Abstract
Introduction
Ulcerative colitis (UC) is a chronic inflammation of colon in which the innermost tissue of colon and rectum develops deep-rooted inflammation. Diosmetin is the aglycone of the flavonoid glycoside diosmin, commonly found in citrus fruits. Therapeutically diosmetin is indicated to demonstrate anticancer, antimicrobial, antioxidant, oestrogenic and anti-inflammatory activity.
Methods
In this research, we studied the action of diosmetin on TNBS (2,4,6-trinitrobenzene sulfonic acid)-induced UC in rats. Male Wister rats were anesthetised with pentobarbital and TNBS introduced by performing an enema. Diosmetin treatment was provided through oral gavage for the next 28 days. Animals were sacrificed on the 29th day and colon tissues were collected for further examinations.
Results
Diosmetin treatment decreased colonic ulceration dramatically and decreased the percentage of inflammation in the colonic mucosa. Depletion of the TNBS assisted of superoxide dismutase and catalase was substantially restricted, while lipid peroxidation was recorded in the colonic tissue as malondialdehyde content was also decreased. After treatment with diosmetin, the occurrence of TNF-α, IL-6and NF-κB was considerably lowered and the number of apoptotic cells observed was significantly reduced.
Conclusion
Taken together, these observations demonstrated the potential of diosmetin against ulcer formation and development.
Introduction
Ulcerative colitis (UC) is a recurrent inflammation of the colon, a complex disease triggered by the association between genetic and environmental factors that impair mucosal homoeostasis and activate abnormal immune responses. 1 UC has grown into a global burden owing to its high incidence in developed countries and the dramatic rise in developing countries.2,3 There is a deterioration of the intestinal epithelial barrier during colitis progression which favours the entrance of pathogens into the tissue from the intestinal lumen. 4 Additionally, UC is also considered as a result of alteration in the equilibrium between intestinal microbiota and mucosal immunity that leads to severe intestinal inflammation. 5 Additionally, enterotoxin secretion causes an increase in the permeability of intestinal mucosa, and production of immunosuppressive protein leads to immune dysfunction. The intestinal epithelial cells are invaded by increasing populations of harmful bacteria and their excessive growth alter the metabolic and energy metabolism that further stimulates intestinal inflammation and damage to the intestinal mucosa. Indeed, reduction of the intestinal mucosal barrier function, the shield function of the intestinal wall, and increased translocation of intestinal microbiota damages the intestinal mucosal barrier, further leads to vicious cycle and aggravates the intestinal inflammatory response. 6 Consequently, inflammatory cells are therefore recruited through the damaged colonic mucosa where they trigger pro-inflammatory cytokines and extinguish the invasive pathogens, primarily via the development of reactive oxygen species. 7 Though the pathogenesis of UC is still unidentified, current findings supports that the importance of irregular immune control and abnormal cytokine system, associated with the in-depth assessment of patho-mechanisms of the disease condition. UC is normally characterized by both local and systemic inflammation with significant elevation of the circulating pro-inflammatory cytokines levels including TNF-α from macrophages, and associated with increased levels of IL-6 and IL-1β cytokines which further re-activates NF-κB.8,9 Increasing evidence suggested the significant role of oxidative stress in the pathogenesis and development of UC. 10 It has been reported that the increased production of reactive oxygen species (ROS) is due to neutrophils and macrophages, 11 which further instigate inflammation with a declined plasma level of endogenous antioxidants. 12 Taken together of all the aspects of UC pathogenesis, treatment modalities with antioxidative and anti-inflammatory effects could be investigated for successful therapy. The available treatment strategies for the therapy of UC includes the use of immunosuppressive drugs such as methotrexate, tacrolimus, cyclosporine, and 6-mercaptopurine, anti-inflammatory agents like 5-aminosalycylic acid and corticosteroids, and biologics resembling to vedolozumab, infliximab, natalizumab and certolizumab. 13 However, high price, inconsistent efficacy, high recurrence rate and severe side effects including cramps, abdominal pain, fever and rashes restricted their applications in clinical practice. 14 In this condition, novel agents must be developed with greater therapeutic outcome with fewer side effects at a minimum cost. Considering these facts, compounds from natural products considered as promising and fruitful in the treatment of UC. Various investigations suggested that different active compounds, such as polyphenols, flavonoids and alkaloids have attributed efficacy in ulcerative colitis. 15
Diosmetin (3′,5,7-trihydroxy-4′-methoxyflavone) is a flavone aglycone found in citrusspecies, 16 olive leaves. 17 Diosmetin exerts anticancer,18–20 anti-inflammatory, 21 antiapoptotic, 22 antimutagenic 23 and antibacterial 24 effects. Diosmetin provides a strong antioxidant activity in human monocytes through the prevention of intracellular ROS generation and the formation of malondialdehyde (MDA), associated with increased activity of the intracellular antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx). 25 Diosmetin also provides an inhibitory activity against lipopolysaccharide (LPS)-induced acute lung injury through the suppression of inflammatory cell infiltration and pro-inflammatory cytokines overproduction in broncho-alveolar lavage fluid. 26 These outcomes clearly indicate the strong anti-inflammatory property of diosmetin. Thus, considering the number of biological activities of diosmetin and as no scientific researches has been carried out with diosmetin in the therapy of ulcerative colitis, this work is designed for the evaluation of intestinal anti-inflammatory activity and the mechanism of action of diosmetin, via in vivo animal experimentation.
Materials and methods
Animals
Male Wistar rats, 180–200g and 9–10 weeks of age, were obtained from the Department of Laboratory of Animal Sciences of Inner Mongolia Medical University. The rats were housed in plastic container and kept in standard atmospheric conditions at a temperature of 22–24°C, and relative humidity of 20% with a 12-h light/dark cycle, with free access to food and water during the entire study. The animals were acclimatized at the specified condition for 1 week prior to the experiments. The recommendations by Ethics Committee of Animal Sciences of Inner Mongolia Medical University with Approval Number: IMXR-2020(07) were strictly obeyed during the animal experimentation.
Chemicals
All reagents used for the experiment were of analytical grade. Diosmetin (purity>98%) was purchased from PureOne Biotechnology (Shanghai, China). 2,4,6-trinitrobenzene sulfonic acid (TNBS), biotinylated horseradish peroxidase, and 3,3′-diaminobenzidine (DAB) were purchased from Sigma Chemical, Co. (St. Louis, MO,USA). The assay test kits for the measurement of oxidative stress namely, superoxide dismutase (SOD), MPO, MDA were supplied by Multisciences (Lianke) Biotech, Co., Ltd. (Hangzhou, China). Anti-rat antibody for IL-6, NF-κB, TNF-α and goat anti-rabbit IgG secondary antibody were bought from Genetex, Inc. (Irvine, CA, USA). An immunohistochemistry kit was procured from Biovision, Inc. (Milpitas, CA, USA), and an apoptosis detection kit was purchased from Takara Bio Inc. (Shiga, Japan). Other reagents were purchased from local firms in their purest forms.
Induction of ulcerative colitis in rats
The local enema with TNBS was used to obtain ulcerative colitis as mentioned by27,28 with minor adjustments. After 24 h of fasting, the rats were anesthetized using pentobarbital (50 mg/kg). Enema was carried out on the rats by PE50 catheter with the head-down position (4 cm near the anus) through inserting 100 μL of the TNBS solution (2.5% w/v in 50% ethanol). Then the tail was lifted and maintained the head side down position for 10 min to avoid the leakage and to maintain the uniform distribution of solution in the rectum followed by injection. This treatment with TNBS was continued for 3 successive days for the induction of the model.
Dose selection with grouping and treatment
According to the previously suggested report, the dose of diosmetin was selected. Reports suggest that a small dose of diosmetin protects against oxidative stress injury in myocardial ischaemia and inhibits cardiomyocyte apoptosis in rats. 29
Fifty rats were randomly allocated to five groups (10 rats/group): Normal control (NC) group: Rats treated with normal saline by oral gavage. Experimental group: Rat treated with 100 μL of TBNS by using PE50 catheter in anus. Diosmetin (50 mg/kg)-treated colitis group: Rat treated with diosmetin (50 mg/kg/day) by oral gavage for 28 days daily. Diosmetin (100 mg/kg)-treated colitis group: Rat treated with diosmetin (100 mg/kg/day) by oral gavage for 28 days daily. Diosmetin (200 mg/kg)-treated colitis group: Rat treated with diosmetin (200 mg/kg/day) by oral gavage for 28 days daily.
Evaluation of colon weight-to-length ratio
The colons were excised from the rats followed by the longitudinal dissection of the abdomen, and rinsed under running water to remove the faeces. Colons were mounted on non-absorbent surfaces and were blindly evaluated for weight-to-length ratios. Excised colon tissues were preserved at −80°C.
Macroscopic assessment of colonic damage
According to the process described by Ballester and Naeini, the colon tissues were rated from 0 to 100% on the basis of increasing order of severity, macroscopically noticeable injuries such as thickening, shortening, hyperaemia and necrosis.30,31 The mean scores were reported as the Disease Activity Index (DAI) of the colon.
Microscopic assessment of colonic damage
The samples of the colon tissue from the distal portion were fixed by 10% formaldehyde, embedded in liquid paraffin and transversely cut into sections of 5 μm thickness by microtome. Then, the section mounted on glass slides were soaked in xylene for deparaffinization for 5 min and then put into 100%, 95%, 85% and 70% ethanol for 1 min each followed by washing with deionized water. Subsequently, the haematoxylin solution and eosin solution were added dropwise for staining for 5 min, respectively. After dehydration, the sections were soaked in xylene solution for transparentizing for 10 min, followed by mounting with neutral resin covered with cover slip.
Assessment of antioxidant enzymes in the colon
Colon tissue were homogenized by a Potter-Elvehjem homogenizer (Ultra-Turrax T25, Janke and Kunkel IKA-Labortechnik, Staufen, Germany) on ice-cold Tris-HCl buffer (0.01 M, pH 7.4) to prepare a 10% homogenate, that was utilized for antioxidant assays of superoxide dismutase (SOD), catalase activity (CAT) and malondialdehyde (MDA) in the colons.
Superoxide dismutase (SOD)
The SOD activity was evaluated as per the method mentioned by Misra and Fridovich 32 with slight modification. Briefly, 500 μL of the tissue homogenate was treated with 750 μL ethanol (96% v/v) and 150 μL of ice-cold chloroform followed by centrifugation at 2000 rpm for 20 min at 25°C. Then, 500 μL EDTA (0.6 mM) and 1 mL carbonate bicarbonate buffer (0.1 M, pH 10.2) were added to 500 μL supernatant. 50 μL of adrenaline (1.3 mM) was added to initiate the reaction. At 480 nm, the absorbance was recorded against a blank for 4 min using the ultraviolet–visible spectrophotometer.
The formula for the assessment of percentage inhibition of auto-oxidation of adrenaline
The enzyme activity was expressed as unit per mg protein.
One unit of the enzyme activity was defined as the amount of enzyme that inhibits the auto-oxidation of adrenaline by 50% at 25°C
Catalase (CAT)
Catalase activity of the colon was evaluated by the method described by Sinha
33
with a few adjustments. The assay mixture contains 1 mL (0.01 M) phosphate buffer (pH 7.0), 500 μL H2O2 (1.18 M) and 400 μL of water, which was added to 100 μL tissue supernatant and incubated for 5 min at 28°C to begin the reaction. A 2 mL acetic acid-dichromate mixture composed of 3:1 ratio of glacial acetic acid and 5% potassium dichromate was added to terminate the reaction. Absorbance of the chromic acetate was determined at 620 nm. One unit of catalase activity was defined as the amount of enzyme needed to decompose 1 μmol H2O2 per min per mg protein at 25°C and pH 7.0. The enzyme activity was expressed in terms of its molar extinction coefficient of 39.4 M−1cm−1
Malondialdehyde (MDA)
Malondialdehydeis a by-product of lipid peroxidation and its level in the colon was determined as per the method described by Heath and Parker. 34 Briefly, a 3 mL mixture of 20% trichloro acetic acid (TCA) and 0.5% thiobarbituric acid (TBA) was added to 1 mL of the tissue extract. Then, it was heated at 95°C for 30 min, cooled in an ice bath and centrifuged at 2000 rpm for 10 min. Absorbance of the MDA-TBA complex was recorded at 532 nm against the blank by a UV spectrophotometer. The MDA extinction coefficient (1:56×10−5M−1cm−1) was used to evaluate the MDA concentration (nmol/mg protein).
Immunohistochemical analysis of TNF-α, IL-6 and NF-κB
The tissues were fixed in formalin fixed and embedded in paraffin then cut into sections of 5 μm thickness which was mounted in the glass slides. The tissue sections were then deparaffinised and rehydrated by ethanol. Then, 1% H2O2 was added to terminate the endogenous peroxidase activity followed by the addition of 5% normal goat serum for 30 min in order to decrease nonspecific binding sites causing unnecessary background staining. Thereafter, the sections were incubated with 50 μL of rat polyclonal ligand affinity-purified rabbit anti-rat IL-6, TNF-α and NF-κB primary antibodies overnight at 4°C. Antibody detection was performed with the Histostain-Plus Bulk kit (Invitrogen) against rabbit IgG, and 3,3′diaminobenzidine (DAB) was used to visualize the product. Subsequently, the slides were submerged in ddH2O two times for 5 min each. All slides were counterstained with haematoxylin, rinsed, dehydrated, mounted with cover slips and photographed with an Olympus C-5050 digital camera mounted on Olympus BX51 microscope. Scoring was performed by two investigators using coded slides. The labelling index was calculated as a percentage of IL-6, TNF-α and NF-κB positive cells per total number of cells counted.
Apoptosis assay
The apoptosis assay was performed by In Situ Apoptosis Detection Kit from Takara Bio Inc. (MK500) as per the manufacturer’s protocol, which was developed by DAB as chromogen and methyl green was used as counter stain. The slides were cleaned, desiccated and mounted. The reddish brown-stained nuclei were identified as the apoptotic cells. 35
Statistical analysis
Data were reported as mean ± standard error mean (SEM). Statistical testing was conducted using t-test and one-way variance analysis (ANOVA) measured by the application (GraphPad prism), accompanied by post hoc test (Dunnet’s t test), the differences were denoted as statistically significant at
Results
Effect of diosmetin on body weight on colitic rats
The colonic inflammatory response is stimulated through the intrarectal application of TNBS in rats which causes passing of loose stool (with or without occult blood) and loss of body weight. A gradual weight increase (0–4.04%) in the naïve control rats was recorded during the 28 days. But unlike the naïve control, the rats with TNBS-induced colitis showed a reduction in body weight from day 0 to day 28. In contrary, diosmetin-treated rats showed gradual increase in the body weights with the increasing dose of diosmetin from 50 mg/kg 200 mg/kg (Figure 1). Effect of diosmetin therapy on the body weights of TNBS-induced colitic rats.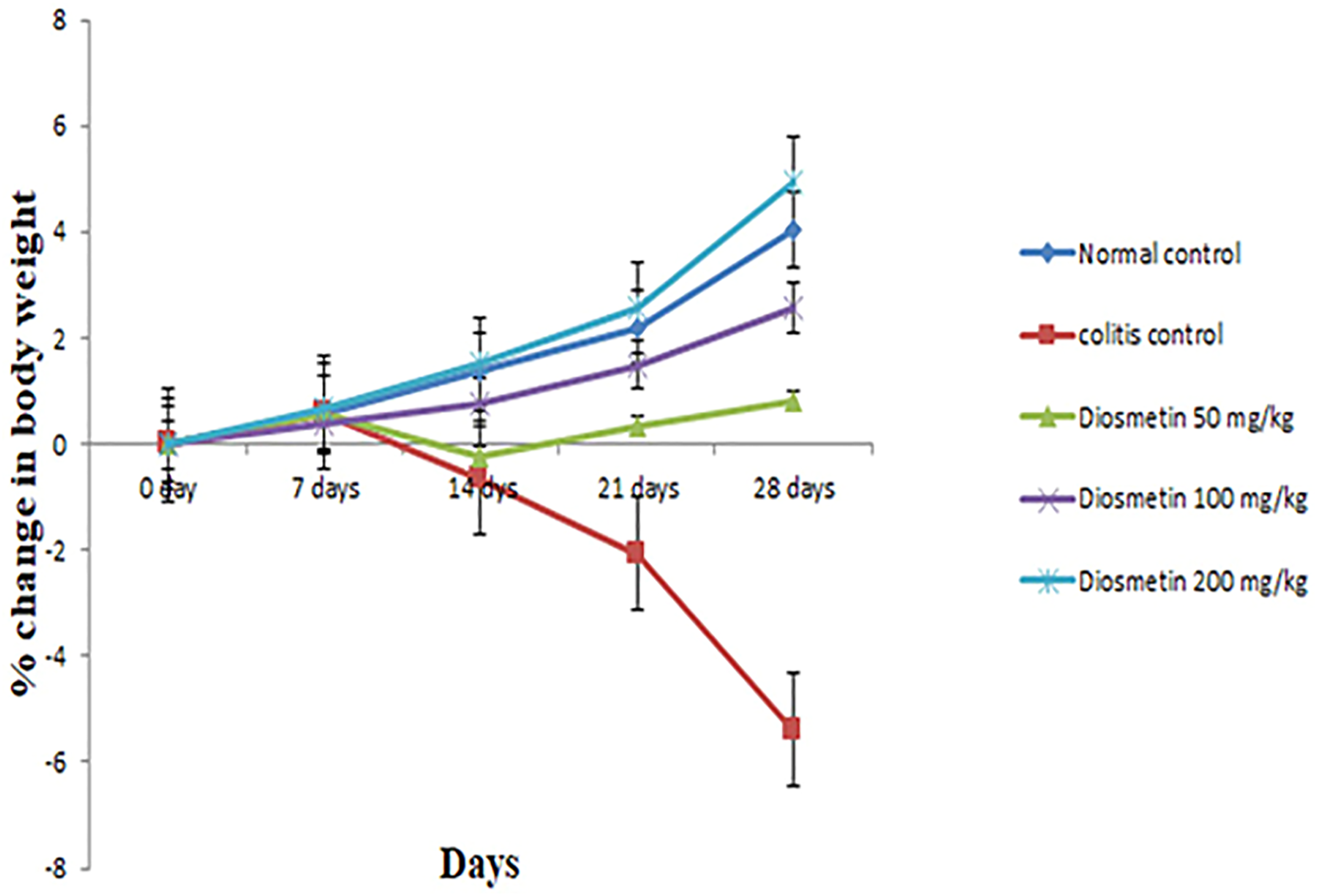
Effect of diosmetin on colon weight-to-length ratio in TNBS-induced colitic rats
The increase in colon weight/length ratio attributed to the increase in inflammatory cell infiltration, vascular permeability and oedema.
36
The mean colon weight/length ratio of normal control rats showed 0.155 g/cm showed which was significantly increased in the experimental rats with the value of 0.761( Effect of diosmetin on colon weight/length ratio of TNBS colitic rats. Results were presented as mean ± SEM. ∗
Similarly, diosmetin treatment at 100 and 200 mg/kg doses drastically decreases the mean colon weight/length ratio to 0.552 g/cm and 0.16 (
Effect of diosmetin on macroscopic scores of TNBS-induced colitic rats
There was not any sign of colitis in the normal control rats (Figure 3). Colitis was induced as described in Method. Rats were euthanized on day 28 and the colons were excised, longitudinally opened, washed and examined (a) normal control, (b) colitis control, (c) 50 mg/kg diosmetin therapy, (d)100 mg/kg diosmetin therapy, (e) 200 mg/kg diosmetin therapy.
However, a colonic inflammation was noticed due to the intrarectal administration of TNBS which are associated with increased neutrophil infiltration, massive necrosis of mucosal and submucosal layers, submucosal ulceration, increase in vascular dilation, and oedema clearly observed as reddened and necrotic areas. Similarly, rats treated with 50 and 100 mg/kg diosmetin exhibited moderate colonic inflammation with minor ulceration, whereas such inflammatory areas were not observed for animals treated with 200 mg/kg diosmetin.
The characteristics of colonic inflammation such as thickening, shortening, hyperaemia and necrosis were quantified as a Disease Activity Index (DAI) (Figure 4), ranging between 0 and 100% in ascending order of severity. Effect of diosmetin on macroscopic score of colons of TNBS-induced colitic rats. Results were presented as mean ± SEM. ∗∗
Naïve control rats reported perfectly healthy colon with a DAI of 0%. In comparison to the naïve control rats, the group with TNBS-induced colitis causes severe colonic inflammation characterized by hyperaemia, thickening, shortening and oedema with DAI of 88.65% (
Effect of diosmetin on histopathology of colon in TNBS-induced colitic rats
The colonic mucosa of the normal control rats demonstrated standard histopathological architecture including mucosa, submucosa, muscularis mucosa and intact gastric cells (Figure 5(a)). Conversely, colons of experimental rats (Figure 5(b)) showed large necrotic areas along with abscesses in the colonic mucosa and infiltration of inflammatory cells into the submucosa. Alternatively, the histopathological study of rats treated with 50 mg/kg and 100 mg/kg diosmetin (Figure 5(c) and (d)) presented with mild ruptures and inflammatory infiltration. The 200 mg/kg diosmetin-treated rats (Figure 5(e)) however demonstrated standard histopathological architecture similar to that of the normal control animals. Effect of diosmetin therapy on microscopic changes in the colons of TNBS-induced colitic rats. (a) Normal control, Mm represents muscularis mucosa; m represents mucosa; sm represents submucosa and Gc represents goblet cells; (b) colitis control, ab represents abscesses; inf represents infiltration of inflammatory cells; (c) 50 mg/kg diosmetin therapy, r represents rupture; (d) 100 mg/kg diosmetin therapy; (e) 200 mg/kg diosmetin therapy. (H&E) 40X magnification.
Epithelial cell loss and gross colonic injury were significantly decreased in all the diosmetin-treated groups. These observations are as par with our previous results and demonstrate the capability of diosmetin to reverse the changes caused by TNBS-induced colitis.
Effect of diosmetin on the activity of superoxide dismutase and catalase on TNBS-induced colitic rats
Effect of diosmetin on SOD, CAT and MDA levels of TNBS-induced colitic animals.

Results were presented as mean ± SEM and statistically compared using student’s t test.
∗Significant difference between experimental group versus diosmetin treatment group (
∗∗Significant difference between experimental group versus diosmetin treatment group (
$Significant difference between normal control group versus experimental group (
$Significant difference between normal control group vs. 50 mg and 100 mg/kg diosmetin-treated groups (
The mean CAT activity (nmol/min/mg protein) of naive control rats was 10.15 nmol/min/mg protein (Table 1) which was significantly decreased (
Effect of diosmetin on Malondialdehyde (MDA) Levels in TNBS-induced colitic rats
MDA levels in naïve control rats were noted at 2.15 nmol/mg protein which was significantly increased (
Following administration of 50 mg/kg diosmetin, MDA levels were significantly decreased (
Effect of diosmetin on the expressions of TNF-α, IL-6 and NF-κB on the colonic mucosa of TNBS-induced colitic rats
The normal colonic mucosa of the rats [Figure 6a(i)] denoted 4.59% cells expressing TNF-α which was significantly increased ( Effect of diosmetin therapy on (i) TNF-□, (ii) IL-6 and (iii) NF-κB expressions in the colons of TNBS-induced colitic rats. (iv) Effect of diosmetin therapy on apoptotic changes in the colons of TNBS-induced colitic rats. (a) Normal control, (b) colitis control, (c) 50 mg/kg diosmetin therapy, (d) 100 mg/kg diosmetin therapy, (e) 200 mg/kg diosmetin therapy. Arrows represent the expressions of specific indicated markers detected through specific anti-bodies. 40X magnification. Effect of diosmetin therapy on (iv) apoptosis in the colons of TNBS-induced colitic rats. (a) Normal control, (b) colitis control, (c) 50 mg/kg diosmetin therapy, (d) 100 mg/kg diosmetin therapy, (e) 200 mg/kg diosmetin therapy. Brown colouring denotes apoptotic cells. 40X magnification. Effect of diosmetin on TNF-α, IL-6 and NF-κB levels of TNBS-induced colitic animals. §Each score represents the results of 6 slides per rat and 6 rats per group, mean ± S.E. ( *Significant difference between diosmetin-treated and experimental group ( **Significance difference between diosmetin-treated and experimental control ( $Significant difference between normal control and experimental group ( $Significant difference between normal control and diosmetin 50 and 100 mg/kg treated groups (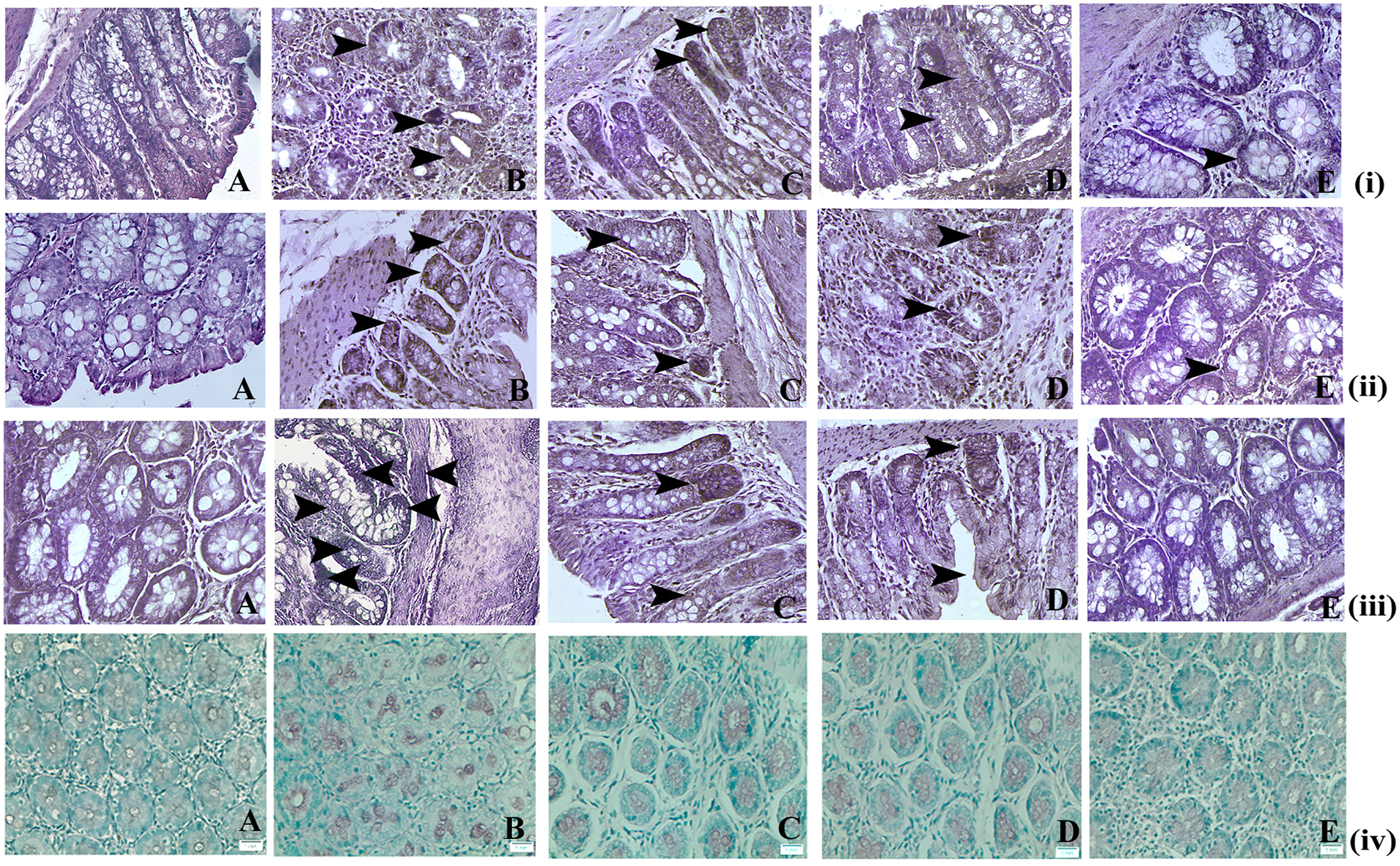
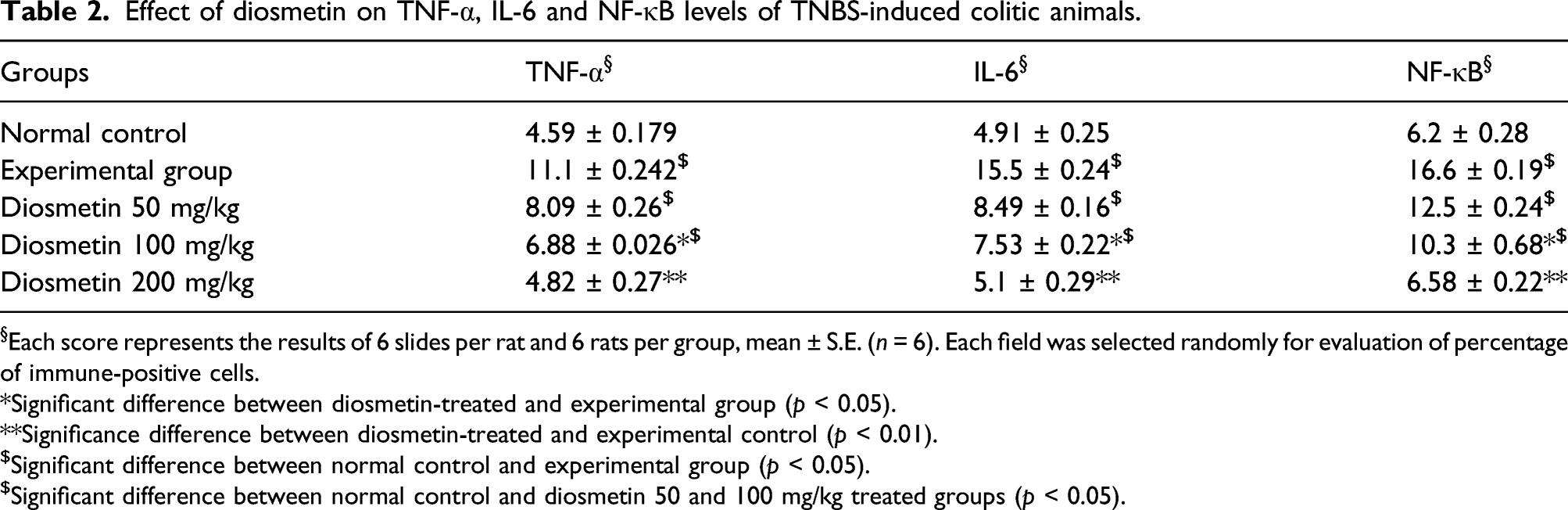
The colonic mucosa of the naïve control rats [Figure 6a(ii)] denoted 4.91% cells expressing IL-6 which was significantly increased (
The colonic mucosa of the naïve control rats [Figure 6a(iii)] denoted 6.2% cells expressing NF-κB which was significantly increased (
Following diosmetin treatment at 50 mg/kg [Figure 6c(iii)], there was an insignificant decline in the expressions of NF-κB to 12.5%, whereas 100 mg/kg diosmetin [Figure 6d(iii)]-supplemented groups expressed 10.3% of cells with IL-6 immune positive stains indicating a significant difference (
Taking all these observations together, we can assume that diosmetin treatment significantly decreased all inflammatory parameters like TNF-α, IL-6 and NF-κB in the colonic mucosa of the TNBS-treated colitic rats.
Effect of diosmetin on apoptosis in the colonic mucosa of TNBS-induced colitic rats
The appearance of brown stain, developed by DAB chromogen, coinciding with the condensed chromatin of apoptotic bodies demonstrated the TUNEL positive apoptotic cells. A moderate level of apoptosis was observed in the animals of normal control group [Figure 6a(iv)], whereas in the TNBS administered colon tissues an increased incidence of apoptosis was recorded [Figure 6b(iv)]. In the TNBS-induced colitis group, 15 apoptotic cells were found on an average in a field of about 700 cells. On the other hand, in the diosmetin-treated group, a decrease in apoptotic cells was observed [Figure 6c–e(iv)] with a reduction of 10–12 cells per 700 cells at a dose level of 200 mg/kg [Figure 6e(iv)].
Apoptosis in colon.
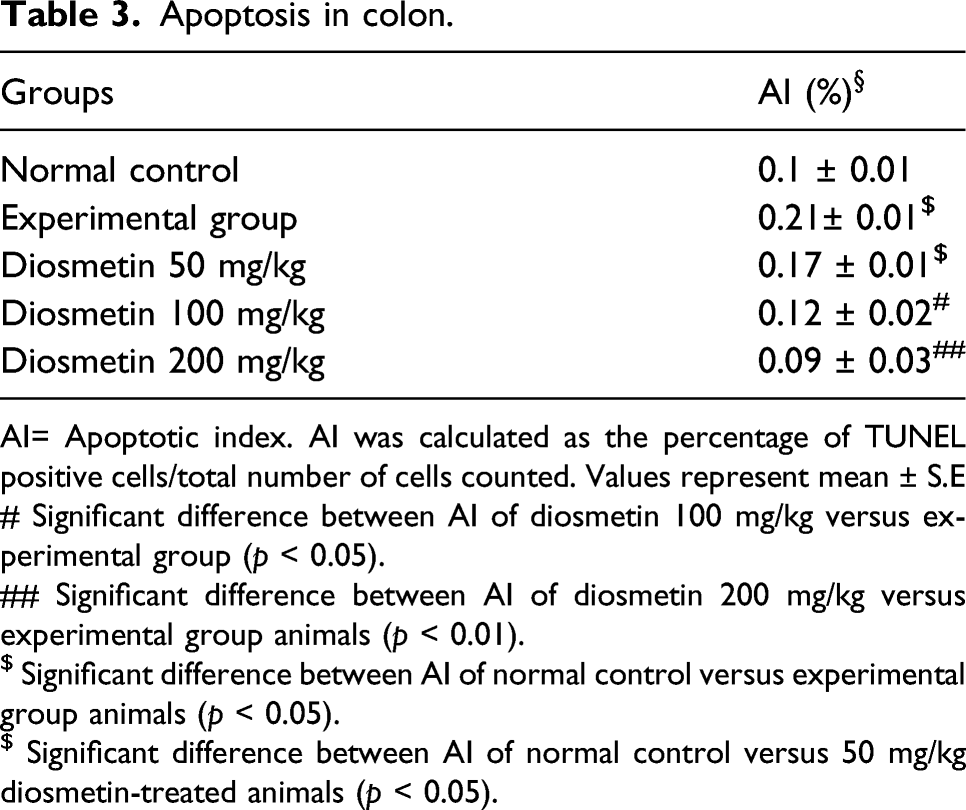
AI= Apoptotic index. AI was calculated as the percentage of TUNEL positive cells/total number of cells counted. Values represent mean ± S.E
# Significant difference between AI of diosmetin 100 mg/kg versus experimental group (
## Significant difference between AI of diosmetin 200 mg/kg versus experimental group animals (
$ Significant difference between AI of normal control versus experimental group animals (
$ Significant difference between AI of normal control versus 50 mg/kg diosmetin-treated animals (
AI was significantly increased in from 0.01 in normal control rats to 0.02 in TNBS experimental groups (
Discussion
Numerous intestinal inflammatory animal methodologies have been introduced and can be classified into four groups: spontaneous colitis, inducible colitis, genetically modified models and adoptive transfer models. 37 However, colitis caused by trinitrobenzene sulfonic acid (TNBS) has been regularly used for the screening of drugs.38–40 TNBS is a type of hapten, which preferentially binds to endogenous proteins in the colonic mucosa and triggers the activation of a local immunologic response in a macrophage and T cell-mediated pathway. 41
The intrarectal administration of TNBS greatly decreased the body weight of rats in this study. The decrease in the body weights of the animals could be attributed to several factors like deficiency in nutrition arising from diminished appetite, food intolerance or malabsorption coupled with rapid loss of body fluids due to colorectal bleeding and diarrhoea. TNF-and IL-6 also have a significant role in weight loss in colitis by producing neuropeptides that inhibit appetite and promote cachexia. 42
A significant improvement in the colon weight/length ratio of rats was discovered upon macroscopic inspection of the colon. Macroscopic changes of the colonic mucosa in ulcerative colitis can be credited to extreme oedema of the skin, necrosis, hyperplasia of goblet cells and inflammatory invasion of cells.43,44 Well-formed faecal pellets without visible blood or mucus stains were observed upon macroscopic analysis of the intestinal content of diosmetin-treated rats, thus denoting the significance of unblemished mucus layers of the colon in these groups. The effect of diosmetin on the colon mucosa may be further responsible for the prevention of abnormal bleeding in faecal pellets indicating towards the successful demonstration of anti-ulcerative potential of the drug.45–47 Diosmetin treatment further ensures the repair of TNBS-induced epithelial damage, consequently preventing diarrhoea and blood loss through faeces. 48 Thus, the preservation of the colonic mucosa by diosmetin assured the elimination of colonic ulceration and decreased inflammatory scores in ulcerative colitis. Histopathological examination of the colonic mucosa denoted that diosmetin therapy in TNBS-induced animals re-established the cellular architecture of the overall colonic mucosa and prevented further invasion of inflammatory cells, congestion, ulceration, degradation, necrosis and hyperplasia. These results also suggest that diosmetin can protect the colonic mucosa of the rats and decrease the progression of ulcerative colitis.
Superoxide dismutase (SOD) and catalase (CAT) are fundamental antioxidant enzymes in animals. As a result of free radical-induced oxidative damage, these enzymes levels are decreased in colonic tissues associated with ulcerative colitis. 49 SOD serves as a protection against ulcerative damage by mediating superoxide anion dismutation and inhibiting lipid peroxidation. CAT, on the other hand, is found in the peroxisomes and catalyses the conversion of hydrogen peroxide, a cytotoxic chemical, into water and oxygen. SOD ameliorates TNBS-induced colitis by reducing oxidative stress and leucocyte recruitment into the inflamed intestine. Hence, significant increase in the SOD activities of the TNBS-induced colitic tissues following diosmetin supplementation bear impartial evidence to its antioxidant potential which is as per the research investigated by Segui and his research team. 50 Diosmetin supplementation significantly increased SOD and CAT activities in TNBS-induced colon tissues, confirming its antioxidant capacity, which is crucial for its anti-inflammatory action.
Malondialdehyde (MDA) is a direct result of lipid peroxidation in the tissue. MDA levels of plasma rise dramatically in ulcerative colitis, and this is seen as an effective diagnosis for people with inflammation in the intestinal tract. 51 Natural products with antioxidant activity are advantageous in such cases because lipid peroxidation take place during oxidative stress. 52 Diosmetin treatment significantly inhibits MDA in the colon tissues leading us to conclude that it is capable of reducing the oxidative stresses in TNBS-induced colitic rats.
In the pathogenesis of inflammatory bowel disease, tumour necrosis factor alpha (TNF-a) and interleukin 6 (IL-6) play important roles.53,54 They influence the immune system of the mucosa, modify epithelial integrity, and organize invasion and release of neutrophils and macrophages, resulting in colonic injury. TNF-α causes increased endothelial cell permeability, pyrexia, algesia, cachexia and leucocyte formation as well as mobilization for the promotion of prostaglandins. IL-6 promotes the synthesis of acute-phase proteins and the activation of the complement system, resulting in the release of C3 and C5. C5 and C3 are essential in neutrophil chemotaxis, leucocyte activation or generation of additional mediators, and mast cell activation to produce histamine and heparin 55 TNF-α, IL-6, COX-2 and their upstream signal receptor, NF-κB, have recently become successful new anti-inflammatory targets for IBD associated UC therapy. 56 In the current investigation, the elevation of TNF-α, IL-6 and NF-κB levels in the TNBS experimental rats relative to their decreased levels in the rats treated with diosmetin indicates the compound’s antiulcerative capacity. Treatment of rats with diosmetin not only substantially inhibited the elevation of inflammatory markers like TNF-α, IL-6 and NF-κB but also resulted in gross colon safety as evident from the histopathological studies.
Apoptosis is a form of controlled cell death that keeps the balance of healthy cell survival and cell death in check. Findings suggest that elevated cytokine activity, such as TNF, IL, and interferon family members, causes apoptosis in intestinal epithelial cells. 57 During acute UC inflammatory events, elevated apoptosis of intestinal epithelial cells has been reported that can impair intestinal mucosal structure and barrier function subsequently causing inflammation. 58 Discovering the best ways to reduce colonic epithelial cell death and restoration of mucosal tissues has been the focus of UC treatment. Diosmetin therapy significantly reduced apoptosis in the TNBS-induced colitic animals thus orienting us to conclude that diosmetin is capable of restoring the damages made to the colonic mucosa and bringing out the balance of apoptosis. Despite the advancements of this research, there are some limitations associated with this study that need to be investigated for the better assessment of protective role of diosmetin against TNBS-induced ulcerative colitis. Some of the important limitations are commencement of power analysis, gender-specific effect, food and water intake rate, assessment of various other inflammatory markers like serum amyloid A, alpha-1-acid glycoprotein, ceruloplasmin, hepcidin, haptoglobin and human colitis cell line study. Taken together, these findings will aid in a more accurate assessment of diosmetin in the human cellular microenvironment.
The results of the present experiment are representative of the protective capacity of diosmetin to suppress TNF-α, IL-6 and NF-κB activity against the adverse effect of TNBS-induced colitis in rats. SOD and CAT levels in the colonic mucosa were raised as well as decreased MDA helps us identify the antioxidant capabilities of diosmetin in ulcerative colitis. The outcomes of this study indicate significant proof of concept for the prospective use of diosmetin therapy to inhibit, reverse or postpone ulcerative colitis development, and so this could be a potential clinical contender in near future.
Conclusion
In conclusion, the present study represents the protective effect of diosmetin against TNBS-induced ulcerative colitis through the suppression of TNF-α, IL-6 and NF-κB. Additionally, it also caused the elevation of SOD and CAT levels in the colonic mucosa and depletion of MDA which further confirms the antioxidant activity of diosmetin in ulcerative colitis. Thus, the result of this experiment firmly concludes the potential utilization of diosmetin to inhibit, reverse or postpone the development of ulcerative colitis which surely could be a prospective clinical candidate in recent future.
Footnotes
Author contributions
Yu X designed, planned and wrote the manuscript. Liu Y executed the experiments, analysed the data and wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The recommendations by Ethics Committee of Animal Sciences of Inner Mongolia Medical University with Approval Number: IMXR-2020(07) were strictly obeyed during the animal experimentation.
Animal welfare
The present study followed institutional guidelines for humane animal treatment and complied with relevant legislation.
