Abstract
Ten of 40 cows died within 48 h of gaining access to a barn in which various chemicals were stored. Some of the surviving cows exhibited drooling, muscle tremors, and agitation. Postmortem examinations of 2 cows were performed in the field, and revealed nonspecific, moderate-to-severe pulmonary congestion. Liver and rumen contents, each from a different cow, were analyzed using a qualitative, multi-residue GC-MS method validated for the detection of pesticides and other chemical analytes. Using this method, extracts from the liver and rumen content samples were compared to atrazine (neat standard) and matrix-matched, control samples fortified with atrazine. GC-MS analysis detected atrazine at 215 m/z (NIST match >97%) with a retention time of ~13 min in liver and rumen content samples from our case. Detection of atrazine in the samples from the cows in this herd, combined with the clinical history, indicate that atrazine toxicity was the likely cause of clinical signs and death observed in this herd.
Atrazine, a triazine herbicide, is commonly used in agricultural and roadway application to control broadleaf weeds, specifically in corn, sugarcane, and sorghum fields.2,3,6 Atrazine has a low oral intoxication level in mammals, when used under proper conditions.4,5 However, intoxication has occurred in animals that gained access to formulated atrazine and ingested large quantities. Textbook signs of intoxication include anorexia, locomotor disturbances, tachypnea, irritability, and hypersensitivity. 4 Clinical signs reported in cases and studies of toxic ingestion by cattle included hyperthermia, increased pulse and respiratory rate, ataxia, tenesmus, anorexia, stiffness, muscle spasms in the hindquarters, drooling, and death.8,9,11 Gross lesions included congestion of kidneys, lungs, and liver, enlarged adrenal glands, epicardial petechiae, and hemorrhage in skeletal muscle, thymus, thyroid glands, and heart.9,11
In December 2018, the Heeke Animal Disease Diagnostic Laboratory (ADDL), Southern Indiana Purdue Agricultural Center (Dubois, IN, USA), was contacted for diagnostic advice concerning a herd of 40 beef cows that broke into an old barn that contained various pesticides and herbicides, including carbaryl (Sevin dust; Bayer) and glyphosate (Roundup; Bayer). After further investigation, it was discovered that atrazine powder was also stored in the barn. Within 24 h of the incident, 4 cows had died. After another day, 6 additional cows were dead. The remaining cows had signs of drooling, muscle tremors, and agitation. Within 24 h of death, a postmortem examination of cows in the field revealed nonspecific, moderate-to-severe pulmonary congestion. Liver and rumen contents from 2 cows were submitted to the Toxicology Section of the Indiana ADDL (West Lafayette, IN, USA) for analysis by gas chromatography–mass spectrometry (GC-MS; Thermo Scientific).
Liver (5 g) and rumen contents (25 g) were individually homogenized in 10 mL of acetonitrile (Thermo Fisher) and 1 g of sodium chloride (MilliporeSigma; 10:1, v/w) and the homogenate centrifuged at ~1,640 × g for 5 min. The supernatant was aliquoted; an additional 5 mL of acetonitrile was added to the solids, and the sample was homogenized and centrifuged as described above. The supernatants from each homogenization were combined. Sodium sulfate (1 g; EMD) was added to dry the supernatant, which was nitrogen (Indiana Oxygen) evaporated to ~1 mL. The extracts were applied to conditioned solid-phase extraction cartridges (ENVI-Carb-II/PSA; Supelco). The eluates were evaporated to dryness under nitrogen and reconstituted with 0.5 mL of acetonitrile:toluene (3:1; Thermo Fisher) for GC-MS analysis. The GC instrument parameters included an injector port temperature of 220°C, a column flow of 1.0 mL/min, and a column temperature program initially set at 90°C (held 0.10 min) with a 7.1°C/min ramp to 300°C. Atrazine standard, quality controls, and diagnostic samples (1 µL of each) were injected onto a VF-5ms column (30 m × 0.32 mm ID; df = 0.25; Varian). Election ionization MS scans from 40–650 m/z were used to monitor ions.
The result of the initial GC-MS screen indicated that the samples were negative for carbamate (including carbaryl), organophosphate, and organochlorine insecticides. GC-MS analysis of extracts from the liver and rumen content samples detected atrazine at a retention time of ~13 min (Figs. 1, 2). Key molecular ions detected for confirmation included 215 m/z (molecular ion), 200 m/z (base peak), 58 m/z, and 173 m/z (Figs. 3, 4). National Institute of Standards and Industry (NIST; https://chemdata.nist.gov/) library matches to atrazine for both samples were >97%. Analysis of the atrazine neat standard (100 ppm; MilliporeSigma) revealed a retention time of 13.07 min, and key molecular ions detected were 215 m/z (molecular ion), 200 m/z (base peak), 58 m/z, and 173 m/z (Figs. 5, 6). GC-MS analysis of control liver fortified with atrazine (positive control) revealed a retention time of 13.07 min with a NIST library match of 97%. Major ions detected for confirmation included 215, 200, 58, and 173 m/z (Figs. 7, 8). The presence of possible metabolites of atrazine was not investigated.
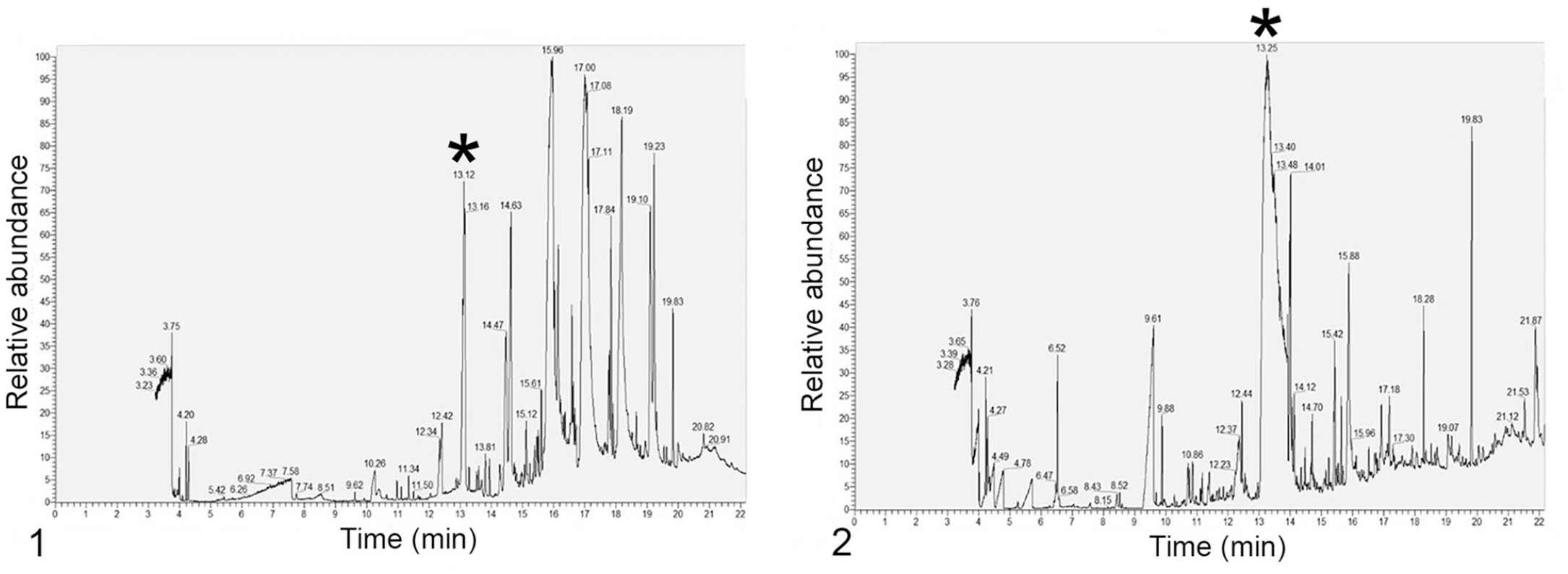
Gas chromatography chromatograms of liver and rumen contents.
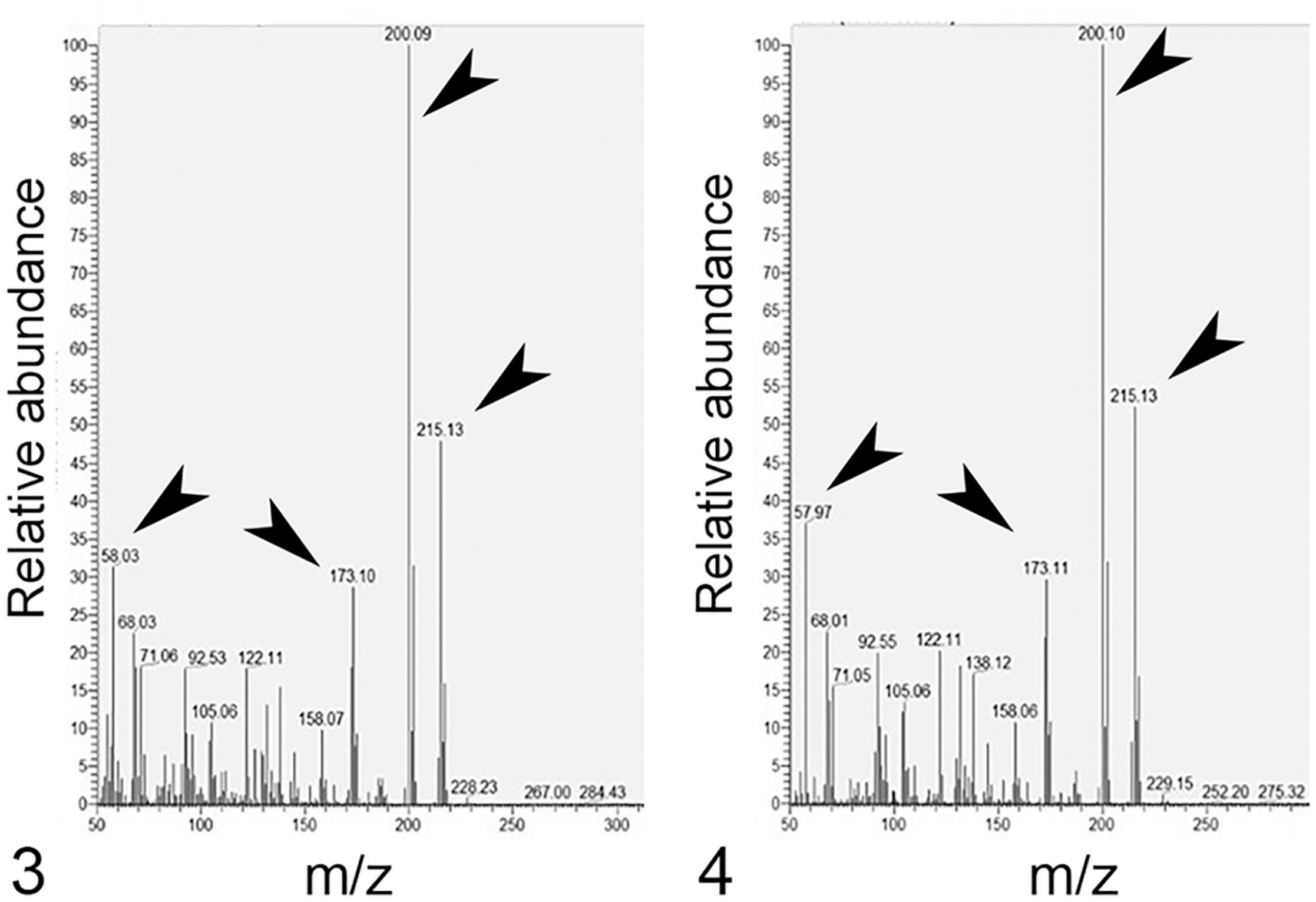
Mass spectra of atrazine detected in liver and rumen contents.
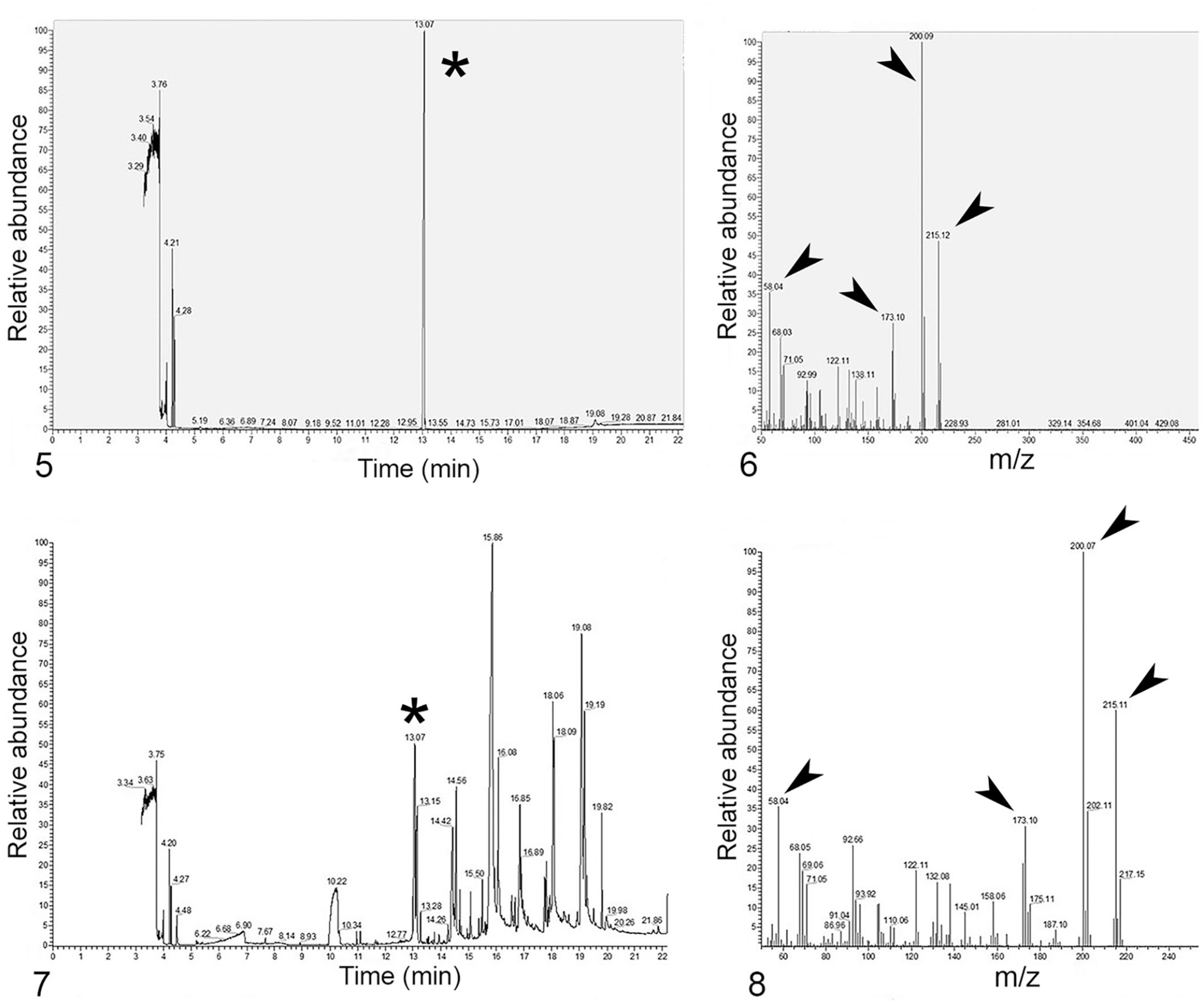
Gas chromatography (GC) chromatograms and mass spectra of atrazine standard and positive control liver.
In one reported case of atrazine intoxication in cattle, 8 of a herd of 73 cattle became acutely ill, and 1 cow died within 6 to 8 h, after consuming ~4, 2.3-kg (5-lb) bags of atrazine (AAtrex 80 w wettable powder [76% atrazine]; Ciba-Geigy). 8 Six more cows died 3 d post-exposure and exhibited clinical signs of anorexia, tenesmus, drooling, weakness, and a stiff gait. The first cow that died was submitted for diagnostic examination. At autopsy, this cow had froth present in the trachea and edematous lungs. Rumen contents, liver, and kidney from this cow were analyzed for atrazine. The rumen contents contained 12,300 ppm atrazine, and replicate analyses of tissue samples revealed average atrazine levels of 75–79 ppm in the liver and 53–67 ppm atrazine in the kidney.
A few studies have been conducted to better understand the effects of atrazine on cattle in experimental settings. In one study, investigators tried to determine if natural grazing of hay treated with atrazine at normal usage concentrations would have an adverse effect on cattle. 7 Six Holstein–Friesian calves were either fed hay sprayed with atrazine at 22.5 kg/ha, hay sprayed with atrazine at 44.9 kg/ha of hay, or untreated hay for 4 wk with a new batch of hay sprayed each week. With respect to the 22.5-kg/ha application, the concentration of atrazine in the hay over the course of the 4-wk period was 342–651 ppm, resulting in an average daily intake of 560 mg/kg. For the 44.9-kg/ha application, the concentration of atrazine in the hay over the 4-wk feeding period was 454–1,029 ppm, resulting in an average daily intake of 840–1,230 mg/kg of atrazine in the calves. There were no clinical signs or adverse effects observed in the calves, and no significant clinical chemistry or CBC abnormalities or histologic lesions. Therefore, it was concluded that cattle grazing on forage sprayed with atrazine at the maximum, recommended use rate did not result in toxicologic effects.
In another study, yearling cattle were dosed orally via drench or with capsules at doses of 10–250 mg/kg with a wettable powder containing 80% atrazine. 11 The toxic dose in the study occurred with 8 doses via drenching or 2 doses via capsule at 25 mg/kg. Signs of intoxication seen at this dosage included muscle spasms in the hindquarters, anorexia, and stilted stance and gait. Lesions found during autopsy included congestion of the liver, lungs, and kidneys, and epicardial petechiae. A single dose of 250 mg/kg administered via drench to one animal resulted in death.
In a third study, 6 cattle were orally administered 500 mg/kg of AAtrex 80 w (~400 mg/kg of atrazine). 9 Within 12–24 h, the cattle had clinical signs of intoxication including slightly increased body temperature, increased respiration and pulse rates, scours, stiffness, ataxia, and drooling. Three cattle were treated with activated charcoal (454 g/animal) at 4, 24, 48, and 72 h after ingestion. The 3 treated cattle survived; the 3 untreated died 2–3 d after ingestion. Lesions found during autopsy of the untreated cattle included hemorrhage in skeletal muscle, thymus, and thyroid glands, along with ecchymotic hemorrhages in the heart, and congestion of the vessels in the brain and the kidneys.
These experimental studies and clinical cases demonstrate that under conditions of normal use, ingestion of atrazine in small quantities over time does not appear to cause overt health issues. However, acute ingestion of large quantities of atrazine, similar to the case received at the ADDL, can cause intoxication.
Given the widespread use of atrazine in commercial agriculture, much study has been devoted to potential environmental intoxication by this chemical and any health issues related to chronic, low-dose exposure, such as carcinogenesis and endocrine disruption. Thus, relatively fewer studies using dosages that might be encountered in a situation of acute toxicity are available. Regulatory agencies within the United States consider atrazine as only mildly acutely toxic for laboratory animals exposed via the oral route, with LD50 values for adult rats exposed via oral gavage of 672–1,471 mg/kg, depending on the sex of rat and the study cited. 1
The mechanism of action of atrazine is not fully understood; however, studies suggest that atrazine may cause toxicity via cellular oxidative stress and mitochondrial dysfunction. In a review of studies using biomarkers of oxidative stress in organisms treated with atrazine or the related triazine herbicide terbuthylazine, the authors concluded that, in general, rodent studies supported the idea that atrazine compromised antioxidant enzyme systems. 12 Studies included in that review used dosages of <0.1–400 mg/kg, indicating that some studies were meant to model chronic, low-dose exposure, whereas others approached situations similar to acute, high-dose exposure. 12 Studies reported in 2017 and 2018 using quail as an animal model have investigated organ-specific responses to atrazine at levels up to 500 mg/kg. In these studies, the authors concluded that atrazine induced mitochondrial dysfunction and oxidative stress in the kidney and liver via modulating the Nrf2 pathway, and in the cerebrum via activation of the NXR response.10,13,14 Thus, multi-organ mitochondrial dysfunction and oxidative stress likely contribute to acute toxicity in cases of high-dose atrazine ingestion.
We have documented here that the qualitative, multi-residue GC-MS method validated for the detection of pesticides and other chemical analytes can isolate and confirm the presence of atrazine in liver and rumen contents. Given the reported clinical signs and gross lesions, as well as detection of atrazine within the clinical samples, atrazine intoxication was considered the likely cause of death and clinical signs observed in this herd.
Footnotes
Acknowledgements
This manuscript was presented within the toxicology section of the 2019 AAVLD annual conference.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
