Abstract
An 8-year-old, crossbred beef cow was referred to the Indiana Animal Disease Diagnostic Laboratory at Purdue University for a complete necropsy in October 2009. The cow was the sixth to die in a 7-day period. Affected cows were reportedly stumbling and became weak, excitable, and recumbent. Histologically, myonecrosis was severe in the skeletal muscles and mild in the heart and tongue. According to the submitter, exposure to a poisonous plant was suspected, and a plant specimen received from this case was identified as white snakeroot (Ageratina altissima). Using the white snakeroot specimen, a gas chromatography–mass spectrometry analytical method for the detection of tremetone and dehydrotremetone (2 components of white snakeroot) was developed. Both tremetone and dehydrotremetone were detected in the plant specimen. Dehydrotremetone was recovered from the liver, while neither component was recovered in the rumen content. In the past, because of the lack of standard reference material, the diagnosis of white snakeroot poisoning was based mainly on history of exposure and the presence of the plant in the rumen. The analytical method described herein can be used to document exposure to tremetone or dehydrotremetone in cases of suspected white snakeroot poisoning when coupled with the appropriate clinical signs and lesions.
White snakeroot (Ageratina altissima, previously Eupatorium rugosum) is common throughout the eastern half of the United States and is known to contain benzofuran compounds and sterols. 4 Livestock that ingest the plant may exhibit signs of weight loss and trembling with progression to recumbency and death. 1 Constipation, a noticeable odor to the breath and urine, excessive salivation and dyspnea have also been reported, 6 with onset of clinical signs varying from less than 2 days to up to 3 weeks after ingestion of white snakeroot. 5 Severe skeletal muscle necrosis accounts for the trembling; the death of the animal is attributed to the cardiotoxic effects. Inadequate feed and exceptionally hot, dry summers increase the risk of consumption of this otherwise relatively unpalatable plant.
White snakeroot plants, easily identified by their white flowering cluster, are common along tree-lined pastures or creeks. Because the toxic compounds pass through milk, calves may be poisoned by suckling, and human beings that consume the milk are at risk of “milk sickness.” 5 The toxic component of white snakeroot may also be present in the meat (muscle tissue) from intoxicated animals. 2
In October 2009, an 8-year-old crossbred beef cow from northwestern Indiana was found dead with no history of disease or exposure to known toxins. According to the submitter, the cow was the sixth to die in a 7-day period. Affected cows were reportedly stumbling and became weak, excitable, and recumbent. The owner suspected plant toxicosis and submitted 3 plant specimens along with the carcass of the cow. One plant was identified as white snakeroot (Fig. 1); the other 2 were nontoxic weeds.

White snakeroot (Ageratina altissima) from the pasture.
At necropsy, skeletal muscles, including semimembranosus, semitendinosus, and psoas, contained irregularly shaped and poorly defined tan foci. Intermuscular fascia around the psoas muscles was expanded by clear, yellow, gelatinous material. In the heart, tan streaks extended from the endocardium through ventricular and interventricular myocardium. Other organs, including the liver, were unremarkable grossly. Histologically, many myocytes throughout sections of the psoas and caudal thigh musculature were swollen with loss of cross striations, vacuolated sarcoplasm, and hypereosinophilic sarcoplasm (Fig. 2). Some affected myofibers were infiltrated by a few macrophages or neutrophils (Fig. 3). Collapsed myotubes contained proteinaceous debris and macrophages, and were surrounded by macrophages and fewer lymphocytes. Muscle interstitium was expanded by edema. The extent of inflammation and myonecrosis varied markedly within and among sections. Rare individual cardiac myocytes were necrotic, with loss of cross striations, sarcoplasmic vacuolation, and densely eosinophilic contraction bands. Lymphocytes and macrophages were scattered throughout the myocardial interstitium. Hepatocytes throughout the liver contained clear cytoplasmic vacuoles, consistent with lipid. In some lobules, centrilobular hepatocytes were more severely affected.
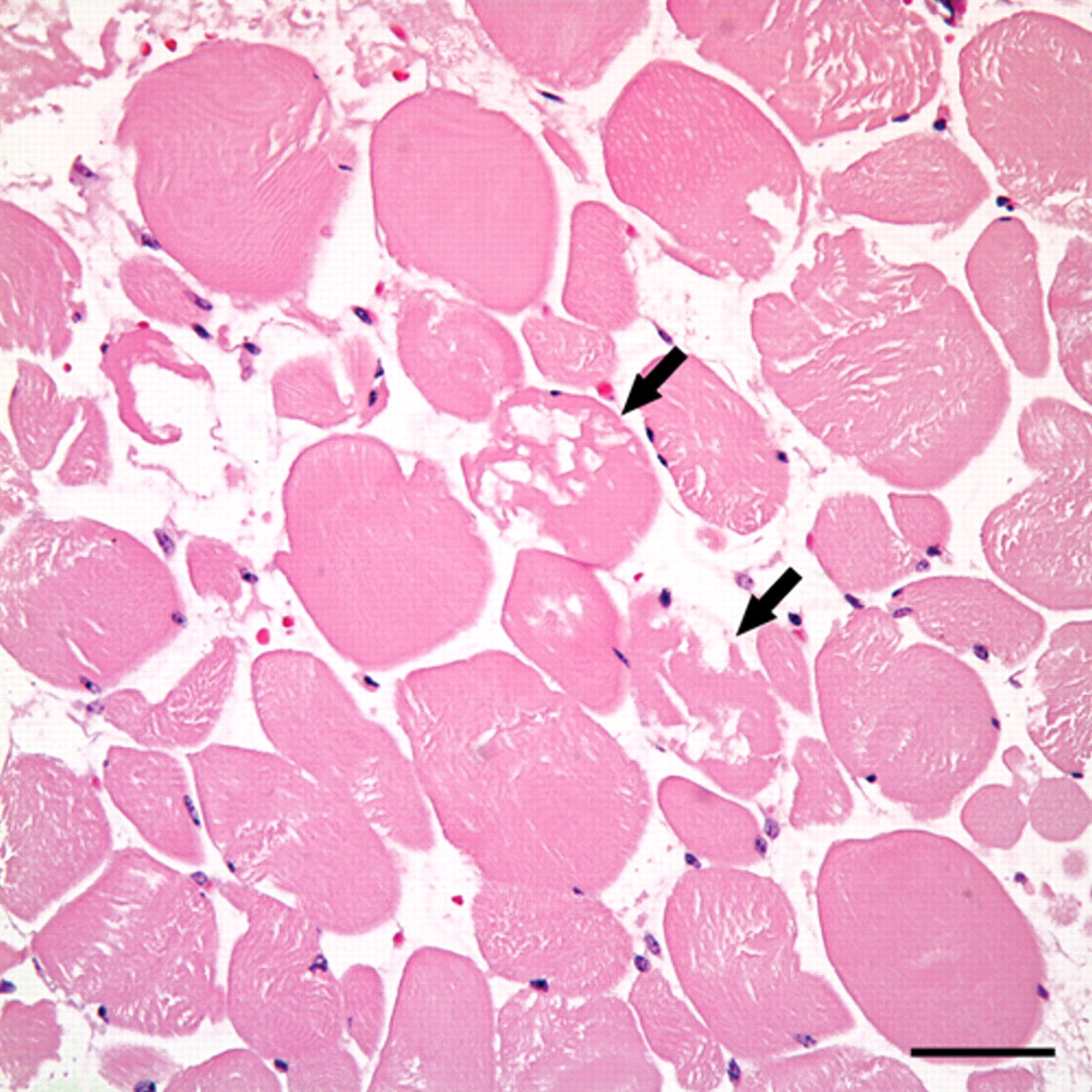
Acute skeletal myonecrosis. Individual myocytes have hypereosinophilic and vacuolated sarcoplasm, and lack cross striations (arrows). Hematoxylin and eosin. Bar = 50 µm.
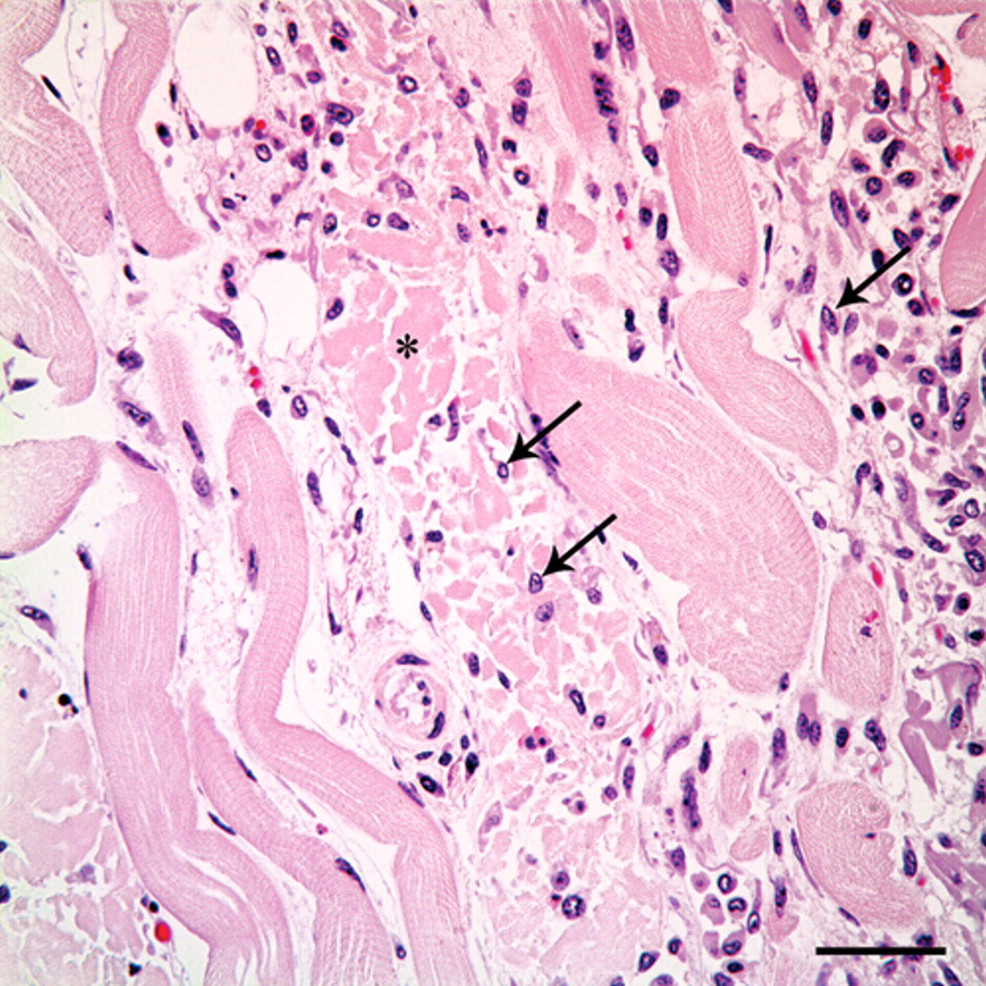
Subacute myonecrosis. Skeletal myocytes have hypereosinophilic and fragmented sarcoplasm, and lack cross striations (asterisk). Multiple macrophages infiltrate necrotic myocytes (arrows). Hematoxylin and eosin. Bar = 50 µm.
The rumen content was visually inspected for white snakeroot plant material, but none was identifiable; therefore, an analytical method was developed to confirm white snakeroot intoxication. Specifically, the developed qualitative method detected 2 known chemical components of white snakeroot: tremetone and dehydrotremetone. Several white snakeroot plants were ground into fine particles and extracted with chloroform. a The extract was concentrated and reconstituted prior to gas chromatography–mass spectrometry (GC-MS) b analysis. The GC-MS was equipped with a 30 m × 0.25 mm × 0.25 µm capillary column. c Chromatographic analyses were performed using the following operating parameters: helium carrier gas, 1 ml/min; injector port temperature, 220°C; initial oven temperature, 100°C held for 0.10 min, followed by a ramp of 7.1°C/min to 300°C, holding for 1.90 min for a total run time of 30.17 min. The mass spectrometer operating temperatures for the ion trap, manifold, and transfer line were 150°C, 40°C, and 220°C, respectively. Mass spectra were acquired in electron impact mode, with ionization delayed during the first 3 min of acquisition. The mass range acquired was 40–650 m/z (mass-to-charge ratio) with an emission current set at 10 µA (microamperes) and maximum ionization time of 25,000 µsec.
Mass fragments were compared to previously published data 3 (Table 1). Two major chromatographic peaks were present in the extract of the white snakeroot reference plant (Fig. 4). The largest peak eluted at 14.7 min and generated a mass spectrum with m/z 187 (relative intensity: 100), 202 (84), and 159 (35). The second largest peak eluted at 15.2 min and generated a mass spectrum with m/z 185 (100), 200 (82), 157 (12). The chromatographic retention times and mass spectral data of the sample were compared to tremetone and dehydrotremetone reference standards. d The retention times were equivalent to those of the standards, and the spectral data were found to match 92% and 94% of the qualifier ions for tremetone and dehydrotremetone, respectively.
Mass spectral data for tremetone and dehydrotremetone.*
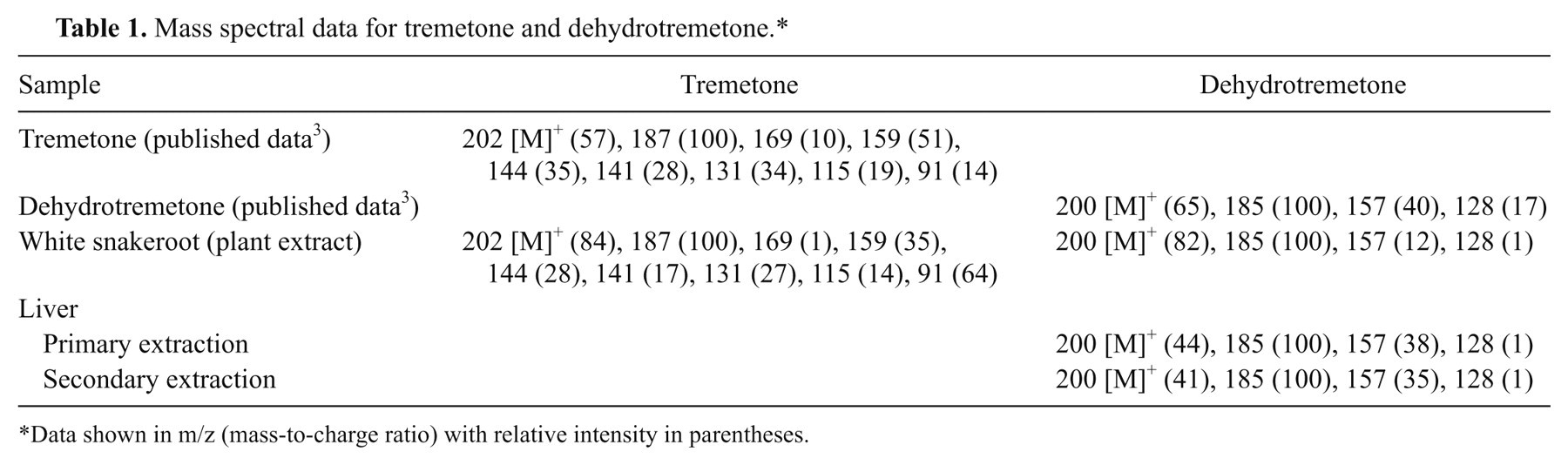
Data shown in m/z (mass-to-charge ratio) with relative intensity in parentheses.
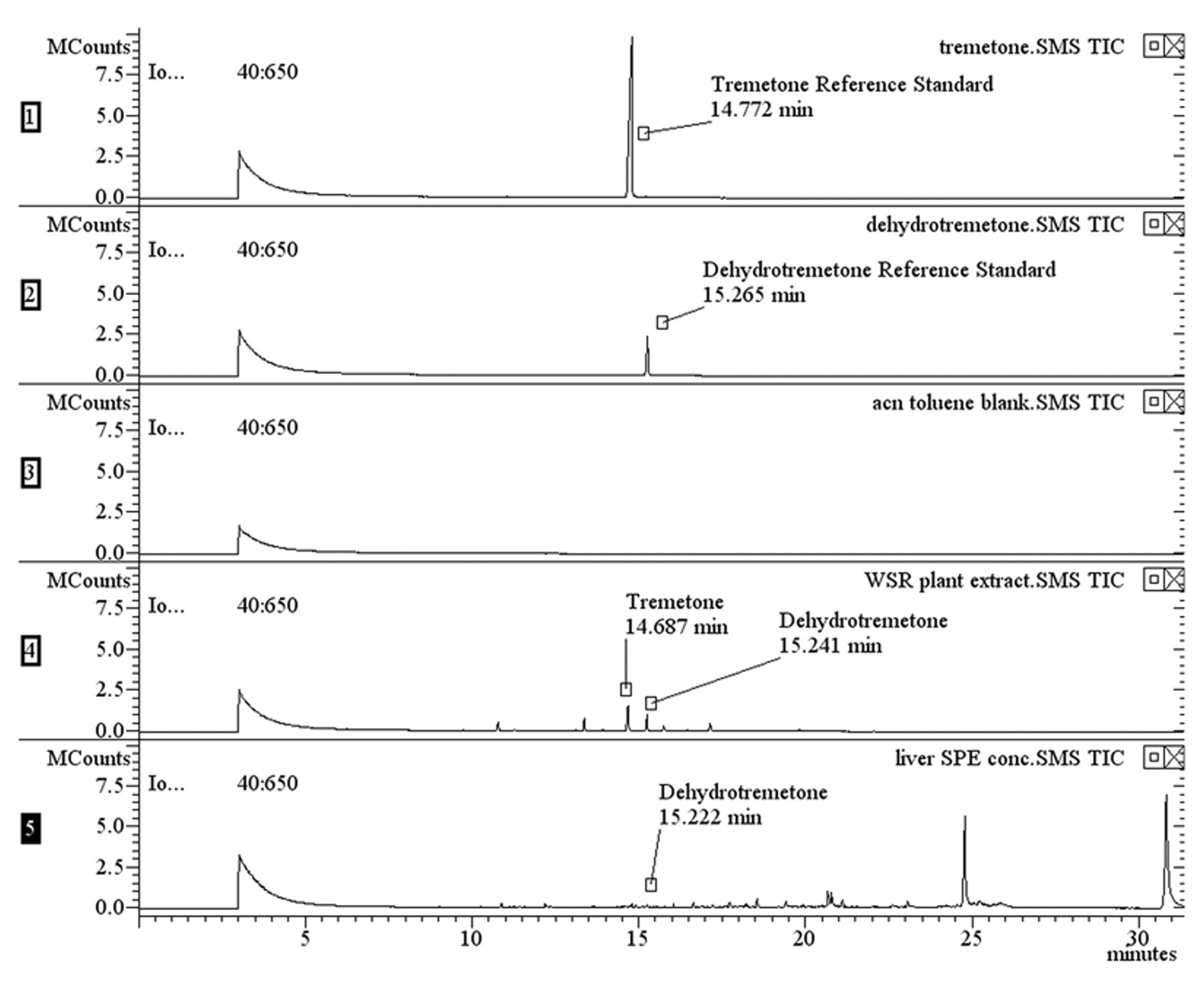
Chromatogram of reference standards, blank, plant extract, and liver.
Rumen content was extracted using the same protocol as the plant material; however, neither tremetone nor dehydrotremetone were detected. This could be attributed to sampling bias, sampling size, or the fact that white snakeroot plant material was no longer present in the rumen.
Liver was extracted with a 9-to-1 ratio of hexane e -to-acetone. f The extract was concentrated and fatty acids and other nonspecific analytes removed using a dual-layer solid phase extraction (SPE) cartridge. g Recovery of tremetone and dehydrotremetone was verified using the SPE protocol on the reference standards and the white snakeroot plant extract. Both analytes were recovered using the SPE cartridge and were detectable by GC-MS. Although several election impact mass fragments observed in the liver matched those of tremetone, the ion intensities were not strong enough to determine a positive match to the reference standard. However, a peak eluted at 15.2 min that had a mass spectrum with m/z (relative intensity) 185 (100), 200 (44), 157 (38). The chromatographic retention time and mass spectral data were compared to the dehydrotremetone reference standard. d The retention time was equivalent to the standard, and the spectral data was found to be a 73% match (Figs. 4, 5). A secondary extraction of the liver solids with chloroform a and SPE cleanup, as described previously, confirmed the presence of dehydrotremetone. A peak at 15.2 min that generated a mass spectrum with m/z (relative intensity) 185 (100), 200 (41), 157 (35) was observed. The spectral data were compared to the dehydrotremetone reference standard d and was found to be a 74% match. Therefore, dehydrotremetone was detected in the liver from the cow, and white snakeroot toxicosis was confirmed.
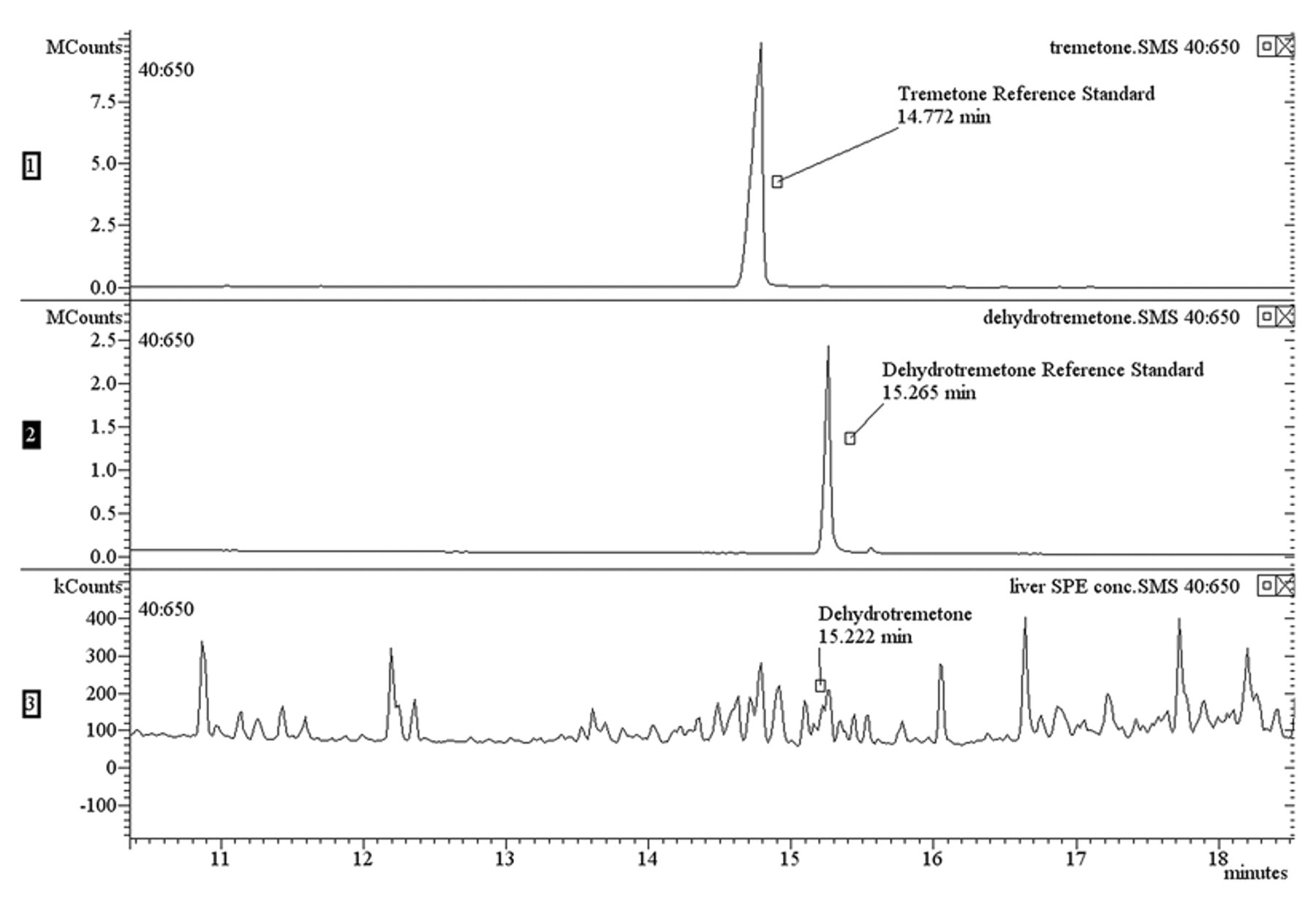
Expanded region of liver chromatogram showing dehydrotremetone.
Until the development of the GC-MS analytical method described herein for the detection of tremetone and dehydrotremetone and the availability of standard reference material, the diagnosis of white snakeroot toxicosis has been based mainly on characteristic histologic lesions in myocardium and skeletal muscle and known exposure to the plant. In ruminants, the intact plant or fragments of the plant can often be detected by visual examination alone; however, confirmatory chemical assays of liver may be required when the rumen content contains no visible white snakeroot or in cases submitted from nonruminant species.
Footnotes
Acknowledgements
The authors would like to thank the USDA-ARS Poisonous Plant Research Laboratory for donation of the standard reference material.
a.
Honeywell International, Morristown, NJ.
b.
Agilent Technologies Inc., Santa Clara, CA.
c.
FactorFour™ VF-5ms, Agilent Technologies Inc., Santa Clara, CA.
d.
U.S. Department of Agriculture, Agricultural Research Service, Poisonous Plant Research Laboratory, Logan, UT.
e.
Mallinckrodt Baker Inc., Phillipsburg, NJ.
f.
VWR International, Radnor, PA.
g.
Supelco Supelclean™ ENVI-Carb™ II/PSA, Sigma-Aldrich, St. Louis, MO.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
