Abstract
Methamphetamine abuse is one of the most medical and social problems many countries face. In spite of the ban on the use of methamphetamine, it is widely available in Iran’s drug black market. There are many analytical methods for the detection of methamphetamine in biological specimen. Oral fluid has become a popular specimen to test for the presence of methamphetamine. The purpose of the present study was to develop a method for the extraction and detection of methamphetamine in oral fluid samples using liquid–liquid extraction (LLE) and gas chromatography/mass spectrometry (GC/MS) methods. An analytical study was designed in that blank and 50 authentic oral fluid samples were collected to be first extracted by LLE and subsequently analysed by GC/MS. The method was fully validated and showed an excellent intra- and inter-assay precision (reflex sympathetic dystrophy ˂ 10%) for external quality control samples. Recovery with LLE methods was 96%. Limit of detection and limit of quantitation were 5 and 15 ng/mL, respectively. The method showed high selectivity, no additional peak due to interfering substances in samples was observed. The introduced method was sensitive, accurate and precise enough for the extraction of methamphetamine from oral fluid samples in forensic toxicology laboratories.
Keywords
Introduction
Methamphetamine abuse has been an important social problem in many countries since its assent to peak popularity especially in young population. 1 It can be one of the contributory factors to significant percentage of fatal road traffic collisions. 2 Law enforcement agencies and police authorities have focused on the investigation of prosecuted and arrested persons accused of violence, criminal offences or road traffic accidents for methamphetamine abuse. 3 Routine analysis of biological samples such as blood, urine and hair are performed in professional forensic toxicology laboratories as an indicator of drug use and abuse. 4 However, other biological samples such as oral fluid may be used. Oral fluid has many advantages over other conventional biological samples since sample collection is easy and less invasive to donors’ privacy, avoiding sample manipulation and adulteration due to direct supervision and offers information about recent methamphetamine abuse. 5 –7 Therefore, oral fluid gains its importance for drugs of abuse testing. 8 Drug rehabilitation clinics, workplace, prisons and roadside police officers use oral fluid for drugs of abuse testing. 9 Sensitive, specific and reliable methods such as gas chromatography/mass spectrometry (GC/MS) are widely used in forensic toxicology laboratories for the detection of amphetamine type stimulants (ATSs) in oral fluid. 7 Yet, sample preparation methods in order to matrix effect reduction prior to instrumental analysis are an important task and time consuming. GC/MS was used for the analysis of ATSs in blood, serum, oral fluid and urine samples by Kankaanpää et al. 10 Pujadas et al. described GC/MS method for identifying and quantifying psychoactive drugs in oral fluid using solid-phase extraction (SPE) as pre-analytical sample preparation. 11 Souza et al. developed and validated a method for the identification and quantification of amphetamine, methamphetamine, fenproporex, diethylpropion and methylphenidate in oral fluid. They had used solid-phase microextraction (SPME) and GC/MS techniques in their study. 7
Despite the large number of studies describing sophisticated processes for sample preparation using SPE in order to be subsequently analysed for ATS, 7,10,11 there is still a need for a rapid, labour saving and efficient extraction method for oral fluid analysis. The main goal of the present study was to develop and validate a convenient and simple liquid–liquid extraction (LLE) method for the extraction of methamphetamine from oral fluid and analyse the extraction product with GC/MS technique.
Methods
Chemicals and reagents
Methamphetamine hydrochloride (HCl) was purchased from Lipomed Pharmaceutical (Arlesheim, Switzerland). Amantadine HCl was donated by Amin Pharmaceutical Company (Isfahan, Iran). Water, n-hexane and n-heptane were high-performance liquid chromatography (HPLC) grade and purchased from Darmestadt, Merck (Germany). Potassium hydroxide (KOH) and sodium bicarbonate (NaHCO3) were Reagent Plus®, 99.9% and were supplied by Sigma Aldrich (Germany). Heptafluorobutyric acid (HFBA) in derivatization grade was obtained from Sigma Aldrich.
Oral fluid samples
Blank oral fluid samples were collected from healthy volunteer donors with no history of substance abuse, by spitting after chewing gum for 2 min.
For minimizing the matrix effects, all oral fluid samples were mixed and frozen at −20°C until the day of analysis.
Preparation of standard and working solutions
Freshly prepared stock solutions of 1 mg/mL of MA and 100 µg/mL of amantadine were prepared in HPLC-grade methanol and stored at −20°C. From this stock solution of MA, working solutions of 10 and 1 µg/mL were made and used for spiking into blank oral fluid samples and for calibration curve preparation.
Sample preparation, extraction and derivatization procedures
Frozen oral fluid samples were thawed quickly and vortexed for 1 min. The oral fluid samples were centrifuged for 5 min at 9000 r/min and the supernatant was transferred into a clean glass tube. One-hundred microlitres of amantadine (100 µg/mL) was added as internal standard, the mixture was alkalinized by adding KOH (0.5 M) and pH was adjusted to 11–12.
Extraction was continued by adding n-hexane (3 mL) and shaking for 20 min. The organic phase was separated by immersion of tubes into a freezing bath (−30°C). After the addition of 100 µL methanol/HCl (99:1 v/v) the solvent was evaporated to dryness under gentle stream of nitrogen at room temperature. The extract was reconstituted with 100 µL n-heptane and vortexed for 1 min. To achieve a complete derivatization reaction after adding 25 µL of HFBA, the recent mixture was vortexed for 3 min. The excess amount of HFBA was deactivated with 500 µL of 10% NaHCO3 and vortexed for 1 min. Organic layer was separated by putting the glass tubes in freezing bath. This layer was transferred into a vial, crimp-capped tightly and analysed using GC/MS.
Internal standard selection
Selection of an internal standard is important for the precision of a method, 12 also it improves the data quality. 13,14 Internal standards are substances with chemical characteristics similar to the analyte of interest. Internal standards are used in the analyses procedures in which the quantity of the analyte of interest varies from run to run or sample preparation steps cause the reduction in the concentration of proposed analyte. 15 Amantadine was selected as an internal standard in the present study as it shows the most rational criteria that have a positive impact on its selection. Amantadine shows similar extraction characteristics, retention times, stability, detector response and chemical structure as the analyte of interest, methamphetamine. Amantadine was added to all method validation samples at final concentration of 100 µg/mL.
GC/MS analysis conditions
GC/MS was carried out on an Agilent 5975 mass series that was coupled with Agilent 6850 gas chromatograph. Agilent MS Chem Station software was used for instrument control and data acquisition. Agilent 5-MS capillary column (30 m, 0.25 mm I.D, 0.25 µm film thickness) was employed throughout the study.
The chromatographic conditions for GC method were as following:
Helium (99.999%) constant flow of 1.5 mL/min, inlet temperature 250°C, injection volume of 1 µL (splitless) were used. The oven temperature was programmed at 90°C (held for 1 min) followed by 20°C min−1 ramp to 280°C and held for 5 min.
Mass source and quadrupole temperatures were set at 230°C and 150°C, respectively. The ion source was operated in full scan and selected ion monitoring (SIM) mode both together. In full scan mode, scan range was 40–500 m/z, ions selected for quantitative analysis were 118, 254 for MA and 290 for amantadine.
Method validation procedures
Suitability of the method was evaluated by assessment of linearity, limit of detection (LOD), limit of quantitation (LOQ), selectivity, accuracy, matrix effect, precision, repeatability, recovery and carry over. Data were subjected to one-way analysis of variance.
Linearity
A quantitative method is linear when obtained test results are proportional to the amount of analyte in the studied matrix. 16,17 The linearity of introduced method was evaluated using six concentrations 15, 30, 50, 75, 150 and 200 ng/mL with triplicate measurements for each. Least square method was used for the preparation of the regression line and expressed as correlation coefficient (R 2 > 0.99).
Detection and quantification limits
LOD was calculated as the concentration with a signal/noise = 3, whereas LOQ was evaluated as the concentration with a signal/noise = 10. Blank oral fluids were spiked with low and decreasing methamphetamine concentrations until signal/noise of about 3 was achieved. LOQ was defined in the same manner.
Selectivity
An analysis method is selective when it can determine a particular drug in the analysed matrix without interference with other drugs in the medium. 18 Selectivity experiment was carried out for the investigation of the presence of drugs other than methamphetamine in oral fluid samples. Tramadol, methadone, morphine, codeine and diazepam were spiked into blank oral fluid samples at 20 µg/mL of each and extracted by proposed method.
Intra- and inter-day accuracy and precision
If the observed results of an analytical method is in agreement with the true concentration of the analyte in the sample the method is accurate. Accuracy of quality control (QC) samples remained within 99.7–100% of the target concentrations. Random errors are measured as precision, or may be expressed as repeatability. 17 QC samples were prepared by spiking blank oral fluids at three concentrations (low = 20, middle = 80, high = 160 ng/mL) of methamphetamine. Inter-day precision was achieved in a single run, analyzing triplicate of QC samples (n = 9). Intraday assay was performed by analyzing QC samples over 3 consecutive days (n = 27). Precision was expressed as RSD of concentrations for QC samples. Precision had not exceeded 10% of the relative standard deviation.
Extraction efficiency (recovery)
The efficiency of the validated method for recovery of MA from oral fluid has been evaluated taking into consideration the possible losses of analyte during preparation processes. For the estimation of relative recovery of MA in LLE method, 20, 80 and 160 ng/mL of MA were spiked into blank oral fluid samples and extracted with the proposed method. Peak areas of spiked oral fluid extracts were compared to peak areas obtained from unextracted standards at the same concentration.
Carry-over assessment
In order to assess carry-over as a validation parameter, blank oral fluid samples were injected immediately after the analysis of the highest point of the calibration curve concentrations three times on each of 3 days of the validation protocol. Any probable peak present at the retention time of target analyte was evaluated.
Authentic oral fluid collection and analysis
To assess the suitability of the validated method for the analysis of authentic samples, 1 mL of oral fluid samples were collected from 50 individuals in drug abuse rehabilitation centres Tehran, Iran. An inclusion criterion for sample collection was positive screening test results for methamphetamine in urine samples. These specimens were collected in a 6 months period by spitting in test tubes. The specimens were transferred to the laboratory and stored at −20°C until the day of analysis. Real samples were extracted as described and MA concentration was calculated for each sample separately.
Results
The present study was conducted to validate a suitable LLE method for the extraction of MA from oral fluid samples.
Method validation results
Linearity
Linear calibration curve was obtained for MA with a correlation coefficient (R
2) higher than 0.99 and RSD ˂ 10%. Linear regression equation was

Linear calibration curve for methamphetamine at 15, 30, 50, 75, 100, 150 and 200 ng/mL concentrations.
Detection and quantification limits
The method has proved to be sensitive. The LOD and LOQ for MA in oral fluid sample were 5 and 15 ng/mL, respectively. LOQ was determined by performing six replicate analyses at each concentration level (15–200 ng/mL).
Selectivity
The method showed high selectivity, no additional peak due to substances in samples that could interfere with the analyte under investigation was observed. Figure 2 shows the chromatogram of oral fluid sample spiked with tramadol, methadone, morphine, codeine and diazepam.

Chromatogram of authentic oral fluid sample spiked with tramadol, methadone, morphine, codeine and diazepam for selectivity assessment.
Intra- and inter-day accuracy and precision
Validated method was accurate and precise enough. Precision was expressed by inter-day and intraday precision. The intraday accuracy was determined to be 99.7–100% by performing tree replicate analyses at each QC concentration level (20, 80 and 160 ng/mL). Table 1 shows a summary of intra- and inter-day validation parameters.
Validation parameters for quantitative analysis of methamphetamine in oral fluid samples.
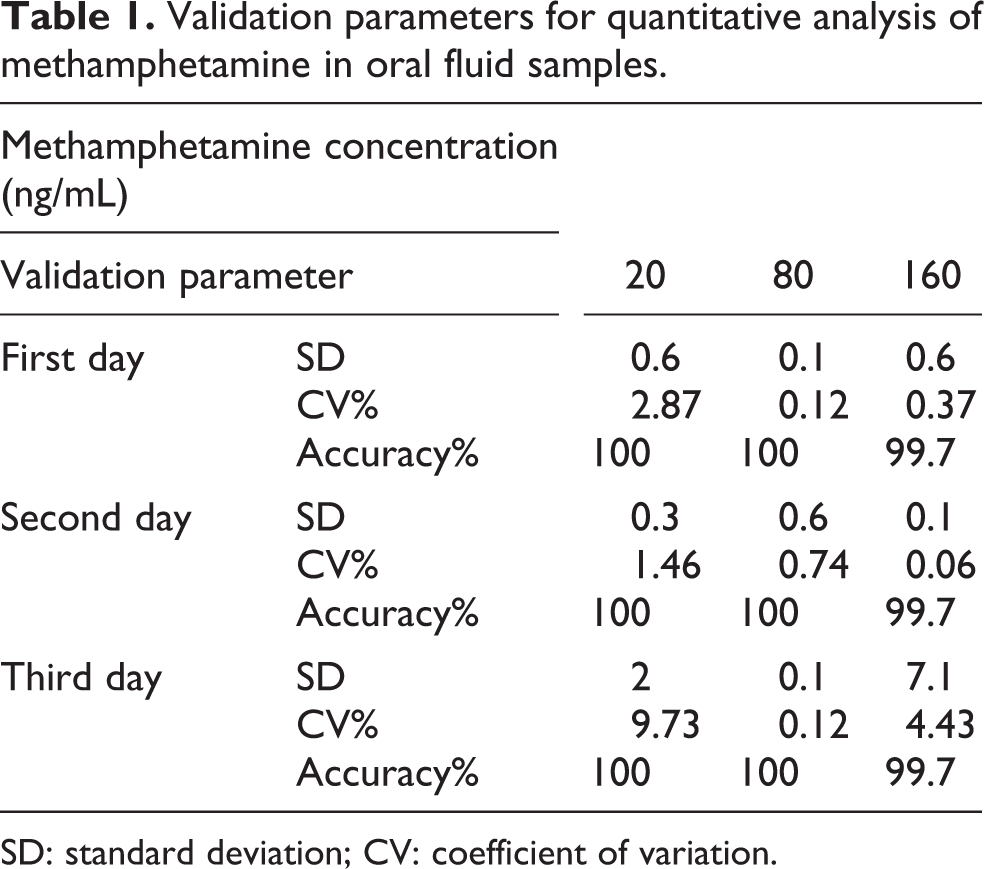
SD: standard deviation; CV: coefficient of variation.
Extraction efficiency (recovery)
Absolute recovery (mean ± SD) was obtained after LLE of different concentration levels of MA in oral fluid samples. Recovery of MA was estimated to be 96%.
Carry-over assessment
Following the injection of nine blank samples after the highest QC concentration, no peaks were observed, indicating the absence of carry-over during analysis.
Authentic oral fluid analysis results
Fifty real oral fluid samples were analysed by the validated method. The concentrations of MA were calculated as peak height ratio (methamphetamine/amantadine). MA was detected in all 50 authentic samples in concentration ranges of 20.412–441.370 ng/mL. The concentration range of a calibration should frame the expected concentration range of the analyte in the sample. It should be noted that the highest concentration was achieved by serial dilution of the sample to lie within the calibration curve concentration ranges. Twenty samples contained amphetamine. Tramadol and methadone were detected in 8 and 4 samples, respectively.
Discussion
The purpose of the present study was to design a quick, sensitive and accurate LLE method for the extraction and subsequently detection of MA from oral fluid sample using GC/MS technique.
According to the findings of the present study the validated method showed acceptable results for linearity, LOD, LOQ, precision, accuracy and selectivity for the qualitative and quantitative analysis of MA in oral fluid samples. LOD and LOQ obtained in the present assay were in accordance with the Substance Abuse and Mental Health Services Administration guideline for oral fluid testing to detect methamphetamine. 19 LOQ for MA was 15 ng/mL in the present study whereas it was 5 ng/mL in the study of Drummer et al. 20 Also they had used LLE solvent (butyl chloride) and derivatization reagent (pentafluoropropionyl anhydride) different from the present study. 20
It should be taken into consideration that oral fluid, as an unconventional sample can be a suitable alternative for blood in pharmacokinetic studies of substance abuse. Detection of drugs in oral fluid is an indicator that the subject is under the influence of drug and recent drug use. 3,21 There are some reports that suggest oral fluid as a substitute for urine in drug abuse testing due to its lower refusal collection rate when assessing drug impaired individuals. 3,11,22,23
Several methods have been developed for the detection and quantification of drugs of abuse in oral fluid and other biological samples. 10 Preparation of oral fluid samples for analysis by instrumental methods is an important step to get reliable results. Many studies use solid phase extraction (SPE), SPME whereas the others use one or two steps LLE and hollow fiber liquid-phase microextraction methods, followed by derivatization process. 6,10,12,20,24
In accordance with the results obtained from other studies, the recovery of MA from oral fluid samples was about 96% in the present study. Langel et al. in a study conducted on oral fluid samples for the detection of multiple drugs simultaneously found that LLE and SPE had acceptable extraction efficiency. 25 Scheidweiler and Huestis recorded 86.5% recovery for methamphetamine from oral fluid using SPE as extraction and gas chromatographic–electron impact ionization mass spectrometry as confirmatory methods. 26 n-Heptane was chosen as an extraction solvent in such a way that an aliquot of the solvent phase was used to inject to GC/MS directly. Extraction, back extraction steps and addition of hydrochloric acid can avoid the loss of MA. This method was used by the study of Lillsunde and Korte. 27 No heating step is needed in the present validated method. This method has been used in the previous studies too. 10 They had used single-step extraction using toluene and stated that there were no interfering substances in the procedure. However, two-step LLE method using hexane and heptane were used in the present study.
A substance should be volatile to be analysed using GC method. Derivatization of MA with HFBA renders MA to be sufficiently volatile to be eluted at suitable temperature without any structural decomposition. 28 ATSs show base peaks at low masses, which can interfere with biological background. Derivatization can resolve the problem by changing the base peaks of the parent compound. 10
Progressive methods have been attributed to greater use of labour saving derivatization reagents. In accordance with our study, Kankaanpää et al. had used HFBA as derivatization reagent. 10 Amphetamine and methamphetamine show little discriminative mass spectra. Derivatization improves resolution, sensitivity and selectivity of the analysis method for the detection of these low-molecular weight amphetamines. 7 Derivatization with HFBA needs no standing or heating period. 27 Excess reagents in the analysis procedure should be inactivated in order to prevent the degradation of GC column. 29 Peters et al. had used sodium phosphate solution to remove the excess reagents. 30 Excess amount of HFBA was inactivated by adding 10% NaHCO3 in the present study.
Deuterated forms of drugs had been used as internal standard for the detection and quantitation of 49 licit and illicit drugs in oral fluid samples using SPE as pretreatment technique in the study of Wylie et al. Samples were subjected to confirmation analysis using liquid chromatography/MS/MS and GC/MS methods. 31 However, amantadine was used as internal standard in the present study instead of high-cost deuterated forms of amphetamines.
Street methamphetamine is an impure cocktail containing different active pharmaceutical ingredients and other impurities added to them intentionally. 32 These drugs may be detected in the laboratory analysis process in addition to methamphetamine. Moreover, many substance abusers use different drugs to achieve their full desired effects. 33,34 Therefore the method was validated for the simultaneous detection of methamphetamine and a number of drugs with a high potential for abuse such as tramadol, methadone, morphine, codeine and diazepam as the probable interfering active pharmaceutical ingredients.
Preparative LLE method and GC/MS were validated for the detection of MA in oral fluid samples in the presence of other drugs without any interference. Validation parameters have shown that the designed method is linear, specific and sensitive. Recovery, precision and accuracy are in accordance with the accepted limits for the method validation in biological studies. 35 Total run time was less than 10 min. Due to many advantages of oral fluid, this method can be used in forensic toxicology laboratories.
Indeed oral fluid has many advantages for monitoring drug levels in comparison to conventional samples (blood, urine). Oral fluid gathering is a non-invasive procedure that is easy and less prone to adulteration. Due to high blood perfusion of salivary glands, drugs in blood transfer rapidly to saliva. Oral fluid has more acidic pH than plasma, thus basic drugs such as methamphetamine (pK a = 10.1) are found with high concentrations in oral fluid compared to plasma within minutes of parenteral drug administration.
Interpretation of oral fluid test results is important in forensic cases. There is a high correspondence of drug concentrations in oral fluid to blood; this fact makes oral fluid a suitable sample to be tested for the detection of recent drug exposure, possible drug induced risky behaviour and antisocial effects. 36 We should say that we have encountered some limitations to perform the present study. Sample collection from rehabilitation centres was very difficult. In some instances, mouth dryness was the main cause of insufficient amounts of oral fluid gathering.
We recommend more studies to validate other inexpensive and reliable sample preparation methods for the extraction of other drugs of abuse such as mephedrone and cathinone derivatives in unconventional samples such hair and oral fluid.
Conclusions
Validated method in the present study has significant potential to be used in forensic toxicology laboratories and other settings including prisons, emergency, drug rehabilitation centers, workplace and in other situations where there are evidences to support reasonable suspicion for methamphetamine abuse.
Footnotes
Acknowledgments
The authors wish to thank the kind personnel of the Toxicology Department, Legal Medicine Research Center, Legal Medicine Organization, Tehran, Iran for their cooperation in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
