Abstract
Alpha-amanitin (α-AMA), the primary toxin of Amanita phalloides, is known to cause nephrotoxicity and hepatotoxicity. Resveratrol is an antioxidant that has shown efficacy in many nephrotoxicity models. The aim of this study was to investigate the effects of resveratrol against the early and late stages of α-AMA-induced nephrotoxicity, compared to those of silibinin, a well-known antidote for poisoning by α-AMA-containing mushrooms. Mice kidney tissues were obtained from five groups: (1) α-AMA + NS (simultaneous administration of α-AMA and normal saline), (2) α-AMA + SR (simultaneous administration of α-AMA and resveratrol), (3) α-AMA + 12R (resveratrol administration 12 h after α-AMA administration), (4) α-AMA + 24R (resveratrol administration 24 h after α-AMA administration), and (5) α-AMA + Sil (simultaneous administration of α-AMA and silibinin). Histomorphological and biochemical analyses were performed to evaluate kidney damage and oxidant–antioxidant status in the kidney. Scores of renal histomorphological damage decreased significantly in the early resveratrol treatment groups (α-AMA + SR and α-AMA + 12R), compared to those in the α-AMA + NS group (p < 0.05). Catalase levels increased significantly in the α-AMA + SR group, compared to those in the α-AMA + NS group (p < 0.001). Early resveratrol administration within 12 h after α-AMA ingestion may reverse the effects of α-AMA-induced nephrotoxicity, partly through its antioxidant action, thereby suggesting its potential as a treatment for poisoning by α-AMA-containing mushrooms.
Introduction
Cyclopeptide-containing mushrooms are the primary mushrooms involved in deaths from mushroom poisoning. Amanita phalloides is the most commonly found cyclopeptide-containing mushrooms. 1 The main toxins of these mushrooms include amatoxins, phallotoxins, and virotoxins, with the heat-stable bicyclic octapeptide alpha-amanitin (α-AMA) as the primary toxin. 2,3 α-AMA is a highly potent toxin; ingestion of just one mushroom can lead to toxicity, resulting in severe morbidity or mortality in a healthy individual. 4 α-AMA enters hepatocytes via the organic anion transporter peptide (OATP) 1B3. Inhibition of RNA polymerase II activity and mRNA expression and reduction of protein synthesis represent the main toxic mechanism of α-AMA in eukaryotic cells. 2,4 Clinically, patients are asymptomatic at the first 8–12 h after A. phalloides ingestion. Watery diarrhea, colic-like abdominal pain, and vomiting occur within 24 h after ingestion (the gastrointestinal period). Liver enzyme levels increase in the third cytotoxic period between 24 and 48 h. Finally, hepatic and renal failure are observed in the fourth (fulminant) period between 2 and 4 days after ingestion. 5
Although α-AMA is well-known to target the liver, this toxin is not metabolized in the liver and is eliminated in large quantities by the kidneys via urine. 2 Its level in kidney tissue is also found to be much higher than that in the liver. 2,4 Therefore, acute tubular necrosis followed by renal failure is one of the most important consequences of α-AMA poisoning. Studies have determined oxidative stress and inflammation as the main mechanisms of α-AMA-induced nephrotoxicity. 6 –11
Silibinin exerts antidote effect against A. phalloides poisoning by acting as an OATP1 inhibitor. Although penicillin G has been used as an alternative antidote to silibinin, there is no strong evidence of its effectiveness against mushroom poisonings. Despite these treatments, mortality rate in A. phalloides type mushrooms poisoning is still high. 2,12 Therefore, new treatment alternatives are needed.
Resveratrol is a compound containing non-flavonoid stilbenes found in edible plants, such as red grapes, peanuts, pine, and legumes. 13 It exhibits protective and therapeutic effects in broad-spectrum pathologies, such as neuronal, renal, cardiovascular, and neoplastic conditions. 14 Resveratrol has been shown to exhibit protective and therapeutic effects through anti-inflammatory and antioxidant activities in many models of nephrotoxicity and hepatotoxicity, such as those induced by gentamicin, cisplatin, and methotrexate. 15 –17
Recently, the therapeutic efficacy of resveratrol has been shown in a mice model of α-AMA-induced hepatotoxicity. 18 However, its therapeutic effect on α-AMA-induced nephrotoxicity has not been elucidated. Therefore, this study aimed to investigate the therapeutic effects of resveratrol against both the early and late stages of α-AMA-induced nephrotoxicity in vivo.
Materials and methods
Materials
Archival paraffin-embedded kidney samples, frozen kidney samples (−80°C), and frozen serum samples (−20°C) were used in this study. These samples were obtained from BALB/c mice in a previous study (44/2014). 18 Dokuz Eylul University Multidisciplinary Laboratory Animal Studies Local Ethic Committee approved the study in terms of using previously collected mouse kidney tissues and blood specimens (DEU HADYEK, 21/2016). The study was conducted in the Department of Pharmacology, Dokuz Eylul University between April and October 2017.
Experimental groups
α-AMA at 1.4 mg/kg dose intraperitoneally (i.p.) was confirmed to exert hepatotoxic effects in the previous study 18 ; thus, it was determined whether it exerts nephrotoxicity before starting this study. The nephrotoxic effect of α-AMA at 1.4 mg/kg dosage was evaluated histomorphologically by comparing with the vehicle group in which dimethyl sulfoxide (DMSO) was administrated to mice, which was used as a solvent for resveratrol and silibinin. To examine the effects of resveratrol (i.p.) on both the early and late stages of α-AMA-induced nephrotoxicity, the samples were divided into five groups: α-AMA + NS (simultaneous administration of α-AMA and normal saline), α-AMA + SR (simultaneous administration of α-AMA and resveratrol), α-AMA + 12R (resveratrol administration 12 h after α-AMA), α-AMA + 24R (resveratrol administration 24 h after α-AMA), and α-AMA + Sil (simultaneous administration of α-AMA and silibinin). The resveratrol and silibinin doses used were 30 mg/kg and 5 mg/kg via administrated i.p. route, respectively. 19,20 In the 48-h experimental protocol, resveratrol was administered at 12-h intervals, whereas silibinin was administered at 6-h intervals. 18 All injections were administered intraperitoneally, as in previous studies. 6,7,15 –20 At the end of the 48 h, mice were euthanized under ether anesthesia.
Histomorphological examination
Kidney tissues were examined under light microscopy (Olympus BX-50; Olympus, Japan). Five-micrometer-thick sections were obtained and stained with hematoxylin–eosin (H&E). Next, structural changes were examined in 100 glomerular proximal tubules in each sample, whereas tubular atrophy, tubular brushed edge loss, tubular dilatation, cast formation, vacuolization, mononuclear cell infiltration, erythrocyte extravasation, changes in renal corpuscular morphology, and changes in the interstitial area were evaluated semi-quantitatively. Renal histological damage was scored from 0 to 3 (0: denotes no change, 1: change affecting <25% tubular damage, 2: change affecting 25–50% of tubules, 3: change affecting >50% of tubules). 21
Assessment of renal function
Serum urea and creatinine levels were measured using the methods recommended by the International Federation of Clinical Chemistry (IFCC) on a Hitachi 902 analyzer (Roche, Basel/Switzerland). The results were expressed as mg/dL.
Preparation of kidney tissue for analysis of oxidant–antioxidant status
Kidney tissues (0.1 g) were homogenized in 0.1 mM Tris/HCl buffer (pH 7.4) containing 0.5% Triton X-100, 0.1 mg/ml phenylmethanesulfonyl fluoride, and 5 mM β-mercaptoethanol, by using a tissue lyser (TissueLyser II; Qiagen, UK). Homogenates were centrifuged for 10 min at 2500 × g. Next, the supernatants were collected for malondialdehyde (MDA) analysis, and the homogenates were further centrifuged at 14,000× g for 5 min (4°C) to obtain supernatant for superoxide dismutase (SOD) assay. 22 The homogenates for catalase and glutathione peroxidase (Gpx) analyses were prepared according to the instructions in each kit, as described below.
Determination of MDA level in kidney tissue
MDA levels in the kidney were analyzed by a high-performance liquid chromatography 23 method that depends on the formation of MDA-thiobarbituric acid complex. The samples were injected onto a column (Nucleosil 100-5; Macherey-Nagel Inc., Diiren, Germany), and the intensity of the complex was fluorometrically measured at excitation and emission wavelengths of 515 and 553 nm, respectively. A standard calibration curve was constructed by using 1,1,3,3-tetraethoxypropane (Sigma, St. Louis, Missouri, USA). Data were expressed as µmol/µg protein.
Determination of SOD activity in kidney tissue
SOD activity in kidney homogenates was determined by using a colorimetric assay kit (K335-100; BioVision, Milpitas, California, USA) according to the manufacturer’s instructions. The principle of this assay was based on the reduction of water-soluble tetrazolium (WST-1) salt to WST-1 formazan with superoxide anion, which was generated by xanthine oxidase. Inhibition of this reaction by SOD was evaluated at 450 nm. Data were expressed as U/mg protein.
Determination of catalase activity in kidney tissue
Catalase activity in kidney homogenates was analyzed by a colorimetric assay kit (K773-100; BioVision) according to the manufacturer’s instructions. First, H2O2 was converted to water and oxygen by catalase. Second, OxiRed™ probe was added to the solution to generate a product that can react with unconverted H2O2. Finally, the reaction was monitored at 570 nm. Catalase activity is inversely proportional to the signal. Data were expressed as U/mg protein.
Determination of Gpx activity in kidney tissue
Gpx activity was measured by a colorimetric assay kit (K762-100; BioVision) according to the manufacturer’s instructions. Briefly, a reaction mixture containing glutathione, NADPH, glutathione reductase, and assay buffer was prepared. Cumene hydroperoxide (CuOOH) was added to the reaction mixture to initiate the enzymatic reaction. Changes in absorbance were monitored at 340 nm. Data were expressed as U/mg protein.
Protein assay
Protein levels in kidney tissue were measured by the bicinchoninic acid (Sigma, Munich, Germany) method. Standard calibration curve was generated by using bovine serum albumin. 24
Statistical analysis
Statistical analysis of biochemical parameters and antioxidant enzymes were performed using one-way analysis of variance. Tukey–Kramer multiple comparisons test was performed to determine differences between groups. Statistical analyses of histomorphological parameters were performed by the nonparametric Mann Whitney-U and Kruskal Wallis tests (GraphPad Prism version 5.00 for Windows, GraphPad Software, San Diego, California USA). Data were shown as mean ± standard deviation. p < 0.05 was considered statistically significant.
Results
Confirmation of nephrotoxicity
As shown in Figure 1, 1.4 mg/kg α-AMA resulted in marked increases in mononuclear cell infiltration, tubule dilatation, and loss of brush border (Figure 1(a)). Also, histomorphological damage score was found to be significantly increased in α-AMA group (2.7 ± 0.2) compared to the vehicle group (0.6 ± 0.03) (p < 0.01) (Figure 1(b)).

(a) Representative histochemical stainings (H&E) micrographs of α-AMA at 1.4 mg/kg dose (a1,a2) and Dimethyl sulfoxide (DMSO) (b1,b2). Mononuclear cell infiltration (*), brush border loss and proximal tubular cell debris in the tubular lumen (Þ), and proteinous material accumulation in tubule lumen (®). (b) Scores of renal histomorphological damage in the kidney tissue. Data are expressed as Mean ± SD. **p < 0.01 vs vehicle group.
Histomorphological examination
The results of histomorphological examination of kidney tissues in all groups are presented in Figure 2(a). Kidney tissues from the α-AMA + NS group showed abnormal morphology characterized with mononuclear cell infiltration, tubular dilatation, proteinaceous substance accumulation, brush border loss, and cell debris in the proximal tubular lumen. However, mononuclear cell infiltration and proximal tubular cell debris decreased in the α-AMA + SR and α-AMA + 12R groups, compared to those in the α-AMA + NS group (Figure 2(a)). Consistent with these histomorphological findings, renal damage scores decreased significantly in the α-AMA + SR (2.14 ± 0.14) and α-AMA + 12R (2.11 ± 0.20) groups, compared to that in the α-AMA + NS (2.8 ± 0.1) group (p < 0.01 and p < 0.05, respectively). There was no difference in the late resveratrol (α-AMA + 24R, 2.6 ± 0.2) and silibinin (α-AMA + Sil, 2.2 ± 0.3) treatment groups compared to the α-AMA + NS group (p > 0.05) (Figure 2(b)).
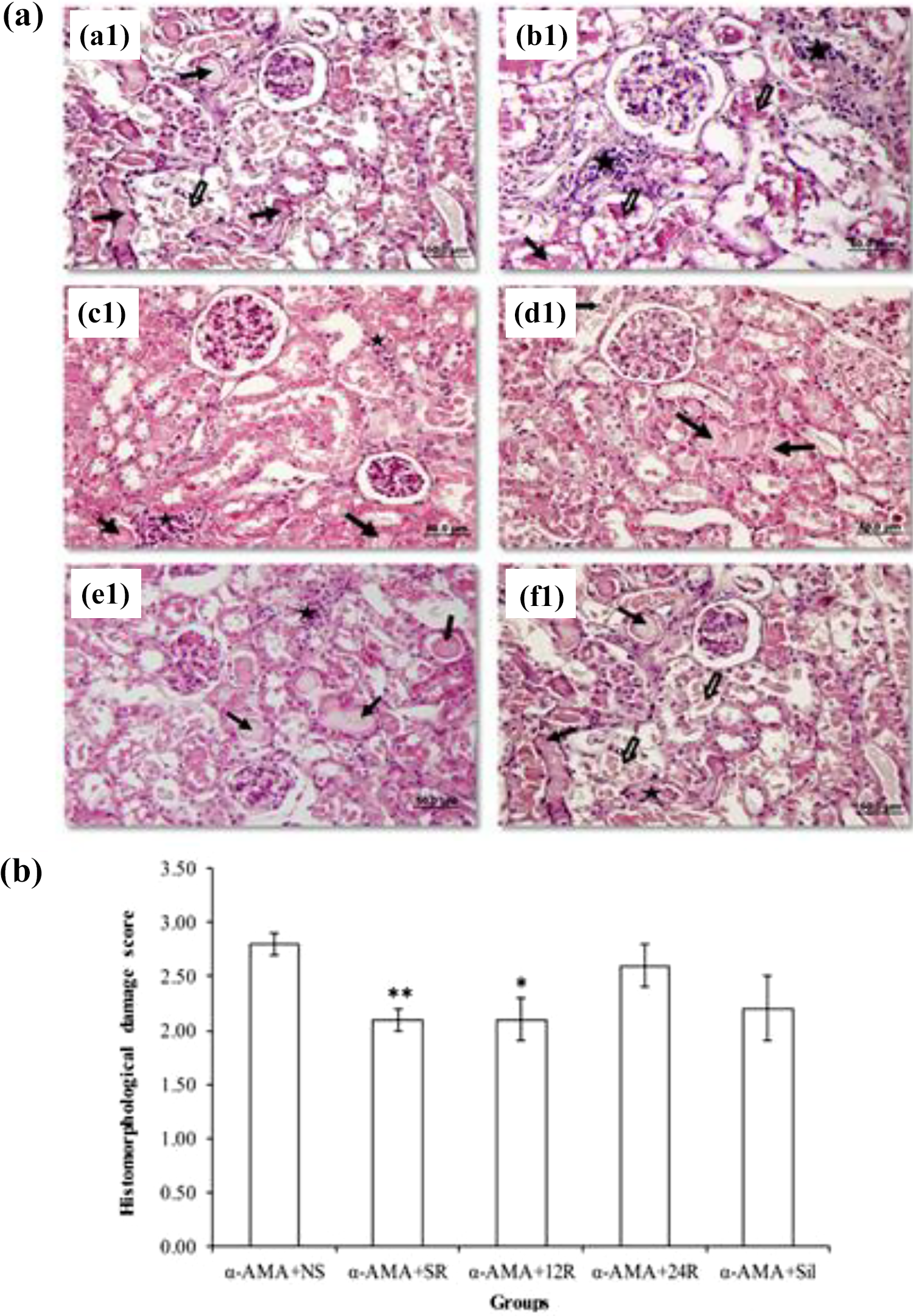
(a) Photomicrographs of H&E stained kidney tissue sections. Mononuclear cell infiltration (*), brush border loss and proximal tubular cell debris (⇒), and proteinous material accumulation in tubule lumen (→) in the α-AMA + NS group (a1, b1). Mononuclear cell infiltration and proximal tubular cell debris decreased in the α-AMA + SR and AMA + 12R groups compared to those in the α-AMA + NS group (c1, d1). In the α-AMA + 24R and α-AMA + Sil group, all these parameters were marked as in α-AMA + NS group (e1, f1). (b) Scores of renal histomorphological damage in the experimental groups. Data are expressed as mean ± SD. *p < 0.05 versus α-AMA + NS group, **p < 0.01 versus α-AMA + NS group. α-AMA: alpha-amanitin; H&E: hematoxylin–eosin; α-AMA + NS: simultaneous administration of α-AMA and normal saline; α-AMA + SR: simultaneous administration of α-AMA and resveratrol; α-AMA + 12R: resveratrol administration 12 h after α-AMA; α-AMA + 24R: resveratrol administration 24 h after α-AMA; α-AMA + Sil: simultaneous administration of α-AMA and silibinin.
Renal functions
There was no difference among the experimental groups in the serum concentrations of urea and creatinine (Table 1).
Serum urea and creatinine levels in the experimental groups.a

α-AMA + NS: simultaneous administration of α-AMA and normal saline; α-AMA + SR: simultaneous administration of α-AMA and resveratrol; α-AMA + 12R: resveratrol administration 12 h after α-AMA; α-AMA + 24R: resveratrol administration 24 h after α-AMA; α-AMA + Sil: simultaneous administration of α-AMA and silibinin.
a Data are expressed as mean ± SD.
Tissue MDA levels
Tissue MDA levels decreased in α-AMA + SR, α-AMA + 12R, and α-AMA + 24R groups (5.2 ± 1.0, 5.2 ± 1.0, and 4.5 ± 1.2 µmol/mg protein, respectively) compared to that in the α-AMA + NS group (11.7 ± 1.3 µmol/mg protein) (Figure 3(a)) (p < 0.01).

Oxidant/antioxidant status in the kidney tissue of the experimental groups. (a) MDA, (b) SOD, (c) catalase, and (d) Gpx levels in the experimental groups. Data are expressed as mean ± SD. *p < 0.05 versus α-AMA + NS group, **p < 0.01 versus α-AMA + NS group, and ***p < 0.001 versus α-AMA + NS group; # p < 0.05 versus α-AMA + SR group, ## p < 0.01 versus α-AMA + SR group, ### p < 0.001 versus α-AMA + SR group, and &p<0.05 versus α-AMA + 12R group. α-AMA: alpha-amanitin; MDA: malondialdehyde; SOD: superoxide dismutase; Gpx: glutathione peroxidase; α-AMA + NS: simultaneous administration of α-AMA and normal saline; α-AMA + SR: simultaneous administration of α-AMA and resveratrol; α-AMA + 12R: resveratrol administration 12 h after α-AMA.
Tissue SOD, catalase, and Gpx activities
Tissue SOD activity increased in the α-AMA + SR (0.4 ± 0.03 U/mg protein) and α-AMA + 12R (0.4 ± 0.03 U/mg protein) groups, compared to that in the α-AMA + NS group (0.2 ± 0.03 U/mg protein) (p < 0.05). However, SOD activity decreased in the α-AMA + 24R (0.2 ± 0.03 U/mg protein) group compared to those in the α-AMA + SR (0.4 ± 0.03 U/mg protein) and α-AMA + 12R (0.4 ± 0.03 U/mg protein) groups (p < 0.05) (Figure 3(b)).
Tissue catalase activity increased in the α-AMA + SR (0.6 ± 0.03 U/mg protein) group, compared to that in the α-AMA + NS (0.3 ± 0.03 U/mg protein) group (p < 0.001). However, catalase levels decreased in the α-AMA + 12R (0.4 ± 0.03 U/mg protein), α-AMA + 24R (0.4 ± 0.07 U/mg protein) (p < 0.01), and α-AMA + Sil (0.4 ± 0.01 U/mg protein) groups, compared to that in the α-AMA + SR (0.6 ± 0.03 U/mg protein) group (p < 0.01, p < 0.01, and p < 0.001, respectively) (Figure 3(c)). On the other hand, there were no significant differences in tissue Gpx activity level among groups (p > 0.05) (Figure 3(d)).
Discussion
In the present study, the therapeutic effects of resveratrol on both the early and late stages of α-AMA-induced nephrotoxicity was investigated and compared with those of silibinin. The results showed that resveratrol might be effective against α-AMA-induced nephrotoxicity, especially at the early stage. This effect might be related, in part, to its antioxidant effect.
Upon ingestion of A. phalloides, α-AMA is absorbed by the gastrointestinal tract and enters hepatocytes via OATP1B3, subsequently causing severe hepatotoxicity and resulting in mortality. 5 In addition, α-AMA targets the kidney after ingestion. Previous studies showed that α-AMA levels are higher in the kidney than in the liver, 2,4 thereby causing nephrotoxicity. It was reported that the mechanism underlying α-AMA-induced nephrotoxicity involves oxidative stress and inflammation. 6 –11 In clinical practice, the most commonly used antidote for A. phalloides poisoning is silibinin. 12 Although the therapeutic effect of silibinin against nephrotoxicity induced by polymyxin E and cyclosporine has been investigated in several experimental models, 25,26 there is no information on its effects in an α-AMA-induced nephrotoxicity model. Therefore, we hypothesized that resveratrol, which has strong antioxidant and anti-inflammatory activities, is effective against α-AMA nephrotoxicity. Firstly, 1.4 mg/kg α-AMA, which was previously confirmed to exert hepatotoxicity, 18 was found to induce histomorphological damage in kidney tissue. These findings were consistent with those of previous experimental and clinical studies on A. phalloides poisoning. 6 –8,27 –29
The beneficial effect of resveratrol on the kidney is well reported in various experimental models of nephrotoxicity. 15,17,30 In this study, resveratrol administration, especially within 12 h after α-AMA ingestion, decreased oxidative stress, resulting in significant improvements in renal histomorphological damage and antioxidant enzyme activities in the kidney. Thus, it was more effective in reversing the effects of α-AMA-induced nephrotoxicity if it was administered within 12 h after A. phalloides ingestion. Although concentrations of serum urea and creatinine were found to be decreased in the early resveratrol treatment groups (α-AMA + SR and α-AMA + 12R) compared to the α-AMA + NS group, the difference was not significant. Garcia et al. evaluated the influence of polymyxin B 31 and also combined administration of polymyxin B and methylprednisolone in counteracting α-AMA-induced nephrotoxicity. 11 They reported that although there was no difference in serum urea and creatinine concentrations between α-AMA and treatment groups in both studies for 24-h protocol, treatment resulted in histomorphological improvement in renal damage. 11,31 It means that histomorphological changes may not be sufficient enough to induce significant changes in the serum concentrations of urea and creatinine at the dose levels and duration of the study.
Level of MDA, which is a product of lipid peroxidation, was measured to evaluate oxidative stress. MDA levels in kidney tissue significantly decreased in all mice treated with resveratrol. Resveratrol also increased SOD and catalase activities in the group treated with early resveratrol administration, suggesting its antioxidant activity. These findings supported the results of previous studies on the antioxidant and anti-inflammatory effects of resveratrol on nephrotoxicity models. 16,30 –36 Histomorphological analysis showed significantly decreased mononuclear cell infiltration in early resveratrol treatment groups but anti-inflammation parameters could not be evaluated in this study. Therefore, we concluded that the nephroprotective effects of resveratrol might be attributed mainly to its antioxidant effect. In contrast, silibinin, compared to resveratrol, did not reverse α-AMA-induced nephrotoxicity. Several studies have shown the protective effect of silibinin in experimental nephrotoxicity models. However, not only those studies were different in terms of nephrotoxicity models used, but also the dose of silibinin was higher than that of the dose administered in this study. 25,26 Although silibinin has been shown to exhibit protective effect in several nephrotoxicity models, this effect is not particularly significant in clinical practice because most patients are presented to the emergency department at the late stages of mushroom poisoning. 37 Therefore, even when administered simultaneously with α-AMA ingestion, silibinin still cannot completely prevent nephrotoxicity.
Conclusion
Poisonings with A. phalloides type mushrooms which contain α-AMA is a very important health problem inducing nephrotoxicity as well as hepatotoxicity. Resveratrol exhibits therapeutic effect in an experimental α-AMA-induced nephrotoxicity model. Early resveratrol administration may reverse the effects of α-AMA-induced nephrotoxicity, partly through its antioxidant effect. However, further studies are required to identify the molecular mechanisms underlying the preventive effects of resveratrol against poisoning by α-AMA-containing mushrooms. The results will contribute to the planning of further studies in which resveratrol can be used as an alternative antidote for mushroom poisonings.
Limitations
Resveratrol’s anti-inflammatory effect was verified on the basis of histomorphological data alone. Anti-inflammatory parameters could not be measured.
Footnotes
Authors’ note
This study was presented as a poster at the 43rd FEBS Congress held in Prague in 2018. FEBS Journal, pp. 09–149. This research was partially carried out at the Dokuz Eylul University Medical School Learning Resources Center Research Laboratory (R-LAB).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Dokuz Eylul University Research Foundation (2017.KB.SAG.010).
