Abstract
Chronic use of nucleoside reverse transcriptase inhibitors (NRTIs) in managing human immunodeficiency virus (HIV) infection has been associated with several complications. Available management options for these complications have yielded controversial results, thus the need to urgently find newer alternatives. Naringin, a plant-derived flavonoid, has been shown to possess antioxidant and antiapoptotic properties which can be exploited in managing NRTI-induced complications. This study therefore investigated the effects of naringin on some NRTI-induced complications. Forty-nine rats (200–250 g) were divided into seven groups and were orally treated with stavudine (d4T)-only, d4T + naringin, d4T + vitamin E, zidovudine (AZT)-only, AZT + naringin, AZT + vitamin E, and distilled water, respectively. Drugs were administered once daily for 56 days, and oral glucose tolerance tests conducted on day 54 of the experiments and rats were thereafter sacrificed on day 56 by halothane overdose. Plasma samples and the left gastrocnemius muscles were stored at −80°C for further analysis. There was significant glucose intolerance, insulin resistance, oxidative stress, and apoptosis in the skeletal muscles of AZT- or d4T-only–treated rats. Naringin, however, significantly reduced fasting blood glucose and fasting plasma insulin concentrations, mitigated glucose intolerance, and insulin resistance in addition to reducing malondialdehyde and carbonyl protein concentrations when coadministered with either NRTIs. Furthermore, naringin improved antioxidant enzyme activities, reduced skeletal muscle BCL-2-associated X protein expression, and improved B-cell lymphoma-2 protein expression compared to AZT- or d4T-only–treated rats. Naringin ameliorated AZT- and d4T-induced complications and therefore should be further investigated as a possible nutritional supplement in managing HIV infection.
Introduction
The management of human immunodeficiency virus (HIV) infection has evolved over the years from monotherapy to the present day administration of combination therapy, involving at least three antiretroviral agents. 1 Nucleoside reverse transcriptase inhibitors (NRTIs), protease inhibitors (PIs), integrase inhibitors, and non-nucleoside reverse transcriptase inhibitors are classes of antiretroviral drugs that have been incorporated as components of highly active antiretroviral therapy (HAART) regimens. 2 Although HAART has markedly reduced HIV-associated morbidity and mortality, its administration has been complicated by an increase in the incidence of metabolic abnormalities. 2,3 Most of these metabolic complications are attributed to NRTIs which make up the backbone of HAART and exert their detrimental effects in tissues and organs with high energy requirements or turnover, such as the skeletal muscles and adipocytes. 4 –6
Zidovudine (AZT) and stavudine (d4T) are NRTIs that have traditionally been used in the management of HIV infection. 7 –9 Although the World Health Organization recommended its withdrawal from HAART regimens due to associated life-threatening lactic acidosis and debilitating lipodystrophy, d4T remains one of the main drugs in the management of HIV in resource-poor countries, owing to its reduced cost and relative availability compared to the preferred alternatives. 7 AZT, on the other hand, has continued to maintain its relevance in the prevention of mother-to-child transmission of HIV during pregnancy and at birth and also as immediate prophylaxis following accidental viral exposure. 9,10
Glucose intolerance, insulin resistance, peripheral neuropathy, and myopathy are some metabolic complications associated with AZT and d4T administration. 7,11 –13 Although the precise mechanisms underlying the development of these complications are yet to be clearly elucidated, cellular oxidative damage and apoptotic changes are thought to underlie their development. 12 Furthermore, most of the high energy-requiring organs and those tissues with high energy turnover such as the skeletal muscles and liver have been observed to have a large population of mitochondria which is often the target of NRTI action intracellularly. 14,15 Apart from its other intracellular side effects, NRTIs cause a reduction in mitochondrial population and protein content in addition to increasing apoptotic changes and reducing the skeletal muscle size (a sign that has been taken as one of the indicators of myopathy in patients treated with NRTIs). 14 –16
So far, no standard management guidelines have been established in the management of these complications of NRTIs. Measures, such as symptomatic management of complications and withdrawal or substitution of the offending NRTI, have been employed in addressing these complications with minimal level of success. 17,18 Furthermore, results of supplements such as carnitine and vitamins have not been convincing, thus necessitating further research into more promising treatment options. 15,19,20
The role of antioxidants in modifying the progression of several disease conditions associated with free radical generation is the subject of several ongoing research. Naringin (4′,5,7-trihydroxy flavonone-7-rhamnoglucoside) is a plant-derived flavonoid found in grapefruit and most other citrus fruits. 21,22 Its antioxidant, antiapoptotic, antimutagenic, anti-inflammatory, free radical quenching, and antihyperglycemic effects have been previously elucidated. 22 Although there is paucity of information on the effects of naringin on myopathy, citrus-derived polyphenols, which consists of flavonoids among other constituents, have been shown to prevent skeletal muscle catabolism and probable skeletal muscle mass loss. 23 Furthermore, Dhanya et al. showed the ameliorative effects on oxidative stress and glucose uptake in skeletal muscle cells that were pretreated with naringin. 24 Since oxidative stress and apoptotic changes have been suggested as mechanisms underlying the development of these metabolic complications in the skeletal muscles of NRTI-treated individuals, we therefore sought to investigate the potentials of naringin in mitigating these side effects.
Materials and methods
Animals
Male Wistar rats (Rattus norvegicus) weighing between 200 and 250 g were purchased from and maintained within the facilities of the Biomedical Resources Unit of the University of KwaZulu-Natal, South Africa. They were exposed to a 12-h light/12-h dark cycle, humidity of 55 ± 5%, temperature of 23 ± 2°C, and allowed free access to water and fed with standard rat chow ad libitum. Study protocols were approved by the Animal Research Ethics Committee of UKZN (ethics reference number: 008/14/animal), and animals were treated with humane care according to the National Institutes of Health revised principles for laboratory animal care. 25
Drugs and chemicals
All chemicals were of analytical grade and sourced from Sigma-Aldrich chemicals (St. Louis, Montana, USA), while antibodies were purchased from Santa Cruz Biotechnology Inc. (Dallas, Texas, USA). AZT and d4T were procured from Aspen Pharmacare® (South Africa), while vitamin E was purchased from Pharma-Natura (Pty) Ltd (South Africa).
Determination of sample size
The number of rats included in each treatment group was estimated using the formula 26 :
One rat was added in each group to cater for mortalities that might arise from oral dosing and drug exposure.
Exposure to drugs and treatment protocol
Forty-nine male Wistar rats were randomly divided into seven groups (n = 7) and daily treated with either 50 mg kg−1 body weight(BW) of d4T (groups A, B, and C) or 100 mg kg−1 BW of AZT (groups D, E, and F), respectively, by oral gavage (Table 1). 27 –30 Additionally, rats in groups B and E were similarly treated with 50 mg kg−1 BW of naringin, while rats in groups C and F had 100 mg kg−1 BW of vitamin E coadministered, respectively. Rats in group G served as the controls and were similarly treated with 3.0 ml kg−1 BW of distilled water. 27,30
Animal treatment protocol over 56 days.

d4T: stavudine; AZT: zidovudine; BW: body weight.
Glucose tolerance tests
Glucose tolerance tests (GTTs) were carried out on the 54th day of drug treatment following an overnight fast. Blood samples for determination of fasting plasma insulin (FPI) were taken, while baseline fasting blood glucose (FBG) concentrations were measured with a glucometer (in vitro diagnosticum; OneTouch Select®; Lifescan Inc., Milpitas, California, USA) via tail prick and rats were thereafter treated orally with 3.0 mg kg−1 of glucose in distilled water. Blood glucose concentrations were measured at 15, 30, 60, 90, and 120 min, respectively, post-glucose load, while blood–glucose–time plots were graphically constructed and area under the curve (AUC) calculated and expressed as 30 :

Animal sacrifice
Following 56 days of drug administration, rats were killed by halothane overdose (5 vol% in oxygen) after an overnight fast and blood withdrawn by cardiac puncture, centrifuged at 3000 r min−1 for 10 min, and separated plasma samples stored at −80°C for further biochemical analysis. 31 The gastrocnemius muscle from each rat’s left hind limb was promptly surgically excised; rinsed in phosphate-buffered saline solution, blotted dry, weighed, snap frozen in liquid nitrogen; and stored at −80°C for further biochemical analysis.
Biochemical analysis
Determination of FPI concentrations
FPI concentrations were analyzed using the ultrasensitive rat insulin enzyme-linked immunoassay kit (DRG Instruments GmbH, Marburg, Germany) as per the manufacturer’s instructions.
Evaluation of insulin resistance
Insulin resistance was determined using the homeostasis model assessment of insulin resistance (HOMA-IR) formula:

where FPI is fasting plasma insulin (μ IU ml−1) and FBG is fasting blood glucose (mmol l−1). 32,33
Determination of antioxidant enzyme activities
Activities of skeletal muscles glutathione peroxidase (GPx) and manganese superoxide dismutase (MnSOD) enzymes were determined using commercially available kits from Cayman chemicals (Ann Arbor, Michigan, USA) following the manufacturer’s instructions.
Determination of skeletal muscle malondialdehyde concentrations
Skeletal muscle malondialdehyde (MDA) concentrations were determined using the modified method of Halliwell and Chirico. 34 Briefly, 200 µl of supernatant from homogenization of 100 mg of skeletal muscle were sequentially added to 500 µl of 2% of phosphoric acid (H3PO4), 400 μl of 7% H3PO4, and 400 µl of butylated hydroxytoluene/thiobarbituric acid (BHT/TBA) solutions in a set of clean glass test tubes, respectively. In a counterpart set of eight clean fresh test tubes, 200 µl each of serial dilutions of MDA standard were added to 500 µl of 2% H3PO4 400 μl of 7% H3PO4 and 400 µl of BHT/TBA solutions, respectively. Reaction was initiated in each tube by adding 200 μl of 1 M hydrochloric acid solution, and the contents of each tube were then boiled for 15 min at 100°C with constant agitation. Tubes were allowed to cool down to room temperature, then contents thoroughly mixed with 1.5 ml of n-butanol, followed by aliquoting 200 μl of the top phase into a clean 96-well microtiter plate in triplicates. Absorbance was read at 532 nm on a Bio-tek Synergy™ HT Multi-Detection Microplate Reader (MTX Lab Systems Inc., Vienna, Virginia, USA), while an extinction coefficient of 1.56 × 105 M−1 cm− 1 was utilized in calculating the MDA concentration in each sample.
Determination of skeletal muscle protein carbonyl concentrations
Skeletal muscle protein carbonyl concentrations were determined using commercially available kits from Cayman chemicals (Ann Arbor, Michigan, USA) and reactions carried out as per the manufacturer’s instructions.
Detection of apoptotic protein expression
Expressions of BCL-2-associated X (Bax) and B-cell lymphoma-2 (BCl-2) proteins were detected by the Western blot method. Briefly, skeletal muscle were homogenized in a 1:9 solution of ice-cold radioimmunoprecipitation assay buffer containing 1.0% triton X-100, 1.0% PI cocktail, 0.5% sodium deoxycholate, 150 mM sodium chloride, 0.1% sodium dodecyl sulfate, 1% Nonidet P-40, and 50 mM Tris (pH 8.0) and spun in an ice-cold centrifuge for 20 min at 12,000 r min−1. Protein estimation was performed using Bradford assay protocol and samples thereafter adjusted for equal loading. 35 Denatured protein samples (35 µg each) were loaded in each well of a 10% polyacrylamide gel and resolved at room temperature by electrophoresis at 150 mV for 1.5 h. Separated proteins were transferred onto nitrocellulose membranes at 100 mV for 1 h and membrane incubated in blocking solution containing 5% bovine serum albumin in Tris-buffered saline (TBS) for 1 h at room temperature. Membranes were probed overnight at 4°C with a 1:100 dilution of either anti-BCL or anti-Bax primary antibodies, respectively. Membranes were then subjected to 5× vigorous washes in TBS solution containing 1% Tween-20 (TBS-T) and thereafter probed with a 1:1000 dilution of appropriate horseradish peroxidase-conjugated secondary antibodies. Membranes were subsequently washed five times in TBS-T, developed using the Lumiglo chemiluminiscent reagent (Cell Signaling Technology Inc., Danvers, Massachusetts, USA) and viewed with Biorad’s ChemiDoc imager (Bio-Rad Laboratories Inc., Hercules, California, USA). Densitometry was carried out using the ImageLab® software (Bio-Rad Laboratories Inc., Hercules, California, USA).
Statistical analysis
All data obtained are presented as mean ± standard error of the mean. Statistical difference between groups was determined with the Student’s t-test using the Graph Pad Prism® version 5.0 statistical package. Values of p less than 0.05 were taken as the statistical level of significance.
Results
Effects of naringin on BW, skeletal muscle index, and glycemic parameters
Treatment with AZT- or d4T-only produced significantly (p < 0.05) reduced total BW and skeletal muscle index (SKI; an index of myopathy, calculated as the ratio of the gastrocnemius muscle weight relative to the total BW) compared to controls, while naringin significantly (p < 0.05) improved BW and SKI in AZT- or d4T-treated rats, respectively (Table 2). Vitamin E, on the other hand, significantly (p < 0.05) improved SKI and BW among d4T-treated rats but not AZT-treated rats, respectively.
Change in BW and SKI.a

AZT: zidovudine; NAR: naringin; VITE: vitamin E; d4T: stavudine; BW: body weight; SKI: skeletal muscle index.
aValues are expressed as mean ± SEM. Change in BW is calculated as total BW on day 56 − total BW on day 0 of NRTI treatment, and SKI is calculated as the ratio between weights of gastrocnemius muscle/total BW on day 56 of NRTI treatment × 100.
b p < 0.05 compared to controls.
c p < 0.05 compared to AZT.
d p < 0.05 compared to d4T.
e p < 0.001 compared to controls.
f p < 0.01 compared to AZT.
g p < 0.01 compared to d4T.
Furthermore, there were significant (p < 0.05) increases in FBG concentrations and AUC units of GTT in AZT- or d4T-only–treated rats compared to controls suggesting glucose intolerance following treatment with NRTIs only (Figures 1(a) and (b) and 2(a) and (b)). Naringin, however, significantly (p < 0.05) decreased AUC units of GTT but not FBG concentrations in AZT- or d4T-treated rats, while vitamin E significantly (p < 0.05) reduced FBG concentrations in AZT- or d4T-treated rats and AUC units of GTT in AZT-treated rats but not d4T-treated rats, respectively.
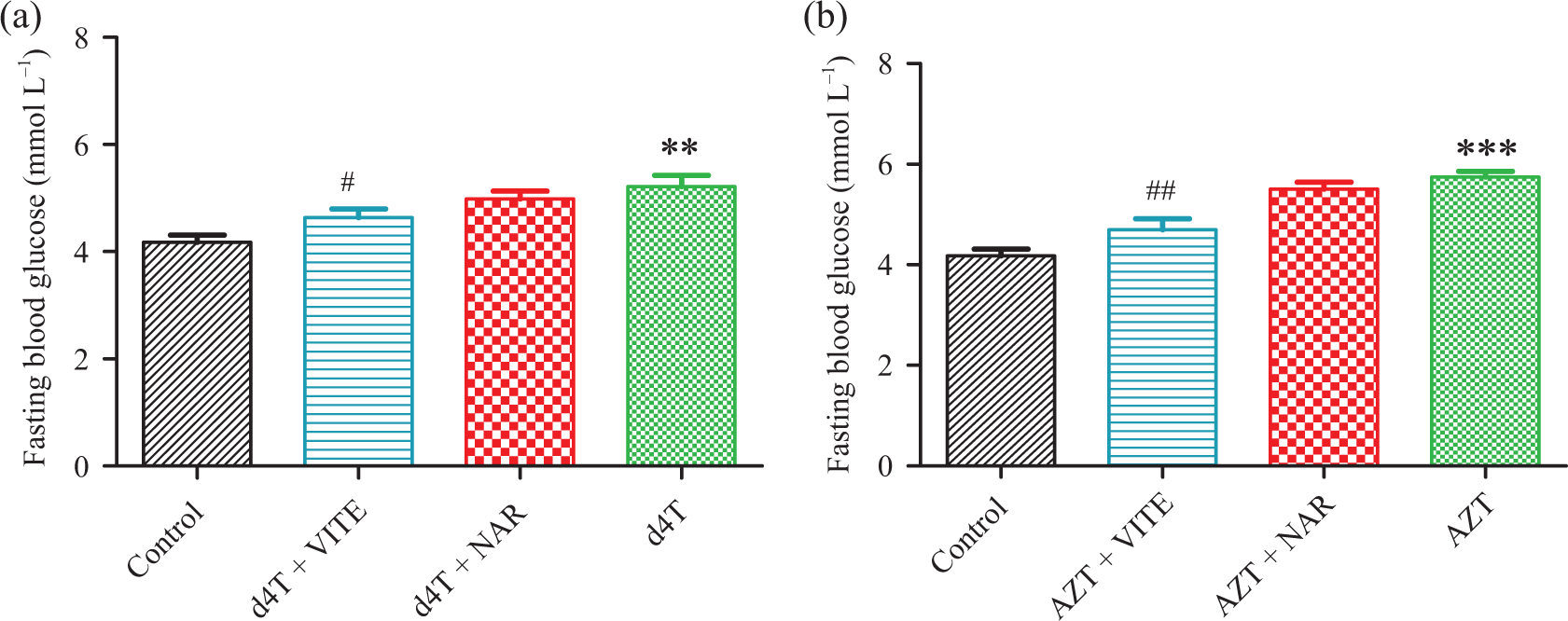
FBG concentrations in d4T- or AZT-treated rats, respectively. (a) d4T-treated rats (**p < 0.01: compared to controls; #p < 0.05: compared to d4T-only) and (b) AZT-treated rats (***p < 0.001: compared to controls; ##p < 0.01: compared to AZT-only). FBG: fasting blood glucose; d4T: stavudine; AZT: zidovudine.

AUC units of GTT in d4T- or AZT-treated rats, respectively. (a) d4T-treated rats (***p < 0.001: compared to controls; # p < 0.05, ### p < 0.001: compared to d4T-only) and (b) AZT-treated rats (*p < 0.05: compared to controls; # p < 0.05: compared to AZT-only). d4T: stavudine; AZT: zidovudine; AUC: area under the curve; GTT: glucose tolerance tests.
Effects of naringin on insulin resistance
Markers of insulin resistance (FPI concentrations and HOMA-IR values) were significantly (p < 0.01) elevated by AZT- or d4T-only treatment compared to controls (Figures 3(a) and (b) and 4(a) and (b)). Naringin or vitamin E, however, significantly (p < 0.05) reduced FPI concentrations in both AZT- or d4T-treated rats (Figure 3(a) and (b)), while naringin, but not vitamin E, caused significantly (p < 0.001) reduced HOMA-IR values in the AZT-treated rats only (Figure 4(b)).

FPI concentrations in d4T- or AZT-treated rats, respectively. (a) d4T-treated rats (***p < 0.001: compared to controls; # p < 0.05: compared to d4T-only) and (b) AZT-treated rats (***p < 0.001: compared to controls; ### p < 0.001: compared to AZT-only). d4T: stavudine; AZT: zidovudine; FPI: fasting plasma insulin.
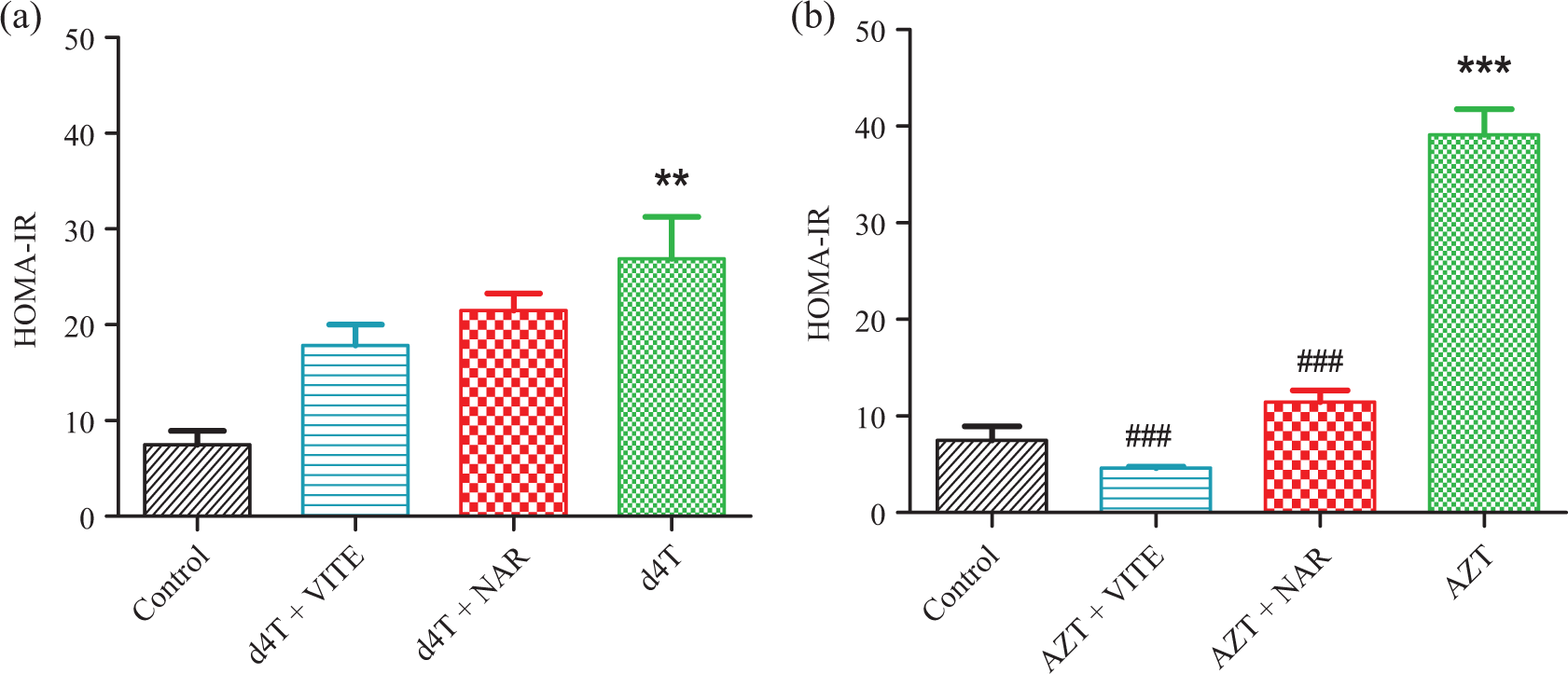
HOMA-IR index of insulin resistance in d4T- or AZT-treated rats, respectively. (a) d4T-treated rats (**p < 0.01: compared to controls) and (b) AZT-treated rats (***p < 0.001: compared to controls; ### p < 0.001: compared to AZT-only). d4T: stavudine; AZT: zidovudine; HOMA-IR: homeostasis model assessment of insulin resistance.
Effects of naringin on oxidative stress
Administration of AZT- and d4T-only caused significantly (p < 0.05) increased MDA (Figure 5(a) and (b)) and protein carbonyls concentrations MDA (Figure 6(a) and (b)) coupled with decreased antioxidant enzyme (GPx and MnSOD) activities (Figures 7 and 8(a) and (b)) compared to controls. Coadministration of either naringin or vitamin E with d4T or AZT, however, significantly (p < 0.05) reduced MDA and protein carbonyls concentrations in addition to significantly (p < 0.05) elevating GPx and MnSOD activities compared with d4T- or AZT-only–treated rats, respectively.

MDA concentrations in the skeletal muscles of d4T or AZT-treated rats, respectively. (a) d4T-treated rats (***p < 0.001: compared to controls; # p < 0.05, ### p < 0.001: compared to d4T-only) and (b) AZT-treated rats (*p < 0.05: compared to controls; ## p < 0.01: compared to AZT-only). d4T: stavudine; AZT: zidovudine; MDA: malondialdehyde.

Protein carbonyls concentrations in the skeletal muscles of d4T- or AZT-treated rats, respectively. (a) d4T-treated rats (***p < 0.001: compared to controls; ## p < 0.01, ### p < 0.001: compared to d4T-only) and (b) AZT-treated rats (***p < 0.001: compared to controls; ### p < 0.001: compared to AZT-only). d4T: stavudine; AZT: zidovudine.

GPx enzyme activity in the skeletal muscles of rats, respectively. (a) d4T-treated rats (***p < 0.001: compared to controls; # p < 0.05: compared to d4T-only) and (b) AZT-treated rats (***p < 0.001: compared to controls; # p < 0.05, ### p < 0.001: compared to AZT-only). d4T: stavudine; AZT: zidovudine; GPx: glutathione peroxidase.
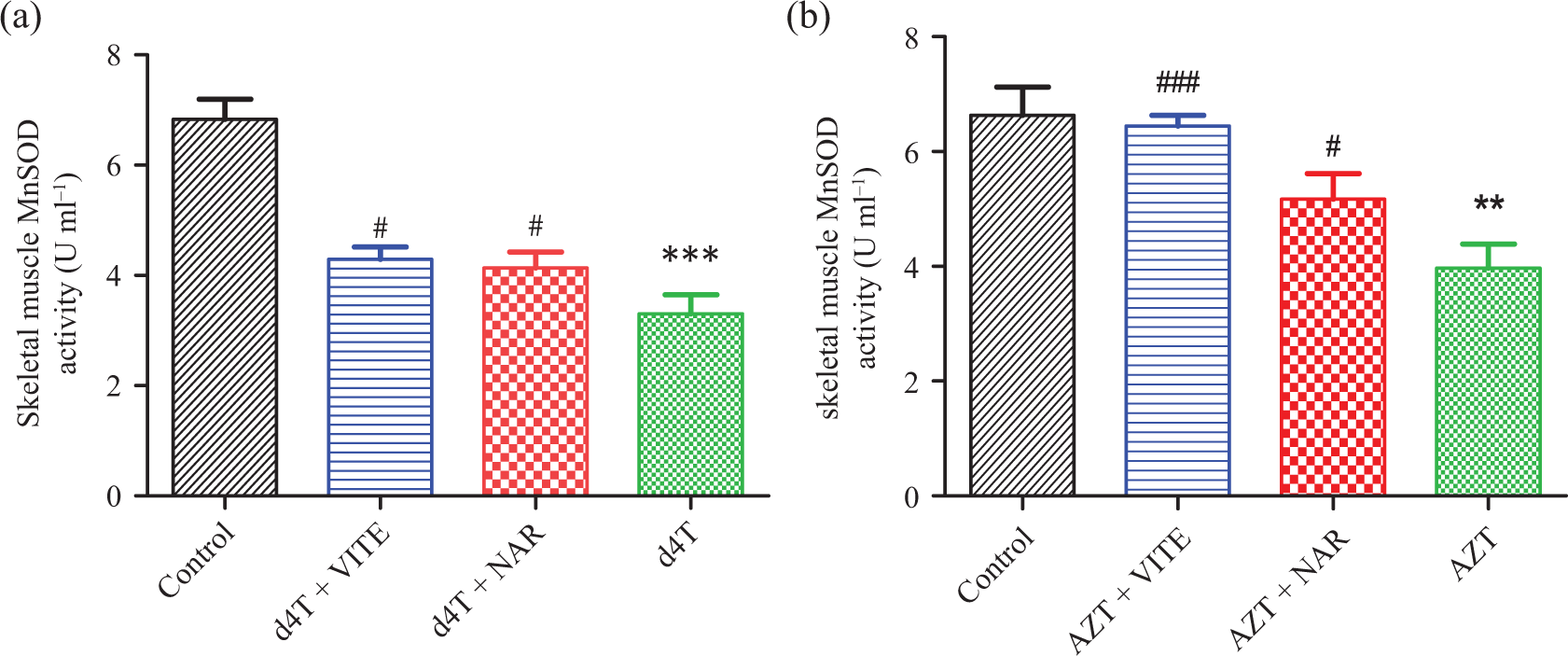
MnSOD enzyme activity in the skeletal muscles of rats, respectively. (a) d4T-treated rats (***p < 0.001: compared to controls; # p < 0.05: compared to d4T-only) and (b) AZT-treated rats (**p < 0.01: compared to controls; # p < 0.05, ### p < 0.001: compared to AZT-only). d4T: stavudine; AZT: zidovudine; MnSOD: manganese superoxide dismutase enzyme.
Effects of naringin on apoptotic changes
AZT- or d4T-only treatment caused significantly (p < 0.001) increased expression of proapoptotic protein (Bax) while significantly (p < 0.001) reducing the expression of antiapoptotic protein BCl-2, respectively, compared to the controls (Figure 9(a) to (f)). Naringin or vitamin E, on the other hand, significantly decreased Bax protein expression and increased BCl-2 protein expression compared to AZT- or d4T-only–treated rats, respectively. Furthermore, there was significant (p < 0.05) elevation of the Bax:BCl-2 ratios in d4T- or AZT-only–treated rats compared to controls (Figure 9(g) and (h)). Administering either naringin or vitamin E with each of the NRTIs, however, reduced this ratio compared to d4T- or AZT-only–treated rats.

Expression of apoptotic proteins (BCL-2 and Bax): (a) and (b) show representative gels of BCl-2 and Bax proteins expression; (c) and (d) show densitometry scans of BCl-2 protein expression normalized to beta actin expression; (e) and (f) show densitometry scans of Bax protein expression normalized to beta actin expression, while (g) and (h) show the ratio of Bax to BCl-2 proteins. (c), (e), and (g) show d4T-treated rats (***p < 0.001: compared to controls; # p < 0.05, ## p < 0.01, ### p < 0.001: compared to d4T) and (d), (f), and (h) AZT-treated rats (***p < 0.001: compared to controls; ### p < 0.001: compared to AZT), respectively. d4T: stavudine; AZT: zidovudine; BCL-2: B-cell lymphoma-2; Bax: BCL-2 associated X.
Discussion
The use of HAART in the management of HIV has revolutionized outcomes of the infection, however, with the advent of the combination therapy came a concomitant increase in associated metabolic complications including, but not limited to, diabetes mellitus, impaired glucose tolerance, insulin resistance, myopathy, and lipodystrophy among others. 14 Most of these complications are associated with the use of NRTIs, which are the major component of most HAART regimen. NRTIs have been suggested to cause these metabolic complications through their inhibitory effects on polymerase-γ (Pol-γ), the only mitochondrial enzyme that regulates mitochondrial DNA (mtDNA) replication and repair, leading to mtDNA depletion and mutation. 14,36 Apart from being class specific, these metabolic complications have been shown to be organ specific, with each NRTI demonstrating predilection for specific target organs. 37,38 While AZT is known to cause myopathy and bone marrow suppression, d4T is associated with severe lipodystrophy which predisposes to insulin resistance and impaired glucose tolerance which are risk factors for future development of diabetes mellitus and cardiovascular complications. 2,38 –40 In this study, AZT- or d4T-only–treated rats developed glucose intolerance and insulin resistance evidenced by significantly elevated FBG concentrations (Figure 1(a) and (b) and AUC units of GTT (Figure 2(a) and (b)) in addition to significant increases in FPI (Figure 3(a) and (b)) and calculated HOMA-IR values (Figure 4(a) and (b)), respectively, compared to controls. This confirms the development of a rat model of NRTI-induced metabolic complications. Furthermore, there was significant reduction in the skeletal muscle mass evidenced by reduced calculated SKI values (Table 2). Indeed, a reduction in the skeletal muscle mass has previously been taken as a marker of myopathy, thus suggesting the development of possible myopathy in our rat model. 41
Of the various suggested underlying mechanisms put forward, oxidative stress or cellular oxidative damage has been identified to be consistently associated with the development of these complications. 2 Inhibitory effect of NRTIs on Pol-γ, resulting in mtDNA depletion and/or mutation, causes reduced mitochondrial protein content, of which the mitochondrial electron transport chain (ETC) proteins are of particular interest. 2,14 NRTIs have been shown to inhibit complexes I and IV of the mitochondrial ETC, with a resultant increase in reactive oxygen species (ROS) production and reduction in adenosine triphosphate (ATP) generation. 2,42 Increased oxidative damage to intracellular lipids, proteins, and nucleic acids with the production of MDA, carbonyl proteins, and nucleic acid adducts, respectively, occurs consequent upon an increase in mitochondrial ROS production. 19 An estimation of MDA, carbonyl proteins, and nucleic acid adducts concentrations has taken as an indirect measure of the severity of ROS production within a particular system, while a direct measurement of free radicals produced within a system which can be affected by various intracellular and extracellular factors can also be undertaken. 19 In this study, AZT- or d4T-only–treated rats showed severe oxidative stress evidenced by increased concentrations of skeletal muscle MDA (Figure 5(a) and (b)) and carbonyl proteins (Figure 6(a) and (b)), respectively, thus suggesting an increase in oxidative stress in the skeletal muscles of our rat model. Antioxidant enzyme proteins such as superoxide dismutase and GPx are important factors in regulating intracellular ROS content as they rapidly convert free radicals into less toxic molecules intracellularly. 43 Increased oxidative stress accompanying the use of NRTIs has been associated with a marked reduction in the activities of these antioxidant enzymes as well as a reduction in the expression of the antioxidant genes with a concomitant increase in uncounterracted ROS activity intracellularly. 14
Attempts at combating these side effects of NRTIs have led to the trial of various agents including mitochondrial supplements, such as coenzyme-Q, carnitine, and antioxidants, such as vitamin E and C, with controversial/inconclusive results, 44 thus necessitating further research of potential agents that will prove beneficial in ameliorating or preventing these complications associated with NRTI administration. The beneficial effects of dietary sources of antioxidants in ameliorating or preventing oxidative stress-related conditions are the subject of most research efforts in this dispensation. Naringin is a plant-derived flavonoid mostly found in citrus fruits, rice, oregano, and tomatoes and has been attributed as the reason for the bitter taste of grapefruit. 22,45 Its antioxidant, antiapoptotic, free radical quenching, and antihyperglycemic effects have been previously elucidated. 22,30,46,47 In this study, rats treated with either naringin or vitamin E (which served a positive control for naringin’ effects) demonstrated markedly reduced markers of oxidative stress as there was significantly reduced skeletal muscle MDA (Figure 5(a) and (b)) and carbonyl protein concentrations (Figure 6(a) and (b)) coupled with an elevation of MnSOD and GPx activities (Figures 7(a) and (b) and 8(a) and (b)), respectively. Naringin has previously been shown to increase antioxidant gene expression and improve the mitochondrial, ETC complex IV protein expression in the liver of NRTI-treated rats. 48 Although we currently did not investigate ETC protein expression or activities, the results herein obtained suggest that naringin might have mitigated oxidative stress in AZT or d4T treated by increasing the complex IV expression or by increasing the antioxidant gene expression, thereby facilitating the mopping up of free radicals produced consequent upon NRTI use.
Furthermore, there were significantly decreased BCl-2 protein (Figure 9(c) and (d)) and increased Bax protein (Figure 9(e) and (f)) expression, coupled with increased Bax/BCl-2 ratios (Figure 9(g) and (h)), respectively in the skeletal muscles of AZT- or d4T-only–treated rats. The increase of these known indices of apoptosis suggests occurrence of apoptosis in the skeletal muscles in our model. NRTIs are known to cause apoptosis due to cellular energy starvation from reduced ATP production and this underlies the development of most complications of NRTIs, including myopathy. 49 Naringin’s antiapoptotic effects have been demonstrated and in the present study, it significantly reversed these apoptotic indices similarly to vitamin E in NRTI-treated rats. 22,50
Although we have demonstrated preliminary beneficial effects of naringin in a setting of NRTI-induced oxidative stress, deeper studies investigating the effects of naringin on other apoptotic pathways as well as mitochondrial function as it relates to regulation of oxidative stress were not demonstrated. As the mitochondria appears to be a major target of NRTI activities, future studies should therefore focus on factors such as mitochondrial structure and functions as well as other apoptotic pathways in a bid to better understand the effects of naringin on NRTI-induced mitochondrial dysfunction and metabolic complications.
In the present study, naringin reduced AZT- and d4T-induced glucose intolerance, insulin resistance in addition to mitigating oxidative damage, reduction in antioxidant enzyme activities and alterations in apoptotic markers in rats’ skeletal muscles. At a period where a solution to these complications of HAART is urgently required, naringin could be a potentially successful candidate nutritional supplement in the treatment of complications arising from HAART in HIV-positive individuals.
Footnotes
Acknowledgments
We are grateful to the staff of the Biomedical Resources Unit, University of KwaZulu-Natal, South Africa, for their technical assistance. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contributions
OOA conceptualized the research idea, carried out the animal handling as well as laboratory investigations involved in this work. OOA also analyzed the data obtained and provided the framework for the manuscript. OAA contributed to the research idea, planning, and execution of the research and also contributed to the writing of the manuscript, while PMOO provided supervision and contributed to the research design.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

