Abstract
Prolonged zidovudine (AZT) treatment in HIV-infected and AIDS patients is shown to induce liver toxicity leading to complications. Therapeutic regimen that could encounter this adverse effect is unavailable and management of toxicity is often symptomatic or is limited to withdrawal of therapy. In the present investigation, we evaluated the alleviating properties of silibinin (SBN), a flavanolignan obtained from Silybum marianum against subacute AZT-induced hepatotoxicity and oxidative stress in rats. AZT treatment (50 mg/kg body weight (b.w.) periorally (p.o.), daily for 45 days) caused highly significant increases in alanine transaminase, alkaline phosphatase, argininosuccinic acid lyase and bilirubin in serum. Oxidative stress is shown by a highly significant increase in lipid peroxidase and total carbonyl content and decrease in catalase and protein thiols in the liver tissue. Hyperlipidaemia is indicated by highly significant increase in total lipids and free fatty acid in serum. Evaluation of liver by haematoxylin and eosin staining shows parenchymal cell enlargement, inflammatory changes and increase in sinusoidal spaces. Simultaneous treatment of SBN (100 mg/kg b.w. p.o., daily for 45 days) significantly protected the liver against hepatotoxicity, oxidative stress and hyperlipidaemia induced by AZT, and this alleviating property is attributed to hepatoprotective, membrane-stabilizing, antioxidant and free radical scavenging properties of SBN.
Keywords
Introduction
Zidovudine (AZT) is a nucleoside analogue, which is commonly used in combination with reverse and non-reverse nucleoside transcriptase inhibitors, protease inhibitors and antitubercular drugs for the treatment of HIV-infected patients, pregnant women and their infants. 1,2 It is a prodrug that requires to be converted to its active form to produce therapeutic effects and is mediated through phosphorylation by cellular thymidine kinase enzyme. 3 AZT treatment is shown to develop headache, nausea, vomiting, confusion, gastrointestinal disturbances, anaemia, neutropenia and alternation in haematological parameters within the first few weeks of ingestion in humans. 1,3 Long-term AZT therapy has been documented to produce myopathy, lactic acidosis, lipid droplet accumulation in muscles and cytochrome c oxidase deficiency. 4 –6 Complication of prolonged AZT treatment is the development of varying degrees and frequencies of hepatotoxicity marked as elevation in liver enzymes, hepatic failure, hepatomegaly and hepatic steatosis. 1,7,8 The incidence of severe hepatic abnormality is reported to occur in 5–10 per 100 persons, and some studies have shown development of hepatotoxicity in as much as 30% of recipients of antiretroviral drugs containing AZT. 9 –11 Experimental studies conducted in rats using higher dose of AZT has demonstrated increase in liver enzymes and total bilirubin. 12 The cause for the above hepatic lesions upon AZT administration in humans is not clearly established and management of therapy is poorly documented. In case of severe liver toxicity, withdrawal of AZT treatment is recommended. 13,14 The mechanism of AZT-induced liver toxicity is not clearly defined in the literature. However, it is postulated that release of peroxides and oxidative stress-inducing agents from the mitochondria during metabolism of AZT play a major role in alteration of oxidative phosphorylation coupling in the liver, which is said to mediate the development of AZT-induced hepatotoxicity. 15,16 A standard hepatoprotective agent useful to prevent AZT-induced hepatotoxicity and oxidative stress is yet to be identified.
Silibinin (SBN) is a natural polyphenolic flavonoid, which is a mixture of two diastereoisomers (containing silibinin A, B and isosilybin A, B) in 1:1 proportion, and it is a major ingredient (constituting around 60–70%) of the active principle silymarin that is isolated from the medicinal plant Silybum marianum, commonly called ‘milk thistle’. 17,18 Use of milk thistle has been documented systematically in ancient medical literature (as early as 77 AD) until the present time. 18 This plant has been indicated as ‘excellent for carrying off bile’ and effective in removing the obstruction of liver and spleen. 19 The German Commission E recommends silymarin for the treatment of dyspeptic complaints, toxin-induced liver damage, liver cirrhosis and supportive therapy for chronic inflammatory conditions of the liver. 20 Silymarin and SBN have been proved to offer protection against alcohol, carbon tetrachloride, thalium, phenylhydrazine and many other drugs- and chemicals-induced liver injury. 17 Hence, both silymarin and SBN are advocated for the prevention of various liver diseases like cirrhosis, chronic hepatitis, steatohepatitis, gall stones, jaundice and hepatic fibrosis induced by alcohol as well as by various xenobiotics in humans and experimental animals. 21–22 We have demonstrated the ameliorating properties of SBN against N-nitrosodimethylamine-induced hepatic fibrosis in rats. 23,24
Although short-term and long-term administration of AZT is reported to induce liver toxicity in as much as 30% of its recipients, 11 studies regarding the protective properties of SBN against this deleterious adversity are unavailable. The objective of this study is to evaluate the hepatoprotective and antioxidant properties of SBN against AZT-induced hepatotoxicity in rats. We hereby report the development of hepatocellular degenerative changes, hyperlipidaemia and oxidative stress in rats on subacute administration of AZT and its mitigation by simultaneous treatment with SBN.
Materials and methods
Chemicals
Silibinin (SBN), 1,1,3,3-tetramethoxypropane malondialdehyde (MDA), thiobarbituric acid, argininosuccinate sodium, 2,4-dichloro-1-napthol, bilirubin, palmitic acid, triolein and 5,5-dithio-bis-2-nitrobenzoate were purchased from M/s Sigma Aldrich Chemicals (St Louis, Missouri, USA). AZT was procured as gratis from the Director, Central Drug Testing Laboratory, Government of India, Chennai, Tamil Nadu, India. All the other chemicals used for various assay procedures were purchased locally and were of analytical grade.
Animals
Wistar albino rats of either sex, weighing (150 ± 20 g) aged 3–4 months procured from Institutional Animal House facility were used in this study. They were maintained at controlled environmental conditions (temperature: 24 ± 2°C; relative humidity: 50–60%; 12-h dark–12-h light cycle) in polypropylene cages over husk beddings and were provided standard pellet diet and water ad libitum. Permission from Institutional Animal Ethical Committee was obtained prior to experimentation (IAEC No: 02/010/2011).
Experimental protocol
The rats were divided at random into five groups (n = 6 in each group), and the control group received saline throughout the study period. The second group of rats were treated with propylene glycol 2000 grade (vehicle), third group was treated with AZT alone and the fourth group was administered SBN simultaneously with AZT till the end of experimentation. The fifth group was administered SBN alone. AZT and SBN were administered at 50 and 100 mg/kg body weight (b.w.), respectively. The therapeutic dose of AZT prescribed to humans 25,26 was administered to the rats based on the calculation of body surface area. 27 The dosage of SBN was selected based on our previous studies. 23,24 AZT, SBN and saline were given orally, daily for 45 days and the volume of their administration were maintained at 0.5 ml/100 g b.w. of the animal. Whilst AZT was dissolved in saline, SBN was placed in propylene glycol and administered as suspension.
In this study, we have restricted the presentation of AZT treatment to the subacute period for 45 days because we observed that the above drug-induced hepatotoxicity was at its peak level (as determined by the status of marker enzymes of hepatotoxicity and bilirubin) on day 45, which declined significantly on day 60 till day 90, (but maintaining steady state level of hepatotoxicity) as compared to day 45 (data not shown).
Sample collection and preparation
All the groups of rats were fasted overnight and subjected to mild ether anaesthesia 24 h after the last dose, that is, (on day 46). Blood was quickly withdrawn (2–2.5 ml) from retro-orbital plexus using capillary tube into clean centrifuge tubes. The blood was allowed to clot and then centrifuged at 3000 r/min for 15 min to obtain the serum (1–1.5 ml). The sera were separated and stored (at −20°C) until further use. Rats were then killed by cervical dislocation and livers were quickly excised, washed in saline to remove blood clot and other tissue debris and blotted to dryness. The liver tissue homogenates were prepared (1%) using Tris–hydrochloric acid buffer (0.1 M; pH 7.4) and they were centrifuged (3000 r/min at 4°C for 15 min). The clear supernatants were separated and stored (at −20°C) until further analysis. All the biochemical estimations were performed within 48 h after killing. Approximately, 5 mm3 of liver tissue slices were removed from caudal lobe and fixed immediately in phosphate-buffered formal saline (0.1 M; pH 7.4) and used for histopathological evaluation.
Determination of ALT activity
The activity of alanine transaminase (ALT) in serum and tissue homogenates were estimated as detailed by Reitman and Frankel. 28 This assay quantifies the pyruvate liberated by this enzyme on treatment with 2,4-dinitrophenyl hydrazine and sodium hydroxide at 520 nm using spectrophotometer.
Determination of ALP activity
The phenol liberated by the enzyme in the presence of the substrate disodium phenyl phosphate at alkaline pH 10 was treated with Folin–Ciocalteu’s phenol reagent and sodium carbonate to yield a blue-coloured complex, which was quantified at 640 nm using spectrophotometer as described by King 29 in serum and liver tissues.
Determination of ASAL activity
The serum argininosuccinic acid lyase (ASAL) activity was quantified as described by Campanini et al. 30 Briefly, the arginine released from the substrate sodium argininosuccinate was allowed to react with a mixture of sodium hypochlorite to form a pink colour, whose optical density was quantified at 515 nm using spectrophotometer.
Serum bilirubin
The serum was treated with methanol and diazo reagent and finally incubated in the dark to form purple-coloured azobilirubin complex, whose intensity was measured spectrophotometrically at 540 nm as described by Malloy and Evelyn. 31
Estimation of FFA
The serum free fatty acid (FFA) was extracted using a mixture containing chloroform, heptane, methanol and copper reagent. The FFA copper complex formed was then treated with diethyldithiocarbamate to yield a yellow colour, whose optical density was measured at 440 nm using spectrophotometer as described by Chromy et al. 32
Estimation of TG
Initially, the serum was vigorously vortexed with isopropanol and alumina to remove the phospholipids and was subsequently saponified using isoproponal and potassium hydroxide. Finally, it was treated with sodium metaperiodate reagent and acetyl acetone to develop yellow colour, whose intensity was read at 405 nm using spectrophotometer as described by Varley et al. 33
Estimation of TL
The serum was treated with concentrated sulphuric acid and then the aliquot of this was subsequently treated with phospho-vanillin reagent to develop amber colour, whose optical density was quantified spectrophotometrically at 540 nm as described by Frings and Dunn. 34
Determination of LPO
MDA formed as the end product during the peroxidation of lipids in the liver tissue was made to react with thiobarbituric acid to form a pink-coloured complex, whose optical density was quantified at 523 nm using spectrophotometer for the assay of lipid peroxidation (LPO) in the liver tissue homogenate as described by Ohkawa et al. 35
CAT activity
The dichromate in acetic acid was reduced to chromic acetate when heated in the presence of hydrogen peroxide (H2O2) by the enzyme present in liver tissue homogenate forming an unstable intermediate, that is, chromic acetate to develop a green colour whose intensity of absorbance was measured at 0, 30 and 60 s at 570 nm as described by Sinha. 36
Protein thiol
The protein thiol content was estimated as described by Vendemiale et al. 37 Briefly, the protein present in liver tissue homogenate was precipitated using sulphosalicylic acid, and the pellet was treated with guanidine hydrochloride and its optical density was measured at 412 nm. The samples were treated with 5,5′-dithio bis-2-nitrobenzoate, and the yellow colour developed was measured again after 30 min at 530 nm using spectrophotometer.
Total carbonyl content
The total carbonyl content of liver tissue homogenate was quantified as described by Levine et al. 38 Initially, the tissue homogenates were treated with 2,4-dinitrophenylhydrazine and subsequently with trichloroacetic acid to precipitate proteins. The protein pellet was washed and dissolved with ethyl acetate and ethanol and then treated with guanidine hydrochloride. The optical density of this product was quantified at 360 nm and again at 380 nm using spectrophotometer.
Assay of total proteins in liver tissue
The total protein content of liver tissues were estimated according to the standard method of Lowry et al. 39 using bovine serum albumin as standard.
Histopathological examination
The phosphate-buffered formal saline (0.1 M; pH 7.4)-fixed liver tissues were washed thoroughly and dehydrated subsequently in ethanol and embedded in paraffin wax. Thin section (4-µm thickness) of liver tissues was cut and stained using haematoxylin and eosin. The sections were permanently mounted on glass slides using DPX mount 40 and were examined under microscope with photographic facility (Motic Images plus version 2.0) and their photomicrographies were taken.
Statistical analysis
The results are presented as mean ± SD. Statistical comparisons of data were performed by one-way analysis of variance (ANOVA). Post hoc multiple comparison tests were employed to assess the degree of significance of difference between the means of various treatment groups employing Tukey’s test using SPSS software (version 16.0). The value of p < 0.05 was considered significant.
Results
Serum marker enzymes and bilirubin
The activities of marker enzymes of hepatotoxicity (ALT, ALP, and ASAL) and bilirubin in AZT- and SBN-treated rats are presented in Table 1. The data show a highly significant (p < 0.001) increase in the activities of ALT and ALP in serum of rats treated with AZT alone. This treatment also produced a significant increase in bilirubin (p < 0.001) as well as in ASAL (p < 0.05) as compared to the saline-treated control. Simultaneous treatment of SBN with AZT significantly (p < 0.001) mitigated the activities of all the above parameters towards normalcy as compared to AZT alone treatment. Propylene glycol and SBN alone treatments did not produce any change in all the parameters investigated in serum and they are comparable with the control.
Effect of AZT and SBN treatments on ALT, ALP, ASAL and bilirubin in serum of rats.a

AZT: zidovudine; SBN: silibinin; ALT: alanine transaminase; ALP: alkaline phosphatase; ASAL: argininosuccinic acid lyase.
aResults are expressed as mean ± SD (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
bControl compared with propylene glycol, AZT, AZT + SBN and SBN treatments.
cp < 0.001: represents very high significant difference.
dp < 0.05: represents significant difference.
ep < 0.01: represents highly significant difference.
fAZT compared with AZT + SBN treatment.
ALT, ALP and protein levels in liver tissue
In the liver tissue, AZT alone treatment produced almost 40% fall (p < 0.01) in the activity of ALT but did not produce any change in the levels of ALP and protein as compared to the control. SBN treatment with AZT significantly (p < 0.05) improved the fall in the status of ALT activity as compared to AZT alone treatment. Propylene glycol and SBN alone treatments did not alter the levels of ALT, ALP and protein in the liver and their levels were on par with the control (Table 2).
Effect of AZT and SBN treatments on ALT, ALP and protein in liver tissue of rats.a

AZT: zidovudine; SBN: silibinin; ALT: alanine transaminase; ALP: alkaline phosphatase.
aResults are expressed as mean ± SD (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
bControl compared with propylene glycol, AZT, AZT + SBN and SBN treatments.
cp < 0.01: represents highly significant difference.
dAZT compared with AZT + SBN treatment.
ep < 0.05: represents significant difference.
Changes in serum lipids
The serum TLs as well as FFAs were elevated highly significantly (p < 0.001) as compared to control in rats treated with AZT alone. This treatment, however, did not produce any change in TGs and its levels were comparable with the control. Simultaneous treatment of SBN with AZT significantly mitigated the increase (p < 0.001) in the levels of total lipids as well as FFAs as compared to AZT alone treatment. There was no change in the levels of all the above serum lipids in rats treated with propylene glycol and SBN alone (Table 3) and their values were comparable with the control.
Effect of AZT and SBN treatments on FFA, TG and TL in serum lipids of rats.a

AZT: zidovudine; SBN: silibinin; ALT: alanine transaminase; FFA: free fatty acid; TG: triglyceride; TL: total lipids.
aResults are expressed as mean ± S.D (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
bControl compared with propylene glycol, AZT, AZT + SBN and SBN treatments.
cp < 0.001: represents very high significant difference.
dAZT compared to AZT + SBN treatment.
Changes in antioxidant parameters
The effect of AZT and SBN treatment on LPO, CAT, protein thiol and total carbonyl content are presented in Table 4. AZT alone treatment produced a highly significant (p < 0.001) increase in MDA and total carbonyl levels (p < 0.001) in the liver tissue as compared to control. The above treatment also produced a highly significant (p < 0.001) fall (by around 40% of control level) in the activity of CAT as well as in protein thiol (by around 25% of control level). SBN treatment with AZT significantly (p < 0.001) mitigated the increase in MDA levels towards normalcy as compared to the AZT alone treatment. This treatment also alleviated the status of total carbonyl content significantly (p < 0.001) as compared to AZT alone treatment, but it did not protect its levels completely towards normalcy as compared to control. Simultaneous treatments of SBN with AZT completely blocked the fall in the status of protein thiol as well as CAT levels towards normalcy and their values were comparable to the control (p < 0.001). Propylene glycol and SBN alone treatments did not produce any change in all the above antioxidant parameters investigated in the liver tissue and they were comparable with the control.
Effect of AZT and SBN treatments on LPO, CAT, protein thiol and carbonyl content in liver tissue of rats.a

aResults are expressed as mean ± SD (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
bnanomoles of MDA formed per minute milligram protein.
cmicromoles of H2O2 utilized per minute milligram protein.
dControl compared to propylene glycol, AZT, AZT + SBN and SBN treatments.
fAZT compared to AZT + SBN treatment.
ep < 0.001: represents very high significant difference.
Liver histopathology
Histopathological examination of liver tissues is presented in Figure 1. The control liver shows normal cellular architecture with hepatic parenchymal cells radiating from central vein and normal sinusoidal space (Figure 1(a)). AZT alone-treated rats show moderate parenchymal cell hypertrophy, dilation of sinusoidal space with mild inflammatory cell infiltration (Figure 1(c)). Simultaneous treatment of SBN with AZT shows considerable reduction in cellular hypertrophy, sinusoidal space and inflammatory cell infiltration (Figure 1(d)). Propylene glycol and SBN alone treatments show normal architecture of the liver and are comparable with the control (Figure 1(b) and (d)).
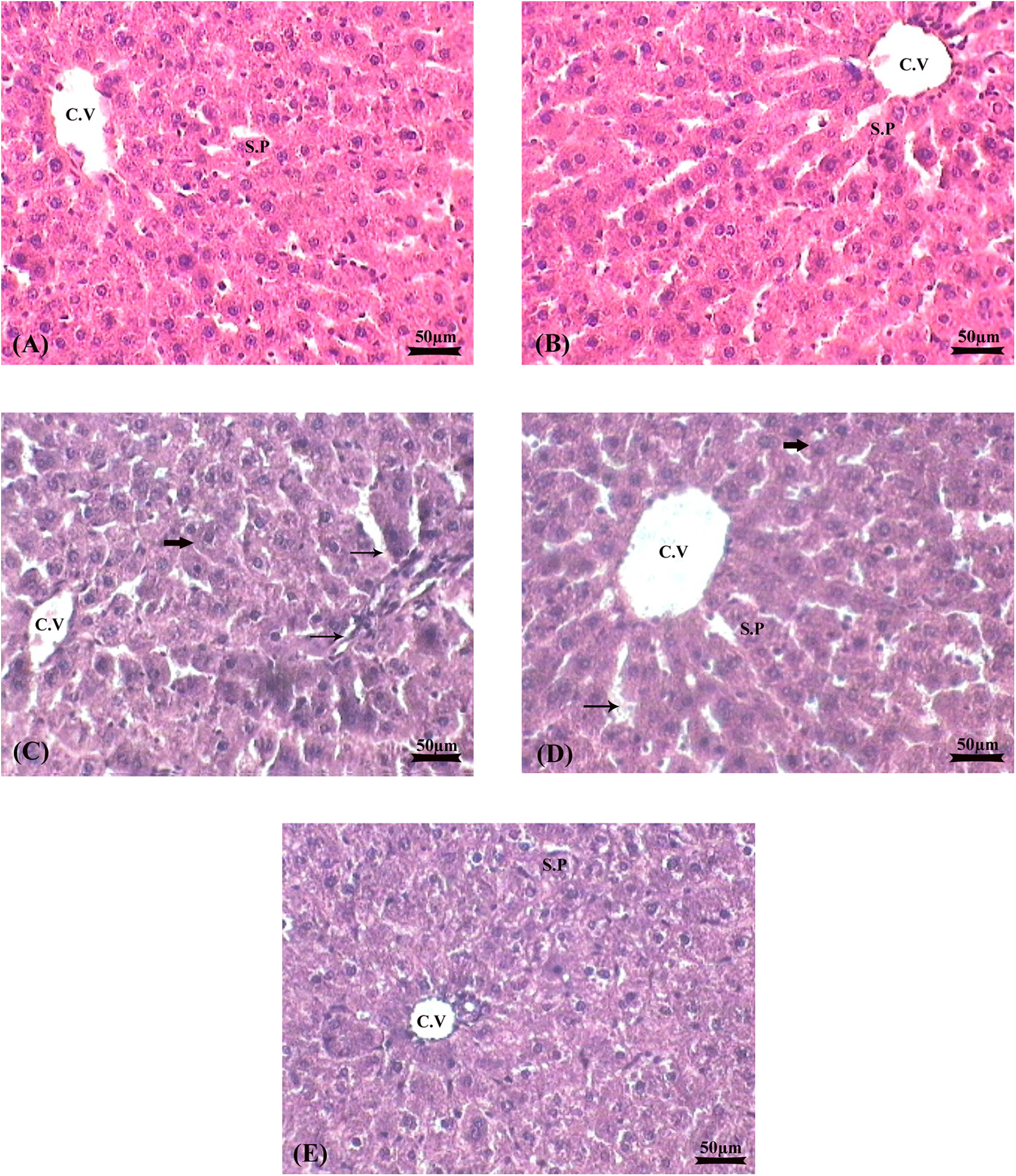
Histopathology of rat liver (H&E). Experimental protocol is described under materials and methods section. (a) Control rats show normal architecture of the liver with hepatocytes radiating from the central vein, sinusoidal space and portal triad; (c) AZT-treated rats show moderate parenchymal cells hypertrophy, dilatation of sinusoidal space with mild inflammatory cell infiltration; (d) (AZT + SBN)-treated rats show reduction in cellular hypertrophy, sinusoidal space and inflammatory cell infiltration; (b and e) Propylene glycol and SBN alone-treated rats show normal architecture, comparable with control. Magnification ×40. Arrows indicate: CV: central vein; SP: sinusoidal space, (⟶) dilatation of sinusoidal space with inflammatory cell infiltration; (⟶) parencymal cell hypertrophy. Optic microscopy: H&E (×40). H&E: haematoxylin and eosin; AZT: zidovudine; SBN: silibinin.
Discussion
Liver toxicity is one of the most relevant adverse drug reactions of AZT, and there is no clear explanation on the mechanism of induction of toxic hepatitis in patients undergoing this therapy because clinical presentations are the same as in other types of toxic hepatitis. 41 AZT antiretroviral treatment have already been shown to significantly increase the levels of transaminases in humans 42 and rats. 12 Transaminases and ALP are sensitive indicators of cellular injury and their elevation in serum is said to occur consequent to necrosis of hepatocytes, leading to their leakage during toxin-induced insult to liver. 43 In this investigation, AZT treatment caused a highly significant increase in ALT and ALP activity in serum accompanied by fall in their activities in liver tissue and our results are in agreement with the above report. The hepatocellular degenerative changes, marked as parenchymal cells hypertrophy, mild inflammatory cell infiltration and sinusoidal dilation observed in the histopathological evaluation in the present work are well corroborated with the biochemical investigations.
The characteristic laboratory finding of cholestasis is the elevation in the levels of serum ALP, the enzyme present in abundance in bile duct epithelium and in the canalicular membrane of the hepatocytes, associated with elevation of serum bilirubin. It is proposed that rupture of bile duct canaliculi leads to the extravasation of bile into the sinusoidal space leading to its accumulation, finally resulting in foamy degeneration of the hepatocytes. Further, obstructive cholestasis is reported to disrupt parenchymal cells resulting in bile leakage. 44 It is an established fact that ALP is a membrane-bound enzyme and its elevation in serum denotes hepatocellular membrane damage. 45 In this study, AZT treatment produced highly significant increase in bilirubin as well as ALP and these observations demonstrate that this toxin-induced insult to the liver could also precipitate biliary obstruction resulting in mild hyperbilirubinemia in addition to hepatocellular necrosis. Elevation of ALP and bilirubin were reported in humans and rats upon subacute or chronic administration of AZT, and our results are in agreement with these reports. 12,43
It is postulated that ASAL, which is present in minute quantity in liver cells, are markedly elevated in certain diseases such as viral hepatitis, chronic hepatitis, cirrhosis, metastasis, hepatomas and cholecytitis. This enzyme is located exclusively in parenchymal cells and is not found in Kupffer cells or any other liver cells, and it is leached out only during cell damage. Hence, an increase in the activity of ASAL is often considered as the more precise indicator of liver toxicity. 30,46,47 In this study, subacute AZT alone treatment caused a significant increase in ASAL activity and this observation also substantiate the contention that AZT treatment produce parenchymal cell damage. Studies regarding elevation of ASAL in AZT treatment have not been reported elsewhere and probably ours is the first report. Alteration in protein metabolism is shown to occur in hepatic dysfunction. Derangement in protein metabolism as well as metabolism of essential amino acids is evidenced by enhanced activities of transaminases during drugs- and chemicals-induced liver damage. 48 In this study, the non-significant fall in liver tissue proteins observed in AZT-treated rats indicates that this toxin affects protein metabolism during progression of hepatocellular dysfunction.
Quantification of serum lipids is considered as valuable biomarkers to assess the impairment of lipid metabolism in the liver. Administration of antiretroviral drugs has been shown to enhance fat accumulation due to their ability to induce lactic acidosis through the channels of acetyl CoA-mediated ketogenesis. 49 Reports on AZT-induced changes in plasma lipids in humans and experimental animals are scanty. In this study, AZT treatment caused a highly significant elevation in serum TLs and FFAs and no change in TGs. Maisonneuve et al. 50 reported no change in plasma TG level in AZT-treated mice and our present results are in agreement with this report. The observation of increase in TLs and FFAs denotes the hyperlipidaemic potential of AZT treatment and these results are not reported elsewhere. The precise mechanism of AZT-induced hyperlipidaemia could not be ascertained, and it is likely that this adversity could be due to enhanced mobilization of depot fats from adipose tissue into the blood stream or might be due to impairment in metabolism of dietary lipids. Further studies, however, are required to understand the precise molecular mechanism of AZT-induced hyperlipidaemia.
Oxidative stress is a common pathological mechanism that is observed during progression of varieties of liver disorders. 51 The breakdown product of lipid peroxidation is MDA and its elevation is proved to be a good biomarker for the evaluation of various drugs- and chemicals-induced oxidative stress in experimental animals. Depletion of antioxidant defence and induction of LPO results in the elevation of MDA levels. 52 Further, free radicals-induced damage to protein thiol has also been implicated as the cause for oxidative stress. Additionally, it is proposed that carbonyl groups are also produced on protein side chains during oxidative stress. 53 In the present investigation, AZT treatment produced a highly significant increase in MDA formation and total carbonyl content. Previous studies have shown LPO and oxidative stress in AZT-treated rats, 12,54 and our results are in agreement with this report. CAT is a heme protein that catalyses the conversion of H2O2 into O2, and it is a major enzyme in the liver tissue that regulates intracellular H2O2 by its destruction in the cells. Thus, CAT protects biological system from oxidative stress induced by drugs and chemicals. 55 The fall in the activity of CAT in AZT-treated rats observed in this study could be due to the involvement of this enzyme towards suppression of oxidative stress. Reports regarding the status of protein thiols and total carbonyl content and their role in oxidative stress induced by AZT are unavailable. In this study, AZT treatment produced a highly significant decrease in protein thiol and increase in total carbonyl content in liver, which are indicators of oxidative stress as reported earlier. 12 Many crucial signalling pathways utilize reversible oxidation and reduction of cysteine thiols of protein as a molecular switch. Some proteins have apparent redox-sensing ability, especially the proteins thiol (CH2–SH) and their oxidation potential are likely to be redox sensitive. 56 Significant reduction in protein thiol content is reported in muscles during oxidative stress. 57 In view of these reports, it is likely that the highly significant fall in the status of protein thiol in AZT-treated rats could be attributed to the oxidative stress-inducing properties of this toxicant.
AZT, a prodrug is a thymidine analogue in which the 3′-OH of thymidine is replaced by an azide group. It is hypothesized that cellular thymidine kinase phosphorylates AZT into its monophosphate, diphosphate and triphosphate (AZT-TP) derivatives. The AZT-TP is a competitive inhibitor of cellular deoxynucleoside-5′-triphosphate, an essential substrate for proviral DNA. 3 It is still unclear and no proof is available to substantiate the generation of free radicals consequence to metabolism of AZT in liver. However, it is postulated that AZT-induced toxicity could be due to alteration in liver mitochondrial DNA, oxidative phosphorylation coupling and changes in fine ultrastructure of liver mitochondria. Moreover, it is suggested that generation of reactive oxygen species, peroxide production and oxidative damage in mitochondria might play a vital role in AZT-induced toxicity. 15,16
In the present investigation, ameliorative effect of AZT-induced hepatocellular degenerative changes, cholestasis, oxidative stress as well as hyperlipidaemia is well evidenced in rats simultaneously treated with SBN and AZT. The hepatoprotective effect is shown by mitigation of increase in the activities of marker enzymes of liver toxicity, bilirubin and protein in serum and fall in the activities of enzymes in liver tissue. Alleviation of oxidative stress is revealed by prevention of increase in LPO, carbonyl content and decrease in CAT and protein thiol. The mitigation of hyperlipidaemia is shown by restoration of increase in plasma TGs and FFAs towards normalcy in rats treated with SBN and simultaneously with AZT. This protective effect of SBN is also demonstrated by histopathological evaluation of liver tissues.
It remains to be elucidated whether simultaneous treatment of SBN in HIV-infected patients taking therapeutic doses of AZT could potentially be alleviated from the adverse effects of AZT-induced hepatotoxicity, oxidative stress and hyperlipidaemia. It is cautioned that phase-III clinical trials are warranted to evaluate the potency of SBN in HIV patients. Several studies have shown the protective role of SBN against various drugs and chemicals-induced liver damage by way of its inherent free radical scavenging and antioxidant property. 17,58 It is reported that the extract of S. marianum, which is rich in SBN, is a membranotropic agent in nature and binds tightly to hepatocellular membrane 59 and thus protects the liver against various drugs and chemicals-induced liver toxicity. Moreover, interaction of SBN and silymarin with polar head groups of phospholipids at the lipid–water interphase of cellular membrane and their counteraction of LPO is said to be the major cause for these bioflavonoids to act as excellent protective agent against LPO, and free radicals-induced peroxidative damage on cellular membrane. 60,61 In view of these reports, it is likely that the above mechanisms would have contributed towards maintenance of cellular integrity and protection against free radicals-induced oxidative damage to the hepatocellular membrane by SBN against AZT-induced hepatocellular degenerative changes, hyperbilirubinemia, hyperlipidaemia and oxidative stress. Further, such mechanism could have also contributed in counteracting the LPO by its inherent antioxidant and free radical scavenging property of SBN.
It is an established fact that cellular injury and inflammatory response rising out of toxicity induced by several agents cause the rapid expression of transcription factors such as nuclear factor κB (NF-κB). 62 Suppression of NF-κB activity is a vital step towards healing inflammatory response, and this effect has been contributed by several pharmacological antioxidant agents. Silymarin treatment (which is rich in SBN) has been shown to suppress both the κB motif of NF-κB DNA-binding activity and its gene expression in hepatic cells. In addition, it blocks translocation of NF-κB (p65 protein to phosphorylation) and its translocation into nucleus without affecting its ability to bind DNA. 63 In view of these reports, it is likely that the ameliorative property of SBN treatment simultaneously with AZT could also be due to the ability of SBN to inhibit the expression of transcription factor NF-κB. However, it is proposed that further studies are warranted to ascertain the precise mechanism of the role of SBN in inhibiting this signal transduction pathway against AZT-induced toxic insult to the liver.
In conclusion, our present results demonstrate the mitigating potentials of SBN against AZT-induced hepatocellular degenerative changes, cholestasis, hyperlipidaemia and oxidative stress induced by AZT by way of its antioxidant, free radical scavenging and membrane-stabilizing property. Whether this beneficial effect could be used for therapeutic purposes against AZT-induced hepatotoxicity in HIV-infected patients warrants further investigation.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The financial assistance provided by UGC, New Delhi, India, by way of Major Research Project grant (UGC, New Delhi letter No. F.37-441/2009 (SR) for conducting this study is greatly acknowledged. The authors thank the Director, Central Drug Testing laboratory, Govt of India, Chennai, Tamil Nadu, India, for providing AZT as gratis.
