Abstract
This study was performed to investigate the biochemical effect of flaxseed oil on oxidative stress and brain monoamines release in streptozotocin-induced diabetic rats. Sixty male albino rats were divided into following four groups (15 for each group): control group, flaxseed oil group, diabetic group, and flaxseed oil-treated diabetic group. Serum glucose, insulin, pentosidine, plasma advanced oxidation protein products (AOPPs), and plasma total antioxidant capacity were estimated. Brain neurotransmitters, malondialdehyde (MDA), and nitric oxide (NO) were also determined. The mean values of serum pentosidine and plasma AOPP showed a significant decrease in treated diabetic group as compared to their values in the diabetic group. Also, brain neurotransmitters levels were improved after treatment with flaxseed. Brain MDA and NO were increased significantly in the diabetic group, while they were significantly decreased after treatment. Brain NO and brain MDA had a significant positive correlation with pentosidine, AOPP, and neurotransmitters. We concluded that flaxseed oil supplementation may be useful in the treatment of brain dysfunction in diabetes.
Introduction
Diabetes mellitus is a complex disease associated with peripheral and central complications. These complications include retinopathy, nephropathy, and neuropathy. 1 Several investigations have confirmed the role of oxidative stress in the development of diabetic-mediated disorders, possibly via the formation of free radicals. 2 Hyperglycemia induces vascular damage probably through a single common pathway, increased intracellular oxidative stress, linking four major mechanisms, namely, the polyol pathway, advanced glycation end products (AGEs) formation, and the protein kinase C—diacylglycerol and the hexosamine pathways. 3
Due to prolonged high glucose levels in diabetes, reactive derivatives are formed via nonenzymatic condensation reaction named Millard reaction between reducing glucose and amine residues of proteins, lipids, or nucleic acids that undergo a series of complex reactions such as rearrangement, dehydration, and condensation to give an irreversible cross-linked complex group of compounds termed AGEs. 4 Examples of some of the best chemically characterized AGE-modified sites in humans with diabetes are carboxymethyllysine, carboxyethyllysine, and pentosidine, which act as markers for the formation and accumulation of AGEs. 5
Diabetes is associated with marked changes in brain monoamines content and transport in both rodents and humans 6 ; however, conflicting results have been reported. Chronic antioxidant therapy may be useful in decreasing the risk of diabetic complications. As an antioxidant, flaxseed oil can cause a significant reduction in blood glucose level 7 and an increase in insulin level in diabetic-treated rats compared to diabetic rats by scavenging reactive oxygen species (ROS). Flaxseed (Linum usitatissimum L.) is a multipurpose crop and its consumption is beneficial for human health. The nutritional components of flaxseed are oil, proteins, lignans, fibers, and vitamins. Additionally, diet supplement with flaxseed ameliorated the antioxidant enzyme activities observed in diabetic rats and significantly decreased malondialdehyde (MDA) levels. 8 This study aimed to investigate the brain oxidative stress induced by streptozotocin (STZ) in rats with consequent changes in brain neurotransmitters and examined the potential protective role of flaxseed oil against the changes due to diabetes.
Materials and methods
Materials
Chemicals
Norepinephrine, dopamine, serotonin, pentosidine high-performance liquid chromatography (HPLC) standards, and STZ were purchased from Sigma Aldrich Chemicals Company (St Louis, Missouri, USA). Flaxseed oil was purchased from local market, Egypt. All other chemicals were of HPLC grade and purchased from Sigma.
Experimental animals
Sixty male Sprague Dawley rats weighing 100–120 g were obtained from the animal house of the National Research Centre (NRC), Giza, Egypt. The animals were housed in individual suspended stainless steel cages at the temperature range of 22 ± 2°C, under a 12-h light/12-h dark cycle, and allowed to acclimatize for a period of 15 days to the experiment; rats were allowed free access to food and water. The guidelines of the ethical care and treatment of the animals followed the regulations of the ethical committee of NRC. The number of ethical committee permission is (10/219). The study was conducted in concordance with relevant European directives.
Methods
Induction of diabetes
STZ was dissolved in 50 mM sodium citrate solution (pH 4.5) containing 150 mM sodium chloride. The solution (6 mg/100 g body weight (b.w.)) was subcutaneously administered in rats. Fasting blood sugar was estimated after 3 days to confirm the development of diabetes mellitus. 9
Experimental design
Sixty male rats were divided into 4 groups, with 15 rats in each group, and were classified as follows: group I (control group), healthy rats received 1.2 ml corn oil/kg b.w./day orally; group II (flaxseed oil group), healthy rats received 1.2 ml flaxseed oil/kg b.w./day orally; group III (diabetic group), diabetic rats received 1.2 ml corn oil/kg b.w./day orally; and group IV (flaxseed oil-treated diabetic group), diabetic rats received 1.2 ml flaxseed oil/kg b.w./day orally 10 by stomach tube.
Samples collection
After the experimental period (8 weeks), animals were kept fasting for 12 h, then anesthetized, and the blood was withdrawn from the retro-orbital venous plexus of the eye using heparinized capillary tubes. Blood samples were collected, left to clot, and then centrifuged at 3000 r/min for 15 min to separate the serum. The serum was stored at −20°C for pentosidine determination. Blood was centrifuged at 2000 r/min for 10 min using a cooling centrifuge. Plasma was separated and immediately frozen for other biochemical parameters. Brains were removed quickly and placed in iced normal saline, perfused with the same solution to remove blood cells, plotted on filter paper, and frozen at −80°C until use for estimation of brain monoamines, MDA, and nitric oxide (NO).
Preparation of tissue homogenate
The frozen tissues were cut into small pieces and homogenized in phosphate buffer (pH 7.4), then centrifuged at 4000 r/min for 15 min at 4°C, and the supernatant was removed for estimation of chemical parameters. 11
Biochemical assays
Freshly prepared serum was used for determination of blood glucose according to Trinder. 12 Plasma insulin level was estimated by enzyme-linked immunosorbent assay (ELISA) kit. 13 Reverse-phase HPLC was used for the determination of the plasma pentosidine content. Briefly, 50 µl of plasma were lyophilized and then hydrolyzed by 50 µl of 6 N hydrochloric acid at 110°C under nitrogen atmosphere for 16 h, subsequently neutralized with 100 µl of 5 N sodium hydroxide and 200 µl of 0.5 M phosphate buffer (pH 7.4), and then filtered through a 0.45-μm Millipore filter (Billerica, Massachusetts, USA) and diluted 20-fold with phosphate-buffered saline. Filtered samples (50 µl) were then injected into C18 reverse phase analytical column (218TP104, Vydac; Separation Group, Hesperia, California, USA) using a fluorescence detector at excitation/emission wavelengths of 335/385 nm 14 by gradient method using auto nitrile/water as mobile phase with a flow rate 1 ml/min. Brain MDA was measured using thiobarbituric acid reaction. 15 The protein content of the homogenate was determined by the method of Lowry et al. 16 using bovine serum albumin as standard. Total antioxidant capacity (TAC) was measured kinetically using commercial kits purchased from Biodiagnostics (Cairo, Egypt). 17 Determination of advanced oxidation protein products (AOPPs) 18 was done with double antibody sandwich ELISA. The kit was obtained from Glory Science Co. Ltd (Del Rio, Texas, USA).
Determination of brain monoamines
Brain homogenate used for the determination of brain norepinephrine, dopamine, and serotonin was carried out using HPLC system, Agilent technologies 1100 series (Santa Clara, California, USA), equipped with a quaternary pump (G131A model). Separation was achieved on octadecylsilyl-reversed phase column (C18, 25 × 0.46 cm2; internal diameter: 5 μm). The mobile phase consisted of potassium phosphate buffer/methanol 97/3 (v/v) and was delivered at a flow rate of 1.5 ml/min. Ultraviolet detection was performed at 270 nm, and the injection volume was 20 µl. The concentration of both catecholamines and serotonin was determined by external standard method using peak areas. Serial dilutions of standards were injected and their peak areas were determined. A linear standard curve was constructed by plotting peak areas versus the corresponding concentrations. The concentration in samples was obtained from the curve. 19
Statistical analysis
All data were expressed as mean ± standard error. Data were analyzed using one-way analysis of variance using Statistical Package for the Social Sciences (Version 12). Duncan’s new multiple range test was used to assess differences between means. Pearson’s correlation test was used to assess correlations between means. A significant difference was considered at the level of p < 0.05.
Results
Diabetes was confirmed by estimating the blood glucose levels in the diabetic and the diabetic flaxseed groups. The diabetic and the diabetic flaxseed groups showed higher glucose levels than the control group (Table 1). There were no significant changes of plasma insulin level in all groups. As shown in Table 2, the serum level of pentosidine was significantly increased in the diabetic rats than in controls, but after treatment by flaxseed oil this value significantly decreased. Plasma AOPP level increases significantly in the diabetic group compared with the control group; on the other hand, plasma TAC level decreases significantly in diabetics group than controls, while the two parameter levels improved after treatment with flaxseed oil (Table 2). The levels of brain MDA and NO were significantly increased in the diabetic group compared with the control group; however, these increases were significantly attenuated after treatment with flaxseed oil. Compared with the control group, brain norepinephrine and dopamine levels were significantly increased in the diabetic group; however, flaxseed oil significantly decreased dopamine level and tended to decrease norepinephrine level compared with the diabetic group (Table 3). On the other hand, there was no significant change in serotonin level in all groups. Our study indicated that there is a positive correlation between MDA, NO, and all parameters in the study (Table 4). Serotonin has not been changed in treated groups.
Blood glucose and insulin in the different studied groups.

Values are expressed as mean ± SE. n denotes number of rats in each group.
ap < 0.05: significant difference compared with diabetic group.
bp < 0.05: significant difference compared with the control group.
Oxidants/antioxidants parameters in the different studied groups.

MDA: malondialdehyde; AOPP: advanced oxidation protein product; NO: nitric oxide; SE: standard error; n: number of rats in each group; TAC: total antioxidant capacity.
Values are expressed as mean ± SE.
ap < 0.05: significant difference compared with diabetic group.
bp < 0.05: significant difference compared with the control group.
Brain monoamines parameters in the different studied groups.
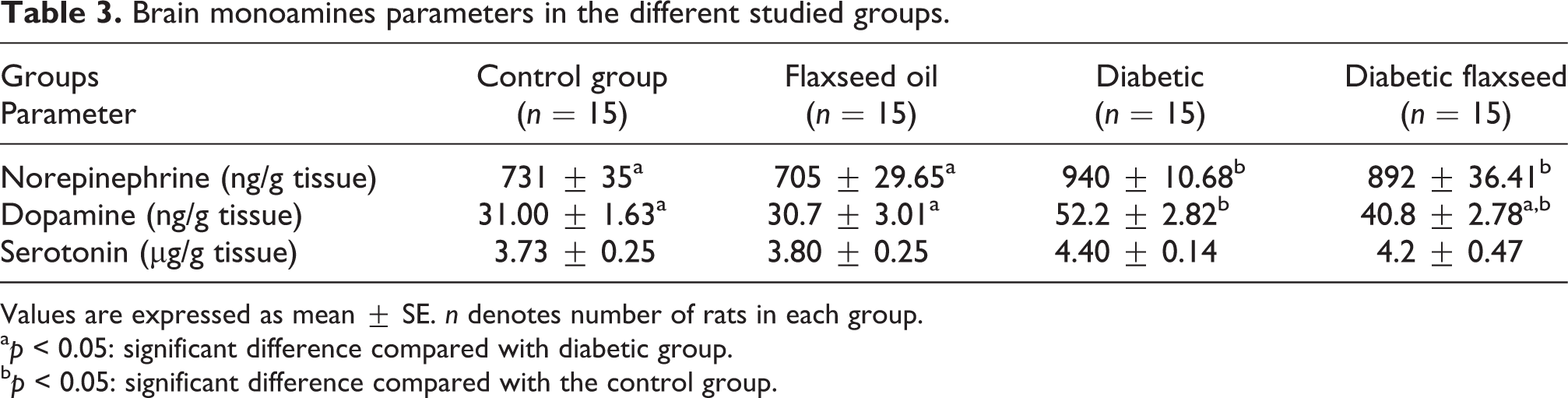
Values are expressed as mean ± SE. n denotes number of rats in each group.
ap < 0.05: significant difference compared with diabetic group.
bp < 0.05: significant difference compared with the control group.
Correlation between MDA, NO, and all parameters in the study.

NO: nitric oxide; MDA: malondialdehyde; AOPP: advanced oxidation protein product.
p < 0.05: correlation is significant.
Discussion
In this study, the rats treated with STZ developed hyperglycemia. The increase in the serum glucose could be due to a decrease in the levels of serum insulin as a result of destruction of the pancreas by STZ and generation of ROS. 20 This study demonstrated that in diabetic rats brain neurotransmitters significantly increased, which may be related to oxidative stress since a positive correlation was observed in this study between MDA, NO, and neurotransmitters. This finding was confirmed by other studies. 19,21
Araki and Nishikawa 22 demonstrated that increased oxidative stress could be one of the common pathogenic factors of diabetic complications (like peripheral neuropathy) as well as in neurodegenerative disorders like Alzheimer’s disease and Parkinson’s disease. 23 Oxidative damage is generally attributed to the formation of highly reactive OH, which leads to severe oxidative damage of the cells’ components like lipids, proteins, and DNA. 24 Efficiency of enzymatic and nonenzymatic antioxidative systems could be detected by the determination of single components of this system or by the so-called TAC. 17
Recent study demonstrated that neurons develop hyperexcitability in STZ-induced diabetic rats; this may be attributed to certain disturbances in neurotransmitters pattern. 21 Consequently, tissue cells isolated from hyperglycemic animals demonstrated increase in norepinephrine and dopamine production/secretion. 25
Firoozrai et al. 26 reported that MDA is produced by breakdown of lipids under oxidative stress conditions. Evidence of lipid peroxidation has been reported to be present in a number of diabetic complications 27 and it significantly contributes to endothelial dysfunction, which is a key event in the pathogenesis of diabetic microvascular disease.
In this study, the lipid peroxidation marker (MDA) was elevated in diabetic rats as reported previously. 28 The increase in lipid peroxidation might be a reflection in enzymatic and nonenzymatic antioxidants of defense systems. 8
This study demonstrated that diabetic rats that were treated with flaxseed oil enriched with omega-3 significantly decrease brain norepinephrine and dopamine levels. In addition, there was a significant decrease in glucose, MDA, and NO levels that indicate a decrease in oxidative stress. The result of this study suggested that flaxseed oil contains a free radical scavenging activity, which could exert a beneficial effect against pathological alterations caused by the presence of
Oxidative stress plays a central role in the pathogenesis of metabolic diseases like diabetes mellitus and its complications (like peripheral neuropathy) due to its great oxygen consumption, high lipid and poor antioxidants defense. Oxygen free radicals attack objects on the polyunsaturated components of membranes and may cause a serious organizational dysfunction within cells and tissues. It has been suggested that the use of omega-3 polyunsaturated fatty acids (PUFAs) may have ameliorating effect on such damage by two possible ways. First, omega-3 PUFA may increase the level of catalase within the peroxisome and in the cytoplasm resulting in enhanced defense against free oxygen radicals. Second, omega-3 PUFAs, which have been supplemented, may be replaced with PUFA components of the membranes that had been attacked by oxygen free radicals. 21
In our study, pentosidine is significantly higher in diabetic rats than the controls. This result is in agreement with Kerkeni et al. 29 who demonstrated that pentosidine might accelerate the development of microvascular complications both by accumulation of AGEs in the vessel walls and by causing endothelial dysfunction, as mediated by the AGE-AGE-receptor axis activity. The mean levels of pentosidine in diabetic group that was modulated with flaxseed oil orally were significantly decreased as compared to the diabetic group but still not reached the levels of the control group. The effectiveness of flaxseed oil in diabetes could be because of its antioxidant activity. 30
Recently, Yamagishi et al. 31 showed the role of AGEs and oxidative stress in vascular complications in diabetes. The authors demonstrated that AGEs induce mesangial cell loss and dysfunction, glomerulosclerosis, and tubulointerstitial fibrosis, taking into account the fact that patients with diabetes are prone to microvascular disease. In the same line, the elevation of AOPP in diabetic rats of this study may be due to the generation of ROS, this result is in agreement with the results of Abou-Seif and Youssef 32 who reported that AOPPs are formed during oxidative stress by the action of chloramines produced by myeloperoxidase in activated neutrophils and is accumulated in biological systems and thus caus damage to biological membranes.
Conclusion
STZ-induced diabetes mellitus is mediated through ROS and prevention of diabetic complications by flaxseed oil is due to its antioxidant activity.
Footnotes
Acknowledgment
Authors are grateful to the National Research Centre, Giza, Egypt, for providing space to carry out this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
