Abstract
Acrylamide (Ac) in the foods and alcohol (Al) in the drinks are unavoidable. Several previous studies demonstrated that these substances which are taken into the body via diet may cause adverse effects in the cells. However, there is no study about how Ac and Al may affect the male reproductive system of the offspring when consumed by the mother during pregnancy and lactation. For this purpose, sexual development in male mice was evaluated after intake of 14 mg/kg Ac and 2 g/kg Al from gestation day 6 to postnatal day (PND) 21. The weight of the offspring was reduced at birth and PND 21 for those exposed to Ac and/or Al. The gonadosomatic index of male offsprings was reduced except for the Ac-treated lactation group. Both substances induced multinuclear giant cells, degenerative cells, atrophic tubules, and maturation-arrested tubules, while decreased Leydig, Sertoli, and spermatogenic cell numbers. Lipid peroxidation level and superoxide dismutase enzyme activity increased in both Al-treated and Ac and Al-treated groups. There was only reduction in the catalase activity during the gestation and lactation periods. These findings suggest that consumption of Ac together with Al may induce impairments on testicular spermatogenesis in male offsprings.
Introduction
Various environmental toxins have effects on the male reproductive system. Recent studies demonstrated that these toxins cause permanent damage to the male reproductive system. 1 The source of these toxins can be dietary or contaminated industrial products. 2 Pregnant women exposed to environmental toxic substances can pass the toxic substances to their offsprings via the placenta (prenatal period) or lactation (postnatal period). Moreover, these substances can cause toxic effects in the offspring. 3,4 Dietary acrylamide (Ac) and alcohol (Al) can pass from the mother to the offspring and exert toxic effects. 5 –8
Ac is a compound resulting from the reaction between carbohydrates and amino acids in nutrients treated at high temperatures (120°C). 9 Especially grain- and starch-containing nutrients have high levels of Ac. 10 Ac is commonly used in dye, paper, textiles, and the cosmetic industry as well as in water purification. 11 Increasing evidence demonstrated that Ac has genotoxic, neurotoxic, carcinogenic, and mutagenic effects and is known to have adverse effects on the male reproductive system. 12 It has been shown in many studies that Ac decreases the testicular weight in adults, and as a result of Ac uptake during gestation and lactation, it can pass to the offspring, increase birth anomalies, decrease organ weight, and cause morphological changes. 7,8,13 –22 Furthermore, Ac decreases sperm counts and causes atrophy in seminiferous tubules and deoxyribose nucleic acid (DNA) fragmentation in germ cells. 16 –18,23
The toxic effects of Al consumption on the male reproductive system were demonstrated by animal and human studies. 24 –27 Al is toxic to the testis and decreases sperm count and motility. 28 Chronic exposure to Al increases oxidative stress and decreases testosterone levels, which in turn leads to testicular atrophy, decreased seminiferous tubule diameter, and a decrease in the number of spermatogenic cells at all stages. 24,26,29 –32 Al consumption decreases reproductive activity by inhibiting spermatogenesis and causing apoptosis in germ cells. 32 –34
Consuming high levels of dietary Ac and Al as a result of our dietary habits, starting from the prenatal period, affects the male reproductive system at different developmental stages. The objectives of this study were to identify the damage exerted on the testes of male offsprings born from mothers exposed to Ac and Al during the gestation and lactation periods and determine the changes resulting from various combinations of Ac and Al. We evaluated the gonadosomatic index and testis histometry, examined the histopathological changes in the testis, and analyzed the level of lipid peroxidation in testicular tissue to identify the reproductive toxicity in male offsprings. Moreover, the activities of catalase (CAT) and superoxide dismutase (SOD), which are parts of the antioxidant system that function against the oxidative stress in the testis, were also analyzed.
Materials and methods
Chemicals
Ac (electrophoresis grade; purity >99%) was obtained from Sigma Chemical Co. (St. Louis, Missouri, USA). Al was purchased from Merck (Whitehouse Station, New Jersey, USA) with 99.9% purity.
Animal husbandry
In this work, we used 30 female Mus musculus Balb/c mice species obtained from the Institute of Experimental Medicine. The average weight of the used pregnant female mice was 25 g. The mice were fed with nutrition and water specific to their species and were kept in an environment at 20–25°C temperature, 50–60% relative humidity, and 12-h:12-h daily light period.
Experimental design
Pregnant mice were separated into 10 groups with 3 mice in each group. All dams were housed individually and body weights were measured daily. Mice were administered by oral gavage 14 mg/kg/day Ac which is the lowest dose that shows the toxic effects on offsprings during maternal exposure 12,18 and/or 2 g/kg/day Al, which is the daily intake dose, 35 from gestation day (GD) 6 to postnatal day (PND) 21 (Figure 1). Control group (C) was exposed to saline (0.9 sodium chloride) during gestation and/or lactation period. 18 Experimental groups that were treated with Ac (14 mg/kg/day) and Al (2 g/kg/day) during gestation and/or lactation are presented in Table 1. The offsprings were weighed at birth and PND 21, and each male offspring was selected at PND 21 for further evaluation. The testis tissues of the offsprings were taken at PND 21 and used for histological and biochemical experiments.

Study design. Time pregnant dams were dosed by oral gavage from GD 6 to PND 21. C: control; G-Ac: exposed to Ac during gestation; L-Ac: exposed to Ac during lactation; GL-Ac: exposed to Ac during gestation and lactation; G-Al: exposed to Al during gestation; L-Al: exposed to Al during lactation; GL-Al: exposed to Al during gestation and lactation; G-Ac + Al: exposed to Ac and Al during gestation; L-Ac + Al: exposed to Ac and Al during lactation; GL-Ac + Al: exposed to acrylamide and Al during gestation and lactation; GD 6: gestation day 6; PND 21: postnatal day 21; Ac: acrylamide; Al: alcohol.
Experimental groups.

Ac: acrylamide; Al: alcohol; GD 6: gestation day 6; PND 21: postnatal day 21; C: control; G-Ac: exposed to Ac during gestation; L-Ac: exposed to Ac during lactation; GL-Ac: exposed to Ac during gestation and lactation; G-Al: exposed to Al during gestation; L-Al: exposed to Al during lactation; GL-Al: exposed to Al during gestation and lactation; G-Ac + Al: exposed to Ac and Al during gestation; L-Ac + Al: exposed to Ac and Al during lactation; GL-Ac + Al: exposed to Ac and Al during gestation and lactation.
Gonadosomatic index
For every experimental group, the testis and the body weights of eight male offsprings were measured, and using the ratio of the testis weight to the body weight, the gonadosomatic index was determined. 36
Histopathology
Testes were fixed with Bouin’s solution for 12 h at 20°C and placed in 70% ethanol. Fixed testes were dehydrated in ethanol series, and cleared in xylene, and finally embedded in paraffin. Paraffin blocks were sectioned at 5 µm thickness, placed onto microscope slides, and stained with hematoxylin–eosin (H&E) for observation of the histological structure. 37
Histometric analysis
For the histometric investigations, the testis samples stained with H&E from eight male offsprings from every group were counted using a standard point counting method. Using 100× magnifications for every individual, 30 areas that contain 100 points were counted. 38 Spermatogonia, primary and secondary spermatocytes, round spermatids, Sertoli, Leydig, and degenerative cells were counted. The numerical density of Sertoli and Leydig cells was determined using Floderus equation. 39 The diameter of tubules and lumens and the height of the germinal epitelium in the sagital sections were determined using an objective micrometer and Image J 1.33u program (National Institutes of Health, USA).
Biochemical analysis
The male offspring testes taken for biochemical investigations were homogenized and sonicated with 1500 r/min within 1 M phosphate-buffered saline buffer. The tissue was centrifuged at +4°C and 10,000g for 20 min, and the supernatants were used in the experiments. Protein content was determined by the method described by Lowry et al. 40 The level of lipid peroxidation was measured by the method of Devasagayam and Tarachand. 41 The malondialdehyde (MDA) content of the sample is expressed as picomoles of MDA formed per milligram protein. The activity of CAT was assayed by the method followed by Sinha. 42 The activity of CAT is expressed as micromolar units of hydrogen peroxide (H2O2) per milligram protein. The activity of SOD was assayed according to the method described by Marklund and Marklund. 43 The enzyme activity is expressed as units per milligram protein.
Statistical analysis
Data are analyzed using analysis of variance.When the F ratio was significant, multiple comparisons were done by Tukey’s test using GraphPad Prism (san diego, California, USA) statistical program to determine the differences among various group means. The values of p < 0.05, p < 0.01, and p < 0.001 were considered statistically significant and were referenced to interpret the results.
Results
Effect on male offspring development
The body weight at birth, the body weight at PND 21, and gonadosomatic index ratios of offsprings in experimental groups that were treated with Ac (14 mg/kg/day) and Al (2 g/kg/day) during gestation and lactation are presented in Table 1. The body weight at birth of male offsprings was significantly decreased in groups treated with Ac and/or Al compared with the control group. The body weight values at PND 21 were decreased in groups GL-Ac, GL-Al, G-Ac + Al, and GL-Ac + Al, when compared with the control group. The gonadosomatic index was reduced significantly in all groups except for the L-Ac group (Table 2).
Avarege male offspring weight and gonadosomatic index (at birth and PND 21) from different groups.a

SEM: standard error of the mean; Ac: acrylamide; Al: alcohol; PND 21: postnatal day 21; C: control; G-Ac: exposed to Ac during gestation; L-Ac: exposed to Ac during lactation; GL-Ac: exposed to Ac during gestation and lactation; G-Al: exposed to Al during gestation; L-Al: exposed to Al during lactation; GL-Al: exposed to Al during gestation and lactation; G-Ac + Al: exposed to Ac and Al during gestation; L-Ac + Al: exposed to Ac and Al during lactation; GL-Ac + Al: exposed to Ac and Al during gestation and lactation.
aValues are given as means ± SEM.
bCompared with control.
c p < 0.05.
d p < 0.01.
e p < 0.001.
fCompared with Ac.
gCompared with Al.
Histopathology
Multinuclear giant cells in the lumen of the seminiferous tubules and structural changes in the connective tissue were observed in G-Ac, L-Ac, and L-Al groups, while deformed connective tissue and atrophic tubules were observed in G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups. In addition, spermatogenesis was mostly arrested at the level of primary spermatocytes in the seminiferous tubules. Similarly, atrophic tubules and structural deformations in testicular connective tissue were seen in all experimental groups during both gestation and lactation periods (Figure 2).

Histopathological changes in testes of offsprings at PND 21 exposed to Ac and Al maternally. (a, e, and i): 0 mg/kg Ac and 0 g/kg Al (untreated control). (b, f, and j): 14 mg/kg Ac. (c, g, and k): 2 g/kg Al. HE stain bars = 30 µM. DC: dejenetative cell, →: connective tissue deformation, *: multinuclear giant cell, ✥: atrophic tubule, •: maturation arrest tubule.
As demonstrated in Table 3, the number of degenerated cells in the testes was higher in all experimental groups compared with the control group. In addition, we observed an increase in the number of degenerated cells in L-Al, G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups, when compared with Ac-treated groups. Moreover, there was a significant increase in G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups, when compared with Al-treated groups. There was no significant difference in the number of atrophic tubules in the testes, when exposed to Ac during gestation and/or lactation periods and Al during only gestation and only lactation periods; however, there was an increase in the number of atrophic tubules in GL-Al, G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups. The number of maturation arrested tubules was higher in groups treated with both Ac and Al during gestation and/or lactation periods (G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups). In all experimental groups, the diameter of the seminiferous tubules and the height of the germinal epithelia were lower compared with the control group. We also observed a decrease in the lumen diameter in the seminiferous tubules only in G-Ac and L-Ac groups when compared with the control group and similarly a decrease in GL-Al groups when compared with Ac-treated group and G-Ac + Al when compared with Al-treated group. However, there was a significant increase in G-Al, L-Al, and L-Ac + Al groups when compared with Ac-treated group in the lumen diameter.
Data for histopathology and morphometry in the testis of male offsprings (at PND 21).a

SEM: standard error of the mean; Ac: acrylamide; Al: alcohol; PND 21: postnatal day 21; C: control; G-Ac: exposed to Ac during gestation; L-Ac: exposed to Ac during lactation; GL-Ac: exposed to Ac during gestation and lactation; G-Al: exposed to Al during gestation; L-Al: exposed to Al during lactation; GL-Al: exposed to Al during gestation and lactation; G-Ac + Al: exposed to Ac and Al during gestation; L-Ac + Al: exposed to Ac and Al during lactation; GL-Ac + Al: exposed to Ac and Al during gestation and lactation.
aValues are given as means ± SEM.
b p < 0.05.
cCompared with control.
d p < 0.001.
e p < 0.01.
fCompared with Ac.
gCompared with Al.
Morphometric analysis
Figure 3 illustrates a decrease in all spermatogenic cell types in all experimental groups compared with the controls. The number of spermatogonia and primary spermatocytes declined in all experimental groups, when compared with the controls. In addition, there was a significant decrease in the Al-treated groups and Ac and Al-treated groups during gestation and/or lactation periods, when compared with the Ac-treated groups (Figures 3(a) and (b)). There was a highly significant reduction in the number of secondary spermatocytes and round spermatids in all experimental groups, when compared with the controls (Figures 3(c) and (d)). Moreover, we observed a decrease in the number of secondary spermatocytes in G-Al, G-Ac + Al, and L-Ac + Al groups, when compared with Ac-treated groups (Figure 3(c)). The number of round spermatids was significantly declined only in groups G-Ac + Al, L-Ac + Al, GL-Al, and GL-Ac + Al, when compared with the Ac-treated groups. In addition, there was a significant decrease in the number of round spermatids in G-Ac + Al and L-Ac + Al groups when compared with Al-treated groups (Figure 3(d)). Figure 4 demostrates that the number of Sertoli and Leydig cells was lower in all groups when compared to the control except for the L-Ac group for Sertoli cell numbers. The number of Sertoli cells was significantly decreased in G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups when compared to the Ac-treated groups (Figure 4(a)). There was a decrease in Leydig cell numbers in L-Al and L-Ac + Al groups when compared to Ac-treated groups (Figure 4(b)).
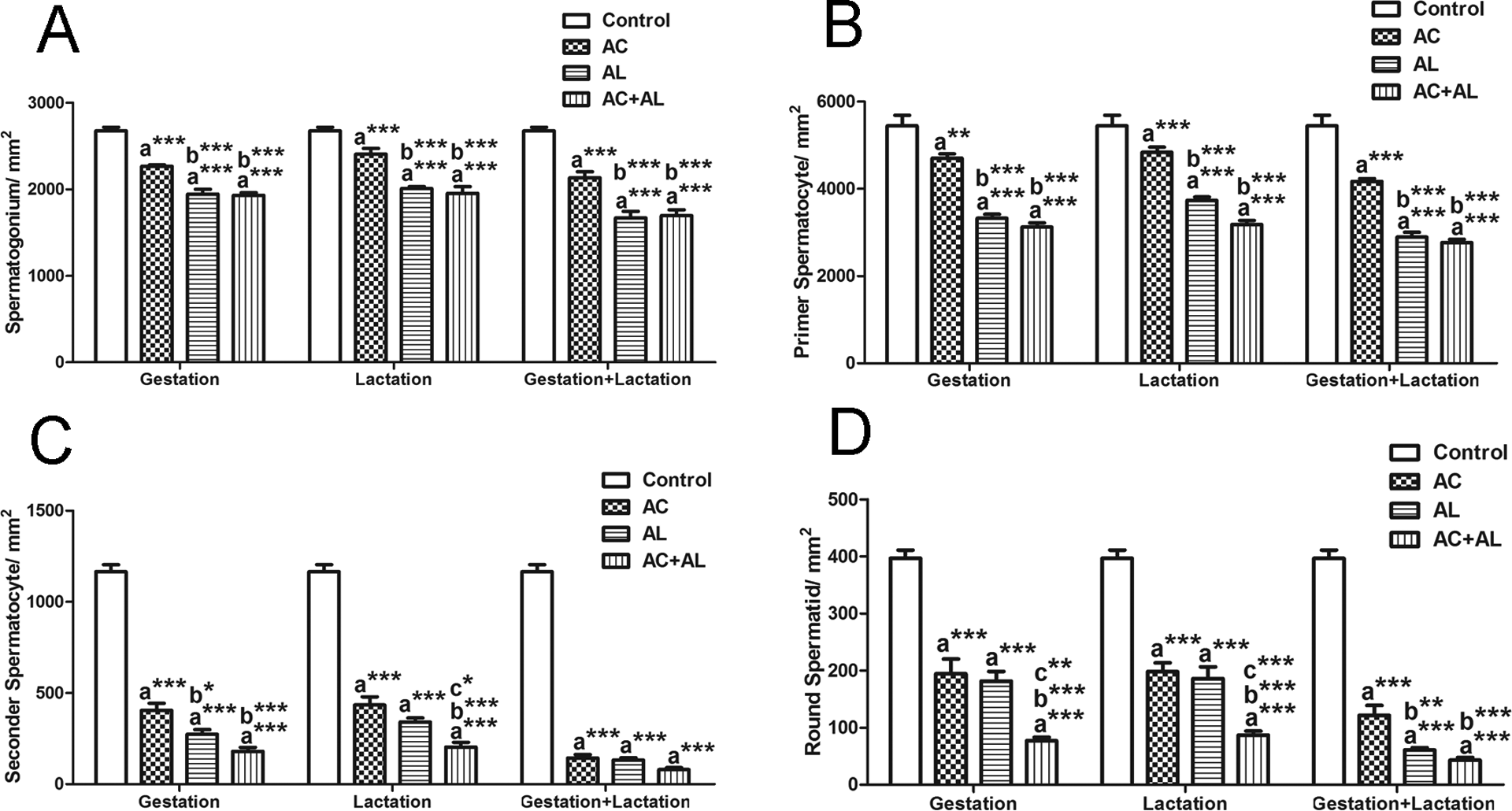
Effect of maternal exposure to Ac and Al on spermatogenic cell count. A: The number of spermatogonia, B: the number of primer spermatocytes, C: the number of secondary spermatocytes, and D: the number of round spermatids. Values are given as mean ± SEM. (a) Compared with control; (b) compared with Ac; and (c) compared with Al; **p < 0.01; ***p < 0.001. Ac: acrylamide; Al: alcohol; SEM: standard error of the mean.

Effect of maternal exposure to Ac and Al on Sertoli (A) and Leydig (B) cell counts. Values are given as mean ± SEM. (a) Compared with control; (b) compared with Ac; and (c) compared with Al; *p < 0.05; **p < 0.01; ***p < 0.001. Ac: acrylamide; Al: alcohol; SEM: standard error of the mean.
The significant increase in the rate of spermatogonia in all experimental groups when compared to the control is shown in Table 4. Primary spermatocyte ratio was increased significantly in all groups except for G-Al group when compared with the control group, while there was a decrease in the rate of secondary spermatocytes and round spermatids in these groups. The rate of spermatogonia increased significantly in Al-treated group compared with the Ac-treated group during gestation; however, the rate of primary spermatocytes decreased significantly in these groups. In addition, there was a reduction in the rate of round spermatids in G-Ac + Al, L-Ac + Al, GL-Al, and GL-Ac + Al groups, when compared with the Ac-treated groups. Moreover, round spermatid ratio was decreased in G-Ac + Al and L-Ac + Al groups when compared with Al-treated group.
Spermatogenic cell rates in seminiferous tubules of male offsprings at PND 21 that were exposed to Ac and Al maternally.a

SEM: standard error of the mean; Ac: acrylamide; Al: alcohol; PND 21: postnatal day 21; C: control; G-Ac: exposed to Ac during gestation; L-Ac: exposed to Ac during lactation; GL-Ac: exposed to Ac during gestation and lactation; G-Al: exposed to Al during gestation; L-Al: exposed to Al during lactation; GL-Al: exposed to Al during gestation and lactation; G-Ac + Al: exposed to Ac and Al during gestation; L-Ac + Al: exposed to Ac and Al during lactation; GL-Ac + Al: exposed to Ac and Al during gestation and lactation.
aValues are given as means ± SEM.
b p < 0.05.
cCompared with control.
dCompared with Ac.
eCompared with Al.
Oxidative stress
The levels of MDA, a lipid peroxidation product, were increased in all groups except for the Ac-treated groups in comparison with the control group. In addition, there was a significant increase in the level of MDA in G-Al, G-Ac + Al, L-Ac + Al, GL-Al, and GL-Ac + Al groups when compared with Ac-treated groups. Moreover, the level of MDA was increased significantly in G-Ac + Al, L-Ac + Al, and GL-AC + Al groups when compared with Al-treated groups (Figure 5(a)). CAT activity was not altered in the experimental groups during only gestation or lactation periods, while the activity decreased in the experimental groups during gestation and lactation periods when compared with the control group (Figure 5(b)). SOD activity was not altered in Ac-treated groups, while the activity was induced only in Al-treated and Ac and Al-treated groups during gestation and/or lactation, when compared with the control group. Furthermore, the activity of SOD enzyme was increases significantly in G-Ac + Al, L-Ac + Al, and GL-Ac + Al groups, when compared with Ac-treated groups (Figure 5(c)).
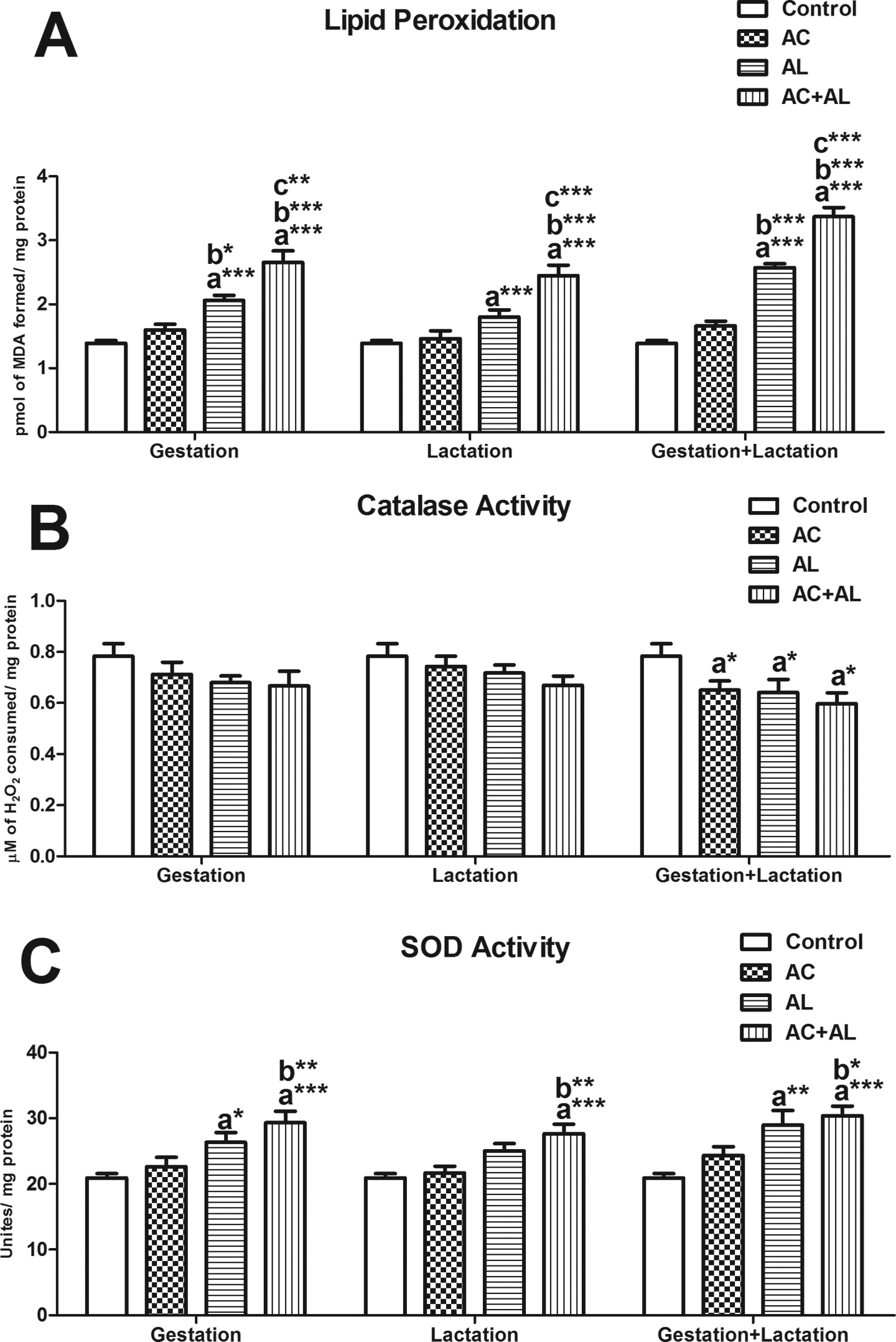
Effect of maternal exposure to Ac and Al on lipid peroxidation (A), catalase (B), and SOD activity (C). Values are given as mean ± SEM. (a) Compared with control; (b) compared with Ac; and (c) compared with Al; *p < 0.05; ***p < 0.001. Ac: acrylamide; Al: alcohol; SEM: standard error of the mean; SOD: superoxide dismutase.
Discussion
In the present study, we first investigated the effects of individual and combined Ac and Al exposure during the gestation and lactation periods on testes of male offsprings. Ac and Al are usually taken up with nutrients. As a result of maternal exposure to Ac and Al individually during gestation and lactation, these substances were passed onto the offspring, resulting in increased birth anomalies, decreased neural development, decreased organ weight, and resulted in morphological changes. 5 –8,13 –22,33 –35,44 –48 Our findings showed that the offsprings’ body weights at birth and PND 21 and gonadosomatic index decreased whose mother was exposed to Ac and Al. The decrease in body weight at birth was more significant in the groups treated with Al during gestation, which suggests that Al is a more effective toxin than Ac. There was also a significant reduction in the gonadosomatic index and body weight in Ac and Al-treated groups during gestation and/or lactation compared with Ac-treated groups, whereas this effect was only significant in treated groups during both gestation and lactation compared with Al-treated groups. These findings suggest that long-term Ac exposure may enhance the toxic effects of Al.
The previous studies demonstrated that individual exposure to Ac and Al decreased the seminiferous tubule diameter and germinal height. 20,45 In our study, maternal Ac and Al exposure during gestation and lactation periods significantly reduced the seminiferous tubule diameter and germinal height, and also the lumen diameter of seminiferous tubule is considerably reduced in Al-treated groups. Ac and Al treatment causes atrophic tubules, maturation-arrested tubules, and degenerative cells. 15 –17,20,22,23,45,46,49 In the current study, the percentages of maturation-arrested tubules and the number of atrophic tubules and degenerative cell numbers are significantly increased particularly in Ac and Al combined groups. These results suggest that maternal Ac and Al exposure during gestation and lactation periods could reduce the testis weight and impair testicular spermatogenesis in fetal and postnatal periods.
Moreover, several studies showed that individual Ac and Al exposure in different periods causes impairments in testicular connective tissue, the presence of spermatogenic cells in the lumen of seminiferous tubules, and vacuolization in spermatogenic cells. 15 –17,20,22,23,45,46,49 –52 They reported that individual exposure to Ac and Al in different periods of time and different doses (10 mg/kg Ac, 15 mg/kg Ac, 60 mg/kg Ac, 5.8 g/kg Al, 30% Al with nutrients and 100 ppm Ac with drinking water) caused connective tissue deformation and vacuolization in spermatogenic cells. 16,20,22,52 Furthermore, Canillioğlu and Ercan 51 reported that individual exposure to Al in gestation and lactation periods was thickening of connective tissue and increased vacuolization, whereas according to Fakoya and Caxton-Martins 45 Al exposure decreased the interstitial tissue. In this study, Ac and Al-treated groups during gestation and/or lactation led to deformities in the connective tissue and caused atrophic tubules and arrested tubules at the primary spermatocyte stage.
Several studies showed that individual exposure to Ac and Al decreases the sperm count, via decreasing the number of cells at certain stages of spermatogenesis. 16,20,22,24,29,53,54 In addition, some studies reported that individual Ac and Al exposure may decrease the Leydig cell number influencing serum testosterone and other hormone levels. 16,20,23,26,33,49,52,54 Moreover, Zhang et al. 53 reported that exposure to Ac (10 mg/kg) caused apoptosis in germ cells. The results of the present study showed that combining Ac and Al treatment during gestation and/or lactation periods reduces the number of spermatogenic, Leydig, and Sertoli cells, which were also reported in the previous studies. 16 –18,20,45,46,49,51 While there was no significant difference in the number of spermatogonia and primary spermatocytes in Ac and Al-treated groups compared to Al-treated groups, there was a significant difference in the number of secondary spermatocytes and early spermatids. These findings suggest that Ac is more effective after the stage of secondary spermatocytes. 49 According to Eftink and Selvidge, 55 Ac inhibits Al dehydrogenase enzymes that function in the biotransformation of Al. The reduction in the number of spermatogenic cells and the increase in degenerated cells in Ac- and Al-treated groups suggest that this might be the underlying reason. According to results of this study, the cells were not able to differentiate into secondary spermatocytes and early spermatids; therefore, spermatogenesis was inhibited. Additionally, we believe that the decrease in the number of Leydig cells is responsible for the inhibition of spermatogenesis. Also, Sertoli cells support spermatogenesis; therefore a decrease in the number of Sertoli cells could result in loss of germ cells. When Ac-treated and Al-treated groups are compared, Al treatment increased the number of spermatogonia and primary spermatocytes and decreased the number of secondary spermatocytes and early spermatids, which suggest that Al is more effective than Ac on inhibition of spermatogenesis gestation and lactation periods.
Oxidative stress can damage various biological molecules, especially DNA, proteins, and lipids. This damage may lead to the formation of reactive oxygen species (ROS) within the cell. 56 ROS causes oxidation of membrane lipids and generates products of lipid peroxidation, which in turn disrupts the membrane integrity. 57 In the present study, we investigated the effects of maternal Ac and Al exposure during gestation and/or lactation periods on lipid peroxidation level in testes. There are various studies investigating the effect of Ac and Al on lipid peroxidation. 21,28,30,31,35,52,58 –63,64 Several recent studies demonstrated that adult Ac exposure obviously increases lipid peroxidation in testes. 52,62,63,64 Another study illustrated that maternal exposure to Ac (10 mg/kg) increases lipid peroxidation levels in the fetal brain. 21 On the contrary, we found that individual Ac exposure did not affect lipid peroxidation levels in the testes. Several earlier studies found that adult Al exposure notably increases lipid peroxidation levels in testes. 28,31,35,58 –61 The current study demonstrated that individual Al exposure increased lipid peroxidation levels during gestation and/or lactation periods. Furthermore, our results indicate that when Ac-treated and Al-treated groups are compared, there was no significant difference in lipid peroxidation during lactation, whereas the level of lipid peroxidation significantly increased during both gestation and lactation periods. The level of lipid peroxidation was significantly higher in the groups treated with both Ac and Al compared to the groups treated with only Ac or Al, suggesting that the combination of these substances has more toxic effect. Furthermore, there was no significant difference in tissue lipid peroxidation levels between the experimental groups treated with Ac and Al during gestation or lactation; however, the level of lipid peroxidation was significantly higher in the group treated with Ac and Al during both periods, which suggests that both substances have a greater effect over the long-term.
Enzymatic and nonenzymatic antioxidants are the natural defense system against free radical-mediated tissue damage in several organs including testes. 65 CAT and SOD are antioxidant enzymes that are involved in scavenging ROS. CAT is one of the enzymes that functions in the removal of H2O2, and it is generally located at the peroxisomes and generates water and oxygen by reacting to combining two H2O2 molecules. 66 The number of studies investigating the effect of Ac on CAT is limited, 22,64 while there are several studies on the effect of Al on CAT. 28,54,58,67 A recent study indicates that adult administration of Ac (10 mg/kg) did not change CAT levels in the testes. 64 CAT is of limited importance in the testes, because there are several isoforms of glutathione peroxidase in this tissue that use glutathione (GSH) as a source of electrons to reduce H2O2 to water. 65 Several studies showed that adult Al exposure decreased (1.6 g/kg Al and 2 g/kg Al) 28,58 or did not alter CAT levels in the testes (3 g/kg Al). 67 Moreover, Dosumu et al. 54 reported that the inhibition of CAT activity (evident in the increased MDA levels) in the Al-only treated group is suggestive of enhanced synthesis of superoxide radicals during Al ingestion. Superoxide radicals have been reported as a powerful inhibitor of CAT. 58 The observation in this study in the means of the reduction of CAT levels in combined Ac and Al-treated groups suggests that Ac enhances the effect of Al. Furthermore, the significant reduction in CAT levels in combined Ac and Al-treated groups during both gestation and lactation periods suggest that these substances are more toxic in the long term.
SOD catalyzes the reactions that remove superoxide radicals. SOD is present in the cytoplasm, between the mitochondrial membranes and within the mitochondrial matrix. 68 There are different reports on decreasing or increasing SOD levels upon Ac treatment. 21,23,62,63,64 There is no consensus in the scientific literature on SOD activity in chronic Al exposure. There are different studies in which SOD activity decreased, remained constant, or increased, and SOD activity differs with respect to the diet and Al consumption. 69 While SOD levels decreased significantly in the previous studies, 21,63,64 we detected an increase in SOD levels in Ac-treated groups similar to studies of Camacho et al. 23 and Yousef and El-Demerdash. 62 Likewise, there was a significant increase in SOD activity in Al-treated groups, while some previous studies shows that SOD levels decreased significantly in Al treatment. 28,58,59 Sridevi et al. 70 also suggest that SOD levels might increase during oxidative stress caused by toxic substances.
In conclusion, the present study indicates that maternal Ac and Al exposure during the gestation and/or lactation period reduce testis weight, the gonadosomatic index, and causes pathological changes in the testes. Additionally, maternal Ac and Al exposure during the gestation and/or lactation period decreases the number of spermatogenic cells, while it increases the number of degenerated cells. Moreover, the level of lipid peroxidation increased significantly after exposure to Ac and Al during the gestation and/or lactation period. Otherwise, Ac and Al did not independently affect the CAT levels during both gestation and lactation; however, CAT levels decreased in groups treated with both substances at both periods. Nonetheless, not all effects are negative. SOD protects the testes from the toxic effects exerted by Ac and/or Al in all groups. Also, Ac and Al exposure were more toxic during gestation compared with the lactation period, and the toxic effects were more significant in groups treated with Ac and/or Al during both periods. In view of importance of human reproductive health and our current dietary habits, it is necessary to further investigate, especially in the level of molecular interaction between Ac and Al exposure during gestation and lactation period in the future.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by Istanbul University Scientific Research Projects (Project numbers: 7503 and 3884).
