Abstract
Acrylamide (ACR), with high prevalence in starchy food, has been associated with the development of several organ toxicities such as immunotoxicity. This study aimed to demonstrate the role of oxidative stress and apoptosis as the mechanisms involved in ACR-induced immunotoxicity in mice. Mice were randomly assigned to six groups and treated as follows: control (normal saline), cyclophosphamide (200 mg kg–1), ACR groups (12.5, 25 and 50mg kg–1, orally), and
Introduction
Immune system plays an important role in protection against infections, diseases, and toxicities caused by several xenobiotic and contaminations. Alterations in the immune pathway harmony play a main role in the pathophysiology of several disorders, such as asthma, autoimmunity, chronic infection, and cancer. 1
Immune system includes a complex network of cells and organs. Spleen and thymus are important organs in immune system’s function. 2
Studies on immunotoxicity revealed that the immune system can be a target for xenobiotic and contaminations in food, such as pesticides and heavy metals. Therefore, chronic exposure to environmental and food contaminant could affect the immune system’s function. 3
Acrylamide (ACR) is a vinyl, water-soluble and colorless crystalline compound used in the production of polymers and copolymers since the 1950s. Interestingly, it was found that ACR can be formed from a reaction between the amino acid asparagine and the carbonyl group of glucose at high temperature (>120) during the Maillard reaction in food processing. 4 In April 2002, the Swedish National Food Authority and the University of Stockholm proved the existence of ACR in heat-treated high-carbohydrate foods including French fries and potato chips, roasted cereals, breads, coffee, and cocoa. 5
Neurotoxic effects of ACR are demonstrated in humans and animal models. Also, researches in rodents showed that ACR can cause reproductive/developmental toxicity, genotoxicity, carcinogenicity, and neurodevelopmental toxicity. 6 International Agency for Research on Cancer categorized ACR as “probably carcinogenic” to humans, too. 7 Recently, the immune toxic effect of ACR has gained attention. Fang et al. showed ACR induced an inhibitory effect on cellular and humoral immunity in mice after 30 days of oral administration. However, they did not present a special mechanism for ACR immunotoxicity. Therefore, further studies are necessary to investigate the detailed mechanisms involved in ACR immunotoxicity. 8
Recent studies suggested the oxidative stress as the mechanism of ACR induces organ toxicity. 9,10 It is well accepted that there is a significant relation between the immune function alteration and oxidative stress. Indeed, it was documented that oxidative stress could induce apoptosis pathway and lead to alteration in the components of immune system. 11
It is well-known that apoptosis acts as a critical factor in cytotoxicity and homeostasis of the cells and tissues of immune system. 12 Impairment in apoptosis process may initiate other important immune disorders such as cancer, AIDS, and autoimmune diseases. 13 Furthermore, the role of apoptosis in immunotoxicity of copper, polychlorinated biphenols, herbicides, and pesticides was clearly shown. 14 –16
Accordingly, in this study, we evaluated the possible relationships between oxidative stress and evoked toxic pathway in mice immune organs after a 30-day oral ACR administration. Also, we assessed the ability of ACR to induce apoptosis in the spleen as the largest tissue of immune system. To confirm the role of oxidative stress in ACR-induced toxicity, the protection effect of
Material and methods
Dosage and treatment procedures
After adaptation for 7 days, 36 Swiss Albino male mice (25–30 g, 6–8 weeks old) were randomly divided into six groups: control group (get normal saline, orally), positive control group (200 mg kg–1 of cyclophosphamide (CP), intraperitoneal injection), different doses of ACR (low dose of ACR: 12.5mg kg–1; medium dose of ACR: 25mg kg–1; and high dose of ACR: 50 mg kg–1 as orally administrated), and
Body/organ weights and hematological parameters
Twenty-four hours after the last dose, the mice were sacrificed, and body and organ weights of the thymus and the spleen were assayed for each animal. Final body weight was evaluated in final day, and organ weights were observed as percentage per final body weight for each animal. For hematological studies, whole blood was collected from the retro-orbital plexus of each mouse by heparinized needle. Then, white blood cell (WBC) count and lymphocytes (LYMs) in peripheral blood were measured with a COULTER Ac·Tdiff2 Hematology Analyzer (Beckman Coulter Inc.). Each animal was checked daily for abnormalities, physical appearance, and mortality. Body weight of mice was measured immediately prior to dosing and at the end of the experiment.
Pathological investigation
After the animals were anesthetized by ketamine, spleen tissues were removed and washed with cold normal saline and fixed in paraformaldehyde solution of 10% for 24 h and then dehydrated in a graded series of ethanol. Also, alcohol was extracted from toluene. We used paraffin in oven for tissues, and the tissues were rapidly saturated by paraffin. After 4 h, blocks samples were fixed on microtome and sections of 3μm thickness were obtained. Then, the sections were transferred to slides. Finally, they were stained with hematoxylin and eosin for assessment with light microscope. 17
Measurement of protein concentration
Protein content was determined in tissues with Bradford method. Bovine serum albumin was used as standard and homogenate samples were mixed with Coomassie blue, and after 10 min, absorbance was determined at 595 nm by a spectrophotometer. 18
Measurement of lipid peroxidation
The lipid peroxidation product (malondialdehyde (MDA)) was quantified by the thiobarbituric acid (TBA) based on the method used by Shokrzadeh et al.
19
Briefly, 0.25 mL of phosphoric acid (0.05 M) was added to 0.2 mL of tissue homogenates and then 0.3 mL of 0.2% TBA. All the samples were placed in a boiling water bath for 30 min. At the end, the tubes were shifted to an ice bath and 0.4 mL of
Measurement of protein carbonyl concentration
Reactive oxygen species (ROS) can attack proteins and modify amino acid (lysine, arginine, proline, and histidine) residues, consequently resulting in the production of carbonylated moieties. Thereby, the content of carbonylated protein can be used as a marker for protein oxidation and damage. 20 Protein carbonyl content was evaluated in both spleen and thymus tissue homogenates by measuring the reactivity of carbonyl groups with 2, 4-DNPH (2,4-Dinitrophenylhydrazine) as described before by Jahani et al. 18 Briefly, 200 μL of tissue homogenates is extracted from 50 μL of 20% (w/v) trichloroacetic acid (TCA). Then, samples are placed at 4°C for 15 min. The precipitates are treated with 500 μL of 0.2% DNPH and 500 μL of hydrochloric acid 2 N for control group, and samples are incubated at room temperature for 1 h with vortexing at 5-min intervals. Then, proteins are precipitated by adding 55 μL of 100% TCA. The microtubes are centrifuged and washed three times with 1000 μL of the ethanol–ethyl acetate mixture. Then, the microtubes are dissolved in 200 μL of 6 M guanidine hydrochloride. Protein carbonyl concentration was determined from the absorbance at 365 nm and expressed as millimolar of carbonyls. 18
Measurement of GSH content
Glutathione (GSH) concentration was determined by 5,5′-dithiobis-2-nitrobenzoic acid (DTNB) as the indicator and by a spectrophotometer. Briefly, tissue homogenates were deprotonated with TCA by centrifugation. Then, 0.1 mL of tissue homogenates was added to 0.1 mol L–1 of phosphate buffers and 0.04% DTNB in a total volume of 3.0 mL (pH = 7.4). Then, the developed yellow color was read at 412 nm on a spectrophotometer (UV-1601 PC; Shimadzu, Japan). A standard curve was drawn using different specified concentrations of GSH solution. With the help of this standard curve, the GSH content was calculated and expressed as nanomolar protein. 19
Evaluation of apoptosis
Splenocytes isolation
Spleens were isolated following the sterile procedures described by Xu et al.
21
Basically, spleens were harvested from animals and transferred to the laboratory in normal saline on ice. Single-cell suspensions of spleen were prepared by homogenizing the organ between the frosted ends of two sterilized microscope slides into a dish containing 5 mL of cold culture medium (500 mL RPMI 1640 with 10% fetal bovine serum (FBS), 2 mM
Detection of apoptosis
Splenocyte apoptosis was measured using annexin V and propidium iodide (PI) cell staining, by flow cytometry with annexin V Apoptosis Detection kit (eBioscience, San Diego, CA, USA, cat number: 88-8005). This assay is based on the ability of the protein annexin V to bind to phosphatidylserine (PS) exposed on the outer membrane leaflet in apoptotic cells. In viable cells, PS is located in the inner membrane leaflet, but upon induction of apoptosis, it is translocated to the outer membrane leaflet and becomes available for annexin V binding. The annexin V assay was performed following the manufacturer’s instructions. Briefly, the cells were washed with PBS and resuspended in binding buffer. Then, 100 μL of cell suspension was mixed with 5 μL of annexin V-FITC and incubated for 10 min in dark at room temperature. The cells were washed with binding buffer, resuspended in 200 μL of binding buffer, and then 5 μL of PI staining solution was added and analyzed by flow cytometry (Partec, Deutschland) within 4 h. 22
Statistical analysis
Results are presented as mean ± SEM (standard error of mean). All statistical analyses were performed using the SPSS software, version 13. Assays were performed in triplicate, and the mean was used for statistical analysis. Statistical significance was determined using the one-way analysis of variance test, followed by the post hoc Tukey’s test. Statistical significance was set at
Results
Results of body/organ weights and hematological parameters
As shown in Table 1, a significant reduction in body and organ weights was observed after ACR administration compared to control group. Similarly, CP(a known immunosuppressive agent) induced a significant reduction in final body weight and spleen weight of mice when compared with animals in the control group. This reduction in thymus weight was not significant (Table 1). The mean WBC count and percentage of LYM of mice in the CP group were significantly lower than those in the control group. Furthermore, the percentage of LYMs decreased after the high dose of ACR administration about 64% compared to that of control group. Although other doses of ACR decrease the count of WBC and percentage of LYM, these depletions were not significant (Table 2).
Effects of ACR on final body and organ weights of mice.a

ACR: acrylamide;
aFinal body weight was evaluated in final day. Organ weights were observed as percentage per body weight. Data were expressed as mean ± SEM.
bSignificantly different from control group (
cSignificantly different from control group (
dSignificantly different from control group (
eSignificantly different from ACR (50 mg kg–1) group (
fSignificantly different from ACR (50 mg kg–1) group (
Effect of ACR on hematology values of mice.a

ACR: acrylamide;
aData were expressed as mean ± SEM.
bSignificantly different from control group (
cSignificantly different from control group (
dSignificantly different from control group (
eSignificantly different from ACR (50 mg kg–1) group (
fSignificantly different from ACR (50 mg kg–1) group (
Pathological manifestation
Spleen is the largest lymphoid organ in the body. Spleen has two different components: the white pulp and the red pulp. The white pulp consists of lymphoid tissue (such as T cells, B cells). The red pulp contains large numbers of sinuses and sinusoid filled with blood. 23 Histological studies showed morphological changes in the spleen of mice after administration of CP and high dose of ACR. There was the structural disorganization in white and red pulps in the spleen after administration of CP and ACR. Also, the congested sinusoid with intracellular hemorrhages was observed. Analysis of white pulp areas showed that ACR and CP treatment reduced white pulp nodules. These findings indicate that ACR induces toxicity in the spleen.
On the other hand, administration of

Hematoxylin- and eosin-stained section of mice spleen (×400); (ACR) spleen from animals treated with ACR (50 mg kg–1) and CP-treated group showing the structural disorganization in white pulp (thin arrow) and the congest sinusoid with intracellular hemorrhages in red pulp (thick arrow); (control) group that received normal saline showing almost normal morphology; (
Results of lipid peroxidation
MDA level, end product of lipid peroxidation, was measured in all treated groups. An ascending trend in MDA concentration was observed in thymus and spleen tissue homogenates of mice after ACR treatment as a dose-dependent manner. Also, after administration of high doses of ACR, the MDA concentration was increased about two times of control group in both tissues. Interestingly,

Effects of ACR on lipid peroxidation in (a) thymus and (b) spleen tissues of mice. Data were expressed as mean ± SEM. *Significantly different from control group (
Results of protein carbonyl concentration
Protein carbonyl concentration in tissue homogenates of mice was increased as a dose-dependent manner after treated with ACR compared to control group. High doses of ACR amplified protein oxidative damage, while the protein carbonyl concentrations in both tissues were more than 1.5 times of control group. Likewise, administration of CP led to the same raise in protein carbonyl concentration in both thymus and spleen tissues compared to control group (Figure 3). Also, the spleen and thymus of the mice that received
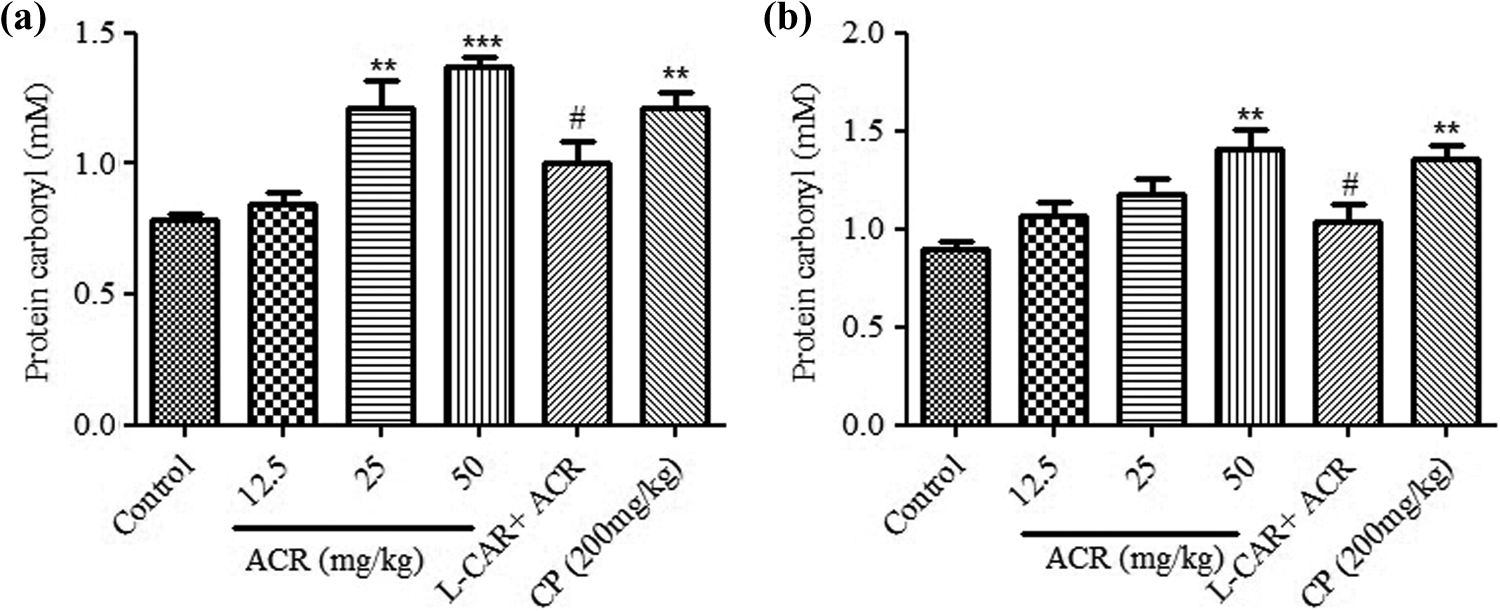
Effects of ACR on protein carbonyl content in (a) thymus and (b) spleen tissues of mice. Data were expressed as mean ± SEM. **Significantly different from control group (
Results of GSH measurement
Another bioindicator of oxidative stress, GSH level, was also determined in treatment group. As shown in Figure 3, a significant and dose dependence lowering tendency in GSH concentration was observed in thymus and spleen tissue homogenates of mice treated with different doses of ACR compared to control group (
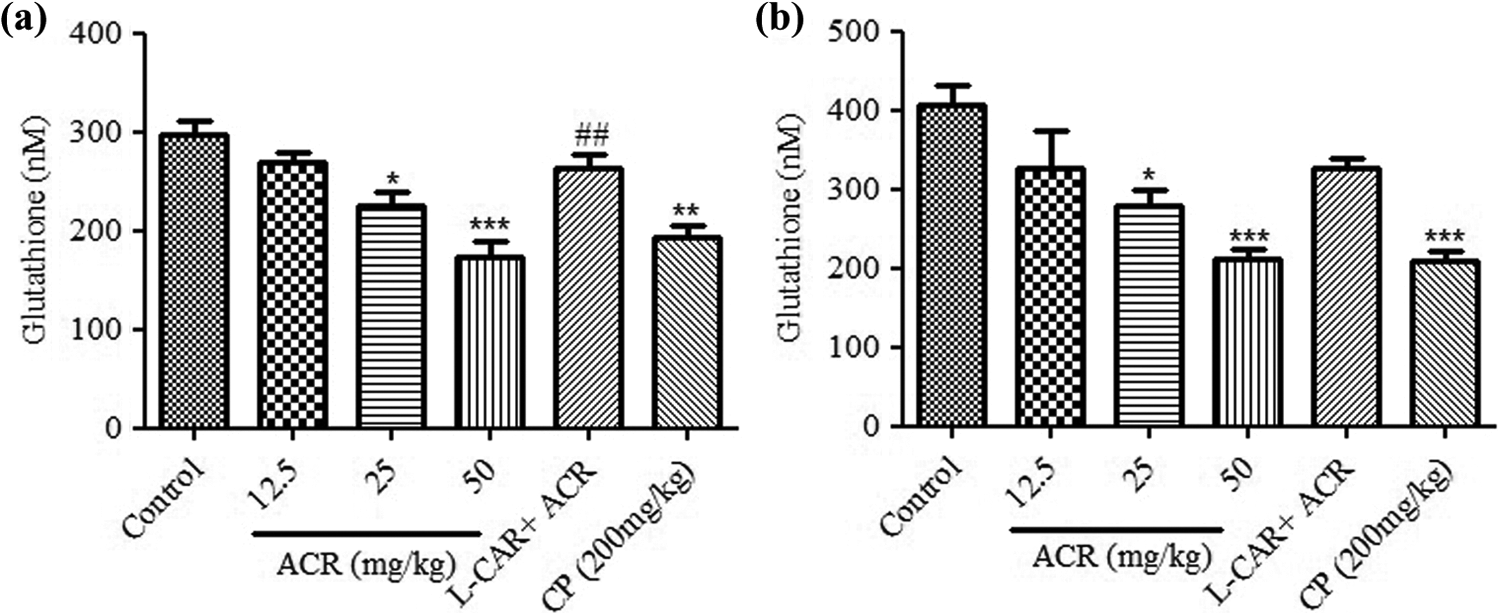
Effects of ACR on glutathione content in (a) thymus and (b) spleen tissues of mice. Data were expressed as mean ± SEM. *Significantly different from control group (
Effect of ACR on apoptosis in splenocytes
The percentage of apoptosis was calculated by flow cytometry analysis. The lower left quadrant of each graph in Figure 5(a) revealed normal cells that shows the minimum absorbance of annexin and PI (annexin V−/PI−), and the upper and lower right quadrants contain the early and end stages of apoptotic population (annexin V+/PI−). Our results indicate that apoptosis in splenocytes increased in a dose-dependent manner after ACR administration. The percentage of early and late stage apoptotic death in splenocytes of mice treated with 25 and 50 mg kg–1 of ACR, respectively, was about five and seven times of control group. Similarly, CP induced apoptosis death in the same way as in the high dose of ACR (Figure 5).

Apoptosis in splenocytes. (a) Analysis of splenocyte apoptosis of annexin V/PI-stained cells detected by flow cytometry. (b) The effect of ACR exposure on splenocyte apoptosis. Mean percentage of apoptotic cells was measured using flow cytometry. Data were expressed as mean ± SEM. ***Significantly different from control group (
The interesting section was where
Discussion
Immune system can be a primary target for many food and environmental pollutants and immunotoxicity often occurs before other toxicities. 24
In this study, we evaluated the potential immunotoxic effects of ACR and the mechanism involved in the pathogenesis of ACR immonotoxicity. Also,
After oral administration of ACR, decrease in weighs of thymus and spleen was observed. Zaidi et al. showed that ACR administration decreased the weight of thymus and spleen in rats. 25 Although changing in body weight is an important nonspecific sign in animal toxicities, it can also reproduce, general health and physiological state in animals. 24 A significant decrease in body weight of ACR-treated mice was observed in our study (26.83 ± 0.94 and 30.18 ± 0.41 g, respectively, for doses of 50 and 25 mg kg–1 of ACR compared to 34.10 ± 0.42 g for control group). The pathological observation confirmed immunotoxic potential of ACR characterized by cell death and atrophy of spleen after administration of ACR. In addition, the counts of LYMs decreased after ACR administration (from 75.77% ± 1.92% for control group to 48.67% ± 8.66% after dose of 50 mg kg–1 of ACR). Besides, there was a reduction in the WBC counts after ACR exposure (from 5.77 ± 0.68 × 10 6 µL–1 for control group to 3.1 ± 0.35 × 10 6 µL–1 after dose of 50 mg kg–1 of ACR). All these evidences confirmed the potential effect of ACR on immune system. These data confirmed the previous evidence about the toxic effects of ACR on immune systems. 8
Apoptosis is a programmed cell death that has essential role in mainlining hemostasis in different parts of body such as immune system. Apoptosis could affect the components of the immune system and cause immune disorders such as AIDS. There are several literatures that show many xenobiotic-inducing apoptosis on immunecompetent cells. 26 For example, glucocorticoids, as immunosuppressive agents, are known to provoke apoptosis of immune cells. 12 Also the role of apoptosis in pesticide-induced immunotoxicity was clearly shown. 27 Thus, the results showed that apoptosis in splenocytes was assessed using flow cytometry after ACR administration. The percentage of apoptotic splenocytes increased significantly in ACR-treated groups. So that, the percentage of apoptosis in splenocytes after administration of high and medium doses of ACR (50 and 25 mg kg–1), respectively, was 26.42% ± 2.05% and 18.43% ± 0.97%, while this value for control group was 3.63% ± 0.52% that is an indicator of ACR cytotoxicity. This is in accordance with previous studies that showed the ACR-induced apoptosis in human astrocytoma cells. 28
Oxidative stress is the main trigger in initiation of apoptosis. Additional ROS and oxidative damage end product are toxic for cells. For example, MDA has toxic effects on cells, which could be linked with cellular macromolecules such as proteins and nucleic acids to induce cell apoptosis.
Furthermore, oxidative stress is known as the main mechanism involved in many toxicological procedures and contributed to immunotoxicity of various agents. 29 For example, relation between oxidative stress and immunotoxicity following exposure to cadmium was shown in zebra fish. 30 Also, previous studies reported increased oxidative stress and decreased antioxidant capacity in some ACR toxicological effects, such as neurotoxicity, reproductive, and genotoxicity. 31 However, in this study, for evaluating the role of oxidative stress in ACR-induced immunotoxicity, we evaluated oxidative stress markers such as lipid peroxidation, GSH level, and protein carbonyl content in thymus and spleen after ACR exposure in mice.
Lipid peroxidation and protein carbonyl are important markers of oxidative stress that significantly increased after ACR exposure in both tissues. In our study, lipid peroxidation was shown by MDA level (end product of lipid peroxidation) that markedly increased to 11.15 ± 0.71 µM after high dose of ACR (50 mg kg–1) compared to 5.71 ± 1.50 µM for control group in thymus tissue. Furthermore, this value was significant in spleen tissue (34.39 ± 1.64 µM for dose of 50 mg kg–1 of ACR in comparison with 20.25 ± 2.09 µM for control group). Increase in lipid peroxidation after ACR exposure is outcome of ACR-induced radical production and attack to membrane and other lipid sections of cells. Protein carbonyl level was also raised in thymus (from 0.79 ± 0.02 mM in control group to 1.22 ± 0.1 mM and 1.37 ± 0.04 mM after, respectively, medium and high doses of ACR) and spleen (from 0.89 ± 0.05 mM in control group to 1.41 ± 0.1 mM after dose of 50 mg kg–1 of ACR. These data are obtained as a result of oxidative damage to protein after ACR administration in spleen and thymus tissues.
Increase in oxidant markers finally leads to attenuation of antioxidant system in cells. On this basis, we measured the level of GSH (as an important intrinsic antioxidant). We showed significant depletion of GSH in thymus (from 297.2 ± 13.51 µM in control group to 225.9 ± 12.84 µM and 173.4 ± 15.16 µM for medium and high doses of ACR) and spleen tissues (from 406.1 ± 26.76 µM in control group to 279.9 ± 20.12 µM and 212.7 ± 10.34 µM for medium and high doses of ACR) after ACR exposure that was along with the increase in lipid peroxidation and protein carbonyl content. Also, depletion of GSH after ACR treatment in human Caco-2 cells was shown by Chen et al, which confirmed the results of our study. 32
It is clearly well-known that using an antioxidant can be valuable for emphasizing the role of oxidative stress as a main-suggested mechanism for ACR-induced immunotoxicity. Many studies support the beneficial effects of
Conclusion
In this study, it was found that ACR exposure led to oxidative damage in spleen and thymus tissues that these alterations induced apoptosis in splenocytes. Finally, immune function of treated mice was affected by oxidative damage and apoptosis. For example, the reduction in counts of WBC and LYM and body/organ weights were shown. Furthermore,
Footnotes
Authors’ note
The data provided in this study were extracted from a part of PhD thesis of Dr Ehsan Zamani.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the research council of Mazandaran University of Medical Sciences, Sari, Iran (registration code: 1822).
