Abstract
Dioscorea bulbifera L., a commonly used medicinal plant in China, is reported to induce hepatotoxicity. The present study is undertaken to investigate the hepatotoxicity induced by diosbulbin B (DB), a diterpene lactone isolated from D. bulbifera L., and to further explore its underlying mechanism. DB was administered to mice at the doses of 0, 16, 32, and 64 mg/kg once daily for 12 consecutive days. Liver injury induced by DB was evidenced by the increased activity of serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP). Liver histological evaluation showed that the mice treated with DB exhibited liver damage with the swelling of hepatocytes. Further results showed that the amount of malondialdehyde (MDA) in the liver was increased in mice treated with DB, while the glutathione amount and the enzymatic activity of glutathione peroxidase (GPx), glutathione-S-transferase (GST), copper/zinc–superoxide dismutase (CuZn-SOD), manganese-SOD (Mn-SOD), and catalase (CAT) were all decreased. DB also decreased the gene expression of CuZn-SOD and CAT. Taken together, our results indicate that oral administration of DB for 12 consecutive days can lead to the oxidative stress liver injury in mice.
Introduction
Dioscorea bulbifera L. is a medicinal plant widely distributed in Asia; and in China, it is generally used in folk medicine to treat thyroid disease (especially goiter) and cancer. 1 However, prolonged chronic exposure to D. bulbifera L. can cause toxic hepatitis during the clinical practice. 2 Our previous studies have shown that ethyl acetate extract isolated from D. bulbifera L. induced liver injury in mice. 3,4 There are already reports demonstrating that diterpenoids are the main chemical components in D. bulbifera L. 5,6 Diosbulbin D, a diterpenoid, can induce cytotoxicity in human normal liver L-02 cells, which suggests that diosbulbin D may be the potential hepatotoxic compound in D. bulbifera L. 7 Diosbulbin B (DB) is another diterpenoid, and our previous study has showed that it was the hepatotoxic compound and highly contained in D. bulbifera L. 3 However, the mechanism of DB-induced liver injury in mice needs further investigation.
Oxidative stress is considered to be involved in the process of many diseases including liver injury induced by carbon tetrachloride, alcohol, acetaminophen, chemotherapeutic agents, and so on. 8 –11 Reactive oxygen species (ROS) produced during the process of liver injury can directly bind to cellular macromolecules like DNA, RNA, or protein and thus lead to cell damage. 12 Cellular critical antioxidant enzymes like superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), glutathione-S-transferase (GST) and low-molecular-weight antioxidants such as glutathione and some plasma protein, play important roles in protecting against ROS-induced cell injury. 13 Our previous studies have already showed that ethyl acetate extract isolated from D. bulbifera L. induced oxidative stress liver injury in mice. 3,4
The present study is designed to observe the hepatotoxicity induced by DB after treating mice for 12 consecutive days and to further explore whether ROS is involved in DB-induced liver injury.
Materials and methods
Experimental animals
Specific pathogen-free male ICR (18–22 g) mice were obtained from the Chinese Academy of Sciences. The mice were fed with a standard laboratory diet and given free access to tap water, kept in a controlled room temperature (22°C ± 1°C), relative humidity (65% ± 10%), and a 12:12-h light–dark cycle. All animals received humane care in compliance with the institutional animal care guidelines approved by the Experimental Animal Ethical Committee of Shanghai University of Traditional Chinese Medicine.
Drugs and reagents
DB (Figure 1) was isolated from the Chinese medicinal herb D. bulbifera L. identified by nuclear magnetic resonance and mass spectrometry and its purity ≥90%. Both reduced glutathione (GSH) and nicotinamide adenine dinucleotide phosphate (NADPH) were purchased from Roche Diagnostics GmbH (Mannheim, German). RevertAid first strand complementary DNA (cDNA) synthesis kit was purchased from Fermentas International Inc. (Ontario, Canada). 2-Thiobarbituric acid (TBA), 5, 5’-dithio-bis (2-nitrobenzoic acid) (DTNB), and other reagents unless indicated were obtained from Sigma Chemical Co. (St. Louis, MO).

Chemical structure of diosbulbin B (DB).
Treatment protocol
Mice were administered intragastrically (i.g.) with DB suspended in 0.5% sodium carboxyl methyl cellulose (CMC-Na) (0, 16, 32, or 64 mg/kg/day) for 12 consecutive days. Peripheral blood samples of each group were collected at 24 h after the last administration of DB. Then, the mice were killed, and their livers were collected and washed with ice-cold saline. Small pieces of livers were separated for histological observation, and the remaining parts were quick-frozen in liquid nitrogen for the further experiments.
Serum biomarkers for liver injury
The blood samples obtained from mice of all groups were allowed to coagulate for 2 h on ice. Serum was then isolated after the centrifugation at 3000g for 15 min. Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were measured according to the reported methods. 14 Serum alkaline phosphatase (ALP) activity was determined using kit obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China).
Histological examination
The slices of the mice livers were fixed in 10% formalin and embedded in paraffin. Samples were subsequently sectioned (5 μM) and stained with hematoxylin–eosin (H&E) and examined by light microscopy (Olympus, Japan) to evaluate liver damage.
Assay of liver MDA amount
Tissues were homogenized in cold phosphate-buffered saline (PBS), and malondialdehyde (MDA) in the tissues was determined by the previous reported method. 15 MDA reacts with TBA to generate a pink-colored product that has an absorbance at 532 nm. The tissue level of MDA based on tissue protein concentration was calculated from the standard curve constructed by the MDA concentration in the range of 0–40 μM.
Assay of liver glutathione amount
The amount of glutathione was measured as described in the previous study. 16 The reaction mixture contained 1 mM EDTA, NADPH (0.24 mM), glutathione reductase (GR) (0.06 units), DTNB (86 μM), and samples. The formed product 5-thio-2-nitrobenzoic acid (TNB) was monitored at 412 nm. The glutathione amount in mouse liver was calculated based on tissue protein concentration.
Assay of GPx, GST, SOD, and CAT activities
Tissue was homogenized in cold PBS, centrifuged at 5,000g for 10 min, and the supernatant was transferred to new tubes to assay GPx, GST, SOD, and CAT activities.
GPx activity was assayed in terms of utilization of glutathione as reaction substrate according to the method reported previously. 17 The tissue activity of GPx was calculated based on tissue protein concentration.
GST activity was measured using 1-chloro-2,4-dinitrophenol (CDNB) as substrate according to the method reported previously. 18 The tissue activity of GST was calculated based on tissue protein concentration.
CuZn-SOD, Mn-SOD and CAT activities were determined according to the methods reported previously 19,20 and calculated based on tissue protein concentration.
Reverse transcription–polymerase chain reaction (RT-PCR) analysis
Total RNA was extracted from liver tissue using a TRIZOL (Life Technologies, USA) reagent according to the manufacturer’s protocol. The single-strand cDNA was synthesized according to the manufacturer’s protocol. Transcripts of the gene for glyceraldehydes-3-phosphate dehydrogenase (GAPDH) were used as an internal control. Sequences of the PCR primers were as follows: CuZn-SOD forward 5′-AAG GCC GTG TGC GTG CTG AA-3′, reverse 5′-CAG GTC TCC AAC ATG CCT CT-3′ (246 bp product). Mn-SOD forward 5′-GCA CAT TAA CGC GCA GAT CA-3′, reverse 5′-AGC CTC CAG CAA CTC TCC TT-3′ (241 bp product). CAT forward 5′-GCA GAT ACC TGT GAA CTG TC-3′, reverse 5′-GTA GAA TGT CCG CAC CTG AG-3′ (229 bp product). The PCR protocol for CuZn-SOD consisted of denaturation at 94°C for 1 min, 35 cycles of denaturation at 94°C for 45 s, annealing at 56°C for 1 min, extension at 72°C for 1 min, and final extension at 72°C for 5 min. The PCR protocol for Mn-SOD and CAT consisted of denaturation at 94°C for 1 min, 35 cycles of denaturation at 94°C for 45 s, annealing at 55°C for 1 min, extension at 72°C for 1 min, and final extension at 72°C for 5 min. The PCR products were electrophoresed in a 2% agarose gel and stained with ethidium bromide. The quantity of PCR products was automatically analyzed by Smart View Bio-electrophoresis Image Analysis system (Version FR-980; FURI Science and Technology Co. Ltd, Shanghai, China).
Statistical analysis
The results were expressed as Means ± standard error of mean (SEM). Differences between groups were assessed by one-way ANOVA with least significance difference (LSD) post hoc test. The analyses were carried out using the SPSS 16.0 software package for Windows. P ≤ 0.05 was considered as statistically significant.
Results
Effects of DB on serum ALT, AST, and ALP
As shown in Figure 2, values of ALT, AST, and ALP in the control group were 13.85 ± 1.82, 22.74 ± 2.16, 17.97 ± 0.35 U/L, respectively. Serum ALT, AST, and ALP activities had no much change after the administration of 16 mg/kg DB for 12 consecutive days. In mice treated with 32 mg/kg and 64 mg/kg DB, the values of ALT, AST, and ALP were all significantly increased as compared with control, that is, the ALT value is 52.05 ± 9.09 U/L (p < 0.01) and 85.43 ± 12.77 U/L (p < 0.001), the AST value is 33.23 ± 4.11 U/L (p < 0.05) and 50.85 ± 4.13 U/L (p < 0.001), and the ALP value is 24.18 ± 2.18 U/L (p < 0.05) and 28.47 ± 3.62 U/L (p < 0.01).
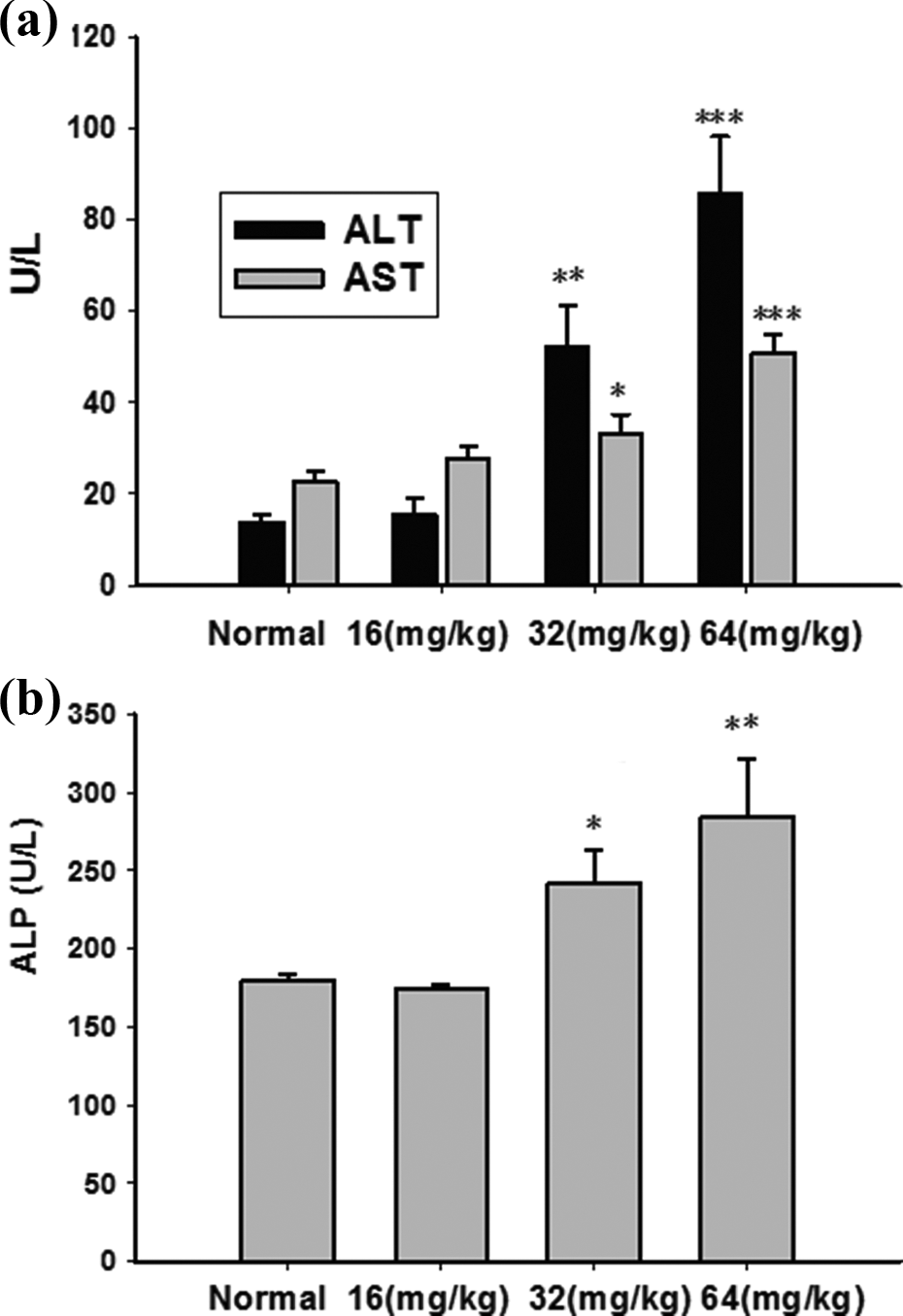
DB increased the activity of serum ALT, AST, and ALP. (a) The serum ALT and AST activity. (b) The serum ALP activity. Data were shown as means ± SEM (n = 10). The significance was estimated by using one-way ANOVA with LSD post hoc test, *p < 0.05, **p < 0.01, ***p < 0.001 in comparison with control group. DB: diosbulbin B; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; SEM: standard error of mean; LSD: least significance difference.
Histological analysis of liver tissue
The results of histological evaluation demonstrated that mice treated with DB (16 mg/kg) showed no difference as compared with control (Figure 3(a), (b)). After exposure to DB (32 mg/kg or 64 mg/kg), the livers almost lost normal architecture with extensive acute hemorrhage, swelling hepatocytes, and widespread necrosis (Figure 3(c), (d)).

Histological evaluation of liver of mice treated with DB for 12 consecutive days. Liver sections were stained with hematoxylin–eosin. (a) Normal group, (b) diosbulbin B (DB) (16 mg/kg) group, (c) DB (32 mg/kg) group, and (d) DB (64 mg/kg) group. (magnification 200×). Arrows indicate necrotic and swelling hepatocytes.
Effects of DB on liver MDA and glutathione amount
MDA is one of the main end products of lipid peroxidation (LPO) and generally served as an index of the LPO intensity. Figure 4A showed that as compared with control group (0.301 ± 0.051 μmol/mg protein), liver MDA amount was increased in mice treated with 64 mg/kg DB (0.436 ± 0.057 μmol/mg protein, p < 0.05) for 12 consecutive days. Glutathione is an important antioxidant that can protect against oxidative stress injury induced by ROS such as free radicals and peroxides. 21 In the present study, liver glutathione amount was decreased in mice treated with 64 mg/kg DB (26.47 ± 0.43 nmol/mg protein, p < 0.05) as compared with control (37.75 ± 3.30 nmol/mg protein; Figure 4(b)).

The amount of MDA and glutathione in livers treated with DB for 12 consecutive days. (a) MDA amount. (b) glutathione amount. Data were shown as means ± SEM (n = 8). The significance was estimated by using one-way ANOVA with LSD post hoc test, *p < 0.05 in comparison with control group. MDA: malondialdehyde; LSD: least significance difference; SEM: standard error of mean.
Effects of DB on the activity of GST and GPx
GST and GPx are intracellular glutathione-related enzymes, and they can cooperate with glutathione to prevent oxidative stress injury. As shown in Figure 5, liver GST activities in mice treated with 32 mg/kg DB (613.64 ± 16.55 U/mg protein; p < 0.01) and 64 mg/kg DB (593.42 ± 25.45 U/mg protein, p < 0.01) were both significantly decreased as compared with control (748.57 ± 25.03 U/mg protein). In addition, liver GPx activities in mice treated with 32 mg/kg DB (35.23 ± 1.26 U/mg protein, p < 0.01) and 64 mg/kg DB (33.67 ± 0.94 U/mg protein, p < 0.01) were both obviously decreased as compared with control (43.05 ± 1.57 U/mg protein).
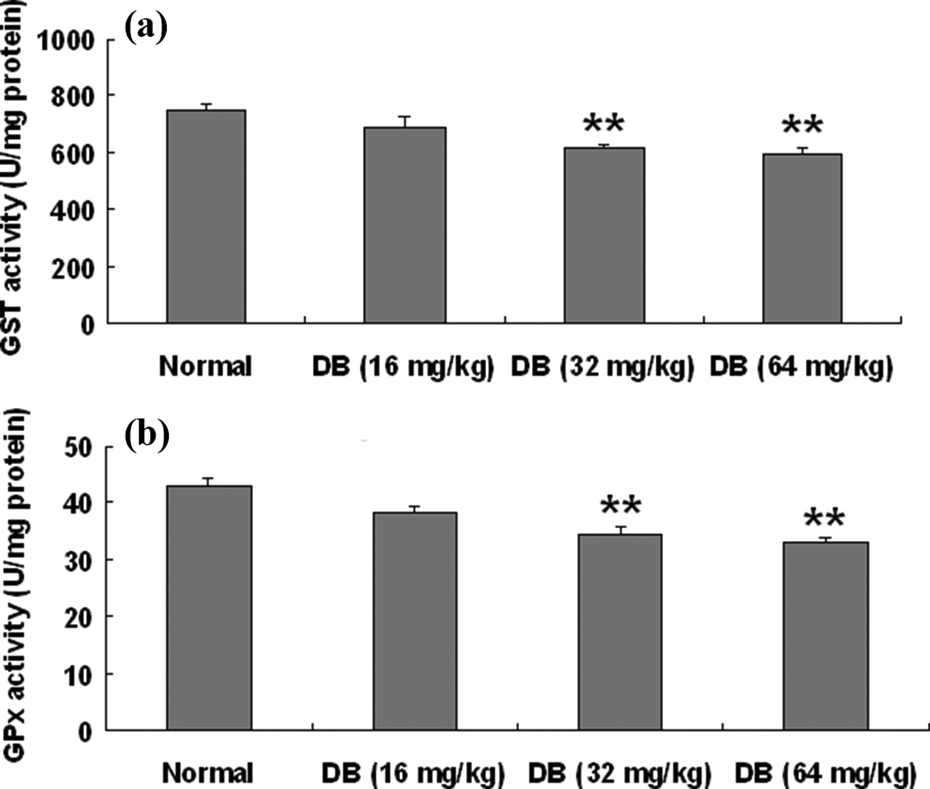
The activities of GST and GPx in liver of mice treated with DB for 12 consecutive days. (a) GST activity. (b) GPx activity. Data were shown as means ± SEM (n = 8). The significance was estimated by using one-way ANOVA with LSD post hoc test, **p < 0.01 in comparison with control group. GST: glutathione-S-transferase; GPx: glutathione peroxidase; LSD: least significance difference; SEM: standard error of mean.
Effects of DB on the activity and expression of SOD and CAT
SOD and CAT are both intracellular important antioxidant enzymes. The results in Figure 6 showed that CAT activity was decreased significantly in mice treated with 32 mg/kg DB (153.18 ± 10.53 U/mg protein, p < 0.01) and 64 mg/kg DB (144.31 ± 8.31 U/mg protein, p < 0.01) as compared with control (215.87 ± 9.21 U/mg protein). CuZn-SOD activity was also decreased significantly in mice treated with 32 mg/kg DB (27.99 ± 1.27 U/mg protein, p < 0.05) and 64 mg/kg DB (27.01 ± 0.86 U/mg protein, p < 0.01) as compared with control (33.77±1.70 U/mg protein), while Mn-SOD was weakly decreased in mice treated with 64 mg/kg DB (13.23±1.33 U/mg protein, p < 0.05) as compared with control (20.07 ± 2.70 U/mg protein). Further, we analyzed the gene expression of CuZn-SOD, Mn-SOD, and CAT. The results in Figure 7 showed that DB decreased the gene expression of CuZn-SOD and CAT but had no effect on the expression of Mn-SOD.

The activities of CuZn-SOD, Mn-SOD, and CAT in liver of mice treated with DB for 12 consecutive days. (a) CuZn-SOD and Mn-SOD activity. (b) CAT activity. Data were shown as mean ± SEM (n = 8). The significance was estimated using one-way ANOVA with LSD post hoc test, *p < 0.05 and **p < 0.01 in comparison with control group. Cu: copper; Zn: zinc; SOD: superoxide dismutase; Mn: manganese; CAT: catalase; LSD: least significance difference; SEM: standard error of mean.

The mRNA expression of CuZn-SOD, Mn-SOD, and CAT in liver of mice treated with DB for 12 consecutive days. (a) Liver mRNA expression of CuZn-SOD, Mn-SOD, CAT, and glyceraldehydes-3-phosphate dehydrogenase (GAPDH) was detected by RT-PCR. The result represents one of three separate experiments, and the level of GAPDH was used as loading control. (b) The amount of CuZn-SOD, Mn-SOD, and CAT expression was normalized to the internal control, GAPDH. Data were shown as means ± SEM. The significance was estimated using one-way ANOVA with LSD post hoc test, *p < 0.05, **p < 0.01 in comparison with control group. Cu: copper; Zn: zinc; SOD: superoxide dismutase; Mn: manganese; CAT: catalase; LSD: least significance difference; SEM: standard error of mean; RT-PCR: reverse transcriptase–polymerase chain reaction.
Discussion
D. bulbifera L. is used to treat thyroid disease and cancer in clinic in China and shows unique clinical benefit. However, the induced hepatotoxicity greatly affects its clinical application. Our previous study has already demonstrated that ethyl acetate extract isolated from D. bulbifera L. and its main compound DB both induced liver injury after treating mice for 24 h. 3 As D. bulbifera L. in clinic is generally a long-term medicine for cancer, so the present study observed the liver injury induced by DB for the consecutive administration of 12 days. In the present study, the results of serum ALT, AST, and ALP analyses suggest that consecutive administration of DB to mice for 12 days will lead to liver injury. Meanwhile, liver histological analysis further confirmed the hepatotoxicity induced by DB. In the previous study, we found that after giving DB (150 mg/kg) for 24 h will lead to liver injury, 3 but the toxic dose is much higher than the toxic dose (32 mg/kg) in the present study. Our results clearly demonstrate that the toxic dose of DB is much lower after being given consecutively for 12 days than given once for 24 h, which reminds us to pay attention to the potential hepatoxicity of D. bulbifera L when used for cancer therapy in clinic.
Oxidative stress is associated with the increased production of oxidizing species or the significant decrease in antioxidants, such as glutathione. 22 Liver MDA amount can reflect the potential injury induced by LPO, and it is generally increased in liver injury induced by some exogenous hepatotoxins like carbon tetrachloride (CCl4) and so on. 23,24 Glutathione is an antioxidant, which prevents damage to important cellular components caused by ROS such as free radicals and peroxides. 25,26 Our results showed that DB increased liver MDA amount and decreased liver glutathione amount. These results indicate that DB can destroy the liver redox balance through exhausting glutathione and thus induces liver oxidative stress injury.
The cytosolic GSTs are found in almost all aerobic species and catalyze the conjugation of glutathione to electrophilic centers on a wide variety of substrates. 27 GPx catalyzes the decomposition of hydrogen peroxide to hydroxides, specifically using glutathione as the electron provider. 28 GST and GPx are both glutathione-dependent antioxidant enzymes. In the present study, DB decreased liver GST and GPx activities, which suggest that DB will also lead to the damage on glutathione-dependent antioxidant enzymes. The results indicate the important role of liver glutathione antioxidant system in protecting against DB-induced liver injury.
During the oxidative stress, superoxide anions are produced. SOD can convert such superoxide anions to hydrogen peroxide or oxygen, while CAT or GPx can further detoxify hydrogen peroxide. 29 There exists several common forms of SOD depending on the metal cofactor that is Cu/Zn-SOD (binds both copper and zinc), Fe or Mn-SOD (binds either iron or manganese), and Ni-SOD (binds nickel), and of which Cu/Zn-SOD and Mn-SOD are the only forms found in yeast and mammals. 30 As the critical antioxidant enzymes, CAT and SOD activities are generally reported to be decreased in hepatotoxin-induced liver injury such as CCl4, ethanol, and so on. 31,32 Our results showed that DB decreased the activities of liver Cu/Zn-SOD, Mn-SOD, and CAT, which further showed the oxidative stress liver injury induced by DB. Furthermore, our results demonstrated that DB-induced decrease of Cu/Zn-SOD and CAT activities was due to the decreased expression of its gene. However, DB had no effect on Mn-SOD gene expression. Thus, whether DB-induced decrease of Mn-SOD activity was due to the change in manganese amount or other mechanism needs further investigation.
In conclusion, the present study shows that oral administration of low-dose DB for 12 consecutive days can induce liver oxidative stress injury in mice. All of these results remind us to pay attention to liver toxicity induced by D. bulbifera L. in clinic, especially when it is used for a long term.
Footnotes
Authors’ Note
The authors Yibo Ma and Chengwei Niu contributed equally to this article.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was financially supported by the Program for New Century Excellent Talents in University (NCET-11-1054), Shanghai Science and Technology Committee Grants (11DZ2272300), and State major science and technology special projects during the 12th five year plan (2012ZX09505001-002).
