Abstract
Sodium nitroprusside (SNP) a nitric oxide (NO) donor has proven toxic effects. Dietary ω-3 polyunsaturated fatty acid (PUFA) has been shown to reduce the severity of numerous ailments. Present study examined whether intake of fish oil (FO)/flaxseed oil (FXO, Omega Nutrition, St Vancouver, Canada) would have protective effect against SNP-induced toxicity. Male Wistar rats (150 ± 10 g) were used in this study. Initially animals were divided into two groups: one fed on normal diet and the other on 15% FO/FXO for 15 days. On the 16th day, SNP (1.5 mg/kg body weight) was administered intraperitoneally for 7 days daily. After 7 days animals were killed, kidneys were harvested for further analysis. SNP induced nephrotoxicity by increasing serum creatinine and blood urea nitrogen, SNP significantly decreased malate dehydrogenase, glucose-6-phosphatase, fructose-1,6-bisphosphatase and malic enzyme but increased lactate dehydrogenase and glucose-6-phosphate dehydrogenase. Brush border membrane enzymes such as alkaline phosphatase, γ-glutamyl transpeptidase and leucine amino peptidase were also decreased. The activity of catalase and glutathione peroxidase decreased concomitantly with increased lipid peroxidation, indicating that the significant kidney damage has been inflicted by SNP. Feeding of FO and FXO with SNP ameliorated the changes in various parameters caused by SNP. The results of the present study suggest that ω-3 PUFA-enriched FO and FXO from seafoods and plant sources, respectively, are similarly effective in reducing SNP-induced nephrotoxicity and oxidative damage. Thus, vegetarians who cannot consume FO can have similar health benefits from plant-derived ω-3 PUFA.
Keywords
Introduction
Sodium nitroprusside (SNP) is a well-known antihypertensive agent due to its ability to donate nitric oxide (NO) molecules in vivo. Although the data are very limited, it has been shown that long-term use of nitrates, for example SNP as donors of NO has the potential to induce many pathophysiological conditions. It has been reported that SNP-induced toxicities are associated with increased generation of reactive oxygen species (ROS). 1 SNP has been documented to induce genotoxicity as assessed by the measurement of micronucleated lymphocytes and to cause DNA strand breaks. 2,3 SNP generates ROS during the redox cycling of nitroprusside. 4 It is converted into nitrite, NO, cyanide and oxygen free radicals such as superoxide and hydroxyl radicals. 4 Since SNP is a NO donor, it can also be converted into peroxynitrite (ONOO) and thus can cause mutagenesis. 5 The potential of SNP to induce apoptosis directly from NO liberation has been well established. 6 Thus, NO and its derivatives (e.g. ONOO) can induce a variety of toxic effects including DNA damage, inhibition of DNA synthesis, cell cycle arrest and inhibition of respiratory chain. 7 –9 On the other hand, SNP has been shown to induce stimulation of glycolysis and glycolytic production of adenosine triphosphate (ATP). 10 However, it may not be sufficient to compensate the decrease in ATP production due to the inhibition of oxidative phosphorylation. Hence total energy production was found to be decreased by SNP and SNP-induced NO formation. Thus, the mitochondrion appears to be the primary site of SNP-induced toxic effects.
Several approaches utilizing different mechanisms have been attempted to reduce chemical- and drug-induced nephrotoxicity and other adverse effects. In past few years, much interest has been centered on the role of naturally occurring dietary substances for the control and prevention of various chronic diseases such as cancer and cardiovascular diseases. ω-3 polyunsaturated fatty acids (PUFAs) have been shown to ameliorate many ailments. 11 –14 Recent studies have shown that ω-3 fatty acids retard the progression of various forms of cancers, depression, arthritis, asthma, cardiovascular and renal disorders. 15,16 Recently it has been documented that ω-3 fatty acids protects against gentamicin- and uranyl nitrate–induced nephrotoxicity. 17,18 Attempts were also made to reduce SNP-induced toxicity with the use of resveratrol and red wine and tea due to their antioxidant properties. 1,19
The present work was designed to study the detailed mechanism of SNP-induced nephrotoxic alterations and other adverse effects and the possible mechanism by which FO/FXO consumption exerts their protective effect in ameliorating SNP-induced nephrotoxicity.
Materials and methods
Chemicals and drugs
Fish oil (FO) was procured from Menhaden, Sigma Chemical Co., USA, FXO from Canada, SNP from Sigma Chemical Co., USA and Griess reagent from Sigma Chemical Co., USA. All other chemicals used were of analytical grade and were purchased either from Sigma Chemical Co. (USA) or from Sisco Research Laboratory (Mumbai, India).
Diet
A nutritionally adequate laboratory pellet diet was obtained from Aashirwaad Industries, Chandigarh (India). Pellets were crushed finely and mixed with (a) 15% FO (b) 15% FXO and stored in airtight containers. Vitamin E as
Experimental design
The animal experiments were conducted according to the guidelines of Committee for Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India. Adult male Wistar rats (8 rats/group) weighing 150 ± 10 g fed with standard rat chow (Aashirwaad Industries, Chandigarh) and water ad libitum were conditioned for 1 week before the start of the experiment. Rats were divided into four groups. They were fed on a normal diet (control and SNP) or diet containing 15% FO (SNP + FO) or 15% FXO (SNP + FXO). After 15 days, SNP was administered to the rats intraperitoneally (1.5 mg/kg body weight/day) for 7 days to all groups except control. Control animals received an equivalent volume of normal saline. The rats were killed 24 h after the last injection under light ether anesthesia. Urine samples were collected for 4 h in standard metabolic cages a day before being killed (Figure 1). Blood samples were collected and the kidneys were harvested and processed for the preparation of homogenates and brush border membrane vesicles (BBMVs) as described below.

Schematic representation of experimental design. C: control; SNP: sodium nitroprusside; FO: fish oil; FXO: flaxseed oil; d: day; ND: normal diet.  : 1.5 mg/kg body weight (bwt) SNP injection (5 days intraperitoneally (i.p.)), _____: 0.9% saline injection (10, i.p.), - - - - - - -: oil diets.
: 1.5 mg/kg body weight (bwt) SNP injection (5 days intraperitoneally (i.p.)), _____: 0.9% saline injection (10, i.p.), - - - - - - -: oil diets.
Serum/urine chemistries
Serum/urine parameters
Serum samples were deproteinated with 3% trichloroacetic acid in a ratio of 1:3, left for 10 min and then centrifuged at 2000
Preparation of homogenates
The kidneys were decapsulated and kept on ice-cold 154 mM NaCl and 5 mM Tris-4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer, pH 7.5. The cortical and medullary regions were carefully separated and homogenized (as mentioned above) in 50 mM mannitol buffer to get 10% (w/v) homogenate. Ten percent of liver homogenate was similarly prepared in 10 mM Tris-HCl buffer, pH 7.5.
One part of the homogenate was centrifuged at 2000
Preparation of BBM
Kidney BBM was prepared from whole cortex homogenate using the MgCl2 precipitation method as described by Yusufi and Dousa. 21 The final membrane preparations were suspended in 300 mM mannitol, pH 7.4. Freshly prepared BBMVs were used for the transport of 32 Pi and some portion was saved and stored at −20°C until further analysis for BBM enzymes. Each sample of BBM was prepared by pooling tissues from 2 to 3 rats.
Assay of carbohydrate metabolism enzymes
The activities of the enzymes involving oxidation of NADH or reduction of NADP were determined spectrophotometrically on Cintra 5 fixed for 340 nm using 3 ml of assay in a 1-cm cuvette at room temperature (28–30°C). The enzyme assays of lactate dehydrogenase (LDH), malate dehydrogenase (MDH), malic enzyme (ME), glucose-6-phosphate dehydrogenase (G6PDH), glucose-6-phosphatase (G6Pase) and fructose-1,6-bisphosphatase (FBPase) activities were studied as described by Khundmiri et al. 20 Hexokinase (HK) was estimated by the method of Crane and Sols and the remaining glucose was measured by method of Nelson-Somogyi. 22,23
Assay of BBM marker enzymes and lysosomal marker enzymes
The activities of alkaline phosphatase (AlkPase), leucine amino peptidase (LAP), γ-glutamyl transferase (GGTase) sucrase and acid phosphatase (ACPase) were determined as described by Farooq et al. 24
Assay of enzymatic and nonenzymatic antioxidants
Superoxide dismutase (SOD) was assayed by the method of Marklund and Marklund. 25 Catalase (CAT) and glutathione peroxidase (GSH-Px) activities were determined by the method of Giri et al. and Flohe and Gunzler, respectively. 26,27 Total –SH was determined by the method of Sedlak and Lindsay and LPO by the method of Ohkawa et al. 28,29
Protein concentrations in the homogenates were determined according to the method of Lowry et al. as modified by Yusufi et al. 30,31
Transport of 32 Pi
Measurement of 32 Pi (Bhabha Atomic Research Center, India) uptake in BBMV(s) was carried out at 25°C by rapid filtration technique as described by Yusufi et al. either in the presence or absence of Na gradient. 32 Uptake was initiated by the addition of 30 μl of incubation medium containing 100 mmol/L mannitol, NaCl/KCl 100 mmol/L, 5 mmol/L Tris-HEPES, pH 7.5, 0.1 mmol/l K2H 32 PO4 to 15 μl BBM suspension (50–100 μg protein) and incubated for the desired time intervals (see Results section). The uptake was stopped by the addition of 3 ml of ice-cold stop solution (containing 135 mmol/l NaCl, 5 mmol/l Tris-HEPES and 10 mM sodium arsenate, pH 7.5) and filtered immediately through 0.45 μm MF-Millipore Membrane, mixed cellulose esters, Hydrophilic (DAWP) Millipore (USA) and washed three times with the stop solution using a Cornwall type syringe (Wheaton, Illinois). Correction for nonspecific binding to filters was made by subtracting the value of corresponding blank obtained by filtration of the incubation buffer without vesicles from all data. The radioactivity of dried filters was measured by liquid scintillation counting (Rackbeta, LKB, Wallac, Sweden) with 10 ml scintillation fluid (Cocktail T, SRL, India).
Determination of tissue nitrite
Tissue nitrite levels were quantified using the Griess assay.
33
Kidney homogenates were centrifuged at 5000
Statistical analyses
All data are expressed as mean ± SEM for at least 4–5 different preparations. Statistical evaluation was performed using one-way analysis of variance. A probability level of
Results
The present work was undertaken to study the detailed mechanism of SNP-induced nephrotoxicity and other deleterious effects and its possible protection by feeding ω-3 fatty acids–enriched oils diets to the rats. To address our hypothesis, the effect of SNP alone and in combination with FO or FXO was determined on nephrotoxicity parameters with serum biomarker enzymes of oxidative stress, BBM and carbohydrate metabolism in rat kidney tissues. Na-dependent phosphate transport was also determined in renal cortical BBM to assess the role of kidney in the conservation of Pi transport under different experimental conditions.
Effect of dietary FO and FXO on SNP-induced nephrotoxicity parameters in serum and urine
In general, the rats remained clinically well throughout the study. There was no significant difference in daily food intake and body weights between control and other experimental rats (data not shown). SNP treatment to control rats resulted in significant increase in serum creatinine (Scr), BUN, cholesterol, glucose and phospholipids but decrease in inorganic phosphate (Pi) compared to control rats (Table 1). These changes where associated with profound phosphaturia, proteinuria and glucosuria (Table 2) accompanied by decrease in creatinine clearance (Table 2). Feeding FO or FXO diet to SNP administered (FOSNP and FXOSNP) rats resulted in significant reversal of various SNP-elicited deleterious effects on serum and urine parameters. Both FO and FXO diets prevented SNP-induced increase in Scr, BUN, glucose and cholesterol and decrease of serum Pi (Table 1). FO/FXO diets appear to greatly improve the renal functions in a similar manner as evident by the increase in creatinine clearance and decrease in the excretion of protein, phosphate and glucose in the urine (Table 2).
Effect of fish oil (FO) and flaxseed oil (FXO) on serum parameters with SNP treatmenta

BUN: blood urea nitrogen; SNP: sodium nitroprusside.
a Results are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control at
c Significantly different from SNP at
Effect of fish oil (FO) and flaxseed oil (FXO) on urine parameters of SNP-treated ratsa

a Results are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control at
c Significantly different from SNP at
Effect of FO and FXO on SNP-induced alterations in metabolic enzymes in renal cortex and medulla
The effect of SNP, FO/FXO diets and their combined treatment was determined on the activities of various enzymes of carbohydrate metabolism in renal cortex and medulla.
Effect of fish oil (FO) and flaxseed oil (FXO) activities of HK, LDH and MDH in homogenates of (a) cortex and (b) medulla with SNP treatmenta

HK: hexokinase; LDH: lactate dehydrogenase; MDH: malate dehydrogenase; SNP: sodium nitroprusside.
a Results (specific activities) are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control at
c Significantly different from SNP at
Effect of fish oil (FO) and flaxseed oil (FXO) on activities of G6Pase, FBPase, G6PDH and ME in homogenates of (a) cortex and (b) medulla with SNP treatmenta
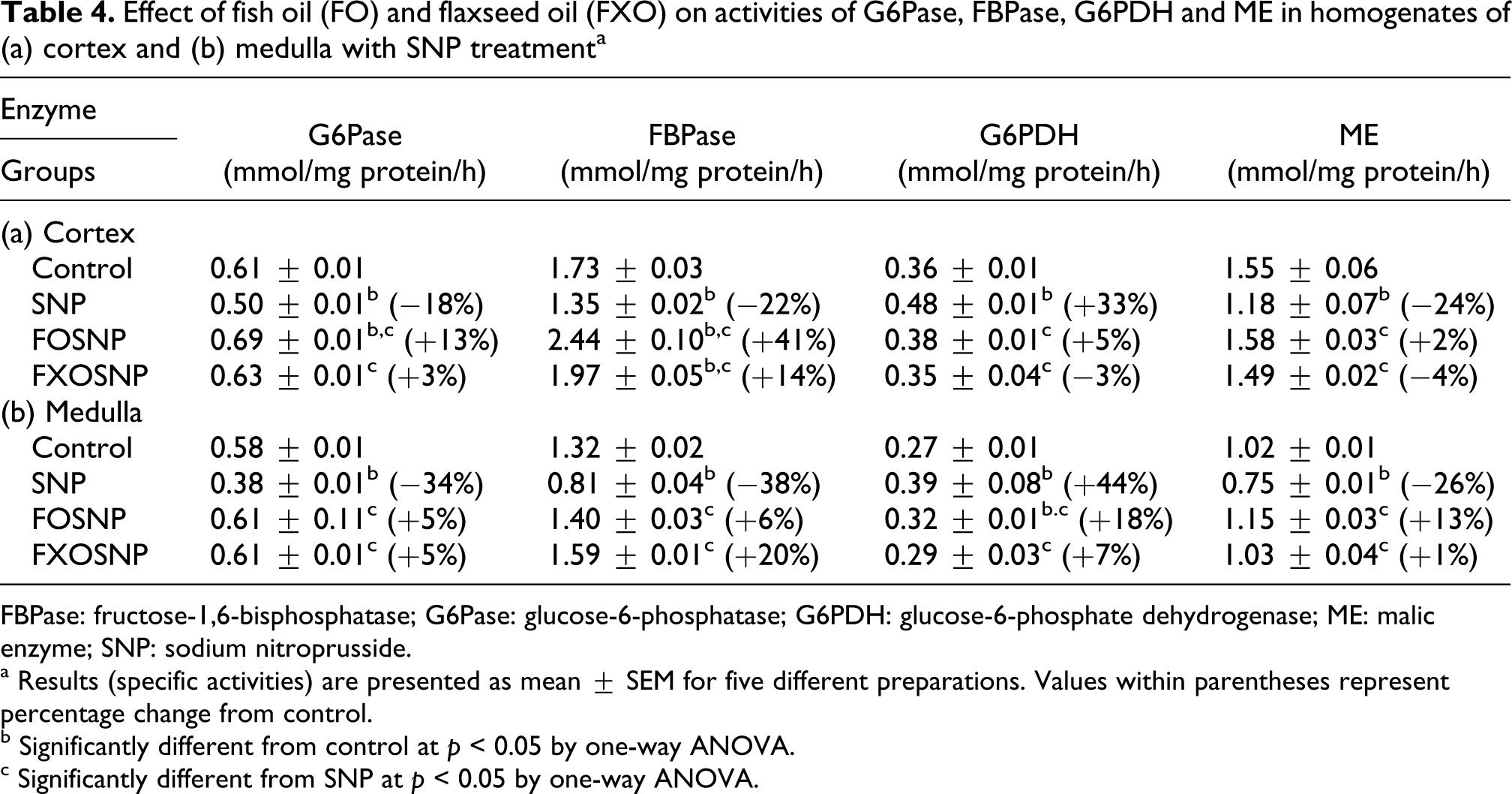
FBPase: fructose-1,6-bisphosphatase; G6Pase: glucose-6-phosphatase; G6PDH: glucose-6-phosphate dehydrogenase; ME: malic enzyme; SNP: sodium nitroprusside.
a Results (specific activities) are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control at
c Significantly different from SNP at
The effect of FO, FXO and SNP combined was also determined on G6PDH and NADP-ME, source of NADPH production needed in various anabolic reactions (Table 4). SNP treatment to control rats resulted in significant increase in G6PDH but decrease in ME activity. SNP-elicited increase in G6PDH activity was normalized to near control values by both FO and FXO diets. SNP-induced ME activity decrease was arrested by both FO and FXO diet.
Effect of dietary FO and FXO on SNP-induced alterations in biomarker enzymes of BBM and lysosomes
To assess the structural integrity of certain organelles, for example plasma membrane (BBM) and lysosomes, the effect of SNP alone and in combination with FO or FXO diets was determined on biomarker enzymes of BBM and lysosomes in the homogenates of renal cortex and medulla and isolated BBM preparations from renal cortex.
Effect of SNP alone and with FO or FXO diet on biomarkers of BBM and lysosomes in the homogenates
The activities of alkaline phosphatase (AlkPase), GGTase), LAP and acid phosphatase were determined under different experimental conditions in the homogenates of renal cortex and medulla (Table 5). SNP treatment to control rats caused significant reduction in the specific activities of AlkPase, GGTase and LAP in cortical homogenate. The prior feeding of FO or FXO diet with SNP treatment prevented SNP-elicited decrease in BBM enzyme activities. As can be seen from the data, SNP-induced decrease in BBM enzyme activities were similarly prevented by FO or FXO diet. The activity of ACPase was also decreased by SNP in cortical homogenates and FO/FXO diet was able to prevent the decrease in enzyme activity in a similar manner (Table 5).
Effect of fish oil (FO) and flaxseed oil (FXO) on biomarkers of BBM and lysosomes in homogenates of (a) cortex and (b) medulla with SNP treatmenta

ACPase: acid phosphatase; AlkPase: alkaline phosphatase; GGTase: γ-glutamyl transpeptidase; LAP: leucine amino peptidase; SNP: sodium nitroprusside.
a Results (specific activities) are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control
c Significantly different from SNP at
The activities of BBM enzymes similar to the cortex were also lowered in the medulla by SNP administration although to a lower extent (Table 5). The consumption of FO/FXO in combination with SNP treatment resulted in the reversal of SNP-induced decrease in AlkPase, GGTase and LAP in the medulla.
Effect of SNP and SNP plus FO and FXO diet on BBM markers in isolated BBMV
The effect of SNP, FO and FXO on BBM marker enzymes was further analyzed in BBMV preparations isolated from the renal cortex (Figure 2). The data show a similar activity pattern of BBM enzymes as observed in cortical homogenates. However, the magnitude of the effects was much more pronounced in BBMV than in cortical homogenates. Activities of AlkPase, GGTase and LAP declined profoundly by SNP treatment compared with the control rats. FO and FXO dietary supplementation similar to the effect in the homogenates appeared to lower the severity of the SNP treatment. SNP-induced decrease in BBM enzyme activities was significantly prevented by dietary FO and to greater extent by FXO diet.

Effect of fish oil (FO) and flaxseed oil (FXO) on activities of alkaline phosphatase (AlkPase), γ-glutamyl transpeptidase (GGTase) and leucine amino peptidase (LAP) in cortical brush border membrane (BBM) with sodium nitroprusside (SNP) treatment. Results (μmol/mg protein/h) are mean ± SEM for five different preparations. *Significantly different from control, †significantly different from SNP: at
Effect of dietary FO and FXO on SNP-induced alterations in antioxidant defense parameters in renal cortex and medulla
Primary components of oxidative stress and cellular injury response include elevation in LPO, depletion of glutathione (GSH) and suppression of antioxidant enzymes. 17,18 To ascertain the role of antioxidant system in SNP-induced toxicity, the effect of SNP was observed on oxidative stress parameters. SNP enhanced LPO and significantly altered antioxidant enzymes both in cortex and medulla, albeit differently (Table 6). LPO measured in terms of malodialdehyde (MDA levels) significantly enhanced in the cortex (+42%) and medulla (+75%) to similar extent, whereas total SH declined in these tissue (−22% to −37%). SNP treatment caused marked increase in SOD (+40%) but decrease in GSH-Px (−29%) and CAT (−18%) activities in the renal cortex. In medulla, however the activity of SOD (−87%), CAT (−35%) and GSH-Px (−42%) significantly decreased by administration of SNP alone.
Effect of fish oil (FO) and flaxseed oil (FXO) on enzymatic and nonenzymatic antioxidant parameters in homogenates of (a) cortex and (b) medulla with SNP treatmenta
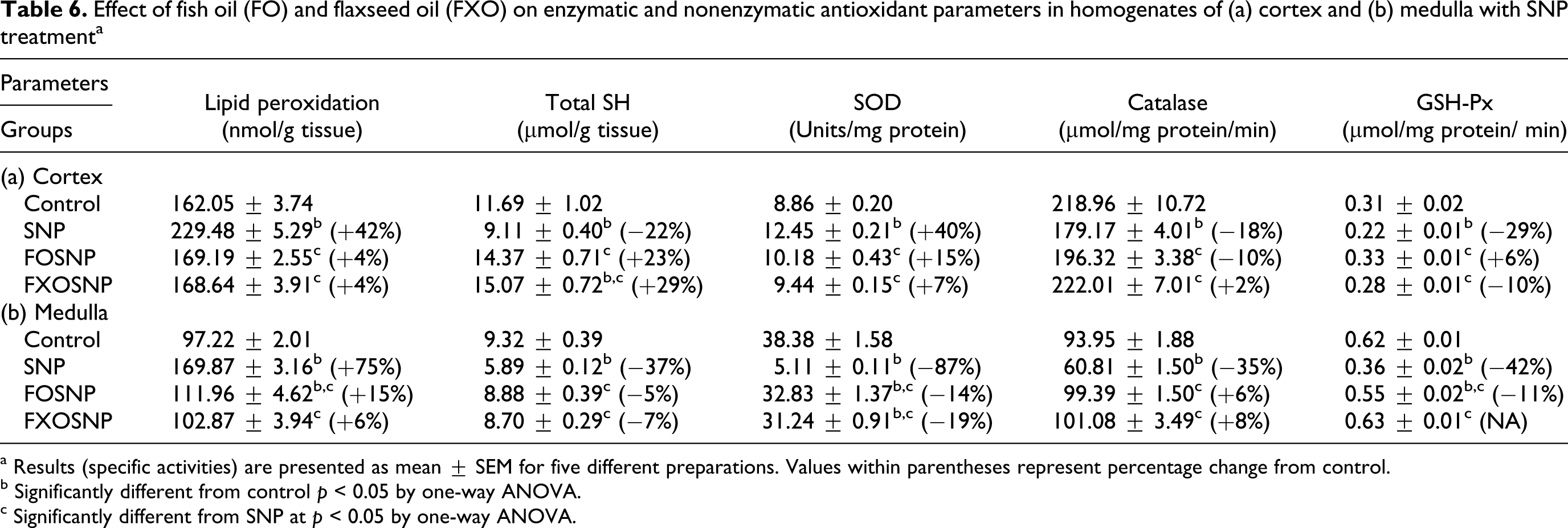
a Results (specific activities) are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control
c Significantly different from SNP at
Since the diet supplemented with FO enriched with ω-3 fatty acids has been shown to reduce gentamicin/uranyl nitrate-induced nephrotoxicity parameters by strengthening antioxidant defense mechanism, 17,18 the protective effect of both FO and FXO enriched in ω-3 fatty acids was determined on SNP-induced oxidative stress parameters. The results indicate that FO and FXO dietary supplementation was able to ameliorate the SNP-induced oxidative damage in both renal cortex and medulla. SNP-induced increase in LPO and decrease in total SH was not observed by feeding FO or FXO diet to SNP-treated rats. The activity of CAT and GSH-Px in the cortex and medulla remained significantly higher in FO/FXO + SNP rats compared to SNP rats. The results indicate marked protection by both FO/FXO diet against SNP-induced oxidative damage to renal tissues.
Effect of dietary FO and FXO on SNP-induced alterations in Na-gradient-dependent transport of 32 Pi in BBMV(s) isolated from renal cortex
The bulk of filtered Pi in the kidney is reabsorbed by its proximal tubule. The Na-gradient-dependent {Naoutside (Nao ) > Nainside (Nai )} transport of Pi in renal proximal tubule across its luminal BBM is an initial and regulatory step. The Pi is transported by secondary active transport mechanism and requires expenditure of energy in the form of ATP, which is generated by cellular metabolism. The uptake of 32 Pi was determined in the presence and absence of Na gradient in the initial uphill phase (30 s) and after equilibrium at 120 min in BBM preparations. The rate of concentrative uphill uptake of 32 Pi in the presence of a Na gradient (NaCl in the medium) was markedly decreased by SNP treatment (Table 7). However, the uptake of 32 Pi at the equilibrium phase (120 min) when Nao = Nai was not significantly different between the two groups. Also Na-independent uptake (in the absence of a Na gradient, when NaCl in the medium was replaced by KCl where Ko > Ki ) of 32 Pi at 30 s and 120 min was also not affected by SNP treatment, indicating specific alterations only when Na gradient was present. When SNP treatment was extended to FO and FXO feeding rats, SNP-induced decrease in 32Pi transport was not observed.
Effect of fish oil (FO) and flaxseed oil (FXO) on uptake of 32 Pi in brush border membrane vesicles (BBMVs) from whole cortex with SNP treatmenta
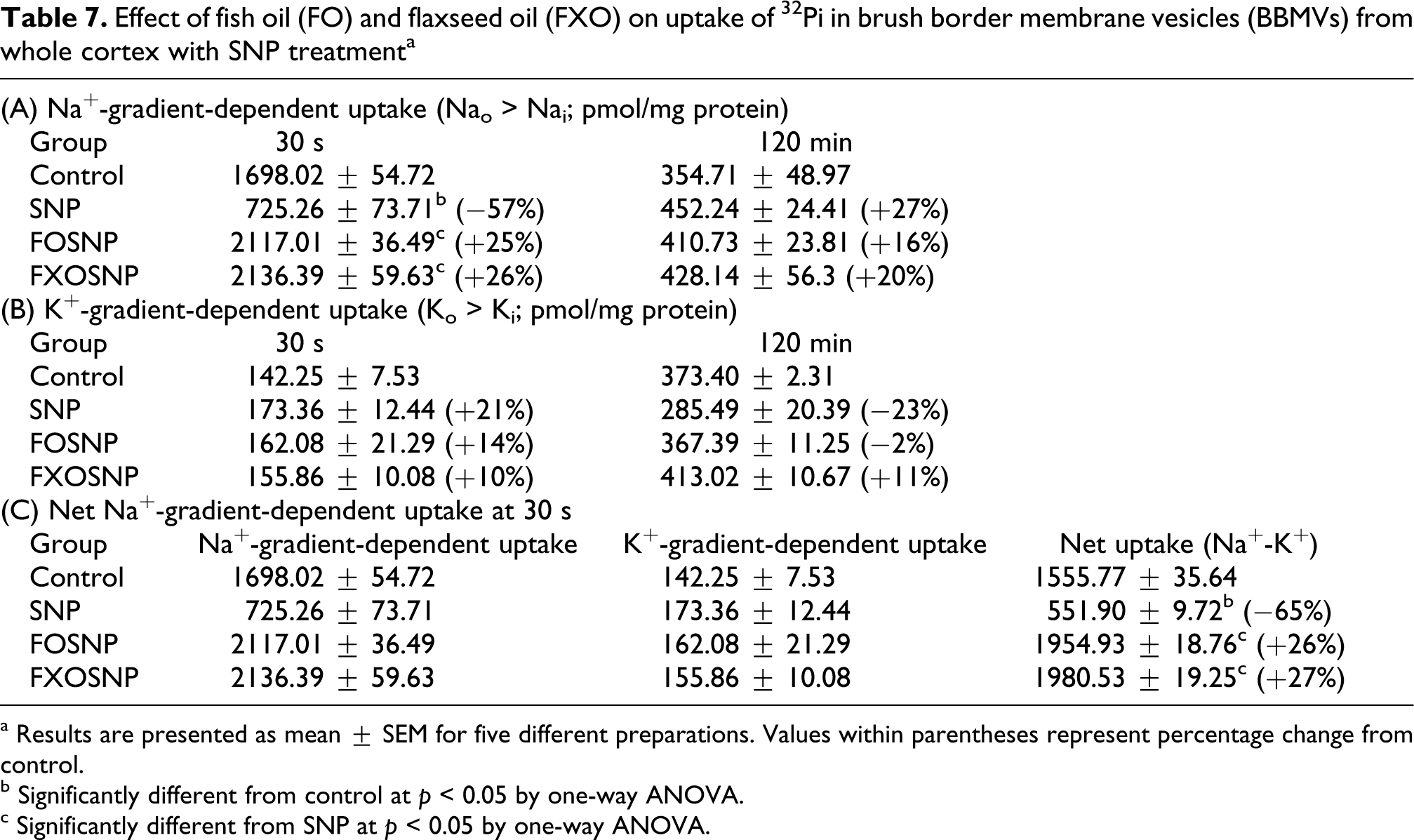
a Results are presented as mean ± SEM for five different preparations. Values within parentheses represent percentage change from control.
b Significantly different from control at
c Significantly different from SNP at
Effect of dietary FO and FXO on SNP-induced alterations in nitrite concentration in renal cortex and medulla
Nitrite is the predominant metabolite of NO in vivo. The effect of SNP, FO and FXO on tissue accumulation of nitrite was determined by the Griess reagent assay as described in the Methods section (Figure 3). SNP significantly increased the nitrite accumulation in all tissues; however, the effect was maximum in renal medulla. Both FO and FXO reduced the amount of nitrite in rat tissues. Thus, we can say that feeding with FO/FXO successfully scavenged excess NO produced by SNP. Further, we can also say that by improving renal function, the renal clearance of nitrite was enhanced, which did not allow nitrite accumulation in the tissues.
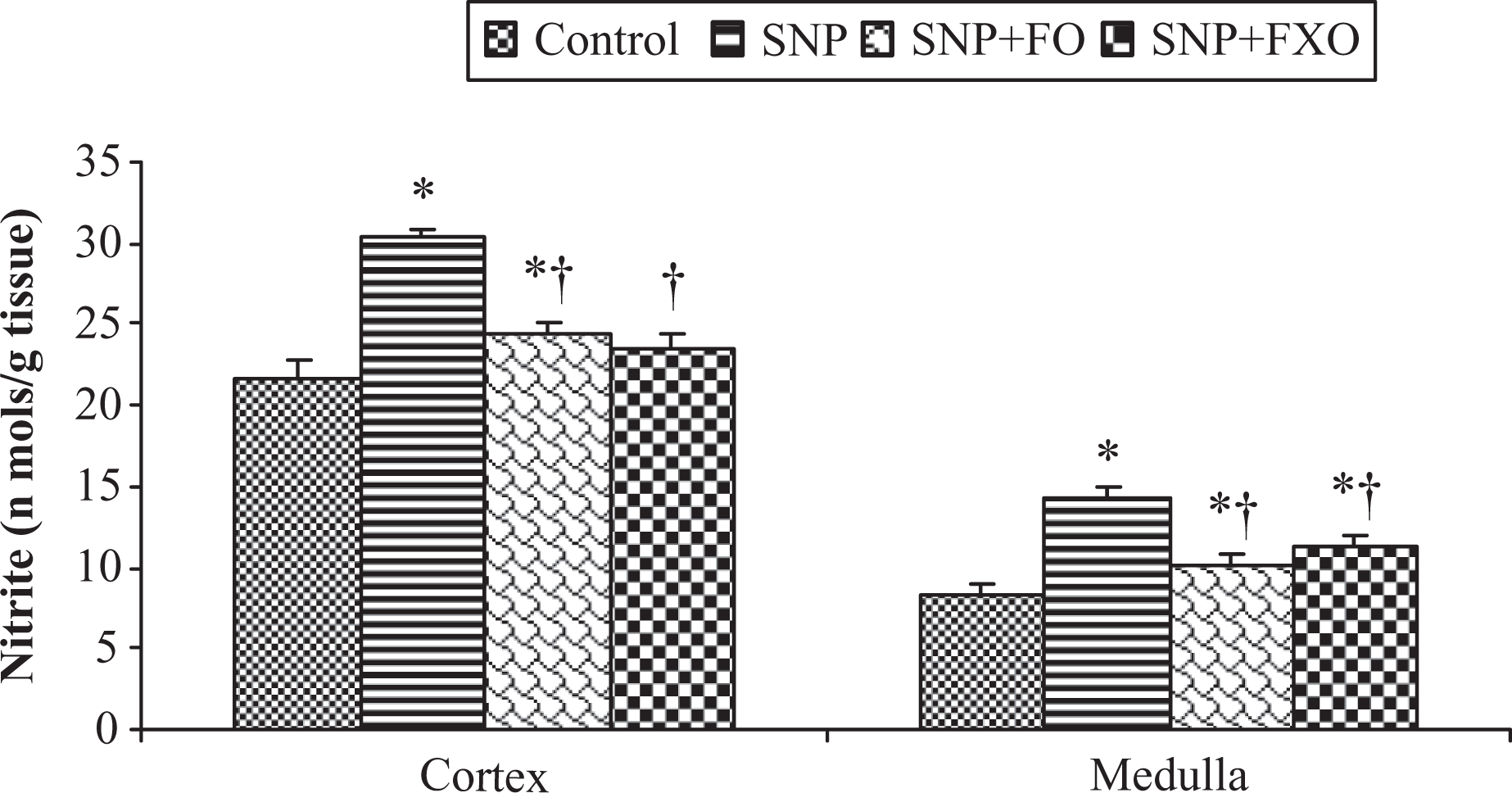
Effect of fish oil (FO) and flaxseed oil (FXO) on tissue nitrite concentration with sodium nitroprusside (SNP) treatment. Results (nmol/g tissue) are mean ± SEM for five different preparations. *Significantly different from control, †significantly different from SNP: at
Discussion
SNP is an NO donor compound that has been used as an antihypertensive agent since 1920s. 34 In contrast to the organic nitrates (e.g. nitroglycerin) which require the presence of highly specific thiol-containing compounds to generate NO, SNP spontaneously generates this product, thus functioning as a prodrug. 35 SNP has been documented to induce, to cause DNA strand breaks, mutagenesis and apoptosis. 2 –6 Thus, NO and its derivatives (e.g. ONOO) can induce a variety of toxic effects including DNA damage, inhibition of DNA synthesis, cell cycle arrest and inhibition of respiratory chain. 7 –9
Several approaches utilizing different mechanisms have been attempted to reduce chemical- and drug-induced nephrotoxicity and other adverse effects. In past few years, much interest has been centered on the role of naturally occurring dietary substances for the control and prevention of various chronic diseases such as cancer and cardiovascular diseases. ω-3 PUFA have been shown to ameliorate many ailments. 11 –14
The present work was designed to study the detailed mechanism of SNP-induced nephrotoxic alterations and other adverse effects and the possible mechanism by which FO/FXO consumption exerts their protective effect in ameliorating SNP-induced nephrotoxicity. The results show that SNP administration produced a typical pattern of nephrotoxicity/hepatotoxicity/gastrotoxicity as indicated by increased Scr, BUN accompanied by massive proteinuria, glucosuria and phosphaturia, indicating that significant kidney damage has occurred and that SNP administration has caused alterations in structure and function of renal proximal tubular membrane. FO when given for 15 days prior to and during SNP administration prevented SNP-induced deleterious effects on various blood/urine parameters and enzyme activities of various pathways in different tissues of rat. SNP-induced increase in Scr/BUN and proteinuria, glucosuria and phosphaturia were all absent in FO-fed SNP-treated rats. SNP-induced alterations in various serum and urine parameters were similarly prevented by FXO diet when given together with SNP. This renoprotective effect was possible by the fact that both FO and FXO alone were able to increase serum phosphate, serum cholesterol, phospholipids, phosphate and reduce, glucose and BUN along with reduction in urinary excretion of Pi and proteins as has been described elsewhere. 17,18
The reabsorption of Na+ ions by proximal tubular BBM is considered to be the major function of the kidney because the transport of other ions and various solutes depends directly or indirectly on Na+ reabsorption. Since these transport depends on structural integrity of BBM and available energy as ATP which is supplied by various metabolic pathways it is imperative that any alterations to these pathways caused by toxic insult would determine the rate of renal transport functions. 20,36 As shown in the Results section, the activities of various enzymes in glycolysis, tricarboxylic acid cycle, gluconeogenesis and hexose monophosphate shunt pathway were differentially altered by SNP treatment. SNP caused significant increase in the activities of LDH and G6PDH and decrease in the activities of MDH (TCA cycle); G6Pase, FBPase (gluconeogenesis) and ME in both renal cortex and medulla albeit to different extent. Although the actual rates of glycolysis and other pathways were not determined, however marked decrease in MDH activity indicates an impaired oxidative metabolism of glucose/fatty acids that will lead to lower ATP production due to mitochondrial dysfunction. These results also indicate a shift in the energy metabolism alternatively from aerobic to anaerobic most likely due to mitochondrial dysfunction as evident by significant increase in LDH. 37 The decrease in TCA cycle enzymes may have caused decrease in gluconeogenic enzymes. This can be explained by the fact that lower TCA cycle enzyme activities especially that of MDH will result in lower oxaloacetate production from malate which is required not only for the continuation of TCA cycle but also for gluconeogenesis. Thus SNP-induced toxicity appeared to be primarily due to renal mitochondrial damage similar to gentamicin/uranyl nitrate/cisplatin (GM/UN/CP) induced. 17,18,38,39
Dietary FO and FXO were also able to prevent SNP-induced decrease/increase in the activities of certain enzymes involved in carbohydrate metabolism both in renal cortex and medulla. These protective effects can be attributed to the fact that both FO and FXO alone were able to effectively enhance the activities of enzymes resulting in the overall improvement of carbohydrate metabolism in renal tissues similar to those reported in rat liver. 17,18,40 ω-3 PUFA have been shown to coordinately regulate the expression of several enzymes involved in carbohydrate and lipid metabolism. 41
Since the BBM of renal proximal tubules and other intracellular organelles such as mitochondria and lysosomes are known targets of free radicals, the structural and functional integrity was assessed by the status of their respective biomarker enzymes. 42 SNP caused significant decrease in the activities of AlkPase, GGTase and LAP (BBM enzymes) in cortical homogenates and to a much greater extent in BBMV preparations. The decrease in BBM enzyme activities might have occurred due to loss of BBM enzyme and other components into lumen followed by their excessive excretion in the urine as reported earlier. 17,18,38 A marked reduction observed in Na-dependent transport of 32 Pi also supports SNP-induced severe damage to BBM of renal proximal tubules. However, SNP caused significant increase in ACPase activity both in the cortex and medulla. Taken together the present results show that SNP treatment indeed significantly altered the structural integrity and functional capacity of renal proximal tubules and particularly BBM and lysosomes as reflected by marked decrease in the activities of BBM enzymes: AlkPase, GGTase, and LAP, Na-dependent transport of 32 Pi across renal BBM and increase in lysosomal enzyme ACPase and certain specific alteration in the enzymes of carbohydrate metabolism.
It has been shown that dietary fatty acids incorporate in the cellular membranes altering structural integrity and functional capacity of plasma membrane and other organelles, leading to altered cellular metabolic activities and those of membrane associated enzymes. 43 Dietary FO/FXO given together with SNP not only prevented/retarded SNP-induced decrease in BBM enzymes, but the activity of AlkPase and/or GGTase remained higher in FOSNP and FXOSNP than in the BBM of control rats. Both FO/FXO also resulted in significant increase in 32Pi transport across the renal BBM in SNP-treated rats. Thus, FO and FXO caused the reversal of SNP-induced alterations because FO/FXO alone have the intrinsic capability to significantly enhance AlkPase and GGTase activities and Pi transport by themselves, suggestive of an overall improvement in renal BBM integrity by FO/FXO diet thus preventing the loss either by lessening the damage caused by SNP or by increasing the regeneration process or both. 44 It appears from the data that SNP by lowering oxidative metabolism lowers ATP production, whereas dietary FO/FXO supplementation increased ATP production by increasing the metabolic activities to support many cellular functions especially in the kidney. Further, FO/FXO diets increased the renal Pi transport suggestive of ω-3-PUFA-induced Pi conservation by the kidneys, which maintains and/or improves metabolic activities of not only kidneys but also other tissues.
Excessive production/accumulation of NO always has the ability to result in the formation of other reactive intermediates of nitrogen. Reactive nitrogen species (RNS) produces cellular injury and necrosis via several mechanisms including peroxidation of membrane lipids, proteins and DNA. 45 Several studies have shown that NO-induced toxicity is engendered by its reaction with superoxide radical to yield the highly toxic ROS ONOO. ONOO then decomposes to form nitrogen dioxide and the hydroxyl free radical leads to LPO, protein oxidation and DNA damage. 45 A major cellular defense against free radicals is provided by SOD and CAT, which together convert superoxide radicals first into H2O2 and then into molecular oxygen and water. Other enzymes, for example GSH-Px uses thiol-reducing power of GSH to reduce oxidized lipids and protein targets of RNS. The present results show that SNP administration to control rats caused severe damage to renal tissues most likely by RNS/ROS generation as apparent by altered activities of the antioxidant enzymes and total-SH content that resulted in increased LPO. As consistent with the previous studies, CAT and GSH-Px were inhibited by SNP-induced NO production. 46,47 Enhanced LPO may result in protein damage and inactivation of membrane-bound enzymes either through direct attack by free radicals or through chemical modification by its end product (MDA). 48 Thus, observed increase in LPO could provide additional explanation for decreased activities of BBM enzymes and other proteins.
The protection against SNP-induced renal dysfunction and decline in activities of BBM enzymes by FO and FXO can be attributed to their intrinsic biochemical and natural antioxidant properties. The ω-3 fatty acids possess antioxidant-enhancing activity, thereby raising the efficiency of antioxidant defense system. 49 These observations were recently confirmed by Priyamvada et al. 17,18
The present biochemical studies clearly demonstrate that long-term SNP administration produces severe nephrotoxicity and causes profound damage to plasma membranes, mitochondria, peroxisomes and so on of renal proximal tubules. The enzymes of oxidative carbohydrate metabolism and gluconeogenesis, BBM, antioxidant defense mechanism and 32 Pi transport capacity appeared to be severely affected by SNP treatment. Most of the SNP-induced effects can be attributed to NO because SNP is a potential NO donor. In contrast, FO and FXO, major source of ω-3 fatty acids, appeared to affect membrane organization and functions by altering membrane fatty acid composition. Both FO and FXO appear to accelerate repair and/or regeneration of injured organelles, for example mitochondria, peroxisomes and increased activity of TCA cycle and BBM as evident by the increased activities of the enzymes of carbohydrate metabolism. Most importantly, by activating endogenous antioxidant defense mechanism FO and FXO provided protection from SNP-induced free radical attack. FO/ω-3 fatty acids-targeted cell cycle regulation/cell signaling may also underlie its protective effects.
Conclusion
We conclude that while SNP elicited deleterious nephrotoxic/hepatotoxic/gastrotoxic effects by causing severe damage to renal mitochondria, BBM and other organelles and by suppressing antioxidant defense mechanism, dietary supplementation with FO/FXO enriched with ω-3 fatty acids caused improvement in nutrition/energy metabolism, BBM integrity, 32 Pi transport capacity and antioxidant defenses and thus prevented SNP-induced various deleterious effects. Since SNP is a NO donor, most of these effects can be attributed to NO-induced RNOS formation. Based on our present observations and already known health benefits, we propose that dietary supplementation of FO/FXO may provide a cushion for a prolonged therapeutic option against SNP-induced nephropathy and other adverse alterations without harmful side effects.
Footnotes
Acknowledgments
Indian Council of Medical Research (ICMR), New Delhi, India is acknowledged for the award of JRF (Junior Research Fellowship) /Senior Research Fellowship to SP and WK. Council of Scientific and Industrial Research (CSIR), New Delhi, India, for awarding JRF to SK.
Funding
Financial support to the Department of Biochemistry, AMU, Aligarh from University Grants Commission (UGC-DRF), Department of Science and Technology (DST-FIST) and a research grant (58/21/ 2001-BMS) from ICMR to ANKY is also gratefully acknowledged.
Declaration of Conflict of Interest
The authors declared no conflicts of interest.
