Abstract
Ulcerative colitis (UC) is a relapsing and remitting inflammatory disease of the colon, with an increasing incidence worldwide. 6-Gingerol (6G) is a bioactive constituent of Zingiber officinale, which has been reported to possess various biological activities. This study was designed to evaluate the role of 6G in chronic UC. Chronic UC was induced in mice by three cycles of 2.5% dextran sulfate sodium (DSS) in drinking water. Each cycle consisted of 7 days of 2.5% DSS followed by 14 days of normal drinking water. 6G (100 mg/kg) and a reference anti-colitis drug sulfasalazine (SZ) (100 mg/kg) were orally administered daily to the mice throughout exposure to three cycles of 2.5% DSS. Administration of 6G and SZ significantly prevented disease activity index and aberrant crypt foci formation in DSS-treated mice. Furthermore, 6G and SZ suppresses immunoexpression of tumor necrosis factor alpha, interleukin-1β, inducible nitric oxide synthase, Regulated on activation, normal T cell expressed and secreted (RANTES), and Monocyte chemoattractant protein-1 (MCP-1) in the DSS-treated mice. 6G effectively protected against colonic oxidative damage by augmenting the antioxidant status with marked decrease in lipid peroxidation levels in DSS-treated mice. Moreover, 6G significantly inhibited nuclear factor kappa B (P65), p38, cyclooxygenase-2, and β-catenin whereas it enhanced IL-10 and adenomatous polyposis coli expression in DSS-treated mice. In conclusion, 6G prevented DSS-induced chronic UC via anti-inflammatory and antioxidative mechanisms and preservation of the Wnt/β-catenin signaling pathway.
Introduction
Ulcerative colitis (UC) is an inflammatory disease of the colon with multifactorial etiology. 1 It is characterized by relapsing and remitting inflammation of the rectum or the entire colon. 2 The mortality rate and incidence of UC is increasing worldwide. 3 Patients with UC suffer morbidity and poor quality of life from symptoms including body weight loss, colonic mucosal ulceration, diarrhea, and rectal bleeding. 4 Several studies have reported the involvement of altered cytokines and chemokines profiles, gut dysbiosis, and oxidative stress in the pathogenesis of UC in human and experimental animals. 5 –7 Although there are advances in armamentarium for the treatment and management of UC, most drugs used for UC treatment are expensive with the potential to induce serious adverse effects when used over a long period of time. 8 Therefore, alternative pharmacological approaches are needed. Various phytocompounds have been demonstrated to amiolerate UC, but their success is limited because most of these phytocompounds were not repeatedly tested on acute and chronic UC. 8,9
Zingiber officinale (Ginger) is a spice with a successful folkloric usage in the treatment of various inflammatory diseases and gastrointestinal disorders. 10,11 It possesses phenolic compounds such as gingerols, shogaols, and paradols that are responsible for its anti-inflammatory, antioxidant, and anticancer activities. 12,13 6-Gingerol (6G) is the most pharmacological active compound of ginger and it has been reported to modulate various inflammatory diseases. 12 Additionally, previous studies from our laboratory and others have demonstrated the attenuating effects of 6G in short-term dextran sulfate sodium (DSS)-induced acute UC in mice. 14,15 Specifically, 6G elicited a site specific modulation of acute UC via reduction of oxidative stress indices, enhancement of endogenous antioxidants, suppression of systemic tumor necrosis factor alpha (TNF-α), and interleukin-1β (IL-1β). 14 However, information on the effects of 6G on the molecular mechanisms associated with the development chronic UC is lacking. Thus, we hypothesized that long-term administration of 6G may be beneficial in chronic UC. In the present study, we evaluated the influence of 6G on chronic UC in mice by assessing biomarkers of oxidative stress, inflammation, and Wnt/β-catenin signaling.
Materials and methods
Chemicals
DSS (molecular weight 37–40 kD) was procured from TdB Consultancy (Uppsala, Sweden); anti-RANTES, anti-MCP-1, anti-TNF-α, anti-cyclooxygenase-2 (COX-2), antinuclear factor kappa B P65 (NF-κB (P65)), anti-inducible nitric oxide synthase (iNOS), anti-p38 (mitogen-activated protein kinase, MAPK), anti-IL-1β, anti-β-catenin, and anti-adenomatous polyposis coli (APC) mouse monoclonal primary antibodies were purchased from ABCAM, UK; and mouse polyclonal secondary antibodies were purchased from Dako (Agilent Technologies, Santa Clara, USA). All other reagents were of analytical grade and obtained from the British Drug Houses (Poole, Dorset, UK).
Isolation of 6G
Ginger rhizomes were purchased from a local vendor in Bodija Market, Ibadan, Nigeria. The plant samples were authenticated by Mr. Dunatus Esimekhuai at the Department of Botany, University of Ibadan, Ibadan, where a sample (voucher specimen number UIH-22390) was deposited in the herbarium. 6G was isolated from the ginger rhizomes and characterized according to the standardized method described by Ajayi et al. 14 —the purity of the isolated 6G was 91%.
Animal husbandry
Thirty-six adult male BALB/c mice weighing 21–24 g were purchased from the Faculty of Veterinary Medicine, University of Ibadan, Nigeria. They were housed in plastic cages and allowed to acclimatize for a week before the commencement of the experiment. The animals were allowed access to standard mice feed and drinking water ad libitum, and subjected to natural photoperiod of 12 h light: 12 h dark cycle. The experimental protocols were carried out after approval and in accordance with the guidelines set by the University of Ibadan Ethical Committee.
Experimental design
The study consisted of six groups of six mice each. Group I (control) mice received corn oil orally at 2mL/kg for 63 days. Group II (DSS only) mice were maintained on normal mice chow for the first 14 days before exposure to three cycles of 2.5% DSS (each cycle is define as 7 days of 2.5% DSS followed by 14 days of normal drinking water). Group III and Group IV mice were orally treated daily with 100 mg/kg of 6G and sulfasalazine (SZ), respectively, throughout exposure to the three cycles of 2.5% DSS. Group V mice received 100 mg/kg of 6G only whereas Group VI mice received 100 mg/kg of SZ only throughout the experiment. All the mice were sacrificed 24 h after the last treatment. The colons were immediately excised, flushed with ice-cold phosphate-buffered saline (PBS), and measured longitudinally. Subsequently, the colons were blotted with filter paper, weighed, and processed for biochemical estimations, immunohistochemistry, and histology.
Assessment of disease activity index
The severity of UC was assessed according to the method described by Cooper et al. 16 by scoring the colon length, the body weight loss, stool consistency, and rectal bleeding. Stool consistency was scored 0 for normal, 2 for loose, and 4 for diarrhea, whereas rectal bleeding was scored 0 for normal, 2 for occult bleeding, and 4 for gross bleeding.
Histology
Colon tissues were fixed in phosphate buffered formalin for 20 h. The tissues were subsequently dehydrated in an increasing concentration of alcohol, cleared twice in xylene with bench top tissue processor (Leica TP102, Illinois, USA), and fixed in paraffin using Leica EG1150H paraffin embedding station (Illinois, USA). Five micrometers of the embedded tissues were sectioned and stained with hematoxylin and eosin. Microanatomy of colon tissue was examined under a light microscope (Nikon Diaphot, New york, USA) and photomicrographs were captured using a digital camera attached to it (Canon D50, New york, USA). Histological assessment was done in a blinded fashion to avoid bias. Histological damage was quantitatively assessed as described by Cooper et al., 16 with slight modifications for the following measures: crypt architecture (normal = 0, severe crypt distortion = 2, severe crypt distortion with loss of entire crypts = 3); extent of inflammatory cell infiltration (normal = 0, slightly dense inflammatory infiltrate = 2, dense inflammatory infiltrate = 3); goblet cell depletion (present = 1, absent = 0); and crypt abscess (absent = 0, present = 1). Total histological damage score for a mouse colon is the sum of each individual score.
Aberrant crypt foci
Aberrant crypt foci were determined in whole mount colon tissues according to the method described by Bird. 17 Briefly, colons were excised, cut opened longitudinally, rinsed with PBS, flattened with the mucosal side facing up, and fixed in phosphate-buffered formalin. The colons were further stained with 0.2% methylene blue and the mucosal surface was examined under a light microscope (Nikon Diaphot, USA) and photomicrographs were captured using a digital camera attached to it (Canon D50, USA).
Assessment of biomarkers of inflammation and oxidative stress
Whole colons were homogenized in ice-cold 0.1M phosphate buffer (pH 7.4) using a teflon homogenizer (Thomas Scientific, Swedesboro, New Jersey, USA), the homogenates were centrifuged at 10,000g for 15 min at 4°C (Thermo Scientific Sorvall™ WX Floor Ultra Centrifuges, Massachusetts, USA), and the supernatant was collected for biochemical estimations. Reduced glutathione (GSH) was determined according to the method of Jollow et al., 18 and superoxide dismutase (SOD) activity was determined according to the method of Misra and Fridovich. 19 Catalase (CAT) activity was determined using hydrogen peroxide as a substrate according to the method of Clairborne. 20 Glutathione peroxidase (GPx) activity was determined according to the method of Rotruck et al., 21 glutathione-S-transferase (GST) was determined according to the method of Habig et al., 22 and lipid peroxidation was quantified as malondialdehyde (MDA) according to the method described by Farombi et al., 23 and expressed as micromoles MDA/mg protein. Nitric oxide (NO) level was determined by measuring the colonic nitrites content, the stable end products of NO. Colonic nitrites content was obtained using a sodium nitrite curve as standard and expressed as μM of nitrites/mg protein as described by Green et al. 24 Myeloperoxidase (MPO) activity was determined in the colon according to the method of Granell et al. 25 MPO activity was expressed as μM H2O2/min/mg protein.
Immunohistochemistry
Immunohistochemistry for TNF-α, COX-2, NF-κB (P65), iNOS, p38, IL-1β, IL-10, RANTES, MCP-1, β-catenin, and APC was performed on 5-µm-thick paraffin-embedded colon tissues sections. The sections were rehydrated in xylene and decreasing concentration of ethanol. Heat-induced epitope retrieval was performed in citrate buffer (pH 6.0) for 20 min followed by immersion in cold water for 10 min. The colon tissue sections were marked with PAP (hydrophobic barrier) pen and the endogenous peroxidase activities in the tissues were blocked with 5% hydrogen peroxide (H2O2) for 5 min in a dark cupboard. The colon tissue sections were subsequently incubated overnight at 4oC with primary monoclonal antibodies in the following dilutions: anti-TNF-α (1/200), anti-COX-2 (1/200), anti-NF-κB (P65) (1/100), anti-iNOS (1/100), anti-p38 (1/1000), anti-IL-1β (1/1000), anti-IL-10 (1/100), anti-RANTES (1/200), anti-MCP-1 (1/100), anti-β-catenin (1/500), and anti-APC (1/100). The slides were thereafter washed with tris buffer saline and further incubated with horse-radish peroxidase labeled anti-mouse polyclonal secondary antibodies (Dako, Agilent Technologies, USA). Antigen–antibody complexes were visualized by adding 0.05% 3, 3-diaminobenzidene on the colon tissue sections countered stained with hematoxylin. The slides were examined under a light microscope (Nikon Diaphot, USA) and photomicrographs were captured using a digital camera attached to it (Canon D50, USA).
Statistical analysis
Data were analyzed using one-way analysis of variance (ANOVA) to compare the various experimental groups followed by Bonferroni’s test to identify significantly different groups (SPSS for Windows, version 17). Values of p < 0.05 were considered significant.
Results
6G reduces phenotypic clinical features of chronic UC
The effects of 6G on the phenotypic clinical features of chronic UC are presented in Figures 1 and 2. At the beginning of the second cycle of DSS administration, mice in the DSS only group developed diarrhea with rectal bleeding that cease upon withdrawal of DSS and reoccurred at the beginning of the third exposure to DSS (Figure 2(a)). There was a significant (p < 0.05) reduction in colon length and colon wet weight in the DSS-exposed mice when compared with other treatment groups (Figure 2(b) to (d)). Administration of 6G or SZ prevented DSS-induced rectal bleeding and diarrhea, and significantly minimized the rate of reduction in body weight, colon weight, and colon length in mice.

Disease activity index.

Influence of 6G and SZ in the development of clinical features of chronic UC. Gross appearance of anus with rectal bleeding (a), decreased colon wet weight (b), shortened colon length (c), and colon length (cm) (d) in mice exposed to DSS alone. Mice administered with 6G or SZ showed significant amelioration in these phenotypic clinical features of chronic UC. Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; SZ: sulfasalazine; DSS: dextran sulfate sodium; UC: ulcerative colitis.
6G ameliorates oxido-inflammatory stress indices in DSS-induced UC
In order to determine the effects of 6G on the level of oxido-inflammatory stress indices in chronic UC, MDA levels, MPO activity, NO concentration, and endogenous antioxidant status were assessed in colonic mucosal homogenate. The effects of 6G on oxidative stress in DSS-induced chronic colitis are presented in Figures 3(c), 3(d), and 4. The MDA level was significantly (p < 0.05) elevated following DSS administration when compared to other treatment groups (Figure 3(d)). This marked elevation of MDA was reduced significantly in mice treated with 6G or SZ prior to DSS exposure. Mice treated with SZ or 6G alone showed no significant changes when compared with the control. The MPO activity and NO concentration in colons of mice administered DSS only were significantly higher as compared to all the treated groups. However, administration of 6G or SZ significantly reduced the MPO activity and NO concentration toward the control values in the DSS-treated mice (Figure 3(a) and (b)). There were significant decreases in SOD and CAT activities in colon of mice treated with DSS alone when compared with other treatment groups (Figure 4(a) and (b)). Administration of 6G or SZ prevented DSS-mediated decrease in SOD and CAT activities; however, these were significant only in the group pretreated with 6G. Mice treated with SZ and 6G alone showed no significant change in SOD and CAT activities when compared with the control.
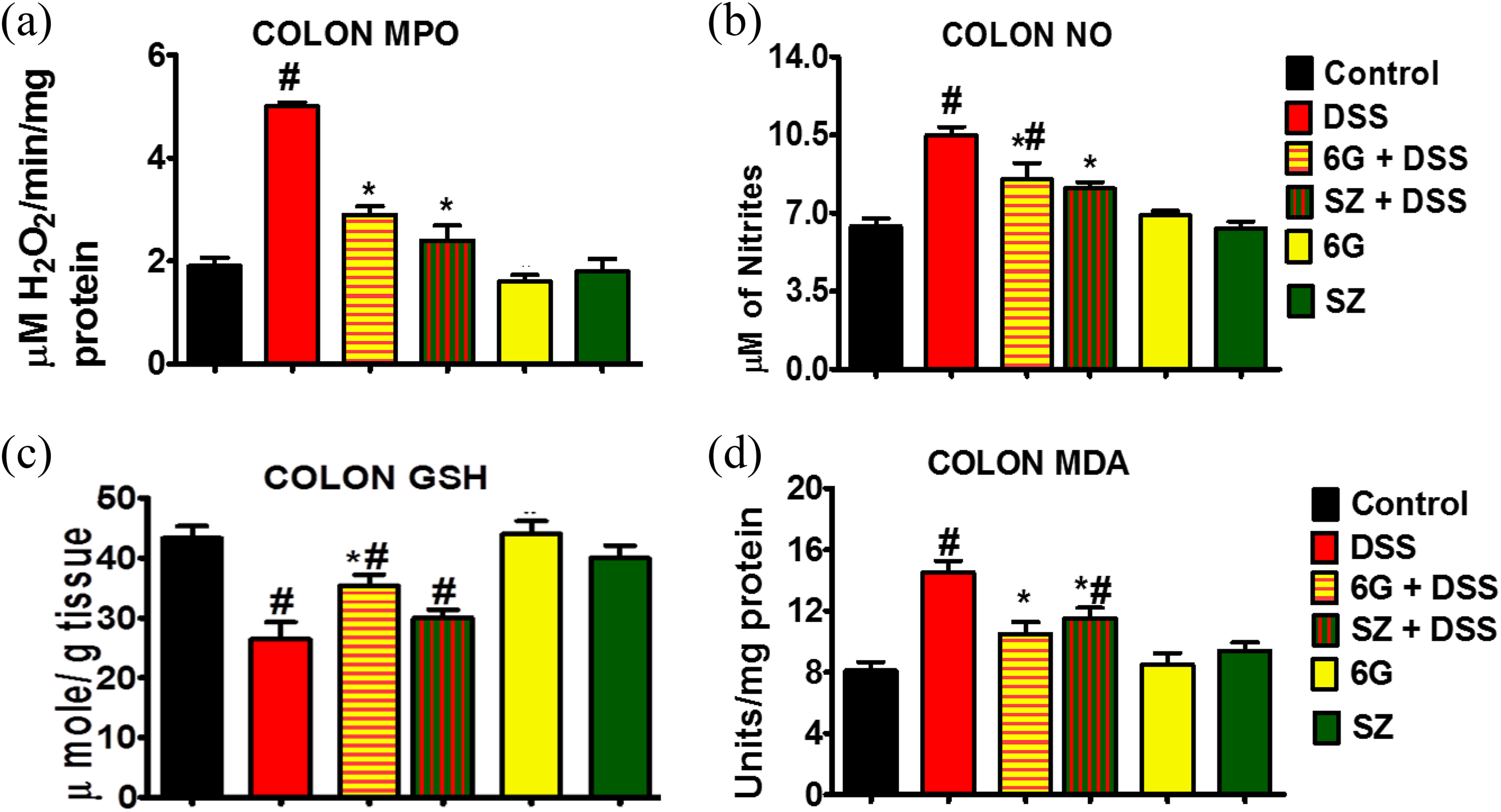
Effects of 6G on oxidative and inflammatory stress indices in mice chronically exposed to DSS. (a) MPO activity, (b)NO concentration, (c) reduced glutathione (GSH) level, and (d) malondialdehyde level. Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; DSS: dextran sulfate sodium; MPO: myeloperoxidase; NO: nitric oxide; GSH: glutathione;

Effects of 6G on free radical scavenging (antioxidant) enzymes in mice chronically exposed to DSS. (a) CAT activity, (b) superoxide dismutase (SOD, (c) GPx, and (d) glutathione-S-transferase activities. Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; DSS: dextran sulfate sodium; CAT: catalase activity; SOD: superoxide dismutase; GPx: glutathione peroxidase.
Oxidative stress burden was further supported by significant decrease in reduced GSH and GSH-dependent enzymes, namely, GPx and GST, in the colon of mice exposed to DSS alone when compared with other treatment groups (Figures 3(c), 4(c), and 4(d)). However, 6G pretreatment significantly increased the activities of GST and GPx as well as GSH level in the DSS-treated mice when compared to DSS alone group.
6G ameliorates histopathological damages in DSS-induced chronic colitis
Histological examination revealed loss of goblet cells, distorted crypts, infiltration of inflammatory cells, and severe colonic mucosal damage with a significant increase in histological damage score in DSS-exposed mice when compared to other treatment groups (Figure 5(a) and (b)). However, 6G or SZ pretreatment prior to DSS challenge significantly ameliorated histological damage score by decreasing colonic neutrophil infiltration and mucosal ulceration. The colon histology of mice treated with 6G alone or SZ alone were normal and comparable to control.

Representative histological signatures of colons of experimental mice. Hematoxylin and eosin staining showed normal colon architecture with no visible lesion following vehicle, 6G, and SZ treatment. DSS treatment showed infiltration of mononuclear cells, cryptisis, erosion of muscularis mucosae, submucosa, laminal propria with crypt abscesses, total absence of goblet, and absorptive cells. DSS-treated mice administered with 6G at 100 mg/kg showed mild infiltration of mononuclear cell; the colon architecture remains intact. DSS mice administered with SZ 100 mg/kg showed normal histological structure with slight neutrophil infiltration on the mucosa and submucosa of the colon (magnification: ×40). 6G: 6-gingerol; SZ: sulfasalazine; DSS: dextran sulfate sodium.
6G suppresses proinflammatory cytokines and chemokines in DSS-induced chronic colitis
Given the established roles of cytokines (TNF-α, IL-1β, and IL-10) and chemokines (RANTES and MCP-1) in the development of UC, we investigated the influence of 6G and SZ on immunohistochemical expression profiles of the aforementioned cytokines and chemokines. Chronic exposure of mice to DSS alone resulted in significant overexpression of TNF-α, IL-β, RANTES, and MCP-1 in the colons of the treated mice (Figures 6–7). Mice pretreated with 6G or SZ before DSS exposure had a significant reduction in expression of RANTES, MCP-1, TNF-α, and IL-β when compared with mice exposed to DSS alone. The expression pattern of these cytokines and chemokines in mice treated with 6G alone and SZ alone were comparable to control. There was a significant reduction in the expression of IL-10 in mice exposed to DSS alone, whereas pretreatment with 6G or SZ prior to DSS challenge increased IL-10 expression (Figure 8).
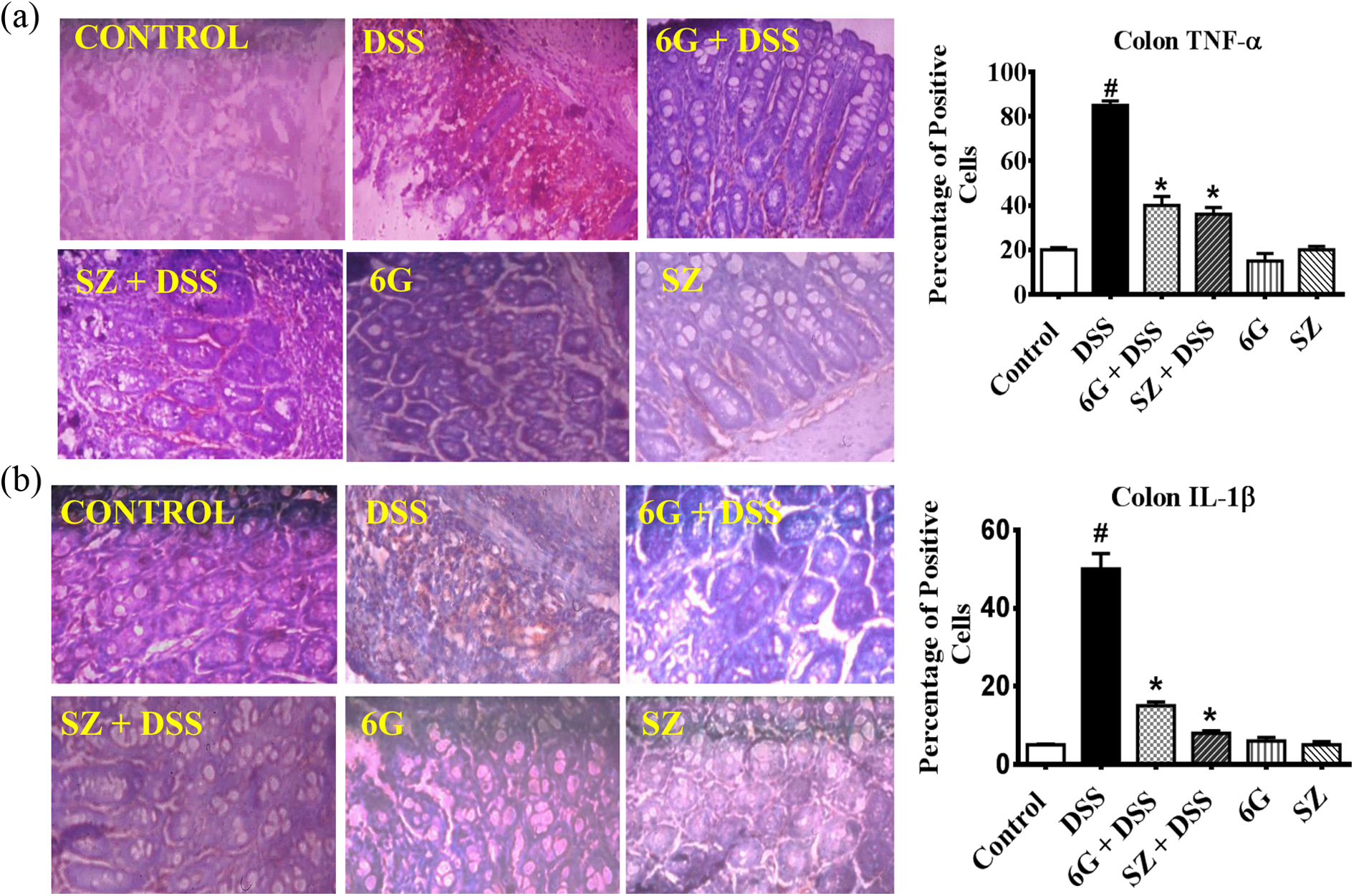
Influence of 6G on colonic (a) TNF α and (b) IL-1β protein expression TNF α and IL-1β expression in mice exposed to DSS. Brown color indicates positive staining for TNF-α and IL-1β antibody on 5-μm section of paraffin-fixed mice colon (magnification: ×100). Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; TNF α: tumor necrosis factor alpha; IL-1β: interleukin-1β.

Influence of 6G on colonic (a) Monocyte chemoattractant protein-1 (MCP-1) and (b) regulated on activation, normal T cell expressed and secreted (RANTES) protein expression in mice exposed to DSS. Brown color indicates positive staining for MCP-1 and RANTES antibody on 5-μm section of paraffin-fixed mice colon (magnification: ×100). Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; DSS: dextran sulfate sodium.
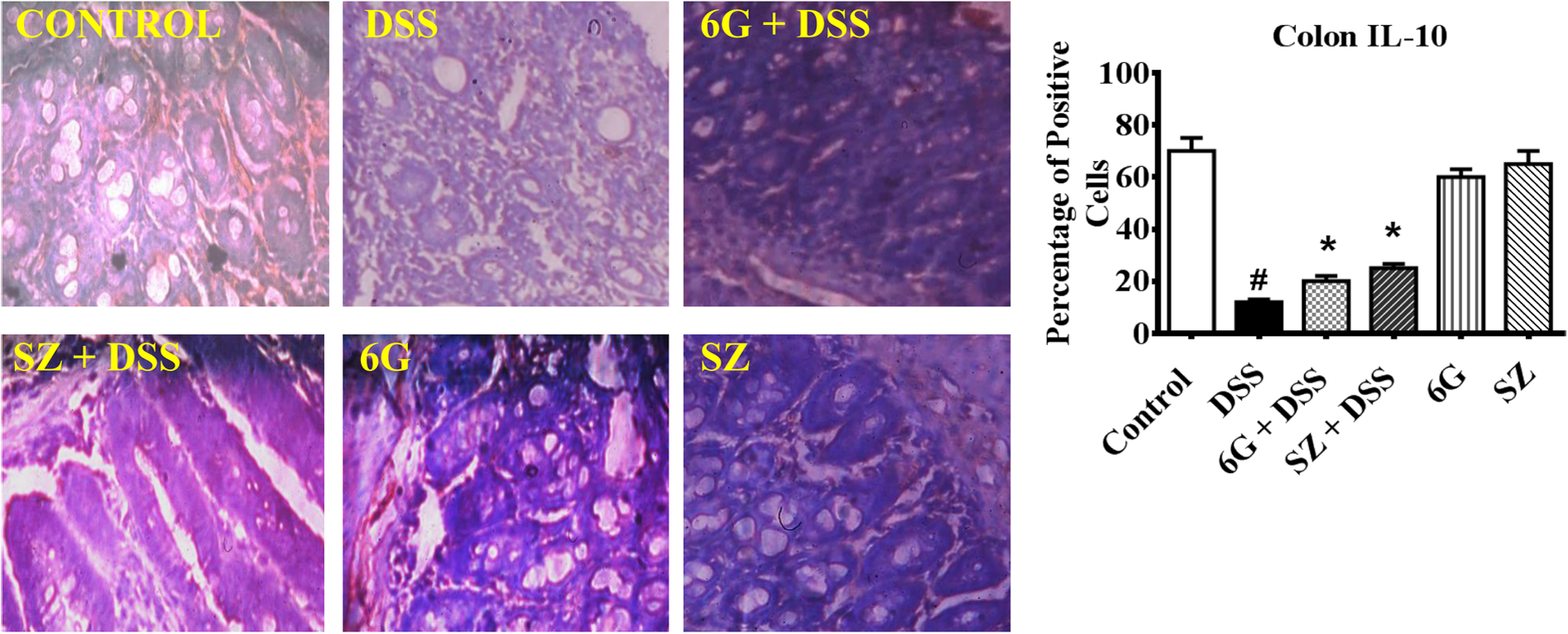
Influence of 6G on colonic IL-10 protein expression in mice chronically exposed to DSS. Brown color indicates positive staining for IL-10 antibody on 5-μm section of paraffin-fixed mice colon (magnification: ×100). Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; DSS: dextran sulfate sodium; IL: interleukin
6G suppresses NF-κB (P65), p38, and iNOS expression in DSS-induced chronic colitis
Further to the suppression of pro-inflammatory cytokines expression by 6G in DSS-exposed mice, we analyzed some of their cellular targets such as NF-κB (P65), p38, and iNOS that catalyze the production of nitric oxide. Figures 9 and 10(b) depict the protein expression of NF-κB (P65), p38, and iNOS in the colons of treated mice. The result showed that the expression of NF-κB (P65), p38, and iNOS were comparable to control in mice administered SZ alone and 6G alone. However, the colons of DSS-exposed mice had increased expression of these proteins when compared with corresponding controls. Interestingly, treatment with 6G or SZ significantly prevented DSS-mediated increase in the expression of these proteins.

Effects of 6G on the protein expression of transcription factor (a) NF-κB p65 and (b) stress stimuli response protein p38in colons of mice chronically exposed to DSS. Brown color indicates positive staining for NF-κB (P65) and p38 antibody on 5-μm section of paraffin-fixed mice colon (magnification: ×100). Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; DSS: dextran sulfate sodium; NF-κB: nuclear factor kappa B.

Effects of 6G on colonic COX-2 (a) and iNOS (b) protein expression in mice chronically exposed to DSS. Brown color indicates positive staining for COX-2 and iNOS antibody on 5-μm section of paraffin-fixed mice colon (magnification: ×100). Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; COX-2: cyclooxygenase-2; DSS: dextran sulfate sodium.
6G ameliorates COX-2 and β-catenin whereas it enhances APC expression in DSS-induced chronic colitis
It is well recognized that β-catenin, a putative regulator of COX-2, plays a crucial role in chronic inflammation. The nuclear translocation of β-catenin is prevented by binding of APC. The results showed a more intense expression of β-catenin and COX-2 whereas a weaker expression of APC was observed in the colons of DSS-exposed mice when compared with all treatment groups (Figures 10(a) and 11). However, administration of 6G or SZ prior to DSS exposure decreased β-catenin and COX-2 expression and increased APC expression in the colon as compared to mice exposed to DSS alone. Mice administered SZ alone and 6G alone exhibited a similar pattern of β-catenin, APC, and COX-2 expression with the control group (Figures 10(a) and 11).

Effects of 6G on colonic β-catenin (a) and APC (b) protein expression in mice chronically exposed to DSS. Brown color indicates positive staining for β-catenin and APC antibody on 5-μm section of paraffin-fixed mice colon (magnification: ×100). Each bar represents mean ± SD of six mice. #: Values differ significantly from control (p < 0.05). *: Values differ significantly from DSS group (p < 0.05). 6G: 6-gingerol; APC: adenomatous polyposis coli; DSS: dextran sulfate sodium.
6G inhibits aberrant crypt foci formation in DSS-induced chronic colitis
There was substantial increase in aberrant crypt foci in the colon tissues of DSS-exposed mice (Figure 12). However, there was a marked decrease in number of aberrant crypt foci in mice that received DSS after 6G or SZ treatment. There was no aberrant crypt foci seen in control and mice treated with 6G alone and SZ alone.

Photomicrographs of whole colon tissue stained with methylene blue for detection of preneoplastic lesion in mice distal colon. Colons of mice exposed to DSS alone showed an intense darkly stained aberrant crypt with thicker epithelial lining and a larger pericryptal zone (red arrow). Normal crypt was observed in control and groups treated with SZ alone and 6G alone. DSS-treated mice administered with 6G or SZ showed normal histological structure but with reduced number of aberrant foci (red arrow) (magnification: × 100) DSS: dextran sulfate sodium; SZ: sulfasalazine; 6G: 6-gingerol.
Discussion
UC is a form of inflammatory bowel disease mediated by dysfunctional colonic mucosal immunity and results to acute inflammation that progresses to chronic repeated episodes of inflammation with higher relapse rate that can result into colorectal cancer if untreated. 26 Several drugs including steroids and biologics are used for the treatment of UC; the drawbacks to these therapies are the exorbitant cost and their serious side effects. 27,28 Most research into the prevention of UC has been directed toward the use of nontoxic plant phytocompounds that modulate colonic immunity and redox signaling via antioxidative and anti-inflammatory mechanisms. 29 –31 We have previously demonstrated the pharmacological effect of 6G in acute colitis through short-term administration of DSS to mice. 14 The effect of long-term administration 6G on chronic colitis was however demonstrated in this study to further elucidate its potential role in relapsing and remitting form of the disease.
Aberrant colonic oxido-inflammatory response which ablates the mucosal that coalesces forming ulcers in the colon are the hallmark of UC. 32,33 The sloughing of this abscesses results in diarrhea and rectal bleeding. 34 As these abscesses heal through acute inflammatory response, the inner muscle layer of the colon undergoes scarring and thickens. Granulation tissue replaces scarred and thickened muscle that resulted from healing and the colon becomes narrowed and shortened. 35 Disease activity index (DAI) is a combined score of body weight loss, stool consistency, and rectal bleeding. Increased DAI score has been reported to correlate with the progression of UC. 36,37 In the present study, DSS-exposed mice presented a significantly high DAI score with reduced colon length. Pretreatment with 6G or SZ delayed in UC progression by reducing DAI score and colon shrinkage in the DSS-treated mice.
The gold standard for UC treatment involves restoration of damage mucosal to normal state. 38 Histomorphometry studies objectively assess the reversion of a damage colonic mucosal to the normal state. 39 The histological damage induced by DSS in animals mirrors those seen in patients with active UC. 40,41 In this study, we observed goblet cell depletion, crypt erosion, infiltration of neutrophils in the lamina propria, and submucosa of mice chronically exposed to DSS alone. These features were consistent with the previous report on DSS-induced UC. 42 The 6G or SZ treatment prevented colon histological architecture damage by reducing the levels of neutrophils infiltration and crypt depletion.
Neutrophils are responsible for additional production of reactive oxygen and nitrogen species that overwhelm the antioxidative capacity of the colonic milieu and subsequently contribute to oxidative damage. 31 Overproduction of reactive oxygen and nitrogen species decreased antioxidant levels and the consequent oxidative stress was reported in the inflamed colonic mucosal of UC patients and animal models. 14,43,44 In this study, oxidative stress was confirmed in DSS-exposed mice by marked decrease in activities of antioxidant enzymes, namely, SOD, CAT, GPx, and GST as well as GSH level. The diminution in colonic antioxidant status was accompanied by a significant increase in MDA levels. Several antioxidant compounds have been found to be efficient in clinical trials and animal models of UC. 8,30,31 The significant reduction in the MDA levels in colons of mice treated with 6G confirmed its well-reported antioxidative properties. 12,45 Ability of 6G or SZ to prevent the massive infiltration of neutrophils, crypt depletion, and ablation of the colonic mucosal might be the possible mechanism by which 6G or SZ protects the integrity of the colonic mucosal barrier. In addition, nitrosative stress was also detected in the present model of chronic colitis. This was evident by increased colonic expression iNOS and concentration of NO. The involvement of iNOS in the development of UC has documented the observation that DSS-induced colitis was attenuated in iNOS -/- mice. 46 Therefore, inhibition of iNOS and NO may be beneficial for the management of UC. 47 In this study, significant decrease in MPO activity, NO concentration, and iNOS expression was evident in mice treated with 6G and SZ. Thus, the present data revealed that 6G and SZ possesses the ability to prevent nitrosative stress associated with chronic UC.
Both TNF-α and IL-1β play an integral role in the pathogenesis of UC. High TNF-α and IL-1β levels have been documented in patients with UC. 48 TNF-α and IL-1β are important components in the pathophysiology of UC and biologic agents that suppress TNF-α and IL-1β production have demonstrated therapeutic potential. 49 Overexpression of TNF-α has shown to compromise tight junction, permitting intestinal penetration of luminal antigens, thus promoting inflammation via induction of NF-κB, a known master switch for redox signaling that activates pro-inflammatory proteins following the nuclear translocation of its P65 sub-unit. 50,51 Several lines of evidence have reported a crosstalk between p38 and NF-κB. 52 Activation of p38 under stress conditions has shown to promote oxido-inflammatory stress and induce apoptosis in intestinal cells. 53 Increase in the levels of NF-κB (P65) and p38 has been reported in humans and animal models of UC. 54 In the present study, 6G and SZ treatment reversed the DSS-mediated increase in expression of TNF-α, IL-1β, NF-κB (P65), and p38 protein expression, demonstrating that 6G and SZ protect the colon by regulating the mediators of inflammation and oxidative stress.
The extent of the inflammatory response and upregulation of oxidative burst in both patients and animal models of UC depends on chemokine expression. 55 Aberrant chemoattraction of leucocyte has been reported in UC. 56 Overexpression of MCP-1 has been implicated in the progression to chronic UC. 57 Furthermore, Some clinical and experimental studies on UC have shown the upregulation of MCP-1 and RANTES in colonic mucosal samples and their homeostatic regulation may be a useful therapeutic interventions for UC. 57,58 The observed decrease in the expression of MCP-1 and RANTES in mice treated with 6G and SZ in the present study indicates their ability to suppress progression to chronic UC and possible prevention of aberrant recruitment of leucocytes in the DSS-exposed mice. Episodic colonic inflammation has been linked to the onset of dysplasia and eventual progression to colorectal cancer. 59 Development of aberrant crypt foci are well known to precede dysplasia, colorectal polyps, and cancer. 43 Aberrant crypt foci with accumulated β-catenin expression have been detected in patients and animal models with long-standing UC. 60,26 In this study, 6G or SZ pretreatment preserved the colon from aberrant crypt foci associated with chronic inflammation following DSS exposure.
Wnt signaling is required for various tissue developmental processes. 61 This pathway is critically regulated by APC, glycogen synthase kinase 3 (GSK 3), and β-catenin protein levels. 62 Activation of the pathway occurs by inactivation of APC which allows the nuclear translocation of β-catenin and downstream activation of its targets genes such as lymphoid enhancer factor, T cell factor, and COX-2. 63 The balance between the expression of β-catenin and a member of its destruction complex APC is vital for the maintenance of intestinal homeostasis. 64 Dsyregulation of Wnt/β-catenin pathway resulting from overexpression β-catenin has been reported in both UC and colitis associated colorectal cancer. 65,66 In the present study, exposure to DSS alone was associated with elevated COX-2 and β-catenin expression, whereas APC expression was decreased, thus suggesting the perturbation of Wnt/β-catenin signaling in the exposed mice. The marked reduction in the β-catenin and COX-2 expression along with increased APC expression in colons of mice treated with either 6G or SZ prior to DSS exposure suggests that 6G and SZ may prevent chronic UC via preservation of the Wnt/β-catenin signaling pathway.
The findings from the present study highlight the protective mechanisms of 6G in DSS-induced chronic UC in mice (Figure 13). The suppression of TNF-α, IL-1β, MCP-1, RANTES, COX-2, and iNOS expression through downregulation of NF-κB (P65) could be a mechanism to explain the anti-inflammatory effect of 6G in DSS-induced chronic UC. Moreover, 6G attenuated colon oxidative damage by augmenting cellular antioxidant and inhibiting lipid peroxidation and p38 protein expression. The preservation of the colon architecture and Wnt/β-catenin signaling pathway is attributable to the maintenance of colonic homeostasis by 6G. In conclusion, the anti-inflammatory and antioxidant effects of 6G are comparable to the reference drug SZ, thus suggesting 6G to be a potential natural remedy for the management of chronic UC.
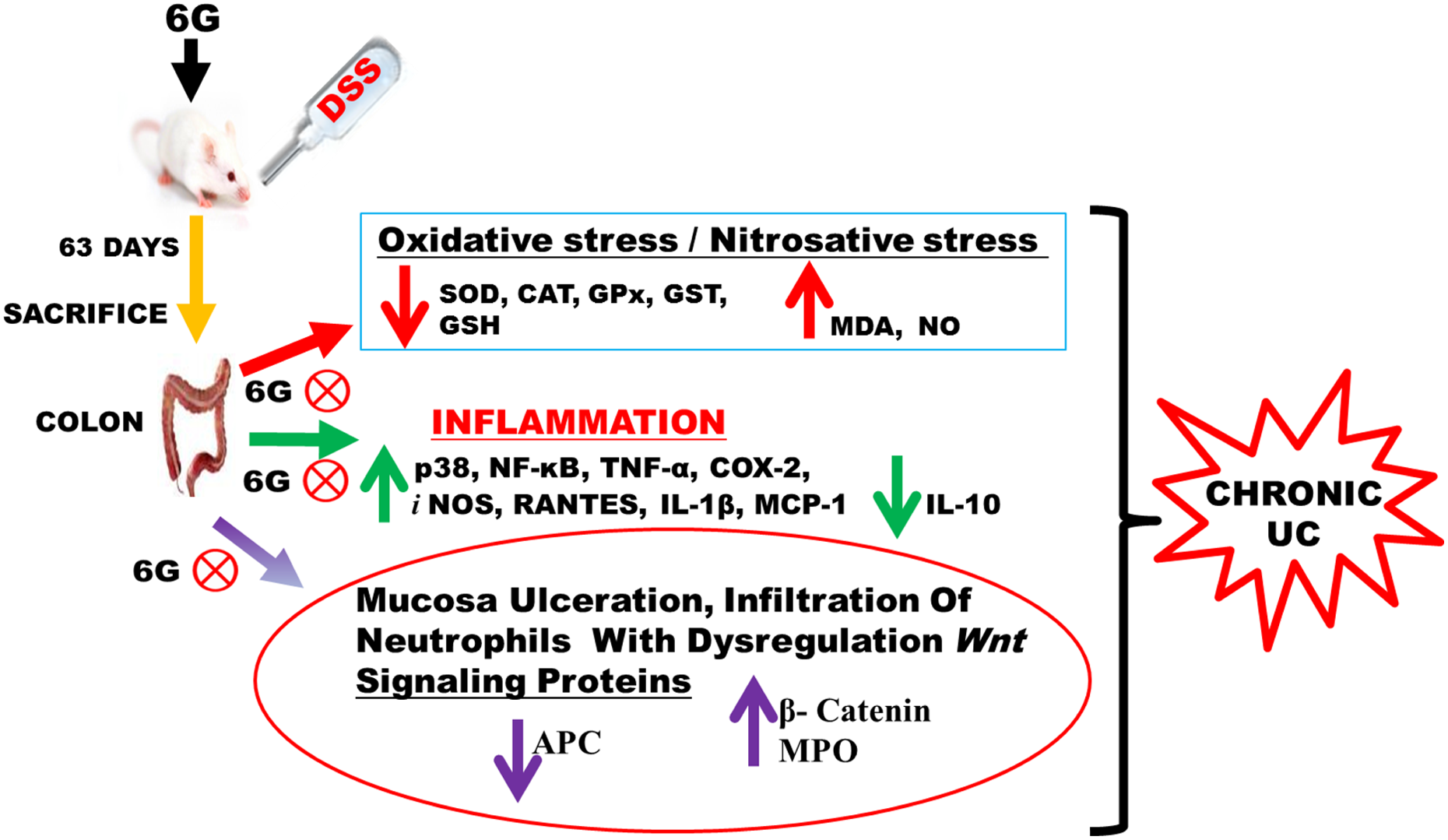
Proposed pathway delineating the cellular and molecular mechanisms of 6G chemoprotection against DSS-mediated chronic colitis in mice. 6G: 6-gingerol; DSS: dextran sulfate sodium.
Footnotes
Acknowledgments
The technical assistance of Dr. Ganiyat K. Oloyede and Mr. C.C. Nwabueze of the Department of Chemistry, University of Ibadan, Ibadan, Nigeria, is appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the TETFUND Institutional Based Research (IBR) 2015 grant awarded to Professor E.O. Farombi.
