Abstract
Cisplatin (CP) is known as one of the most potent chemotherapeutic antitumor drugs. The tissue-specific toxicity of CP in the kidneys is well documented. However, at higher doses less common toxic effects such as hepatotoxicity may arise. Since CP remains one of the most effective antineoplastic drug used in chemotherapy, strategies to protect tissues against CP toxicity are of clinical interest. Recently, ω-3 polyunsaturated fatty acids (PUFAs) from certain plants/seeds notably flaxseed have shown numerous health benefits. In view of this, the present study investigates the protective effect of flaxseed oil (FXO) on CP-induced damage in liver. Rats were pre-fed normal diet and the diet rich in FXO for 10 days and then a single dose of CP (6 mg/kg body weight) was administered intraperitoneally while still on diet. Serum/urine parameters, enzymes of carbohydrate metabolism and oxidative stress were analyzed. CP caused perturbation of the antioxidant defense as reflected by the decrease in the activities of catalase, superoxide dismutase and glutathione peroxidase. Further the activities of various enzymes involved in glycolysis, tricarboxylic acid cycle, gluconeogenesis and hexose monophosphate shunt pathways were determined and were found to be differentially altered by CP treatment. However, these alterations were ameliorated in CP-treated rats fed on FXO. Present results show that dietary supplementation of FXO in CP-treated rats ameliorated CP-induced hepatotoxic and other deleterious effects due to its intrinsic biochemical/antioxidant properties.
Introduction
Cisplatin (cis-diamminedichloroplatinum II (CP)) is one of the most effective and widely used chemotherapeutic agents in the treatment of a variety of human solid tumors. 1 The CP-induced ototoxicity and nephrotoxicity have been very well studied in both clinical and animal research, however hepatotoxicity has been rarely paid attention to. Recent studies have suggested that hepatotoxicity is also a major dose-limiting side effect in CP-based chemotherapy. 2 –4 Continued aggressive high-dose CP chemotherapy necessitates the investigation of ways for prevention of the dose-limiting side effects that inhibit the CP administration at tumoricidal doses. CP chemotherapy induces a fall in plasma antioxidant levels, which may reflect a failure of the antioxidant defense mechanism against oxidative damage induced by commonly used antitumor drugs. 5 A relationship between oxidative stress and CP toxicity has been suggested in many experimental models. Several reports have implicated free radicals and reactive oxygen species (ROS) in the toxicity of CP. CP-induced toxicity is closely associated with an increase in lipid peroxidation (LPO), decreased levels of protein bound sulfhydryl groups and glutathione which indicate the formation of ROS. 6 –8
Until now a large number of studies have been focused on the ways for prevention of CP side effects via supplementation of preventive agents simultaneously. 9,10 The past 3 decades have been a period of rapid expansion in the scientific knowledge of ω-3 PUFAs. The most common way to consume ω-3 fatty acids has been in the form of marine oils like fish. Studies from our laboratory have shown that dietary fish oil (FO) supplementation ameliorates gentamicin and CP-induced damage to kidney and liver, respectively. 4,11 Vegetable sources including grains and oils offer an alternative source for those who are unable to regularly consume fish for religious or other reasons. 12 Flaxseed (Linum usitatissimum) is the richest dietary sources of ω-3 among plant sources. Recently flaxseed has been identified as a significant alternative source of ω-3 fatty acids. 13,14 Dietary supplementation with flaxseed oil (FXO) affects the biochemistry of fatty acid metabolism and thus the balance of proinflammatory mediators and atherogenic lipids, holding a great promise for modulating inflammatory diseases. 15 FXO has also been shown to increase the life span of irradiated mice, suggesting its prophylactic potential against radiation-induced degenerative changes in liver. 16 However, the protective effect of FXO on CP-induced toxicity has not yet been investigated.
Taking into consideration the potential clinical use of CP and numerous health benefits of FXO, the present work was undertaken to study the detailed biochemical events/cellular response/mechanisms of CP-induced hepatotoxicity and its protection by dietary FXO. We hypothesized that FXO would prevent CP-induced hepatotoxicity due to its intrinsic biochemical and antioxidant properties that would lead to improved metabolism and antioxidant defense mechanism of the liver. The results obtained indicate that dietary supplementation with FXO markedly ameliorated CP-induced adverse effects in liver. The activities of carbohydrate metabolism, membrane and antioxidant enzymes were markedly enhanced by FXO feeding to CP-administered rats. These studies support a potential therapeutic use of CP + FXO combination in combating cancer without hepatotoxic and other harmful side effects.
Materials and methods
Chemicals and drugs
Flaxseed oil
Omega Nutrition Canada Inc., Cisplatin (Sigma Chemical Co., USA). All other chemicals used were of analytical grade and were purchased either from Sigma Chemical Co. (St Louis, MO, USA) or from SRL, Mumbai, India.
Diet
A nutritionally adequate laboratory pellet diet was obtained from Aashirwaad Industries, Chandigarh (India). Pellets were crushed finely and mixed with 15% FXO and stored in airtight containers. Vitamin E as
Experimental design
The animal experiments were conducted according to the guidelines of Committee for Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India. Adult male Wistar rats (8 rats/group) weighing between 150 and 200 g were used in the study. Animals were acclimatized to the animal facility for a week on standard rat chow and allowed water ad libitum under controlled conditions of 25 ± 2°C temperature, 50 ± 15% relative humidity and normal photoperiod (12 h dark and light). Four groups of rats entered the study after acclimatization (Figure 1). They were fed on either normal diet (control and CP groups) or diet containing 15% FXO (CPFXO and FXO groups). After 10 days, rats in 2 groups (CP and CPFXO) were administered a single dose of CP intraperitoneally (6 mg/kg body weight) in 0.9% saline. Animals in the control and FXO group received an equivalent amount of normal saline. The rats were killed 4 days after CP administration under light ether anesthesia. Blood and urine samples were collected and liver was removed and processed for the preparation of homogenate as described below. All the preparations and analyses of various parameters were performed simultaneously under similar experimental conditions to avoid any day-to-day variations. Body weights of rats were recorded at the start and completion of the experimental procedures.

Experimental design (ND: normal diet, C: control, FXO: flaxseed oil diet, CP: cisplatin, CPFXO: flaxseed oil + cisplatin treated, i.p.: intraperitoneal; CP injection (⇑); normal saline injection (↑).
Preparation of homogenates
After the completion of treatment schedule, animals were killed, liver was carefully removed from the treated and control animals and homogenized in 0.1 M Tris-HCl buffer (pH 7.5) by a glass-teflon homogenizer (Thomas PA, USA) by passing 5 pulses; at 4°C to make a 10% (w/v) homogenate. The homogenate was then subjected to high-speed Ultra-Turrex Kunkel homogenizer (Type T-25, Janke & Kunkel GMBH & Co. KG. Staufen, Germany) for 3 strokes of 30 s each with an interval of 30 s between each stroke. Homogenate was centrifuged at 2000 r/min at 4°C for 10 min in Beckman J2-M1 (Beckman instruments, Inc Palo Alto, CA, USA) high-speed refrigerated centrifuge to remove the cell debris. The aliquots of homogenates were saved and stored at --20°C for various enzyme activities.
Serum/urine chemistries
Serum samples were deproteinated with 3% trichloroacetic acid in a ratio of 1:3, left for 10 min and then centrifuged at 2000g for 10 min. The protein free supernatant was used to determine inorganic phosphate (Pi) and creatinine. The precipitate was used to quantitate total phospholipids (PLs). Blood urea nitrogen (BUN) and cholesterol levels were determined directly in serum samples. These parameters were determined by standard procedures as mentioned in a previous study. 17 Transaminases, alanine aminotransferase (ALT) and aspartate aminotransferases (AST) were estimated by Reitman and Frankel method using kit from span diagnostics. Glucose was estimated by o-toluidene method using kit from Span diagnostics, Mumbai, India.
Assay of carbohydrate metabolism enzymes
The activities of the enzymes involving oxidation of nicotinamide adenine dinucleotide or reduction of nicotinamide adenine dinucleotide phosphate (NADP) were determined spectrophotometrically on Cintra 5 fixed for 340 nm using 3 ml of assay in a 1-cm cuvette at room temperature (28–30°C). The enzyme activities of lactate dehydrogenase (LDH, E.C.1.1.1.27), malate dehydrogenase (MDH, E.C.1.1.1.37), malic enzyme (ME, E.C.1.1.1.40), glucose-6-phosphate dehydrogenase (G6PDH, E.C.1.1.1.49), glucose-6-phosphatase (G6Pase, E.C.3.1.3.3) and fructose-1,6-bisphosphatase (FBPase, E.C.3.1.3.11) were assayed as described by Khundmiri et al. 17 Hexokinase (HK) was estimated by the method of Crane and Sols 18 and the remaining glucose was measured by method of Nelson. 19
Assay of membrane enzymes and lysosomal marker enzyme
The activities of membrane marker enzymes, alkaline phosphatase (ALP), leucine amino peptidase (LAP), γ-glutamyl transferase (GGTase) and lysosomal marker enzyme, acid phosphatase (ACPase) were determined as described by Farooq et al. 20
Assay of enzymes involved in free radical scavenging
Superoxide dismutase (SOD, E.C.1.15.1.1) was assayed by the method of Marklund and Marklund. 21 Catalase (CAT, E.C.1.11.1.6) and glutathione peroxidase (GSH-Px, E.C. 1.11.1.9) activities were assayed by the method of Giri et al. 22 and Flohe and Gunzler, 23 respectively.
Lipid peroxidation and total –SH group estimation
Total SH groups were determined by the method of Sedlak and Lindsay 24 and LPO by the method of Ohkawa et al. 25
Statistical analyses
All data are expressed as mean ± SEM for at least 4–5 different preparations. Statistical evaluation was conducted by one-way analysis of variance (ANOVA) using Origin 6.1 software. A probability level of p < 0.05 was selected as indicating statistical significance. Most of the changes between various groups were compared with control values for better understanding and clarity.
Results
The present work was undertaken to study detailed mechanism of CP-induced hepatotoxicity and other deleterious effects and its possible protection by feeding ω-3 fatty acids–enriched diet to the rats. To address our hypothesis, the effect of CP alone and in combination with FXO, a rich source of ω-3 fatty acids, was determined on various enzymatic and nonenzymatic parameters of oxidative stress and enzymes of carbohydrate metabolism in rat liver.
Effect of dietary FXO oil on CP-induced alterations in serum and urinary parameters
Results summarized in Tables 1 and 2 show the effect of CP alone and in combination with FXO on blood and urine chemistries.
Effect of flaxseed oil (FXO) on serum parameters with or without CP treatment a

CP: cisplatin treated; CPFXO: flaxseed oil + cisplatin treated; FXO: flaxseed oil diet; BUN: blood urea nitrogen; ALT: alanine aminotransferases; AST: aspartate aminotransferases.
aResults are mean ± SEM for five different experiments.
bSignificantly different from control at p < 0.05 by one-way ANOVA. Values in parentheses represent percent change from control.
cSignificantly different from CP at p < 0.05 by one-way ANOVA.
Effect of flaxseed (FXO) on urine parameters with/without CP treatment a

CP: cisplatin treated; CPFXO: flaxseed oil + cisplatin treated; FXO: flaxseed oil diet.
aResults are mean ± SEM for five different experiments. Values in parentheses represent percent change from control.
bSignificantly different from control at p < 0.05 by one-way ANOVA.
cSignificantly different from CP at p < 0.05 by one-way ANOVA.
The liver dysfunction induced by CP was characterized by the elevated levels of serum transaminases, ALT and AST. CP treatment to control rats resulted in significant increase in serum transaminases (ALT, +106% and AST, +154%), creatinine (Scr, +176%), BUN (+122.31%) and PL (+77%) but decrease in Pi (−17%) and glucose (−79%) compared to control rats. These changes were associated with profound phosphaturia, proteinuria and glucosuria accompanied by decreased creatinine clearance. FXO diet alone caused decrease in Scr, cholesterol and PL accompanied by significant increase in creatinine clearance.
Dietary supplementation of FXO diet prior to and following CP administration resulted in significant reversal of various CP-elicited deleterious effects on serum and urine parameters. FXO prevented CP-induced increase in transaminases, Scr and BUN.
Effect of dietary FXO on CP-induced alterations in membrane enzymes and marker enzyme of lysosomes
To assess the structural integrity of plasma membrane and lysosomes, the effect of CP alone and in combination with FXO diet was determined on membrane and lysosomal enzymes in the liver homogenate (Figure 2). The activities of membrane marker enzymes viz ALP, GGTase and LAP and lysosomal marker enzyme, ACPase, were determined under different experimental conditions in the homogenates of liver (Table 3). CP treatment to control rats caused significant reduction in the specific activities of ALP (−64%), GGTase (−39%) and LAP (−37%) in liver homogenate. CP treatment to FXO fed rats prevented CP elicited decrease in membrane enzyme activities. As can be seen from the data, CP-induced decrease in enzyme activities were prevented by FXO diet. However, the activity of ACPase was increased (+54%) by CP treatment in liver homogenate, while FXO diet was able to prevent the increase in ACPase activity (Table 3).
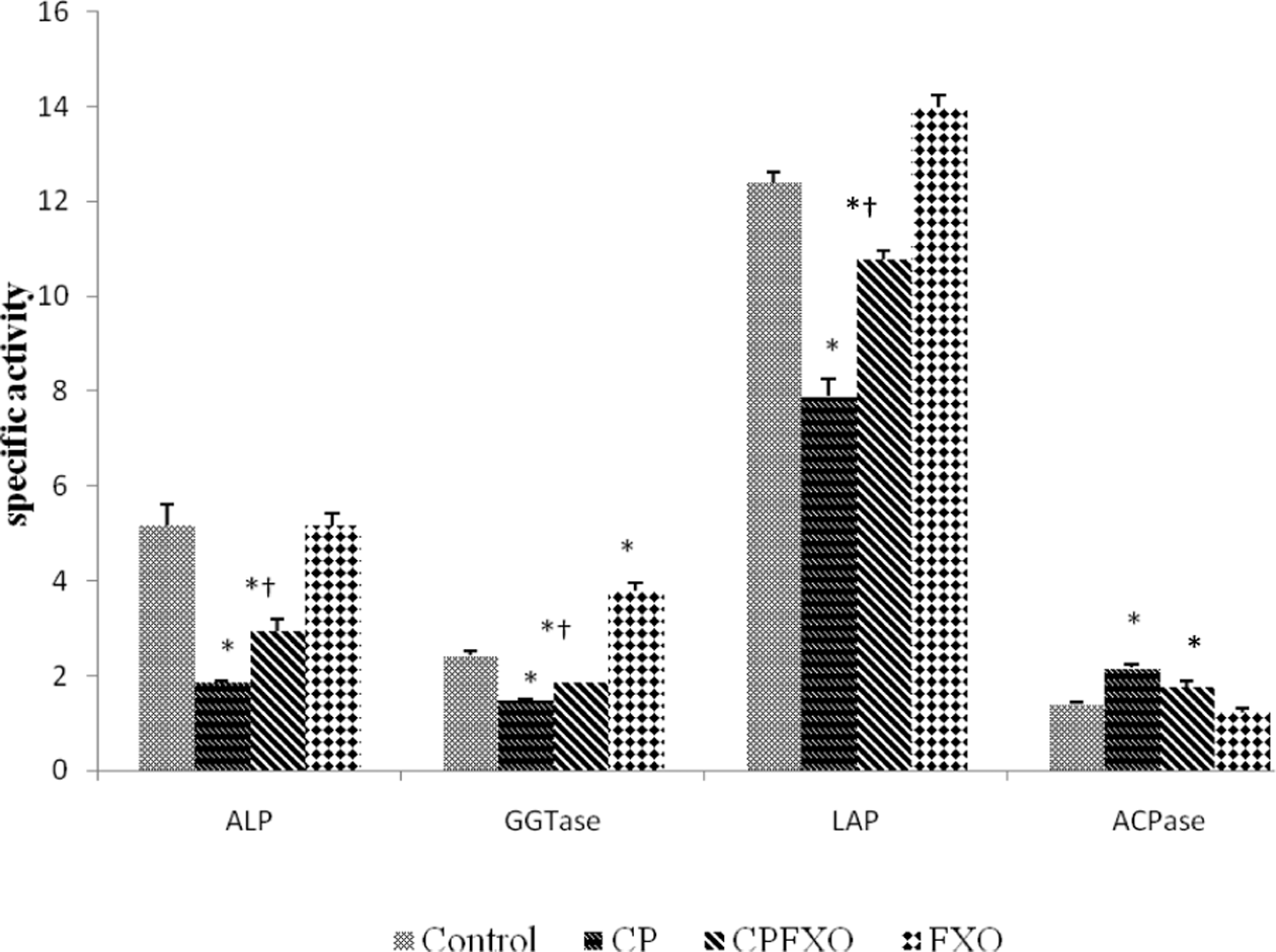
Effect of flaxseed oil (FXO) on the activities of alkaline phosphatase (ALP), γ-glutamyl transferase (GGTase), leucine amino peptidase (LAP) and acid phosphatase (ACPase) in liver homogenate with CP treatment. Results (μmol/mg protein/h) are mean ± SEM for five different preparations. *Significantly different from control, †significantly different from CP: at p < 0.05 by one-way analysis of variance (ANOVA).
Effect of flaxseed oil (FXO) on biomarker enzymes of membrane and lysosomes with/without CP treatment in liver homogenates a

CP: cisplatin treated; CPFXO: flaxseed oil + cisplatin treated; FXO: flaxseed oil diet; ALP: alkaline phosphatase; GGTase: γ-glutamyl transferase; LAP: leucine amonipeptidase; ACPase: acid phosphatase.
aResults (specific activity expressed as μmoles/mg protein/h) are mean ± SEM for five different experiments. Values in parentheses represent percent change from control.
bSignificantly different from control at p < 0.05 by one-way ANOVA.
cSignificantly different from CP at p < 0.05 by one-way ANOVA.
Effect of FXO on CP-induced alterations on metabolic enzymes in liver
The effect of CP, FXO diet and their combined treatment was determined by the activities of various enzymes of carbohydrate metabolism in liver. As shown in Table 4, CP treatment to control rats significantly increased the activities of LDH (+104.76%), HK (+18%) and ME (+52%) but decreased the activities of MDH (−33%), G6Pase (−22), FBPase (−44%) and G6PDH (−49%). In contrast, FXO consumption enhanced the activities of FBPase and G6Pase. Feeding FXO to CP-treated rats arrested CP-induced decline in MDH, FBPase, G6Pase, G6PDH activities and enhancement in LDH, HK and ME activities.
Effect of flaxseed oil (FXO) on carbohydrate metabolic enzymes with/without CP treatment in liver homogenates a

CP: cisplatin treated; CPFXO: flaxseed oil + cisplatin treated; FXO: flaxseed oil diet; LDH: lactate dehydrogenase; MDH: malate dehydrogenase; HK: hexokinase; G6Pase: glucose-6-phosphatase; FBPase: fructose-1,6-bisphosphatase; G6PDH: glucose-6-phosphatase dehydrgenase; ME: malic enzyme.
aResults are mean ± SEM for five different experiments. Values in parentheses represent percent change from control.
bSignificantly different from control at p < 0.05 by one-way ANOVA.
cSignificantly different from CP at p < 0.05 by one-way ANOVA.
Effect of dietary FXO on CP-induced alterations in antioxidant defense parameters in liver
It is evident that ROS generated by various toxicants are important mediators of cellular injury and pathogenesis of various diseases. 26 Antioxidant status is a potential biomarker to determine the physiological state of the cell, tissue or organ. To ascertain the role of antioxidant system in CP-induced toxicity, the effect of CP was observed on oxidative stress parameters. CP enhanced LPO and significantly altered antioxidant enzymes albeit differently (Table 5). LPO measured in terms of malondialdehyde levels significantly enhanced in the (+52%), whereas total –SH declined in the liver (−30%) in CP group. CP treatment caused decrease in SOD (−15%), GSH-Px (−76%) and CAT (−69%) activities. In contrast, FXO consumption increased the activities of antioxidant enzymes albeit to different extents. However, when CP treatment was extended to FXO fed rats (CPFXO group), the decline in SOD, GSH-Px and CAT activities was reduced significantly in comparison to CP group. The results indicate marked protection by FXO diet against CP-induced oxidative damage to renal tissues.
Effect of flaxseed oil (FXO) on enzymatic and nonenzymatic antioxidant parameters with/without CP treatment in liver homogenates a

CP: cisplatin treated; CPFXO: flaxseed oil + cisplatin treated; FXO: flaxseed oil diet; GSH-Px: glutathione peroxidase; LPO: lipid peroxidation; SOD: superoxide dismutase; total –SH: sulfhydryl groups.
aResults are mean ± SEM for five different experiments. Values in parentheses represent percent change from control.
bSignificantly different from control at p < 0.05 by one-way ANOVA.
cSignificantly different from CP at p < 0.05 by one-way ANOVA.
Discussion
CP is a major antineoplastic drug used for the treatment of various forms of cancers. 27,28 The efficacy of CP is limited, however, by its dose-limiting nephrotoxicity. 29 –31 Although CP-induced nephrotoxicity has been very well documented in clinical oncology, hepatotoxicity has been rarely characterized and is less studied. It is known that CP is significantly taken up in human liver and that high doses of the drug produces hepatotoxicity. 32,33 The treatment of tumor cells with CP provokes several responses including membrane peroxidation, dysfunction of mitochondria, inhibition of protein synthesis and DNA damage. 34,35 Formation of free radicals leading to oxidative stress has been shown to be one of the pathogenic mechanisms of the adverse effects of CP in kidney and liver. 36 CP is known to cause histopathological and ultrastructural abnormalities in the liver including inflammatory infiltration, hyperplasia, periportal fibrosis, marked disruption of hepatic cords and dilated blood sinusoids. Many hepatocytes showed karyomegaly and pyknotic nuclei indicating apoptosis. 37,38 Several strategies and agents were utilized to prevent CP-induced toxicity but were not found suitable for clinical practice. 39 –41
The last 3 decades have witnessed a major drift in the scientific community toward the roles of naturally occurring dietary substances for their ability to confer health and physiological benefits. A number of studies have revealed that ω-3 PUFAs have numerous health benefits. The predominant dietary sources of PUFA are plants/seed namely flaxseed and fish/marine foods. ω-3 PUFA-enriched FO has received a great deal of attention as therapeutic options in a variety of clinical situations. 4,42,43 Lately, flaxseed (L. usitatissimum) has been the focus of increased interest in the field of diet and disease research due to the potential health benefits 44 associated with its biologically active component viz ω-3 PUFA. FXO has been shown to prevent lipid disorders and lead-induced neurotoxicity and nephrotoxicity. 45,46 However, the role of FXO in preventing drug-induced toxicity has not yet been investigated.
The present work was undertaken to study detailed mechanism of CP-induced hepatotoxic alterations and possible role of FXO in preventing those deleterious changes in rat liver. Single CP injection caused marked alterations in serum and urine parameters. The present results show that CP administration to control rats caused hepatotoxicity as indicated by elevated levels of transaminases (ALT and AST). Moreover, Scr and BUN were also enhanced with significant decrease in creatinine clearance. These results also show a significant decrease in serum glucose, Pi, PLs and serum cholesterol, accompanied by massive proteinuria, glucosuria and phosphaturia. FXO diet given prior to and following CP administration prevented CP-induced alterations in various serum/urine parameters. Dietary supplementation of FXO prior to and following CP treatment prevented CP elicited increase in levels of Scr, BUN and transaminases. Serum glucose, PLs and phosphate were improved upon CP treatment to FXO fed rats.
Since the brush border membrane of renal proximal tubules has been shown as the major site of CP-induced renal injury, 47,48 the integrity of the membrane was assessed by the status of its biomarker enzymes ALP, GGTase and LAP. CP significantly decreased the activities of ALP, GGTase and LAP in the liver homogenate. The decrease in membrane enzyme activities might have occurred due to the loss of membrane enzyme and other proteinic components, indicating adverse effects of CP on membrane integrity. The present results show that in contrast to CP treatment, dietary supplementation of FXO in control rats caused significant increase in the activities of membrane enzymes in the liver homogenate. Dietary FXO supplementation prior to and following CP treatment prevented/retarded CP-induced decrease in membrane enzymes in the tissue. The activity of lysosomal enzyme ACPase was significantly increased in liver homogenate by CP treatment. Alteration in ACPase activity demonstrate CP-induced loss of lysosomal function. 49,50 However, the CP-induced effect on lysosomal enzyme activity appeared to be ameliorated by dietary FXO supplementation.
To assess the functional aspects, the activities of various metabolic enzymes were determined under different experimental conditions. The activities of various enzymes involved in glycolysis, tricarboxylic acid (TCA) cycle, gluconeogenesis and hexose monophosphate (HMP)-shunt pathways were differentially altered by CP treatment and/or by FXO consumption. CP caused significant increase in LDH and decrease in MDH which was accompanied with a simultaneous increase in HK activity in liver tissue. Although the actual rates of glycolysis or TCA cycle were not determined, marked decrease in MDH activity appears to be due to CP-induced damage to mitochondria. 49,50 A marked increase in LDH activity with simultaneous decline in TCA cycle enzyme, MDH appears to be an adaptive cellular effect in energy metabolism from aerobic metabolism alternatively to anaerobic glycolysis due to CP-induced mitochondrial dysfunction. 49
CP also altered the activities of enzymes of gluconeogenesis and HMP-shunt pathway. The activities of G6Pase, FBPase and G6PDH were profoundly decreased albeit to different extent. However, the activity of NADP ME was increased in the tissue. The present data indicate that CP caused differential effects on different enzymes of carbohydrate metabolism. FXO administration to CP-treated rats resulted in the overall improvement of carbohydrate metabolism as evident by higher activities of MDH and gluconeogenic enzymes in CPFXO as compared to CP group. FXO might have lowered the number of damaged mitochondria or other affected macromolecules or may have increased the number of normally active organelles or macromolecules.
Earlier reports reveal that heavy metals including CP 48,51,52 exert their toxic effects by inducing the generation of ROS. A major cellular defense against ROS is provided by SOD and CAT, which together convert superoxide radicals first to H2O2 and then to molecular oxygen and water. Other enzymes, e.g. GSH-Px, use thiol-reducing power of glutathione to reduce oxidized lipids and protein targets of ROS. However, oxidative stress can occur as a result of either increased ROS generation and/or decrease in antioxidant enzyme system. These antioxidant enzymes protect the cell against cytotoxic ROS. In agreement with the previous studies 4,53 present results show that CP enhanced LPO, an indicator of tissue injury and depleted protein thiols. CP administration to control rats caused severe damage to liver tissue most likely by ROS generation as apparent by perturbation in the antioxidant enzymes (SOD, CAT and GPx-SH) and total-SH content that lead to increased LPO. CP-treated rats fed on FXO-supplemented diet caused significant increase in SOD, CAT and GSH-Px activities accompanied by lower LPO values in liver tissue. The protection against CP by FXO can be attributed to its intrinsic biochemical and natural antioxidant properties. It appears that FXO enriched with ω-3 fatty acids enhanced resistance to free radical attack generated by CP administration. As indicated in Figure 3, our results indicate an overall improvement in metabolic activities, lysosomal integrity and augmentation of antioxidant defenses in CP-treated FXO-fed rats.

Cisplatin (CP)-induced alterations in cell metabolism, lysosomal integrity and oxidative stress and its amelioration by dietary flaxseed oil (FXO): a summary.
We conclude that while CP elicited deleterious hepatotoxic effects by causing severe damage to the plasma membrane, mitochondria and other organelles by suppressing antioxidant defense mechanism, however these effects were ameliorated by dietary supplementation with FXO. Present study thus support the rationale that ω-3 fatty acids–enriched FXO may be effective dietary supplementation to maximize the clinical use of CP in the treatment of various malignancies without hepatotoxic and other side effects.
Footnotes
Acknowledgment
Indian Council of Medical Research (ICMR), New Delhi, India, is acknowledged for the award of JRF (junior research fellowship)/SRF (senior research fellowship) to W.K. University Grants Commission (UGC) for the award of scholarship to A.N. and S.R. and financial support to the department from University Grants Commission (UGC-DRF) is also gratefully acknowledged.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declared no conflicts of interest.
