Abstract
Mono (2-ethylhexyl) phthalate (MEHP) is the principal metabolite of di (2-etylhexyl) phthalate, which is widely used as a plasticizer, especially in medical devices. MEHP has toxic effects on cardiovascular system. The aim of this study was to investigate the possibility that 6-gingerol may inhibit the oxidative DNA damage of MEHP in human umbilical vein endothelial cells (HUVECs) and the potential mechanism. The comet assay was used to monitor DNA strand breaks. We have shown that 6-gingerol significantly reduced the DNA strand breaks caused by MEHP. MEHP increased the levels of reactive oxygen species and malondialdehyde, decreased the level of glutathione and activity of superoxide dismutase, and altered the mitochondrial membrane potential. In addition, DNA damage-associated proteins (p53 and p-Chk2 (T68)) were significantly increased by the treatment of MEHP. Those effects can all be protected by 6-gingerol. The results firmly indicate that 6-gingerol may have a strong protective ability against the DNA damage caused by MEHP in HUVECs, and the mechanism may relate to the antioxidant activity.
Introduction
Evidence from numerous studies indicates that a high consumption of plant-based foods is associated with a significantly lower risk of some chronic diseases, such as coronary artery disease, stroke, diabetes, and cancer. 1 –3 Those agents have antioxidant and anti-inflammatory activities, which contribute to their chemoprotective potential. 4
Ginger (Zingiber officinale) is a common spice that contributes to the taste and flavor of foods and is also an important traditional Chinese medicine. 5 Its extract and major pungent principles have been reported to possess a variety of biological properties. 6-Gingerol was the major pharmacologically active component of ginger (Figure 1). It has diverse pharmacological and physiological effects, including anticancer, antioxidation, anti-inflammation, anticardiovascular disease, and antiplatelet aggregation. 6 As an antioxidant, the properties of 6-gingerol may be related to its abilities to donate electrons and to act as free radical scavengers by the formation of stable phenoxyl radicals. 7 Therefore, 6-gingerol is recommended for prevention of many diseases.

Chemical structure of 6-gingerol.
Phthalates are a class of synthetic plasticizers found in numerous consumer products worldwide, including packaging materials, cosmetics, and medical devices. 8 Di (2-etylhexyl) phthalate (DEHP) is the most common member of phthalates used as a plasticizer, especially in medical devices, such as blood storage bags, cardiopulmonary bypass circuitry, intravenous tubing and bags, hemodialysis systems, and air tubes. 9 When DEHP enters the human body, mono (2-ethylhexyl) phthalate (MEHP) is the principal active monoester metabolite of DEHP in vivo. 10
Since the human DEHP exposure was universal, many investigations were focused on the potential adverse effects of DEHP and MEHP, on the reproductive system, carcinogenicity, liver, kidney, and developmental toxicities. However, the mechanisms are not totally clear. Many data suggest that MEHP induces oxidative stress by disrupting the activities of antioxidant enzymes, which lead to apoptosis, cell toxicity, and DNA damage. 11 –13 DEHP is extracted from the plastic material when it comes into contact with lipophilic biological fluids, especially blood, and then converted to MEHP. However, reports about the MEHP-induced injury of cardiovascular system are limited. In this study, we investigated the possible toxicity of MEHP on cardiovascular system and tested whether the mechanisms are related to oxidative stress.
Human umbilical vein endothelial cells (HUVECs) are cells derived from the endothelium of veins from the umbilical cord. They have played a major role as a model system for the study of the regulation of endothelial cell function and the role of the endothelium in the response of the blood vessel wall to stretch, shear forces, and the development of atherosclerotic plaques and angiogenesis. 14 In our study, HUVECs were used to study the toxic effects of MEHP on cardiovascular system.
As a naturally occurring plant antioxidant, 6-gingerol was used to prevent the toxicity of MEHP in the present study. Since the increased DNA damage was one of the most common outcome of oxidative stress. The aim of this study was to test whether 6-gingerol could modify MEHP-induced DNA damage in a cell system and to determine whether this modification was caused by reducing MEHP-induced oxidative stress. To further confirm MEHP-mediated DNA damage and the related mechanism, we observed the expression of some DNA damage-related proteins by Western blot method. Besides, mitochondrial membrane potential (ΔΨm) was also tested. ΔΨm is an indicator of mitochondrial function, which is closely related with mitochondrial membrane permeability.
Materials and methods
Materials
6-Gingerol (>98% High Performance Liquid Chromatography [HPLC]) was purchased from Medicass Biotechnologies, Co., Ltd (Beijing, China) and dissolved in dimethyl sulfoxide. MEHP, ethidium bromide, 2,7-dichlorofluoroscein diacetate (DCFH-DA), o-phthalaldehyde (OPT), and rhodamine 123 were purchased from Sigma-Aldrich (St Louis, Missouri, USA). p53 and p-Chk2 (T68) antibodies were purchased from Bioworld Technology Inc. (St Louis Park, Minnesota, USA) β-actin antibody was purchased from Santa Cruz (Delaware Avenue Santa Cruz, California, USA). Goat anti-rabbit secondary antibody and goat anti-mouse secondary antibody were purchased from Thermo Fisher Scientific (Waltham, Massachusetts, USA).
Cell culture and treatment
The HUVECs were obtained from Peking Union Medical College (Peking, China) and cultured in Minimum Essential Medium (MEM) containing 10% fetal bovine serum, penicillin (100 IU/mL), streptomycin (100 μg/mL) in a 5% carbon dioxide atmosphere at 37°C. For each experiment, cells were treated with 6-gingerol (10 μM) for 1 h and then exposed to MEHP (10, 20, and 40 μM) for 24 h. 15,16
Comet assay
The HUVECs (1 × 106 cells) were incubated with 6-gingerol (10 μM) at 37°C for 1 h. Then, MEHP (0, 10, 20, and 40 μM) was added for another 24 h. To avoid artifacts resulting from apoptosis and necrosis, Hoechst 33342 (8 μg/mL) was employed to detect apoptotic cells, and trypan blue (50 μg/mL) was employed to detect cell viability. Cell suspensions without apoptotic cells and with cell viabilities >90% were used for the experiment. The assay was performed as described by Singh and Stephens. 17 Cell suspension was mixed with 1% low-melting agarose and placed on slides prelayered with 1.5% regular agarose. Then, the slides were immersed in lysing solution (2.5 M sodium chloride, 100 mM ethylenediaminetetraacetic acid (EDTA), 10 mM Tris, 1% Triton X-100; pH 10) at 4°C for 1 h. After the lysis, the slides were placed in an alkaline solution (1 mM EDTA and 300 mM sodium hydroxide; pH 13) for 20 min to allow DNA unwinding. Then, they were electrophoresed for 30 min at 200 mA. Finally, the slides were viewed using an Olympus BX-51 fluorescent microscope (Japan; excitation filter 549 nm and barrier filter 590 nm). Images were subject to quantitative analysis with Comet Assay Software Project casp-1.2.2 (University of Wroclaw, Poland).
Measurement of intracellular reactive oxygen species
According to the report of Sohn et al., we assessed intracellular reactive oxygen species (ROS) levels using the DCFH-DA method. 18 The cells were pretreated with 6-gingerol (10 μM) for 1 h, and exposed to MEHP for another 24 h. The cells were harvested and suspended in phosphate-buffered saline (PBS) at 5 × 105 cells/mL. After incubated with DCFH-DA (5 μM) at 37°C for 40 min in the darkness, the relative fluorescence intensity of the cell suspensions was measured using a fluorescence spectrophotometer (Hitachi 650-60; Tokyo, Japan). Excitation and barrier wavelengths were 485 nm and 530 nm, respectively.
Measurement of intracellular glutathione
In brief, the HUVECs were incubated with 6-gingerol (10 μM) at 37°C for 1 h before adding MEHP for 24 h. Then, the cells were harvested, washed twice with PBS, and treated with 5% trichloroacetic acid at 4°C for 30 min to extract glutathione (GSH). Finally, the supernatant was mixed with phosphate EDTA buffer (pH 8.3) and OPT (50 μg/mL) solution at 37°C in dark for 15 min. The relative fluorescence intensity was read at an emission wavelength of 420 nm and an excitation wavelength of 350 nm.
Measurement of malondialdehyde content and super oxide dismutase activity
The HUVECs were pretreated with 6-gingerol (10 μM) at 37°C for 1 h and incubated with MEHP for another 24 h. The level of lipid peroxidation in HUVECs was assessed by malondialdehyde (MDA) analysis kit according to the protocol’s instructions (Jiancheng Biotechnology Co., Nanjing, China). The activity of super oxide dismutase (SOD) was detected by SOD analysis kit according to the protocol’s instructions (Jiancheng Biotechnology Co.).
Mitochondrial membrane potential (ΔΨm) assay
ΔΨm was tested using a modified method. 19 After incubated with 6-gingerol and MEHP, the cells were washed twice with PBS. Rhodamine 123 was used for the estimation of mitochondrial membrane potential in the experiment. The cell pellet was mixed with Rhodamine 123 (1.5 μM) at 37°C in a thermostatic bath with gentle shaking for 10 min. The fluorescence was measured at an emission wavelength of 490 nm and an excitation wavelength of 520 nm.
Western blot analysis
The expression of p53 and p-Chk2 (T68) was evaluated by Western blot analysis. At the end of the designated treatments, the protein of the HUVECs was isolated, and BCA protein assay kit was used in protein concentration measurement according to the manufacturer’s instruction. Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) (12%) and transfer of the separated proteins onto a nitrocellulose membrane (Pall Corp., New York City, New York, USA) were performed using standard procedures. The blockage buffer was used in membrane incubating for several hours to reduce nonspecific bindings. After blocking, the membranes were incubated with appropriate primary antibodies for overnight at 4°C and then incubated with the appropriate secondary antibodies at 37°C for 2 h. β-actin antibody was used as a control. Enhanced chemiluminescence was used in protein visualization according to the manufacturer’s instructions.
Statistical analysis
All values were presented as means ± standard deviation. The data were statistically analyzed using one-way analysis of variance and Student–Newman-Keuls test using SPSS v13.0 software. Values of p < 0.05 and p < 0.01 were considered statistically significant.
Results
Effect of 6-gingerol on the DNA strand breaks induced by MEHP
The comet assay was performed to determine the extent of DNA damage. MEHP caused a significant increase in DNA migration compared with that observed with untreated cells (Table 1). In MEHP group, the DNA strand breaks were found to be dose dependent. The highest DNA damage was observed in the 40 μM group as 27.02 ± 3.87% tail DNA and 6.34 ± 2.12 tail moment. When the cells were pretreated with 6-gingerol for 1 h, the comet tails were evident, and the comet tail moment values were significantly decreased compared with that of only MEHP-treated cells.
Protective effect of 6-gingerol against MEHP-induced DNA strand breaks in HUVECs.a

MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells; SD: standard deviation.
aDNA strand breakages were estimated by the comet assay. HUVECs were pretreated with 6-gingerol (10 μM) for 1 h and then exposed to MEHP for 24 h. Results are the mean ± SD (n = 3).
b p < 0.05: Significant difference from control.
c p < 0.01: Significant difference from control.
d p < 0.05: Significant difference from the group treated with MEHP only.
e p < 0.01: Significant difference from the group treated with MEHP only.
Effects of 6-gingerol on ROS formation induced by MEHP
As shown in Figure 2, the fluorescence intensity of ROS increased in MEHP group (20 and 40 μM) in a dose-dependent manner compared with control group (p < 0.01). However, there was no significant difference between the 10 μM MEHP group and the untreated sample. Pretreatment with 6-gingerol produced a significant reduction of DCF fluorescence in HUVECs (p < 0.01).
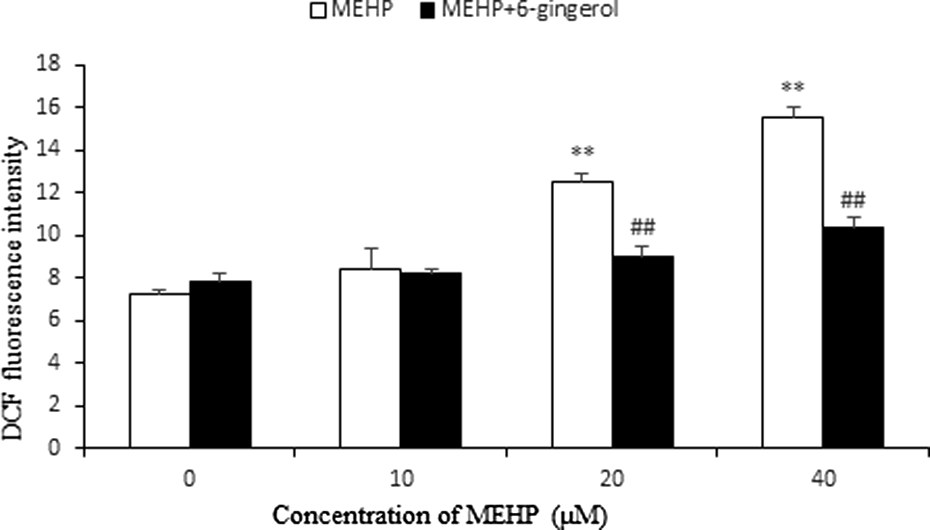
Effect of 6-gingerol on the ROS formation induced by MEHP. HUVECs were pretreated with 6-gingerol (10 μM) for 1 h and then exposed to MEHP for 24 h. Each bar represents mean ± SD of three independent experiments (n = 3). **p < 0.01: significant difference from control and ## p < 0.01: significant difference from the group treated with MEHP only. ROS: reactive oxygen species; MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells; SD: standard deviation.
Effect of 6-gingerol on the GSH depletion induced by MEHP
OPT fluorescence dye was used to evaluate the depletion of GSH. The results (Figure 3) indicate that a striking decrease in intracellular GSH was observed at the MEHP group (10, 20, and 40 μM; p < 0.05). This reduction was significantly and dose dependently prevented by the pretreatment with 6-gingerol (10 μM; p < 0.05).

Effect of 6-gingerol on the GSH depletion induced by MEHP. HUVECs were pretreated with 6-gingerol (10 μM) for 1 h and then exposed to MEHP for 24 h. Each bar represents mean ± SD of three independent experiments (n = 3). *p < 0.05: Significant difference from control and # p < 0.05: significant difference from the group treated with MEHP only. GSH: glutathione; MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells; SD: standard deviation.
Effect of 6-gingerol on MDA and SOD levels after MEHP treatment
The MDA level increased in a dose-dependent manner compared with control group. Pretreatment with 6-gingerol produced a significant reduction of MDA level in HUVECs (p < 0.05; Figure 4). The antioxidant enzyme activity (SOD) is illustrated in Figure 5. Compared with the control group, MEHP-exposed HUVECs possessed significantly less SOD activity, whereas treatment with 6-gingerol effectively upregulated SOD activity (p < 0.05).

Effect of 6-gingerol on the MDA formation induced by MEHP. HUVECs were pretreated with 6-gingerol (10 μM) for 1 h and then exposed to MEHP for 24 h. Each bar represents mean ± SD of three independent experiments (n = 3). *p < 0.05 and **p < 0.01: Significant difference from control; # p < 0.05: significant difference from the group treated with MEHP only. MDA: malondialdehyde; MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells; SD: standard deviation.

Effect of 6-gingerol on the SOD depletion induced by MEHP. HUVECs were pretreated with 6-gingerol (10 μM) for 1 h and then exposed to MEHP for 24 h. Each bar represents mean ± SD of three independent experiments (n = 3). *p < 0.05: Significant difference from control and # p < 0.05: significant difference from the group treated with MEHP only. SOD: superoxide dismutase; MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells; SD: standard deviation.
Effect of 6-gingerol on ΔΨm depletion induced by MEHP
Rhodamine123 as a cationic fluorescent probe has been wildely used for the measurement of ΔΨm. As shown in Figure 6, a statistically significant decrease of Rhodamine 123 fluorescence intensity was observed in cells treated with MEHP (10, 20, and 40 μM) for 24 h (p < 0.05). Pretreatment with 6-gingerol (10 μM) for 1 h produced a significant and dose-dependent increase of Rhodamine 123 fluorescence in the cells (p < 0.05 or p < 0.01).
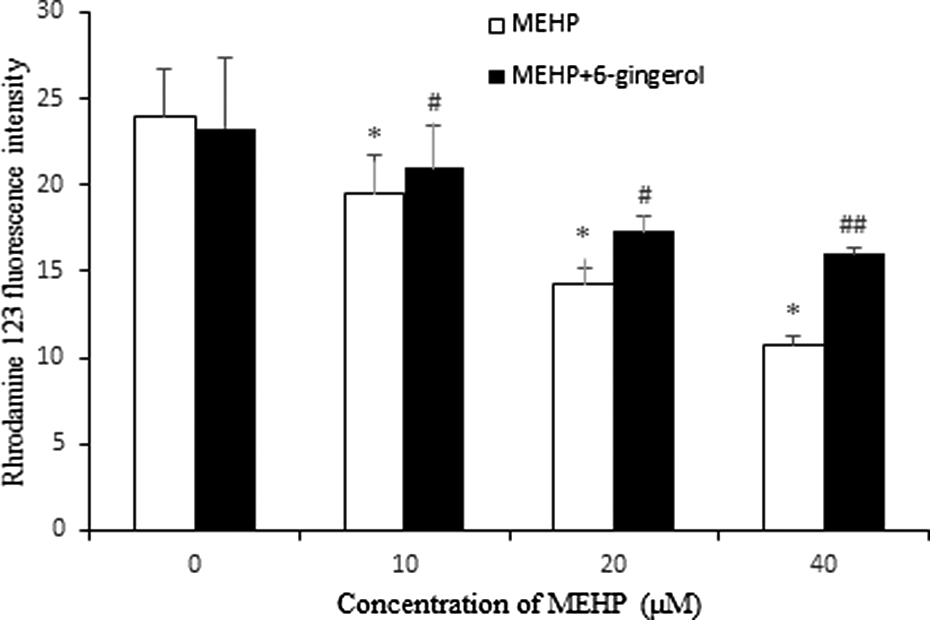
Effect of 6-gingerol on mitochondrial membrane potential. HUVECs were pretreated with 6-gingerol (10 μM) for 1 h, then exposed to MEHP for 24 h, and stained with Rhodamine 123. Each bar represents mean ± SD of three independent experiments (n = 3). *p < 0.05: Significant difference from control; # p < 0.05 and ## p < 0.01: significant difference from the group treated with MEHP only. MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells; SD: standard deviation.
Effect of 6-gingerol on the expression of p-Chk2 (T68) and p53
As illustrated in Figures 7 and 8, the expression of p-Chk2 (T68) and p53 was significantly increased in MEHP-treated cells compared with the control. Similarly, after pretreatment with 6-gingerol, the expression of p53 and p-Chk2 (T68) was significantly decreased compared with those cells treated with MEHP only.

Effects of 6-gingerol on p-Chk2 (T68) expression. HUVECs were treated with 6-gingerol (10 μM) for 1 h and exposed to MEHP for 24 h. (a) The nuclear protein fractions were analyzed by Western blot. β-actin is taken as internal control. (b) Relative expression of p-Chk2 (T68) was expressed as a percentage of β-actin. (**p < 0.01 vs. control and ## p < 0.01 vs. the MEHP group). MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells.

Effects of 6-gingerol on p53 expression. HUVECs were treated with 6-gingerol (10 μM) for 1 h, and exposed to MEHP for 24 h. (a) The nuclear protein fractions were analyzed by Western blot. β-actin is taken as internal control. (b) Relative expression of p53 was expressed as a percentage of β-actin (**p < 0.05 vs. control and ## p < 0.05 vs. the MEHP group). MEHP: mono (2-ethylhexyl) phthalate; HUVECs: human umbilical vein endothelial cells.
Discussion
DEHP, a manufactured chemical, is commonly added to plastics to make them flexible, which is widely used in medical devices. DEHP can be introduced directly into the bloodstream when getting a blood transfusion, receiving medicines through flexible plastic tubing, or having dialysis treatments. 20,21 In most species, DEHP is rapidly hydrolyzed to MEHP and 2-ethylhexanoic acid. Many of the properties of MEHP are like those of DEHP. Since DEHP can enter human blood during certain medical procedures, it seemed necessary to modulate its hazards and set limits.
This study investigated the oxidative DNA damage induced by MEHP in HUVECs using 6-gingerol to prevent the damage and estimated the underlying mechanisms. The DNA damage was evaluated by use of the comet assay, which is a powerful tool for the assessment of DNA breakage at the level of a single cell. 22 In our study, the alkaline comet assay was performed to confirm the presence of DNA strand breaks. The alkaline comet, in which DNA is mobilized under alkaline conditions for DNA denaturation, detects both single-stranded DNA breaks and double-strand breaks (DSBs). 23 Comet assay clearly showed that MEHP-induced DNA strand breaks remarkably in HUVECs, and the damage can be prevented by 6-gingerol.
Oxidative stress is defined as an imbalance between the production of ROS and antioxidant defenses, which results in a series of events including damage to cellular lipids, proteins, or DNA. 24 Some exogenous chemicals may redox cycle following metabolism by the cell, with the subsequent production of electrons that can be transferred to molecular oxygen producing superoxide. ROS may interact with DNA, leading to modification and potentially serious consequences for the cell. 25 Numerous results suggest that ROS may cause gene and chromosome mutations through DNA DSBs, one of the most severe forms of DNA damage. 26 –28 To explore the mechanisms of DNA damage, we investigated the level of ROS using DCFH as a fluorescent probe. The results show that MEHP caused the content of intracellular ROS to increase significantly, which suggest that ROS may play an important role in the induction of DNA damage by MEHP. Furthermore, the ROS level was decreased after the pretreatment of 6-gingerol, indicating that ROS may be involved in the inhibitory effect of 6-gingerol on MEHP-induced DNA damage. Our study also reports that MEHP caused alterations in the mitochondrial membrane potential. Mitochondria are intimately involved in the production of ROS. 29,30 These alterations in the membrane potential may promote the formation of ROS, which was particularly high in the cells exposed to MEHP, and caused DNA damage. MDA is one of the end product of lipid peroxidation induced by ROS and other free radicals. It is a biomarker for oxidative stress. MEHP could increase the MDA level, indicating that the mechanism of DNA damage induced by MEHP may relate to lipid peroxidation.
Antioxidants exert their protective effects not only by scavenging ROS but also by inducing the antioxidants in the cells. Among intracellular antioxidant molecules, GSH is the most abundant intracellular nonprotein thiol in cells, which plays an important role as an antioxidant. 31 GSH has the potential to remove those toxic electrophiles and metals, thereby protecting cells from toxic oxygen products, and keep the cellular environment in a reduced state. 32 In our study, increased generation of ROS and/or depletion of GSH capacity leads to oxidative stress in the HUVECs after treated by MEHP. Oxidative stress can cause DNA damage, lipid peroxidation, and cellular injury. 33 The data from our study showed that the depletion of GSH could be prevented by 6-gingerol, and the increased GSH is able to quench ROS and protect cells from toxic compounds. Antioxidant enzyme activities (SOD) reflect the level of oxidative stress. SOD could attenuate oxidative stress by catalyzing the dismutation process, which converts the superoxide anion into molecular oxygen and hydrogen peroxide. 8 The ability of 6-gingerol to upregulate SOD activity may contribute to the protective role of reducing oxidative stress from MEHP injury.
To further confirm MEHP-mediated DNA damage, the expression of some DNA damage-related proteins was observed in the study. The serine/threonine kinase Chk2 is a key component of the DNA damage response. 34 Under normal condition, Chk2 is present in the nucleus in an inactive monomeric form. 35 After DNA damage, Chk2 is phosphorylated by the enzyme, namely serine/threonine protein kinase ataxia telangiectasia mutated (ATM) on the priming site threonine 68 (T68). T68 is the most important phosphorylation site of Chk2. As the major molecular sensors of DNA damage, ATM can be activated primarily by DNA strand breaks. 36 It will recruit to the site of DNA damage and activate other signaling molecules, including Chk1, Chk2, and p53. 37 When activated, Chk2 phosphorylates many nuclear proteins involved in the process of DNA damage response. p53 has been regarded as an important substrate of p-Chk2. In unstressed cells, p53 has low activity and a short half-life, which allows normal growth of the cells. 38 When DNA damage happens, p53 can be phosphorylated by p-Chk2 and ATM, and it may accumulate in the nucleus and perform its function as transcription factor. 39
In brief, we hypothesize that MEHP may increase the ROS and MDA level, decrease the GSH and SOD level, and alter the mitochondrial membrane potential, which causes DNA strand breaks in HUVECs. When DNA damage is sensed, ATM activates Chk2, and Chk2 activation would lead to p53 stabilization and its accumulation in the nucleus. Therefore, ATM/Chk2/p53 signaling pathway may be activated by DNA strand breaks. Activation of the pathways may modulate other biological outcomes such as apoptosis or cell senescence. 40 However, the pathway can be prevented by 6-gingerol, which suggests 6-gingerol may be involved in the proper coordination of checkpoint and DNA repair processes.
To our knowledge, this is the first study to show the protective effects of 6-gingerol against MEHP-induced DNA damage in vascular endothelial cells. These effects are probably contributed to the antioxidant ability of 6-gingerol. Furthermore, 6-gingerol may prevent the further damage by blocking the ATM/Chk2/p53 pathway. Hence, 6-gingerol has a potential to be used as therapeutic adjuvant in MEHP toxicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81102135), Liaoning Provincial Natural Science Foundation of China (2014023004), China Scholarship Council, and “Dalian Rapidly Enhancing Medical Quality” Training Program.
