Abstract
A mistaken overdose of bupivacaine into systemic circulation may cause severe cardiovascular side effects. The aim of this study was to assess the effects of pretreatment with combined intra venous lipid emulsion (ILE) and high-dose insulin therapy against cardiotoxicity caused by bupivacaine intoxication. The rats were divided into the following three groups: Group B received a saline pretreatment plus a bupivacaine, group L received ILE pretreatment plus a bupivacaine, and in group I, insulin with glucose was infused intravenously, plus ILE pretreatment plus a bupivacaine. The electrocardiogram tracing, invasive arterial pressure, and heart rate (HR) of rats were monitored continuously. Arterial blood gas analysis was performed in all groups. Arterial blood gas analysis revealed that the baseline pH, PaO2, and PaCO2 values were similar between groups (p > 0.05). Widening of Q, R, and S wave complex was found 46.8 ± 16.7, 92.0 ± 5.80, and 106.5 ± 17.9 s after initiation of bupivacaine infusion in groups B, L, and I, respectively. Time elapsed until 25% reduction of HR 127.3 ± 17.7, 248.4 ± 34.1, and 260.1 ± 51.3 s for groups B, L, and I, and 25% reduction of mean arterial pressure 107.6 ± 14.1, 253.2 ± 36.3, and 292 ± 57.7 s for groups B, L, and I, respectively. Arrhythmia was observed after 142.2 ± 27.5, 180.7 ± 17.8, and 190.7 ± 19.2 s for groups B, L, and I, respectively. Finally, asystole occurred after 560.1 ± 76.4, 782.4 ± 63.0, and 882.5 ± 105.1 s for groups B, L, and I, respectively. This finding indicates that the survival time of rats administered pretreatment with ILE plus insulin+glucose and those given ILE was observed to be longer.
Introduction
Bupivacaine is one of the most commonly used local anesthetics (LA) for both central and peripheral blocks in regional anesthesia. 1 A mistaken overdose of bupivacaine into systemic circulation may cause central nervous system dysfunction and asystole that may result in severe cardiovascular side effects. 2,3 In vitro studies have shown that bupivacaine inhibits extracellular K+ flow, stimulation of ventricular myocytes, Ca++ release from sarcoplasmic reticulum, and depolarization of Na+ channels and myocardium. 4,5 Thus, the depolarization of the myocardium reduces axion potential and lengthens the P–R interval and Q, R, and S wave (QRS) duration. 4
Weinberg et al. 6 in a study in 1998 identified that lipid treatment, interestingly and unexpectedly, increased the dose of bupivacaine causing asystole. From this date, the first case of successful treatment with lipid emulsions was reported by Rosenblatt et al. in 2006. 7 Since then there have been many studies published on the positive effects of lipid use for treatment of local anesthesia intoxication. There are even recommendations in many international guidelines on the use of intra venous lipid emulsion (ILE) treatment for local anesthesia intoxication. 8,9 However, some meta-analyses have reported that there may be some side effects linked to rapid infusion of ILE treatment and total ILE amount administered. 10
Insulin increases Ca++ release from sarcoplasmic reticulum and it improves phase 0 depolarization in the heart caused by hypokalemia. As a result of this, insulin improves cardiac output and heart rate (HR) by increasing catecholamine release and Ca++ influx into cardiac muscle cells. 11,12 In addition, the phosphorylation of heat shock protein 27 is induced by insulin. Heat shock protein 27 has been shown to associate with actin microfilaments in adult cardiomyocytes and it is associated with improved myocardial contractility. 13,14 Thus, it eases stimulation of ventricular myocytes and strengthens the contractions during myocardial depression frequently encountered with intoxication. 15 Previous case series have described successful combination of both high-dose insulin and intra venous lipid therapy for some toxicity. 16,17
The aim of this study was to assess the effects of pretreatment with combined ILE and high-dose insulin therapy against cardiotoxicity caused by bupivacaine intoxication in an experimental model of anesthetized rats.
Material and methods
After the study was approved by the Animal Experiments Local Ethical Committee of Zonguldak (Turkey), Bulent Ecevit University (BEUN) Medical School (2016-07-03/02), 24 adult male Wistar Albino rats weighing 300–450 g were randomly divided into three groups. The rats in group B received a saline pretreatment (2 mL/kg in 10 min) plus a bupivacaine infusion (3 mg/kg/min), the rats in group L received lipid emulsion (ClinOleic®; Baxter, Lessines, Belgium) pretreatment (10 mg/kg for 10 min) plus a bupivacaine infusion (3 mg/kg/min), and 100 U/kg of insulin with D-glucose (50 mg/100 mL) was infused intravenously (i.v.) at 0.60 mL/h for 10 min to maintain euglycemia plus lipid emulsion pretreatment (10 mg/kg for 10 min) plus a bupivacaine infusion (3 mg/kg/min) in the rats of group I. All of the rats included in the study were obtained from BEUN Experimental Animals Research Unit, all fed with standard rat pellets and housed in temperature- and humidity-controlled (23 ± 1°C and 55% relative humidity) rooms that were lit on a daily schedule (12:12-h light/dark) until the day of experiment. During the experiment, the care of the laboratory animals was in accord with international guidelines.
Experimental procedure
The animals were anesthetized with 80 mg/kg ketamine hydrochloride and 12 mg/kg xylazine administered intraperitoneally. In this study, we applied an experimental model of anesthetized rats as the same with Hanci et al.’s study. 18 Using sterile techniques, a cannula was inserted into the right femoral vein for drug administration, and then the right femoral artery was cannulated to allow invasive arterial pressure monitoring and measuring arterial blood gas analyze. The electrocardiogram (ECG) tracing, invasive arterial pressure, and HR of the anesthetized rats were monitored and recorded continuously using a monitoring system (Petas KMA 800; Petas, Ankara, Turkey). Body temperature was measured using a rectal probe and maintained at 36.5 ± 0.5°C with an external heat lamp. Arterial blood gas was measured (Abl 800 basic; Radiometer, Copenhagen, Denmark) during a 30-min stabilization period after preparation for surgery. Saline infusion was administered to replace blood that was drawn for blood gas analysis. After the saline infusion was completed, the baseline HR and mean arterial pressure (MAP) of the rats were obtained and recorded. After this measurement was obtained, 2 mL/kg of normal saline was administered i.v. for 10 min (group B), 10 mg/kg of lipid emulsion 20% was administered i.v. for 10 min (group L), or 10 mg/kg of lipid emulsion 20% plus 100 U/kg of insulin with D-glucose (50 mg/100 mL) was infused i.v. at 0.60 mL/h for 10 min using a calibrated electronic infusion pump (Body Guard 323; CME, Lichtenstein, Germany). Hemodynamic measurements were repeated in 5-min intervals until the 15th min after infusion of lipid emulsion or lipid emulsion plus insulin+dextrose or normal saline. Fifteen minutes after the study drugs’ infusions were completed, an intravenous infusion of 0.5% bupivacaine was started a rate of 3 mg/kg/min using a calibrated electronic infusion pump.
Bupivacaine was infused until asystole occurred. At the end of the experiment, the recorded data were evaluated and the timing of the following events was calculated: time to first alteration in QRS complex on ECG trace (defined as an increase of >20% in the duration of the QRS complex); time to first arrhythmia (defined as the first arrhythmia that occurred that was accompanied by an abnormal systole on the arterial pressure tracing); time to 25%, 50%, and 75% reductions in HR relative to baseline; time to 25%, 50%, and 75% reductions in MAP relative to baseline; and time to asystole (defined as the absence of pressure pulses on the arterial pressure tracing). The baseline hemodynamic measurements used in these calculations were obtained before study drug (lipid emulsion or lipid emulsion plus insulin+dextrose or saline) infusion.
The primary outcome of this study was the observing hemodynamic differences from the start of bupivacaine infusion until asystole.
Statistical analysis
In a previous report comparing saline and dexmedetomidine pretreatment in the prevention of bupivacaine-induced cardiotoxicity in rats, the investigators used the same primary outcome as used in this study. 18 On the basis of these data, 18 to achieve a power of 98% and setting 0.05 as the level of significance, we calculated that it was necessary to include eight rats in each group.
All analyses were performed with SPSS (version 17.0; IBM SPSS Inc., Chicago, Illinois, USA). Analysis of variance (ANOVA) or Kruskal–Wallis test was used to determine for differences between three groups. The Tukey test was used as a post hoc test, if the ANOVA test is statistically significant. The Dunn’s test was used for post hoc test after Kruskal–Wallis test. Differences in continuous variables between three groups were compared by paired t test or Wilcoxon signed rank test depending on the distribution of the data. Results were considered significant at p > 0.05. All values are expressed as mean ± standard deviation (SD).
Results
Arterial blood gas analysis revealed that the baseline pH (group B: 7.39 ± 0.28; group L: 7.40 ± 0.34; and group I: 7.38 ± 0.22), PaO2 (group B: 199.8 ± 8.97; group L: 197.4 ± 33.5; and group I: 198.7 ± 19.3 mm Hg), and PaCO2 values (group B: 38.5 ± 4.56; group L: 37.7 ± 5.46; and group I: 37.4 ± 4.72 mm Hg) were similar between groups (p > 0.05).
Baseline HR values (group B: 340.0 ± 18.1; group L: 342.8 ± 25.0; and group I: 342.7 ± 20.5 bpm) and MAP values (group B: 105.6 ± 14.4; group L: 108.0 ± 14.7; and group I: 109.0 ± 15.7 mm Hg) were also similar for three groups (p > 0.05). In −20, −15, −10, −5, and 0 time point, both MAP and HR values were similar between groups (p > 0.05). HR and MAP values were significantly lower in group B than the other two groups in all measurements from 1st to 10th min following bupivacaine infusion (p > 0.05) additionally. There was no statistically significant difference between the two groups in terms of HR and MAP values (p > 0.05). HR and MAP values for groups B, L, and I are included in Figures 1 and 2.
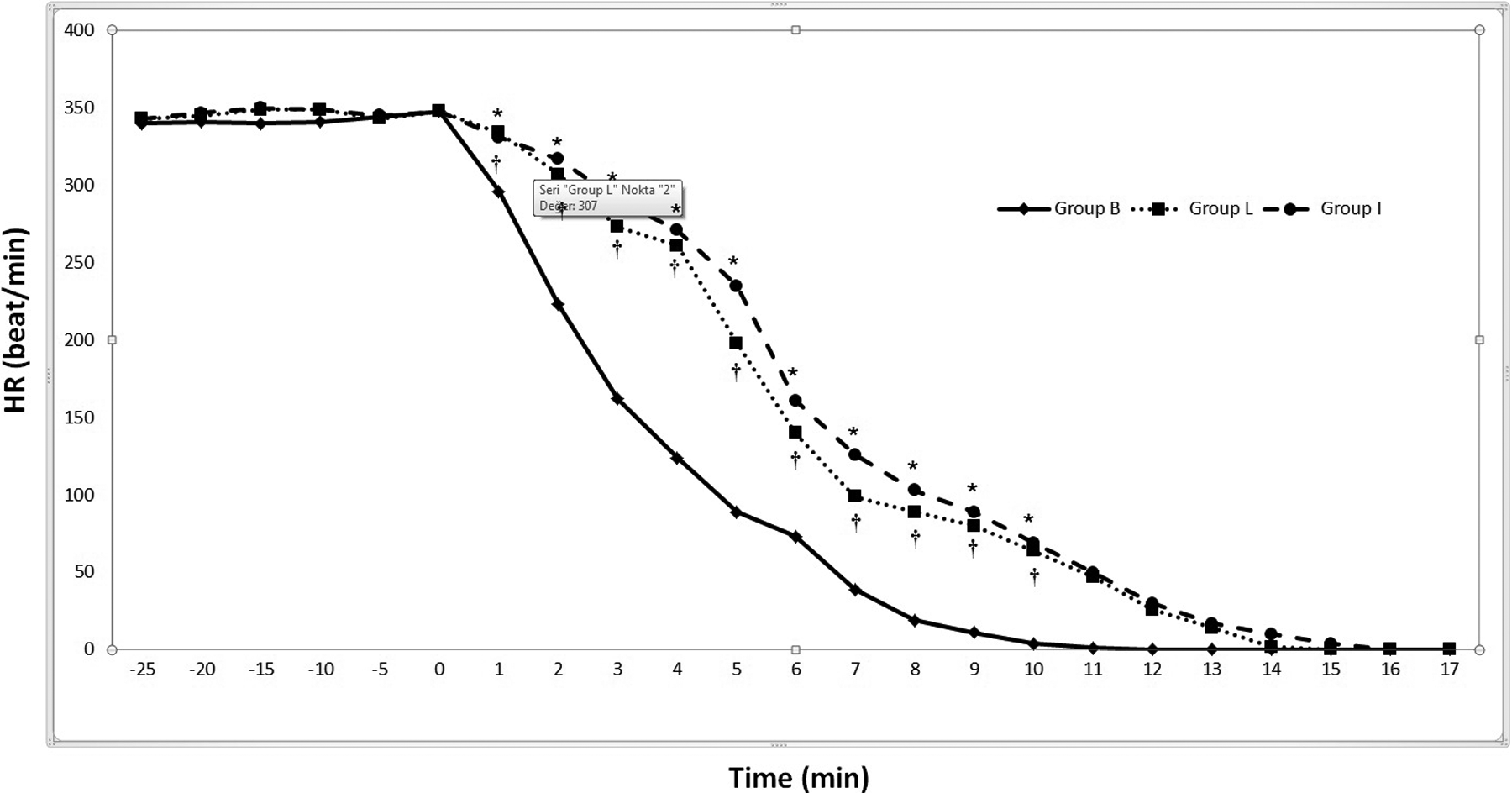
Changes in HR during treatment. Study drug infusion (saline) was started −25th min. Study drug infusion (saline) ended at the −10th min. Bupivacaine infusion was started at 0 min. *p > 0.05, between groups I and B; and †p > 0.05, between groups L and B, assessment of HR values, ANOVA or Kruskal–Wallis test was used to determine for differences between three groups. The Tukey test was used as a post hoc test, if the ANOVA test is statistically significant. The Dunn’s test was used for post hoc test after Kruskal–Wallis test. HR: heart rate; ANOVA: analysis of variance.

Changes in MAP during treatment. Study drug infusion (saline) was started −25th min. Study drug infusion (saline) ended at the −10th min. Bupivacaine infusion was started at 0 min. * p >0.05, between groups I and B; and †p > 0.05, between groups L and B, assessment of HR values, ANOVA or Kruskal–Wallis test was used to determine for differences between three groups. The Tukey test was used as a post hoc test, if the ANOVA test is statistically significant. The Dunn’s test was used for post hoc test after Kruskal–Wallis test. MAP: mean arterial pressure; HR: heart rate; ANOVA: analysis of variance.
The first sign of bupivacaine toxicity is the widening of QRS complex and it was found 46.8 ± 16.7, 92.0 ± 5.80, and 106.5 ± 17.9 s after initiation of bupivacaine infusion in groups B, L, and I, respectively. Other signs of bupivacaine-induced cardiotoxicity, including time elapsed until 25% reduction of HR (127.3 ± 17.7 s for group B, 248.4 ± 34.1 s for group L, and 260.1 ± 51.3 s for group I) and 25% reduction of MAP (107.6 ± 14.1 s for group B, 253.2 ± 36.3 s for group L, and 292 ± 57.7 s for group I). Both 25% reduction of HR and 25% reduction of MAP mean values were statistically significantly lower in group B compared to group L (p < 0.001) and group I (p < 0.001), whereas there was not a significant difference in groups L and I (p > 0.05). Arrhythmia, which is a late sign of bupivacaine toxicity, was observed after 142.2 ± 27.5 s in group B, 180.7 ± 17.8 s in group L, and 190.7 ± 19.2 s in group I. Arrhythmia mean values were statistically significantly lower in group B compared to group L (p = 0.021) and group I (p = 0.006), whereas there was not a significant difference in groups L and I (p = 0.747). Finally, asystole occurred after 560.1 ± 76.4 s in group B, 782.4 ± 63.0 s in group L, and after 882.5 ± 105.1 s in group I. Asystole mean values were statistically significantly lower in group B compared to group L (p < 0.001) and group I (p < 0.001), whereas there was not a significant difference in groups L and I (p = 0.16). All other signs of bupivacaine-induced cardiac toxicity occurred significantly lower in group B compared to group L (p < 0.001) and group I (p < 0.001), whereas there was not a significant difference in groups L and I (p > 0.05). Hemodynamic monitoring and ECG data for groups B, L, and I are included in Table 1 and Figures 1 and 2.
Times to defined end points in the ECG, MAP, and HR after bupivacaine infusion.a
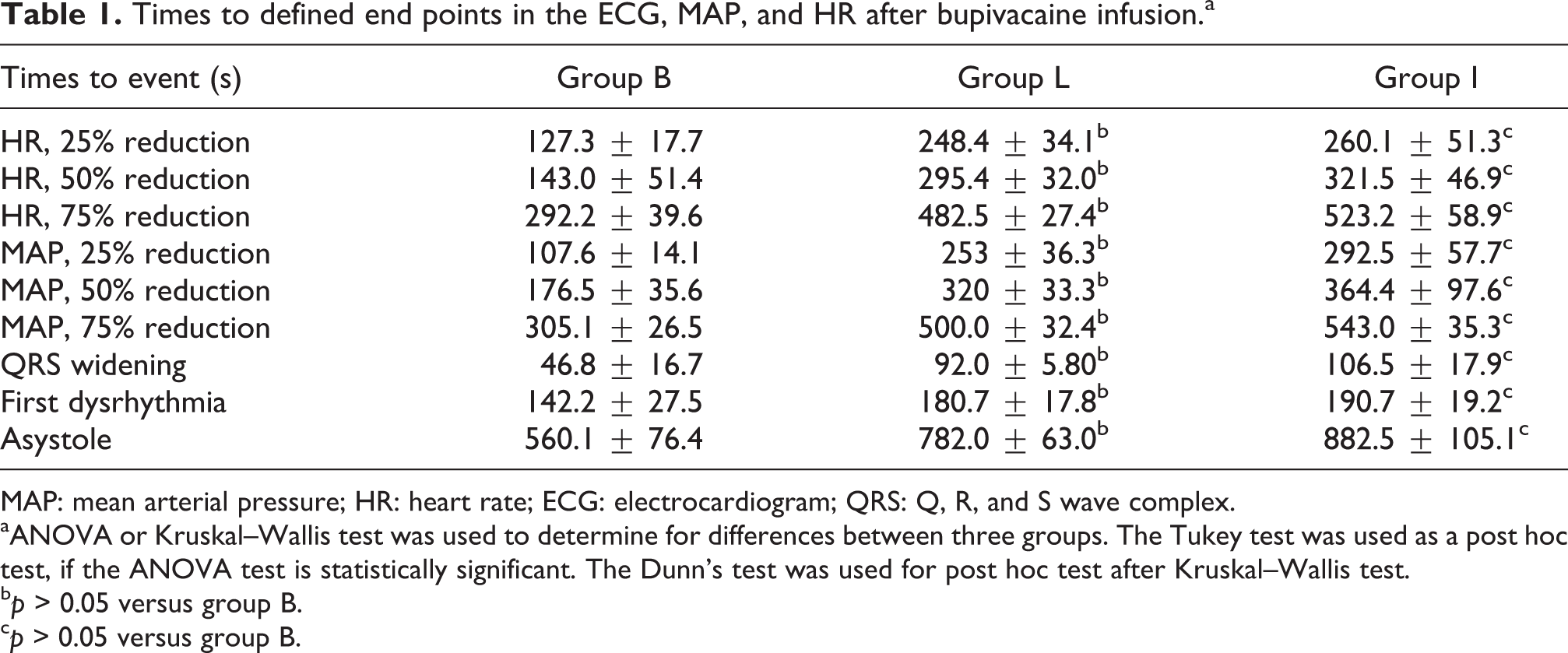
MAP: mean arterial pressure; HR: heart rate; ECG: electrocardiogram; QRS: Q, R, and S wave complex.
aANOVA or Kruskal–Wallis test was used to determine for differences between three groups. The Tukey test was used as a post hoc test, if the ANOVA test is statistically significant. The Dunn’s test was used for post hoc test after Kruskal–Wallis test.
b p > 0.05 versus group B.
c p > 0.05 versus group B.
Discussion
This experimental study showed that lipid and lipid plus insulin+dextrose pretreatment delayed the formation of intoxication linked to bupivacaine and lengthened the duration to asystole. Additionally, this study found that lipid plus insulin+dextrose pretreatment did not result in better durations in terms of formation of bupivacaine intoxication and asystole compared to lipid pretreatment.
In anesthetic practice, many LA agents are used. 1,19 Unwanted i.v. administration of LA may cause an increase in plasma LA concentrations and result in severe cardiovascular side effects. In the 1990s, the mortality rate for LA intoxication was nearly 50% and there was no specific treatment protocol. 20 However, since the middle of the 2000s, i.v. lipid emulsion (ILE) has been successfully used for cardiotoxicity especially linked to bupivacaine. 8,9,21 In fact, in recent times whether in animal models or in case reports, it is reported that ILE may be effective not just for cardiac arrest cases due to only LA agents but also those linked to different medications. 22,23 The use of lipid emulsion in resuscitation increased the values for the dose of bupivacaine required to cause death in 50% of animals after a rapid bolus of bupivacaine administered in a 10-s interval. 6 Additionally, both MAP and HR reduced with bupivacaine infusion. 24 This effect is formed by the inhibition of sodium channels by bupivacaine. 1 With bupivacaine infusion, there is a widening of the QRS complex and a lengthening in correcting QT intervals on the ECG. 25 These effects are due to the delay in repolarization caused by overdose of bupivacaine. In this study, together with these findings, the reduction durations in HR and MAP were defined as cardiotoxicity findings linked to overdose of bupivacaine.
In spite of these hopeful developments, is ILE treatment the only antidote for treatment of cardiotoxicity developing linked to bupivacaine? A recent meta-analysis by Hoegberg et al. found that ILE treatment may reverse some cardiovascular and neurological side effects developing linked to LA toxicity; however, they reported that they did not find persuasive proof showing that ILE was more effective than vasopressor agents or which should be the first treatment for LA toxicity. 26
For regulation of heart muscle contraction disorders observed in toxicity developing due to medications such as LA, beta-blockers, and Ca++ channel blockers, insulin administration is becoming more popular. 11,16,27 Insulin stimulates D-glucose intake in cells in muscles and fat tissue and activates the phosphatidylinositol 3-kinase pathway easing intake of D-glucose within cells. 28 This pathway is suppressed by intoxication with Ca++ channel blockers and is known to be activated by high-dose insulin in vitro. In situations of myocardial stress, insulin increases D-glucose intake within the cell and improves myocardial energy production. Known as an effective inotropic agent, insulin causes vasodilatation in systemic, coronary, and pulmonary veins. Insulin also reduces the formation of free radicals occurring linked to hyperglycemia and suppresses the inflammatory process with protective effect on the heart. 28 In vitro studies have shown that bupivacaine inhibits the extracellular K+ flow, stimulation of ventricular myocytes, Ca++ release from sarcoplasmic reticulum, and depolarization of Na+ channels and myocardium. 5,29 Insulin increases the release of Ca++ from sarcoplasmic reticulum. Insulin also increases phase 0 depolarization of the heart caused by hypokalemia and improves ventricular contractions. 5 A case series by Holger et al. stated that in toxin-linked cardiogenic shock cases, high-dose insulin treatment may be beneficial for cardiotoxicity. 30 Similarly, studies on humans and animals have reported insulin provided inotropic support for cardiotoxicity developing linked to overdose of bupivacaine. 31–32,25
In our study, the group given pretreatment of lipid plus insulin+dextrose did not result in better for cardiotoxicity signs and asystole formation durations linked to overdose of bupivacaine. We think these results are due to the number of rats included in the study. It is clear that due to ethical reasons, no prospective and placebo-control studies on insulin+dextrose or ILE use can ever be performed or will remain at limited levels. 33 However, the need for human studies in the future is clear. Insulin+dextrose increase cytosolic ATP levels and stimulate aerobic metabolism activating the Ca++ and K+ channels and also increase the production of myocardial energy. The result of these events is that insulin has a positive inotropic effect on the heart. 34 An animal study by Yi et al. reported that administration of insulin+dextrose had an ameliorative effect on acute bradycardia and hypotension developing linked to propafenone intoxication. 11
The most important limitation of our study is that after cardiotoxicity findings were created, plasma bupivacaine concentration was not examined. The cardiotoxicity of bupivacaine is known to be dose-linked; however, as bupivacaine was administered in all three groups, this situation did not change the primary outcome. Another limitation of this study is the use of rats with spontaneous respiration in the study model. This situation means the respiratory parameters of rats may have affected the study results. However, before beginning bupivacaine infusion, all rats had arterial blood gas monitored and no rat in the study had any symptoms of hypoxia. Additionally, in terms of blood gas parameters, there was no statistically significant difference between the groups. Since this study did not include a group in which the experiments were conducted with “insulin+dextrose+bupivacaine” only, it is not possible to say whether similar results could be achieved or not if lipid pretreatment was excluded.
Conclusion
In conclusion, the survival time of rats administered pretreatment with lipid plus insulin+dextrose and those given lipids was observed to be longer. Our results are animal study results. Although experimental and controlled studies cannot be performed on humans due to ethical concerns, we believe that in the future case series on this topic will be instructive.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
