fully elucidated. By molecular docking software, we found that genistein have a strong bonding ability with ADORA2A, which suggests genistein could act as the ligand of ADORA2A and have the ability to regulate the signaling pathway.
To test this hypothesis, we identified the protective effect of genistein in IR induced oxidative stress and apoptosis vivo and vitro. Importantly, in vitro experiment, we investigated the role of ADORA2A in renal tubular epithelial cell injure induced by H/R.
Methods
Animal grouping and establishment of renal IR model
30 male mice (7–8 weeks old) were purchased from the Wuhan Institute of Biotechnology (Wuhan, China). The mice were housed in a specific pathogen-free facility at 21–23°C and 50% humidity in a 12 h dark/light cycle with two mice per cage. All mice were allowed free access to water and chow. They were divided randomly into five groups comprised of 6: Sham group, IR group, Vehicel group, Gen-L group and Gen-H group. 7 d before ischemia induction, mice in Gen-L group and Gen-H group respectively received pretreatment with genistein at the concentrations of 2.0 and 4.0 μg/mL, once daily. Mice in Vehicel group were pretreatment with vehicle (0.9% sodium chloride containing 1% dimethyl sulphoxide) without genistein. Establishment of renal ischemia-perfusion injury model: mice were anesthetized with 5% isoflurane for induction and 2% for maintenance. Bilateral dorsal subcostal incisions were made to expose the kidneys and separate the renal pedicle vessels. Bilateral renal pedicles were occluded with non-invasive vascular clamps for 28 min. Then, vascular clamps were released and the incision was sutured. During the operation, mice were placed on 37°C thermostatic electric blanket to maintain body temperature. After 12 h, urine was collected using the metabolic cage. Then, 24 h after surgery, the mice were killed, blood and kidney were harvested for further analysis.
All animal care protocols and experiments were carried out in strict accordance with the guidelines of the China Council on Animal Care and Use and approved by the ethics committee of Yantaishan Hospital.
Hematoxylin and eosin (HE) staining
Kidney tissue samples were fixed by 10% formalin, embedded in paraffin. 4 μm sections were prepared and then stained with HE. Morphological changes were observed with an Olympus BX51 microscope (Olympus, Center Valley, PA) at magnification ×200.
Biochemical measurements
The blood and urine samples were centrifuged. Then, serum creatinine (Scr) levels, serum kidney injury molecule 1 (sKIM-1) and serum neutrophil gelatinase-associated lipocalin (sNGAL), as well as their urinary KIM-1 (uKIM-1) and urinary NGAL (uNGAL) were detected by enzyme-linked immunosorbent assay kit (R&D Systems, Minneapolis, MN, USA).
Measurement of malondialdehyde (MDA) content and superoxide dismutase (SOD) activity
MDA content were measured by the thiobarbituric acid method, and SOD activity was detected by xanthine oxidase according to the instructions of the kit strictly.
Tunel assay
The kidney tissues were embedded in paraffin and sectioned, followed by deparaffinized, rehydrated. Then the tissues were infiltrated with proteinase K at 37°C for 20 min, and treated with TUNEL reagent (Dojindo, Japan). The slides were sealed, and placed in a dark humidified box at 37°C for 1 h. DAPI was used for nuclei staining. The nuclei of TUNEL-positive cells appear brown under an Olympus IX7I fluorescence microscopy (Tokyo, Japan)at magnification ×200. The apoptosis level was evaluated by the percentage of positive cells in the total cell count.
Immunohistochemistry (IHC) staining
The kidney tissues were sequentially deparaffinized, rehydrated and blocked with 10% normal goat serum. After that, the washed cells were incubated with primary antibodies (1:500 dilution), overnight at 4°C. The slides were then incubated with secondary antibodies (Vector lab, Burlingame, CA) for 1 h at room temperature. The sections were then developed with 3,3′-diaminobenzidine and counterstained with hematoxylin. Images were captured using an Olympus IX7I fluorescence microscopy (Tokyo, Japan).
Cell culture and grouping
Rat renal tubular cell lines NRK-52E were supplied by Wuhan Punosei Life Science and Technology Co. LTD. (Wuhan, China). The cells were cultured in low glucose DMEM medium containing 10% FBS and maintained in an incubator with 5% CO2 at 37°C. When the fusion degree reached 70%–80%, cells were maintained in fresh medium containing 1% FBS for 2 h prior to use in the experiments. The experiment was assigned into five groups: control group, hypoxia/reoxygenation (H/R) group, miA2A group, Gen group and Gen + siA2A group.
Establishment of NRK-52E hypoxia/reoxygenation (H/R) model and treatment
In Gen group, cells were pretreatment with genistein at the concentrations of 4.0 μg/mL. In miA2A group, cells were transfected with ADORA2A inhibitor. And in Gen + siA2A group, cells were transfected with ADORA2A mimic and pretreatment with genistein. After 2 h, H/R model were established. For hypoxia, cells were starved for 12 h in serum-free medium, then cultured in glucose-free and serum-free medium in a trigas incubator (94% N2, 5% CO2 and 1.0% O2) at 37°C for 6 h. Subsequently, the cells were changed to complete medium under normoxic conditions for 6 h reoxygenation.
Cell transfection
The overexpressed plasmid of ADORA2A was constructed by inserting the sequence c into the pcDNA 3.1 vector (Invitrogen). The ADORA2A inhibitor, inhibitor-NC, ADORA2A mimic and mimic-NC were constructed by GenePharma Co., Ltd (Shanghai, China). Cell transfection was carried out using Lipofectamine 3000 (Invitrogen) for 48 h.
Cell Counting Kit-8 (CKK8) assay
NRK-52E cells were grown in 96-well plates at 5 × 103/well and incubated for 24 h at 37°C in 5% CO2, different concentrations of genistein were added. After 24 h of cultivation, 10 μL CCK8 reagent (Dojindo, Japan) was added to each well and incubated for 2 h. The absorbance at a wavelength of 450 nm were measured by microplate reader (Bio-Rad, Berkeley, CA, USA).
The 5-ethynyl-2ʹ-deoxyuridine (EdU) cell proliferation assay
NRK-52E cells were grown in 96-well plates at 5 × 103/well and incubated for 24 h at 37°C in 5% CO2. 0.1 mL of 20 μ
Quantitative real-time PCR analysis
Total mRNA was extracted using TRIzol reagent (50 mg/mL) (Invitrogen, USA) according to the manufacturer’s protocol. cDNA was reverse transcribed from RNA using a Reverse Transcription Kit (Invitrogen, USA) according to the manufacturer’s instructions. cDNA amplification by PCR was then performed with a Light Cycler TaqMan Master reaction mix (cat. No. 04535286001 Roche, Switzerland). The amplification conditions were conducted as follows: 95°C for 10 min, followed by 40 cycles of 95°C for 20 s, 60°C for 30 s, and 72°C for 15 s. U6 were used as an internal control. 2-ΔΔCt method was utilized to calculate the relative transcription level of target gene.
Annexin V-FITC/PI assay
Adherent cells were collected and suspended again in 1× binding buffer. Then, 5 μL Annexin V-FITC and 5 μL propidium iodide (PI) were added followed by cultivation at room temperature in the dark for 30 min. Cell apoptosis was evaluated using a flow cytometry system (Becton Dickinson FACS Vantage SE, Sanjose).
Reactive oxygen species (ROS) detection
NRK-52E cells were grown in 24-well plates at 1 × 105/well. 20 μ
Immunofluorescence
NRK-52E cells were grown in 24-well plates at 5 × 103/well and incubated for 24 h. The cells were treated with drugs for 48 h, and washed twice with PBS. Then, 4% paraformaldehyde solution was added to fix for 30 min. Next, the cells were incubated with 0.3% Triton in the dark for 20 min, and blocked with 10% goat serum +10%BSA for 30 min. Anti-ADORA2A antibody (ab105134, 1:200 dilution, Sigma-Aldrich) diluted in blocking buffer at 4°C overnight for 24 h. Goat anti-rabbit fluorescent secondary antibody (ab97040,1:2000 dilution, Sigma-Aldrich) was added to completely cover the cells, and incubated for 30 min at room temperature. Then, fluorescein-labeled secondary antibody and 4,6-diamidino-2-phenylindole (DAPI) stain solutions were added and incubated under dark conditions. Images were captured using Olympus IX7I fluorescence microscopy (Tokyo, Japan) at magnification ×200.
Statistical analysis
Statistical analysis was conducted using SPSS20.0 (IBM, NewYork, USA) for windows. Statistical comparisons between groups were analyzed by one-way analysis of variance (ANOVA). Multiple comparisons were tested by Duncan’ test. All dates were reported as the mean ± SD, and The level of statistical signification was set at p < 0.05. All trials were administered three times.
Results
Genistein alleviates histopathological damage and improves renal function after renal IR in mice
The renal histology was observed by HE staining. As presented in Figure 1(a), tubular epithelial cells were in normal appeared in the control group. Renal IR resulted in significant damage, as evidenced by the renal tubule dilation, brush-border loss, as well as the swelling and necrosis of renal tubular epithelial cells. However, pretreatment with genistein reduced severe renal damage in L-Gen and H-Gen groups compared with that in IR and Vehicel groups. Genistein treatment protects against IRI in mice. (a) Reno-protection of genistein was assessed histologically (H&E staining). Bar = 100 μm. (b, c and d) The blood indicators of renal injury were determined by ELESA. (e and f) The levels of KIM and NGAL in urine were detected by ELESA. **p < 0.01, ***p < 0.001.
To further clarify the protective effects of genistein against renal IRI, biological indicators of renal injury were also examined. Firstly, we observed the level of Scr, sKIM-1 and sNGAL and found that the increasing trends in the IR group were markedly blocked by genistein pretreatment (Figure 1(b), (c) and (d)). The level of uKIM-1 and uNGAL showed a similar trend with sKIM-1 and sNGAL (Figure 1(e) and (f)). These data suggested that pretreatment with genistein could attenuate IR-induced renal dysfunction and pathological damage.
Genistein promote proliferation, attenuated apoptosis and oxidative stress induced by renal IRI in mice
To investigate the role of genistein in renal IRI in vivo, mice were intraperitoneally pretreated with genistein (2 and 4 μg/mL) once daily for 7 days before surgery. We measured the changes in proliferation, apoptosis and oxidative stress. Ki-67 is a main biomarker of proliferation. IHC staining was used to access the level of Ki-67. The results showed that compared with the control group, IR decreased the level of Ki-67, and genistein pretreatment significantly increased the level of Ki-67 (Figure 2(a)). TUNEL staining were performed to evaluate the apoptosis with or without genistein pretreatment. The results showed that compared with the control group, IR increased the percent of tunel positive cells, which was significantly decreased by genistein pretreatment (Figure 2(d)). Bcl-2, Bax and cleaved caspse-3/caspase-3 are important apoptosis biomarkers. As shown in Figure 2(f), (g) and (h), when mice suffered IR, the levels of Bax and cleaved caspse-3/caspase-3 were significantly increased, while expression levels of Bcl-2 molecule was significantly reduced, genistein pretreatment reversed this trend. These results together indicated that genistein pretreatment could promote proliferation and inhibit apoptosis, which showed a protective effect on kidney tissues damage induced by IR. Genistein promotes proliferation, attenuated apoptosis and oxidative stress induced by renal IRI in mice. (a) The levels of Ki-67 were evaluated by IHC staining. Bar = 100 μm. (b and c) MDA content and SOD activity in renal tissues were measured. (d) The apoptosis of renal tubular cell was assessed by Tunel staining. Bar = 100 μm. (e, f and g) The relative expression of Bax and Bcl-2, and Cleaved caspse-3/caspase-3 were evaluated by qRT-PCR. *p < 0.05, **p < 0.01.
Genistein upregulated the expression of ADORA2A in kidney tissues with IR
To determine the potential mechanism by which genistein protects renal IRI, we detected the expression of the ADORA2A signaling pathway in renal by IHC staning and qRT-PCR. As shown in Figure 3, compared with the control group, the expression level of ADORA2A in tissues was decreased markedly, however, pretreatment with genistein upregulated the expression of ADORA2A. The results indicated that genistein might protected kidney tissues from IRI by upregulating the expression of ADORA2A. Effect of genistein on ADORA2A expression. (a and b). ADORA2A expression were determined by IHC staning (Bar = 100 μm). (c). The relative expression of ADORA2A were verified by qRT-PCR. **p < 0.01, ***p < 0.001.
ADORA2A transfection efficiency in NRK-52E cells
To further verify the role of ADORA2A in renal IRI, the NRK-52E cells with overexpression and knockdown of ADORA2A were constructed by Lipofectamine transfection. Transfection efficiency was verified by qRT-PCR. As shown in Figure 4, compared with the NC1 group, the level of ADORA2A in NRK-52E cells showed a significant decline with knockdown efficiency of (0.723 ± 0.045). Compared with the NC2 group, the level of ADORA2A in NRK-52E cells was significantly elevated with transfection efficiency of (1.608 ± 0.033). Transfection efficiency of ADORA2A in NRK-52E cells was validated by qRT-PCR. ***p < 0.001.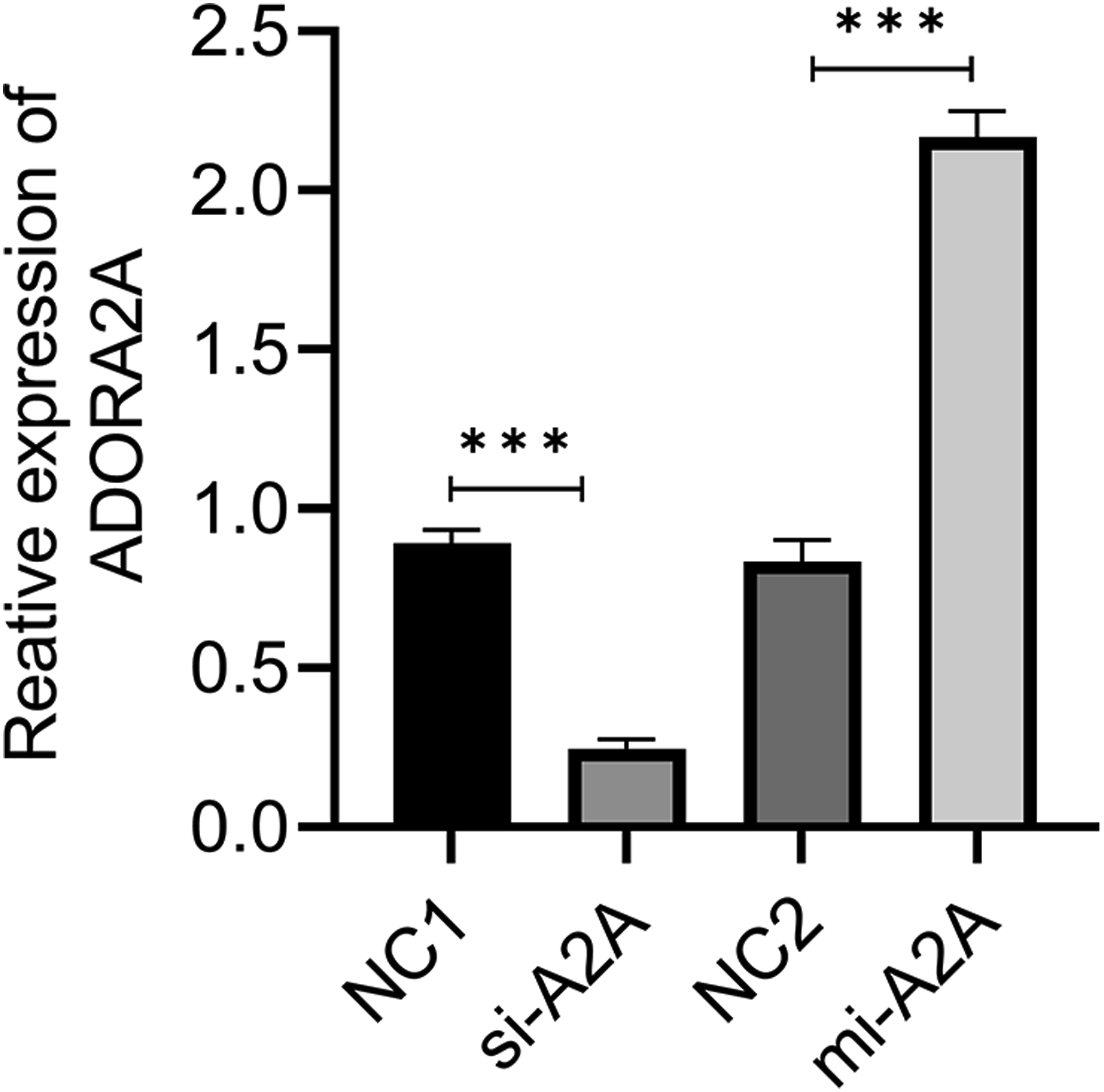
Genistein increases proliferation and inhibits apoptosis in NRK-52E cells with H/R injury via ADORA2A
To explored the Figures 1, 2, 3, 4, 5, 6 and 7 functional significance of ADORA2A in NRK-52E cells with H/R injury, the proliferation and apoptosis were firstly investigated. The CKK8 and EdU assay were used to detect the proliferation ability of NRK-52E cells. Annexin V-FITC/PI assay was used to test the apoptosis. The results (Figures 5 and 6) showed that genistein pretreatment and ADORA2A overexpression reversed the decrease of proliferation and the increase of apoptosis in NRK-52E cells induced by H/R. However, the knockdown of ADORA2A partially weakened this reversal from genistein pretreatment. These results together indicated that genistein pretreatment have a protective effect on NRK-52E cells damage induced by HR via upregulating ADORA2A. Genistein increases proliferation and inhibits apoptosis in NRK-52E cells with H/R injury via ADORA2A. (a and d) Cell proliferation was detected by EDU assay. (c) Cell viability was measured using CCK-8. (B and F) Cell apoptosis was detected by Annexin V-FITC/PI assay. (e, g and h) The levels of Bax and Bcl-2, Cleaved caspse-3/caspase-3 were evaluated by qRT-PCR. **p < 0.01, ***p < 0.001. Genistein attenuates oxidative stress induced by H/R injury via ADORA2A. (a and b) Changes in ROS production were determined by flow cytometry. (c and d) MDA content and SOD activity were evaluated. **p < 0.01, ***p < 0.001.

Genistein attenuates oxidative stress induced by H/R injury via ADORA2A
To determine whether genistein attenuates oxidative stress induced by H/R injury is associated with ADORA2A, the levels of intracellular ROS, MDA and SOD were assessed. As shown in Figure 3, oxidative stress injury was induced by H/R, as indicated by the dramatically upregulation of ROS and MDA, and the significant decrease of SOD activity compared with the control group. Importantly, pretreated genistein and ADORA2A overexpression reversed the changes, while the changes in in Gen group were blocked by the knockdown of ADORA2A. The results demonstrated that genistein could alleviate H/R-induced oxidative abnormalities and restore endogenous antioxidant systems in NRK-52E cells via upregulating ADORA2A.
The expression of ADORA2A in NRK-52E cells with H/R
The expression of the ADORA2A in NRK-52E cells was determined by immunofluorescent staining and qRT-PCR. As shown in Figure 7, compared with the control group, the expression level of ADORA2A in H/R group was decreased markedly. However, pretreatment with genistein upregulated the expression level of ADORA2A, which was weakened by knockdown of ADORA2A. ADORA2A expression in NRK-52E cells. (a and b) ADORA2A expression were determined by immunofluorescent staining. (c) The relative expression of ADORA2A were verified by qRT-PCR, **p < 0.01, ***p < 0.001.
Discussion
In the past decades, significant progress has been made in our understanding of IRI and various pharmaceuticals have been identified to protect against renal IRI.16,17 However, due to the limitations of dose, safety and other factors, there is still no pharmacologic intervention to be successfully applied in clinical studies. 18 In this study, we demonstrated both in vivo and in vitro systems that the genistein was effective to reduce IR or H/R injury by promoting cell proliferation, inhibited oxidative stress and apoptosis. Moreover, our finding in vitro suggested that the mechanisms underlying this protection are largely due to the upregulating of ADORA2A.
Due to high metabolic activity and hypoxia, the most important manifestation of kidney injury after IR is the apoptosis of renal tubular epithelial, which affects the repair of renal tubular epithelial cells, eventually causes irreversible damage. 19 IR induced apoptosis events occur through a series of processes including the generation of free ROS, the activation of inflammatory mediators and leukocyte. 20 Natural phytochemicals due to their multitarget and multimechanism, have attracted extensive attention in renal IRI treatment. 21 Genistein, a kind of polyphenolic non-steroidal compound has been extensively explored in oxidative stress, inflammation and apoptosis. 22 Several studies showed that genistein exerted beneficial effects in animal IR models of different tissues.23,24 Despite this, its protective action against renal IRI and the underlying mechanisms remained unclear until now. In vitro experiment, genistein ameliorated pathological damage to the kidneys and reduced Scr, sKIM-1, sNGAL and uKIM-1, uNGAL levels, suggesting that genistein improved renal pathological damage and dysfunction. Moreover, genistein treatment significantly enhanced cellular proliferation, reduced oxidative stress and apoptosis, firmly supporting the protective effect of renal IRI.
Adenosine, an endogenous nucleoside, was the first signalling molecule s involved in remote ischemic preconditioning. 25 Although extensively recognized for its ability to increase blood flow, effects on hemangiectasis do not sufficiently explain the renal function of adenosine in I/R. 26 Adenosine exerts its biological effects mainly through coupling with any of four G-protein-coupled adenosine receptors designated as A1, A2A, A2B, and A3. 27 Among them, ADORA2A is dominatingly expressed in epithelium and experimental evidence indicates extracellular adenosine acting at ADORA2A represents the protective effects on renal interstitial fibrosis. 28 Also, it is reported that ADORA2A antagonists inhibited or attenuated photoreceptors and RGC apoptosis by modulating the inflammation and oxidative stress in both age-related macular degeneration (AMD) and glaucoma. 29 In vivo experiment, we found that pretreatment with genistein upregulated the reduced ADORA2A induced by IR, indicating the potential negative feedback between degree of renal injury and ADORA2A levels. Further, we simulated the renal IR condition by hypoxia and reoxygenation of renal tubular epithelial cells to investigate the role ADORA2A for genistein against IR injury. The study in vitro revealed that genistein pretreatment or ADORA2A overexpression promoted the proliferation, inhibited the apoptosis and oxidative stress in NRK-52E cells after HR, while the knockdown of ADORA2A partially weakened this reversal in Gen group. All of these studies indicated that genistein could exert multiple physiological processes in renal tubular epithelial cells, including proliferation, apoptosis, and oxidative stress via ADORA2A signaling pathways.
Adenosine, a natural ligand for adenosine receptors, is induced to synthesize during ischemia. 30
Adenosine generation caused increased Akt phosphorylation as well as increased PI3K. 31 A study reported, the specific agonist of ADORA2A receptor activated PI3K in hepatocytes through a cAMP/adenylyl cyclase-dependent pathway. 32 Genistein is also known to be a tyrosine kinase inhibitor, and could influence PI3K/Akt signaling. 33 The PI3K/Akt pathway is a survival pathway, involved in protection against various stressors. 34 Activation of Akt promotes cell survival by modulation Bcl-2 associated death promoter (BAD). 35 The results in our study showed genistein reversed the oxidative stress and apoptosis induced by H/R through its stimulation of ADORA2A expression. Hereby, we believe that that ADORA2A expression or activity could be regulated directly or indirectly by genistein to ameliorate IR-induced kidney. However, the definite mechanism regarding how ADORA2A expression and activity are regulated by genistein in renal cells is still unclearly, and needed to explore further.
In summary, we provided both in vitro and in vivo data demonstrating that genistein could protected against renal IRI by modulating oxidative stress, proliferation and apoptosis of renal tubular epithelial cells. Furthermore, we revealed that genistein targeted ADORA2A to prevent tubular cell damages and deaths. Our study may provide a novel therapeutic target for IRI treatment and present experimental evidence for application of genistein.
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Haozheng Shan, Shuqing Li and Ruigang Diao. The first draft of the manuscript was written by Haiyan He and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
I confirm that all data in this article are available.
