Abstract
Carbon monoxide (CO) poisoning is a significant cause of death especially in developing countries. The current study investigated cardioprotective effects of insulin in CO-poisoned rats. Male rats were exposed to 3000 ppm CO for 1 h. Insulin (100 and 120 U/kg intraperitoneally) was immediately administered after CO exposure and on the next 4 days, on a daily basis (a total of 5 doses). On day 5, animals were euthanized, and the hearts were harvested for Western blotting and histopathological studies. The electrocardiograms (ECG) were recorded postexposure to CO and after the completion of insulin treatment period. Histopathological evaluations showed reduction of myocardial necrosis in insulin-treated animals compared to controls. BAX/BCL2 ratio, as a proapoptotic index, was significantly reduced in treatment groups (p < 0.01). The ECG findings showed no differences among groups; also, compared to control animals, myocardial Akt levels were not markedly affected by insulin. The current study showed that insulin significantly reduces myocardial necrotic and apoptotic indices in CO-poisoned rats.
Introduction
Carbon monoxide (CO) is a by-product of incomplete combustion of fossil fuels that causes serious poisoning in humans. 1 Because of nonirritating, colorless, and tasteless nature of this chemical, it causes a considerable number of deaths annually and is called a “silent killer”. 2 In Iran, which is the second largest producer of natural gas in the world, natural gas is commonly used as a heat source. 3,4 CO poisoning especially affects the organs with greater demand for oxygen such as heart, brain, kidneys, and lungs. 2,5 Heart injury attributed to CO poisoning could be devastating. 2,6,7 CO poisoning leads to acute and chronic toxicity; the latter can cause headache, myalgia, dizziness, or neuropsychological impairment, 1,6 while severe acute CO exposure results in confusion, loss of consciousness, or death. 1 CO affinity for hemoglobin is >200 times higher than that of oxygen and carboxyhemoglobin increment leads to a leftward shift in oxyhemoglobin curve and decreases oxygen delivery to tissues. 8 It should be noted that CO poisoning could occur even at low CO concentrations. 1
Insulin is a polypeptide secreted by the pancreas in response to stimulants including high plasma glucose levels. Besides its role in treating diabetes, insulin preparations were found to have specific cardioprotecting effects following ischemia reperfusion (I/R) injury in animal models. 9 –11 Myocardial salvation was the result of a number of favorable effects including myocardial endothelial nitric oxide synthase (eNOS) increment, coronary vasodilation, and activation of PI3K/PKB/Akt pathway. 12 Akt protein activation by insulin was found capable of reducing apoptosis of cardiomyocytes. 12
The cardioprotective effects of insulin following myocardial I/R injury 9,11,13 may decrease the harmful cardiac effects of CO poisoning. Therefore, we examined the possible cardioprotective effects of insulin in CO-poisoned rats, in terms of myocardial necrosis evaluated by hematoxylin–eosin (H&E) staining, electrocardiographic (ECG) changes, and myocardial Akt protein expression as a cell survival. Also, we determined BAX/ BCL2 ratio because at least in part, apoptosis rate is controlled by the antiapoptotic BCL2 and the proapoptotic BAX proteins. In this regard, overexpression of Bax, which could be triggered by many external stimuli, increases apoptosis rate. Therefore, evaluation of the BCL2 and BAX gene expression balance (i.e. BAX/BCL2 ratio determination) provides an estimate of apoptosis rate. 14
Materials and methods
Animals
In this study, 18 male Wistar rats (aged 8–10 weeks and weighing 200–250 g) obtained from Zabol University of Medical Sciences animal house, Zabol, Iran, were used. Animals were kept under standard conditions (at 25°C with 12-h/12-h light
Chemicals
Akt, BAX, BCL2, anti-β-actin antibodies, and secondary rabbit antibody were purchased from Cell Signaling (Beverly, Massachusetts, USA). Coomassie (Bradford) Protein Assay Kit was purchased from Thermo Scientific (Rockford, Illinois, USA). Regular insulin was purchased from EXIR Pharmaceutical Co. (Tehran, Iran). CO capsule (99.999% purity) was obtained from Darman Gas (Tehran, Iran). For ECG recording, Power Lab (AD Instruments, Bella Vista, New South Wales, Australia) was used.
Experimental groups and study design
Animals were randomly divided into three groups (n = 6 in each group) including control (normal saline-treated) rats, rats treated with 100 U/kg insulin, and or rats treated with 120 U/kg insulin. Insulin doses were selected based on a previously published study, 15 which showed 100 U/kg insulin as the favorable effect on oxidative stress levels as reactive oxygen species are thought to be involved in the pathogenesis of CO-induced injuries.
For induction of poisoning, animals were kept in a 12-L Plexiglas container. The chamber was connected to a CO capsule with an airtight tube and had constant flow and pressure. Before their placement in the container connected to the CO capsule, animals were anesthetized by ketamine (100 mg/kg) and xylazine (10 mg/kg). Rats from all groups were exposed to 3000 ppm CO under 1 atmosphere pressure for 1 h. The concentration of CO was continuously monitored by a CO analyzer (TPI707 CO Analyzer, Korea). 16 After CO exposure and after the completion of insulin treatment period, the ECGs were recorded using an ECG recorder (AD instrument, Australia).
Immediately after CO exposure, animals were removed from the chamber and insulin (100 and 120 U/kg) was administered intraperitoneally (i.p.) along with dextrose (50 mg/kg; i.p.). All insulin-treated animals received 50 mg/kg dextrose for prevention of possible insulin-induced hypoglycemia. Control group animals were injected with the same amount of normal saline. For carboxyhemoglobin level determination, at the end of the exposure period, blood samples were collected from the animals. Insulin administration was continued for the next 4 consecutive days on a daily basis. On day 5, animals were anesthetized by ketamine/xylazine mixture (100/10 mg/kg) and killed. After opening the chest cavity, hearts of animals were excised and collected. Heart samples assigned for pathological analysis were fixed in formalin 10% and sent for histopathological evaluations. For myocardial necrosis evaluation, samples were stained with H&E. The remaining of the hearts were rinsed in saline solution, and about 200 mg of each heart was placed in cryogenic tubes for Western blot analysis.
Samples assigned for Western blot analysis were homogenized with a mechanical homogenizer, centrifuged (at 10,000 rpm and 4°C), and sonicated. Protein content of the supernatant was measured by a Bradford protein assay kit. After dilution, protein levels of all samples were identical. Finally, the samples were either used freshly or stored at −70°C. About 10–20 µl of each sample was loaded on the acrylamide gel. After electrophoresis, the blot was transferred into polyvinylidene fluoride (PVDF) membrane. Next, the PVDF membrane was blocked by 5% skim milk overnight and then treated with primary antibody and then with the secondary antibody linked to horseradish peroxidase. At the end, enhanced chemiluminescence 500–1000 µl (Pierce, USA) was used to detect blots with Syngene Chemidoc, and the blots were analyzed using Gene Tools software (SynGene Company).
Carboxyhemoglobin level assessment
For confirmation of CO poisoning induction, following CO exposure, blood samples were obtained from the rats’ tails and evaluated using a spectrophotometer calibrated for rats’ blood (Jenway 6305; Bibby Scientific Ltd, Staffordshire, UK). 17
ECG recording
ECG in lead I was recorded (Power Lab; AD Instruments, Bella Vista, New South Wales, Australia) after CO exposure and after the completion of the treatment period. ECG was analyzed with focus on ST-segment, T-wave, AV block type 1 and 2, Q wave, premature ventricular contractions, sick sinus syndrome, atrial fibrillation, ventricular tachycardia, and fibrillation.
Histopathological examination
After fixing the hearts in 10% formalin for at least 24 h, slices with 4–5 µm thickness were stained with H&E. Histopathological evaluation was focused on detection of necrotic cells and foci as well as lymphatic infiltration. Pathological findings were categorized into 3 grades based on the severity of insults 16 : grade 1, scattered necrotic cells and/or lymphatic infiltration; grade 2, one or two necrotic foci; and grade 3, more than two necrotic foci 18 (Figure 1).
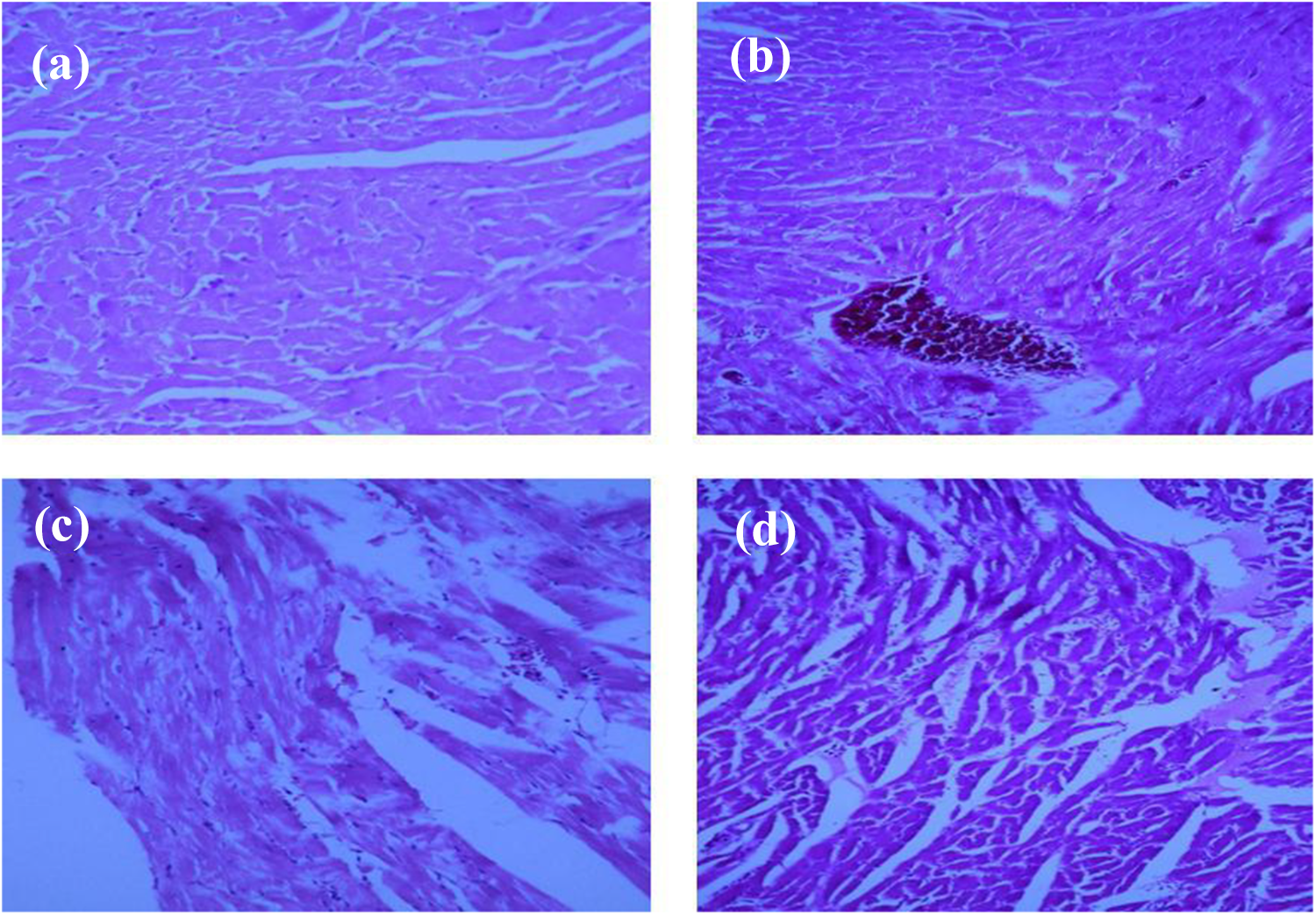
Histopathological findings of rats’ hearts following exposure to 3000 ppm CO in comparison to the intact heart: (a) intact animal, (b) grade 1 pathological findings showing scattered necrotic cells and/or lymphatic infiltration, (c) grade 2 indicating two necrotic foci, and (d) more than two necrotic foci (×40).
Statistical analysis
Data were analyzed using SPSS version 16 (SPSS, Inc., Chicago, Illinois, USA). Two-way analysis of variance was used to compare continuous variables and χ2 and Fisher’s exact test were employed for categorical variables. A p < 0.05 was considered as statistically significant.
Results
Carboxyhemoglobin concentration after exposure to 3000 ppm CO
Mean carboxyhemoglobin serum levels postexposure to 3000 ppm CO for 1 h were found to reach concentrations of 71 ± 8% and were consistent with previous published studies (60–76%). 8,19 Also, it is worth mentioning that, all animals of all groups survived till the end of the experiment; in other words, zero mortality was documented in this study.
Effect of insulin on ECG changes after exposure to 3000 ppm CO
No significant ECG changes were observed between insulin-treated groups and the control group. However, in all CO-exposed groups, ST changes, inverted T waves, premature ventricular contractions, and sinus pauses were observed (Figure 2).

ECG change after 3000 ppm CO poisoning: (a) normal ECG, (b) ST-segment depression, (c) premature ventricular contraction, and (d) sinus pause.
Effect of insulin on histological findings after exposure to 3000 ppm CO
Histopathological evaluation showed considerable differences between control and insulin-treated groups while some samples presented more than one type (i.e. grade) of injury (Table 1)
Histological findings postexposure to 3000 ppm CO in control and insulin-treated rats (100 and 120 U/kg).

a“m/n” indicates that “m” rats out of “n (which is 6 in each group)” rats had the specific grade of histopathological insults.
Effect of insulin on Akt protein levels following exposure to 3000 ppm CO
As shown in Figure 3(a), Akt myocardial protein levels in the control group were not significantly different from those of insulin-treated groups.

Western blot analysis of (a) Akt protein and (b) BAX/BCL2 protein ratio in the heart of rats, 5 days after CO exposure. Band intensities were normalized against β-actin in the same sample. Data are expressed as mean ± standard error of mean. **p < 0.01: significant differences compared to control animals.
Effects of insulin on BAX/BCL2 protein ratio following exposure to 3000 ppm CO
Insulin treatment at both doses of 100 and 120 U/kg significantly reduced myocardial protein BAX/BCL2 ratio in comparison to the control group (p < 0.01; Figure 3(b)).
Discussion
Acute CO poisoning has severe consequences including direct effects on organs that have higher oxygen demand including the heart and brain. 2 Cardiac effects of CO poisoning include necrosis of cardiomyocytes and apoptosis and ECG changes. 6 Recently, considerable attention has been attracted toward the use of insulin in attenuating I/R injury in animal models. 9,11,12 The current study showed that insulin reduced necrosis of cardiomyocytes and apoptotic indices but had no effects on rats’ ECGs and Akt protein levels.
In an I/R setting, insulin exerted vasculoprotective action as reflected by a preserved endothelium-dependent vasorelaxation and reduced endothelial apoptosis. 10 Ma et al. using a canine model showed that insulin treatment accompanied by glucose and potassium infusion reduced endothelial apoptosis via deactivation of caspase 3. 10 Experimental reports have shown that insulin protects the heart against I/R-induced damages through modulation of PI3K/Akt/mTOR pathway. 10,20 Also, it was shown that insulin targets survivin, which is regarded as an antiapoptotic protein and a regulator of apoptosis. 21 The current study offered evidence regarding the antiapoptotic effects of insulin in a cardiac model with similarities to I/R models. BAX is a proapoptotic protein, while BCL2 is an antiapoptotic protein whose low levels lead to caspase activation and apoptosis. 22 BAX/BCL2 ratio is considered an apoptosis indicator. 22 In the current study, insulin treatment significantly reduced the said index.
In studies on endothelial cells, it was found that insulin antiapoptotic effects are mediated through PI3K–Akt–eNOS pathway activation. 10,20 Akt is a pro-survival protein that is important in suppressing I/R-induced apoptosis in cardiomyocytes. 23 It is known that PI3K activation is pivotal to PKB/Akt activation and proapoptotic BAX mitochondrial protein inhibition. 12 Upregulation of Akt transcription inhibits conformational changes in the proapoptotic BAX protein and its translocation to mitochondria, obviating the disruption of mitochondrial inner membrane and activation of caspase-3 18,24 However, in the current study, insulin treatment did not change Akt protein expression. Insulin cardioprotective effects in many settings are considered multifactorial in origin including myocardial apoptosis and infarction extent reduction with a concomitant improvement of cardiac function. 11,20,25 Insulin action in improving energy metabolism and reducing pyruvate synthesis is of particular interest in cases of diminished myocardial oxygen supply such as CO poisoning. 26
In the current study, contrary to the majority of I/R studies, animals were not pretreated with insulin; insulin was administered after CO exposure, a fact that can explain the stable Akt protein levels. At the same time, while insulin had no effects on Akt protein levels, it decreased BAX protein levels; evaluation of the phosphorylated form of Akt protein in future studies may provide a clearer picture of Akt involvement and/or its possible favorable effects on CO poisoning.
Although CO is regarded as a major cause of poisoning-induced death and may lead to acute or delayed neurological consequences (with a prevalence of almost 3–40%), apart from hyperbaric oxygen therapy, no other treatment option is available for treatment of CO-poisoned patients. Experimental studies have shown promising results by using granulocyte colony-stimulating factor, erythropoietin, allopurinol, and so on to protect organs against CO-induced injuries. Nevertheless, clinical applications of these agents need further evaluations. 27
Conclusion
Insulin administration to CO-poisoned rats reduced myocardial pathological insults and significantly decreased the apoptotic index BAX/BCL2 ratio, compared to control animals.
Footnotes
Authors’ note
This work was a PharmD thesis supported by Zabol University of Medical Sciences, Zabol, Iran, and Students Research Committee, Zabol University of Medical Sciences.
Acknowledgments
The authors thank the pathology laboratories at Amiralmomenin Hospital, Zabol University of Medical Sciences, for their kind assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
