Abstract
Dexmedetomidine (DMED), an alpha-2 adrenoreceptor agonist, has been widely used in regional anesthesia procedures. However, the effect of DMED on local anesthetic cardiotoxicity has not been well delineated. This study consisted of two experiments. In experiment A, 42 Sprague–Dawley (SD) rats were randomly divided into 6 groups (n = 7), each group was pretreated with DMED 0 μg kg−1 (D0 group), 1 μg kg−1 (D1 group), 3 μg kg−1 (D3 group), 6 μg kg−1 (D6 group), 12 μg kg−1 (D12 group), and 24 μg kg−1 (D24 group), administered through the right femoral vein. In experiment B, 20 SD rats were randomly divided into 4 groups (n = 5), such as control group, DMED group, yohimbine (YOH) group, and DMED + YOH group. Each subgroup in experiment B was also pretreated similarly as in experiment A. After pretreatment of rats as described above (in experiments A and B), bupivacaine 2.5 mg kg−1 min−1 was infused to induce cardiac arrest. In experiment A, the lethal dose threshold of bupivacaine and plasma bupivacaine concentration in D3 and D6 group were higher than the other groups. In experiment B, there was no interaction between DMED and YOH in lethal dose threshold, arrhythmia time, plasma concentration of bupivacaine, and myocardial content of bupivacaine. DMED doses of 3–6 μg kg−1 elevated the lethal dose threshold of bupivacaine without involvement of the alpha-2 adrenoceptors.
Introduction
Dexmedetomidine (DMED) is a highly selective alpha-2 adrenoceptor agonist that has sedative, anxiolytic, hypnotic, analgesic, and sympatholytic effects. 1 It is a widely used, effective, and safe sedative for the peripartum period, 2 in infants, 3 and the elderly. 4 It also decreases the mortality for patients who undergo cardiac surgery, 5 thereby reducing ischemia/reperfusion injury. 6
Regional anesthesia combined with intravenous DMED administration has demonstrated many advantages in clinical medicine. Candiotti et al. 7 reported DMED is a good option during use in regional anesthesia, demonstrating minimal complications when compared to other pharmacological interventions. Recently, in combination with local anesthetics during regional anesthesia, it has become increasingly popular both in clinical studies 8 and in animal models 9 because of its analgesic properties. Although ultrasound-guided regional anesthesia reduces the incidence of local anesthetic toxicity, 10 the number of cases reported is still increasing. Our study suggests that the application of DMED before local anesthetic administration can delay the occurrence of local anesthetic cardiac toxicity 11 and improve the dose threshold dose regarding seizures. 12 The dose relationship and mechanism of action regarding the ability of DMED to mitigate bupivacaine-induced local anesthetic cardiotoxicity have yet to be delineated.
We hypothesized that there is a dosing level of DMED that increases the lethal threshold for bupivacaine. To test this hypothesis, we established a rat model by administering varied doses of DMED in combination with bupivacaine to induce local anesthetic toxicity. Our primary outcome in this study was the “lethal dose threshold” of bupivacaine that represented the degree of “toxic tolerance” the rats could endure during a background infusion of bupivacaine. Secondary outcomes included arrhythmia time (time to first dropped beat), plasma concentration of bupivacaine, and myocardial content of bupivacaine.
Method
Experimental animals
This study was conducted with the approval of the Wenzhou Medical University’s Animal Care and Use Committee (Wenzhou, China). Healthy, male, specific pathogen-free mature male Sprague–Dawley rats (n = 62) aged 7–8 weeks were purchased from Shanghai SLAC Laboratory Animal Co, Ltd (Shanghai, China) and housed at the Wenzhou Medical University animal center in a standard 12-h reverse day/night cycle at an ambient temperature of 21.5°C in an individual ventilated cages system (Shinva Medical Instrument Co. Ltd, China). The rats were 8–10 weeks of age and weighed 300–400 g when they were entered into the study.
Anesthesia and monitoring
The rats were fasted for 12 h before the experiments, with free access to water. On the day of the experiment, the rats were anesthetized with an intraperitoneal injection of chloral hydrate (350 mg kg−1). They were then intubated via tracheotomy and mechanically ventilated with 1–2% sevoflurane in 100% oxygen using a rodent volume-controlled ventilator (HX-101; TME Technology Co., Ltd, Chengdu, China): tidal volume 6 mL kg−1, respiratory rate 58–62 breaths min−1, and an inspiratory to expiratory ratio 2:3. The right femoral vein and right femoral artery were cannulated. The body temperature was maintained at 38–39°C with an electric blanket. Electrocardiography and the arterial pressure were recorded continuously during the protocol by a MedLab data archiving and retrieval system using U/4C051 (Nanjing Medease Science and Technology Co., Ltd, Jiangsu, China). After completion of all invasive procedures, animals were allowed to stabilize for 15 min at 0.5% sevoflurane and 100% oxygen, after which baseline heart rate (HR) and systolic blood pressure (SBP) were recorded, along with measurement arterial blood gases, which included pH, arterial partial pressure of carbon dioxide (PaCO2), arterial partial pressure of oxygen (PaO2), base excess (BE), bicarbonate (HCO3 −), total carbon dioxide (TCO2), arterial oxygen saturation (SaO2), and lactate.
Grouping of animals and experimental protocols
Experiment A
A total of 42 rats were randomly divided into 6 groups (n = 7 in each group), such as D0, D1, D3, D6, D12, and D24 groups. Each group had the following amount of DMED (P-code: 12815 SML0956-10MG; Sigma-Aldrich Co, St. Louis, Missouri, USA) in 1 mL infused via the right femoral vein, respectively: 0, 1, 3, 6, 12, and 24 μg kg−1 over 10 min. This was followed by an infusion of 2.5 mg kg−1 min−1 bupivacaine (0.5%, P-code: 101524503 B5274-5G; Sigma-Aldrich Co) via the right femoral vein until cardiac arrest (Figure 1). SBP, HR, electrocardiogram, arrhythmia time, and asystole were recorded by MedLab data archiving and retrieval system (Medlab-U/4C051; Nanjing Medease Science and Technology Co., Ltd).

Time line of drug delivery in experiment A. D0, D1, D3, D6, D12, and D24 groups. Each group had the following amount of dexmedetomidine infused, respectively: 0, 1, 3, 6, 12, and 24 μg kg−1 over 10 min. This was followed by an infusion of 2.5 mg kg−1 min−1 bupivacaine until cardiac arrest.
Samples of blood were obtained from the apex of each heart immediately after cardiac arrest. Plasma was separated from blood by centrifugation (4000 r min−1, 10 min). The left ventricle and plasma were stored at −80°C for determination of bupivacaine concentrations. The content of bupivacaine in the heart and the concentration of bupivacaine in the plasma were then measured by liquid chromatography/mass spectrometry (Agilent Technologies, Inc, Santa Clara, California, USA). 13
Experiment B
A total of 20 rats were randomly divided into 4 groups (n = 5), such as control, DMED, yohimbine (YOH), and DMED + YOH groups. Before the infusion of DMED and bupivacaine, YOH (P-code: 101509939 Y3125-1G; Sigma-Aldrich Co) was used to block alpha-2 adrenoceptors. The YOH and DMED + YOH groups had YOH infused intravenously (1 mg kg−1 in 1 mL) via the right femoral vein over 5 min. Alternately, the control and DMED groups had an equivalent volume of normal saline infused over the same period instead. The results in experiment A influenced our decision to choose 3 μg kg−1 as the dose of DMED in experiment B. During the ensuing 10 min, DMED and DMED + YOH groups received an infusion of 3 μg kg−1 DMED (5 mL h−1) via the right femoral vein; control and YOH groups received an equivalent volume of normal saline infusion over the same time period. At the conclusion of the DMED infusion, all groups were administered a constant 0.5% bupivacaine infusion of 2.5 mg kg−1 min−1 through the right femoral vein until cardiac arrest (Figure 2). The methods of data acquisition were similar to experiment A.

Time line of drug delivery in experiment B. DMED + YOH groups had yohimbine infused intravenously (1 mg kg−1 in 1 mL) over 5 min. Control and DMED groups had an equivalent volume of normal saline infused over the same period instead. During the following 10 min, DMED and DMED + YOH groups received an infusion of 3 μg kg−1 dexmedetomidine; control and YOH groups received an equivalent volume of normal saline infusion over the same time period. This was followed by an infusion of 2.5 mg kg−1 min−1 bupivacaine until cardiac arrest. DMED: dexmedetomidine; YOH: yohimbine.
Terms used
Lethal dose threshold refers to the gross dose of bupivacaine infusing before cardiac arrest. Arrhythmia time was defined as the time between the beginning infusion of bupivacaine and the first dropped beat.
Statistical analysis
In experiments A and B, the power analysis was based on results of preliminary experiments (n = 3) comparing the lethal dose in experiment A (D0: 13.6 ± 4.4 mg; D1: 12.1 ± 2.7 mg; D3: 16.7 ± 3.0 mg; D6: 17.2 ± 4.5 mg; D12: 12.3 ± 4.5 mg; D24: 10.0 ± 1.3 mg) and experiment B (control: 11.9 ± 1.4 mg; DMED: 16.7 ± 2.5 mg; YOH: 10.8 ± 0.7 mg; DMED + YOH: 16.4 ± 1.7 mg) yielded a sample size of n = 7 in experiment A and n = 4 in experiment B. Power was set at 0.9; significance criterion was set at 0.05. Considering the animal losses during the preliminary experiment, a sample size of 7 in experiment A and 5 in experiment B was chosen for each group (PASS 11 software; NCSS, Kaysville, Utah, USA).
Statistical analysis was performed using SPSS (version 19.0; Chicago, Illinois, USA) for Windows. Measurement data were presented using mean ± standard deviation (SD). The Shapiro–Wilk test was used to test for normal distribution. Values for baseline parameters, blood gas values, lethal dose threshold, arrhythmia time, the content of bupivacaine in cardiac tissue, and the concentration of bupivacaine in plasma in experiments A and B were all compared by one-way analysis of variance (ANOVA), and Turkey post-tests for data that had homogeneity of variance, or Dunnett T3 post-tests for data that exhibited heterogeneity of variance when indicated by significance of difference. Values for lethal dose threshold, arrhythmia time, the content of bupivacaine in cardiac tissue, and the concentration of bupivacaine in plasma in experiment B were all compared by two-way ANOVA to test interaction between DMED and YOH. Non-normal distribution data such as the BE and TCO2 were analyzed using the Kruskal–Wallis H test. Statistical significance was considered as p < 0.05.
Results
Characteristics of study subjects
Baseline values of weight, SBP, HR, and arterial blood gas values of experiments A and B are demonstrated in Tables 1 and 2. No differences were observed among groups with regard to weight, baseline hemodynamic metrics, or baseline arterial blood gas values.
Baseline values of weight, SBP, HR, and arterial blood gas values in experiment A.a

SBP: systolic blood pressure; HR: heart rate; PaCO2: arterial partial pressure of carbon dioxide; PaO2: arterial partial pressure of oxygen; BE: base excess; HCO3 −: bicarbonate; TCO2: total carbon dioxide; SaO2: arterial oxygen saturation; DMED: dexmedetomidine; YOH: yohimbine; SD: standard deviation.
a Normally distributed data were given as mean ± SD, whereas non-normal distributed data were expressed as median and interquartile values.
Baseline values of weight, SBP, HR, and arterial blood gas values in experiment B.a

SBP: systolic blood pressure; HR: heart rate; PaCO2: arterial partial pressure of carbon dioxide; PaO2: arterial partial pressure of oxygen; BE: base excess; HCO3 −: bicarbonate; TCO2: total carbon dioxide; SaO2: arterial oxygen saturation; DMED: dexmedetomidine; YOH: yohimbine; SD: standard deviation.
a Normally distributed data were given as mean ± SD, whereas non-normal distributed data were expressed as median and interquartile values.
Experiment A
Lethal dose threshold
The lethal dose threshold in D3 and D6 groups was higher than D0, D1, D12, and D24 groups (D3 vs. D0, p = 0.010; D3 vs. D1, p = 0.014; D3 vs. D12, p = 0.001; D3 vs. D24, p < 0.001; D6 vs. D0, p = 0.005; D6 vs. D1, p = 0.007; D6 vs. D12, p < 0.001; D6 vs. D24, p < 0.001). Compared to D0 group, D12 and D24 groups displayed no significant differences in lethal dose threshold (D0 vs. D12, p = 0.940; D0 vs. D24, p = 0.292). D3 group and D6 group displayed no significant differences (p = 1; Figure 3(a)).
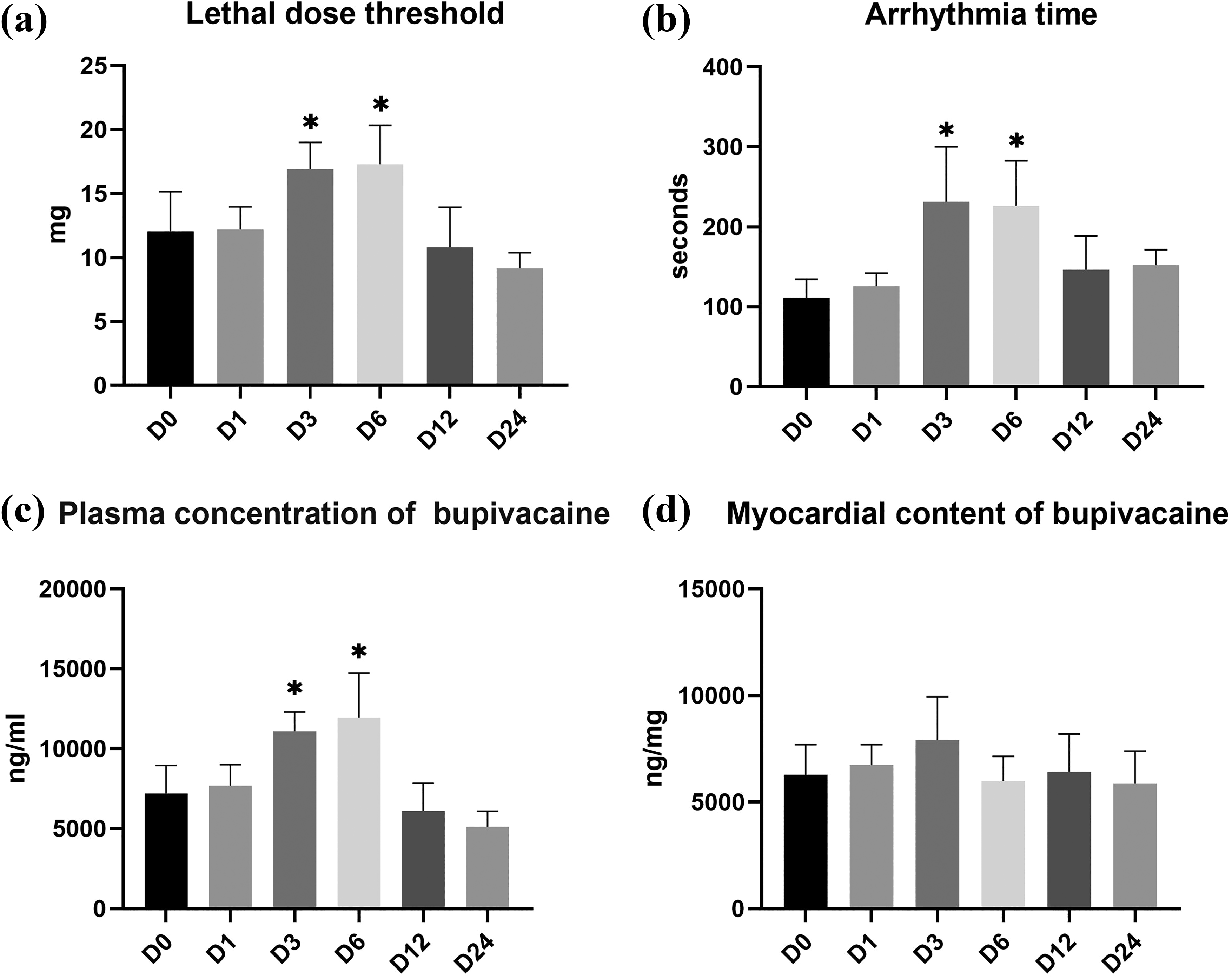
(a–d) Experiment A: lethal dose (a), arrhythmia time (b), the plasma concentration of bupivacaine (c), and myocardial content of bupivacaine (d). Significant differences were demonstrated between the D3, D6, D12, D24, and D0 groups; *p < 0.05 versus the D0 group. (a) D0 versus D3, p = 0.010; D0 versus D6, p = 0.005; (b) D0 versus D3, p = 0.030; D0 versus D6, p = 0.012; (c) D0 versus D3, p = 0.002; D0 versus D6, p < 0.001; (d) No significant differences were demonstrated between the six groups in heart bupivacaine content (p = 0.168).
Arrhythmia time
The arrhythmia times in D3 and D6 groups were longer than in D0 group (D3 vs. D0, p = 0.030; D6 vs. D0, p = 0.012). Compared to D0 group, D1, D12, and D24 groups displayed no significant differences in arrhythmia time (D0 vs. D1, p = 0.917; D0 vs. D12, p = 0.593; D0 vs. D24, p = 0.052). There were no significant differences among D3, D6, D12, and D24 groups in arrhythmia time (p > 0.05; Figure 3(b)).
Plasma concentration of bupivacaine
The plasma concentration of bupivacaine in D3 and D6 groups was higher than D0, D1, D12, and D24 groups (D3 vs. D0, p = 0.002; D3 vs. D1, p = 0.009; D3 vs. D12, p < 0.001; D3 vs. D24, p < 0.001; D6 vs. D0, p < 0.001; D6 vs. D1, p = 0.001; D6 vs. D12, p < 0.001; D6 vs. D24, p < 0.001). Compared to D0 group, D12 and D24 groups displayed no significant differences in plasma concentrations of bupivacaine (D0 vs. D12, p = 0.836; D0 vs. D24, p = 0.239). D3 and D6 groups displayed no significant difference (p = 0.936; Figure 3(c)).
Myocardial content of bupivacaine
There were no significant differences among D0, D3, D6, D12, and D24 groups regarding the myocardial content of bupivacaine (p = 0.168; Figure 3(d)).
Experiment B
Lethal dose threshold
There was no interaction between DMED and YOH in lethal dose threshold (p = 0.608). The lethal dose threshold in DMED and DMED + YOH groups was higher than the control group (DMED vs. control, p = 0.004; DMED + YOH vs. control, p = 0.004). DMED and DMED + YOH groups displayed no significant differences (p = 1; Figure 4(a)).

(a–d) Experiment B: lethal dose (a), arrhythmia time (b), the plasma concentration of bupivacaine (c), and myocardial content of bupivacaine (d). Significant differences were demonstrated between the DMED, YOH, DMED + YOH, and control groups; *p < 0.05 versus control group. (a) DMED versus control, p = 0.004; YOH versus control, p = 0.887; DMED + YOH vs. control, p = 0.004, DMED versus DMED + YOH, p = 1; (b) DMED versus control, p = 0.003; YOH versus control, p = 0.931; DMED + YOH versus control, p = 0.006, DMED versus DMED + YOH, p = 0.985; (c) DMED versus control, p < 0.001; YOH versus control, p = 0.822; DMED + YOH versus control, p = 0.021, DMED versus DMED + YOH, p = 1; (d) No significant differences were demonstrated between the four groups in myocardial bupivacaine content (p = 0.201). DMED: dexmedetomidine; YOH: yohimbine.
Arrhythmia time
There was no interaction between DMED and YOH in arrhythmia time (p = 0.512). The arrhythmia time in DMED and DMED + YOH groups was longer than the control group (DMED vs. control, p = 0.003; DMED + YOH vs. control, p = 0.006). Compared to the control group, the YOH group did not prolong the arrhythmia time (YOH vs. control, p = 0.931). DMED and DMED + YOH groups displayed no significant differences (p = 0.985; Figure 4(b)).
Plasma concentration of bupivacaine
There was no interaction between DMED and YOH in the plasma concentration of bupivacaine (p = 0.799). The plasma concentration of bupivacaine in DMED and DMED + YOH groups was higher than the control group (DMED vs. control, p = 0.02; DMED + YOH vs. control, p = 0.021). Compared to the control group, the YOH group displayed no significant differences in plasma bupivacaine concentration (p = 0.985). DMED and DMED + YOH groups displayed no significant differences (p = 1; Figure 4(c)).
Myocardial content of bupivacaine
There was no interaction between DMED and YOH in myocardial content of bupivacaine (p = 0.475). There were no significant differences among control, DMED, YOH, and DMED + YOH groups in the myocardial content of bupivacaine (p = 0.201; Figure 4(d)).
Discussion
In the current study, doses of 3 and 6 μg kg−1 DMED compared to 0, 1, 12, and 24 μg kg−1 significantly increased the lethal dose threshold of bupivacaine-induced cardiotoxicity and elevated the plasma concentration of bupivacaine at the time of the cardiac arrest. The fact that DMED doses of 3–6 μg kg−1 elevated the lethal dose threshold of bupivacaine and there was no involvement of alpha-2 adrenoceptors is noteworthy.
Kotelko et al. 14 used the widening of the QRS complex to represent the arrhythmia of local anesthetic cardiac toxicity. However, we employed the first dropped beat as the arrhythmia time for the following reasons: compared to the widening of the QRS complex, the occurrence of a dropped beat occurred later than the widening of the QRS and was more representative of impending significant hemodynamic changes; a dropped beat was easily identified and eliminated the potential for error in the initial attempt to recognize a widened QRS complex. In our study, a DMED dose of 3 μg kg−1 prolonged the time to a bupivacaine-induced dropped heartbeat, and this effect was not antagonized by YOH.
Pretreatment with 10 μg kg−1 DMED delays the effects of bupivacaine-induced cardiotoxicity, as reported by Hanci et al. 11 We demonstrated that a DMED dose of 3–6 μg kg−1 delays the cardiotoxic effect of bupivacaine in rats, that is, increasing the lethal dose threshold of bupivacaine. The reasoning as to why larger doses were not protective in experiment A was that large doses of DMED were inherently harmful to hemodynamics. 15 The effect of DMED elevate lethal dose of bupivacaine at 12–24 µg kg−1 may theoretically exist. However, with the increase of dose, the negative effect of DMED on blood pressure and HR is more obvious, which may counteract the effect of reducing the toxicity of bupivacaine.
DMED is a highly selective alpha-2 adrenoceptor agonist. Our research demonstrated that the bupivacaine lethal dose threshold was not antagonized by YOH, thereby demonstrating that alpha-2 adrenoceptors were not involved. There were also some effects induced by DMED which were not via alpha-2 adrenoceptors. For instance, DMED reduced the peak amplitude of compound action potentials, thus extending the duration of peripheral nerve conduction blockade. 16 We found 3–6 μg kg−1 of DMED allowed a higher plasma concentration of bupivacaine than other doses, whereas there were no differences among all groups in the content of bupivacaine, which means each rat had the same tolerance to bupivacaine. Buhrer et al. 17 reported that the dose-sparing effect of DMED can be explained by a DMED-induced decrease in distribution volume and clearance without a major pharmacodynamic interaction. However, in this study, 3–6 μg kg−1 of DMED demonstrated better hemodynamic performance, which suggests that DMED probably delayed the distribution of bupivacaine. As DMED shows the effects of delaying the distribution of lidocaine and mepivacaine in local tissue, 18,19 reducing regional blood flow may play the most important role in decreasing the myocardial content of bupivacaine. Nonetheless, the main mechanism of action is still not clear.
Several shortcomings in our work need mentioning. We could not determine or confirm a mechanism of action in the ability of DMED to mitigate bupivacaine-induced local anesthetic cardiotoxicity. Also, while there are other ways of causing local anesthetic toxicity, such as an overdose, accidental injection into vessels, and regional absorption, we cannot postulate as to whether DMED can be effective in these situations and whether it lessens unsatisfactory outcomes. Future research should (1) delve into the mechanism(s) as to how small dose of DMED pretreatment causes less cardiotoxicity of bupivacaine and (2) initiate exploration into the effect of DMED on other local anesthetics (such as ropivacaine or levobupivacaine) induced cardiotoxicity. In view of the fact that the global volume of surgery has exceeded 300,000,000 cases per annum, 20 we encourage our colleagues to vigorously pursue this line of investigation.
Our study demonstrates that DMED doses of 3–6 μg kg−1 elevated the lethal dose threshold of bupivacaine without involvement of the alpha-2 adrenoceptors. DMED influenced the tissue distribution of bupivacaine, which provide ideas for the prevention and rescue of local anesthetic toxicity.
Supplemental material
Supplemental Material, dropped_beat - Dexmedetomidine elevates the lethal dose threshold of bupivacaine in rats: A dosing study
Supplemental Material, dropped_beat for Dexmedetomidine elevates the lethal dose threshold of bupivacaine in rats: A dosing study by L Pan, Y Zhang, Y He, Z Chen, S Wang, Y Xia, TJ Papadimos, W Lin and X Xu in Human & Experimental Toxicology
Footnotes
Acknowledgment
We would like to thank Xianqin Wang from Wenzhou Medical University for technical assistance. We should also thank the staff in the writing center of Ohio State University of USA for assistance in manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Beijing, China (Grant No: 81670219) and the Wenzhou Science and Technology Bureau, Zhejiang, China (Grant No: Y20180587).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
