Abstract
The present study was designed to investigate aluminium phosphide (ALP)-induced testicular toxicity, including its effects on sperm parameters and histological alterations in Wistar rats, and the possible protective role of hesperidin (HSD). Oral administration of ALP at 1.15 mg/kg body weight (1/10 LD50) for 30 days resulted in a significant increase in testicular malondialdehyde, lipid hydroperoxides, and oxidized protein levels. These indicators of oxidative stress were accompanied by decreased activity of the antioxidant enzymes superoxide dismutase, catalase and glutathione peroxidase, followed by a drastic reduction in the non-enzymatic antioxidant indices of glutathione and total antioxidant capacity when compared to control. Furthermore, ALP treatment produced a marked reduction in sperm count, motility and viability while increasing abnormal sperm morphology and adverse histopathological changes in testis. Co-administration with HSD significantly ameliorated ALP-induced testicular damage by suppressing oxidative stress indices and enhancing antioxidant status while also improving the sperm parameters and histological alterations in ALP-treated rats. The results of the present study indicated that testicular toxic effects of ALP are due to oxidative imbalance and that HSD could be a potential therapeutic agent against ALP-induced testicular damage.
Introduction
Aluminium phosphide (ALP) is a fumigant pesticide that is used in the preservation of agricultural products and processed foods. 1 It has gained extensive use worldwide due to its low cost, efficacy, short half-life and low toxicity of its decomposition products. 2 However, it has proven to be highly toxic to both humans and animals on exposure. 3 Its widespread use poses the hazard of accidental and intentional poisoning to man. Even though ALP owes its notoriety to its deliberate ingestion which has resulted in high number of casualties, 4 accidental and occupational exposures to the pesticide have been reported. 5 –7
ALP toxicity is potentially lethal and affects multiple systems with severe consequences. Its toxic effects are manifested in various forms including cardiotoxicity, 8 neurotoxicity, 9 electrolyte imbalance, 10 hepatotoxicity, 11 metabolic disturbances, 10 haematological toxicity 12 and renal toxicity, 13 among others. The ALP exact mechanism(s) of action has not been well defined. But like other solid phosphides, ALP exerts its toxic effects through the liberation of highly toxic gas, phosphine (PH3), which is released in contact with water, moisture or gastric hydrochloric acid. 4 PH3, a highly reactive radical, when released after the hydrolysis of ALP diffuses to intracellular compartments causing cellular injury through oxidative damage. 14 It is generally reported that oxidative stress is the major contributor to ALP-induced cellular toxicity. 4,15,16 Exposure to ALP down-regulates the activities of both catalase (CAT) and glutathione peroxidase (GPx) while lowering the level of GSH and the total antioxidant capacity (TAC). 4,8 It reportedly stimulates the activity of superoxide dismutase (SOD), in addition to increasing the generation of hydrogen peroxide (H2O2), reactive oxygen species (ROS) and malondialdehyde (MDA). 16
Current management of ALP poisoning has remained mostly supportive, 13 there is, therefore, a need to search for a treatment that can adequately protect against it. Phytochemicals like flavonoids have proven to be useful therapeutic agents in the treatment of the oxidative stress-related injury. Flavonoids are a group of phenolic compounds that are found widely distributed in plants and have proven to possess biological properties. 17 Hesperidin (HSD) is a phytoflavanone that is abundant in citrus fruits like lemon and oranges. 18 It is a flavanone glycoside comprising the flavanone hesperitin and the disaccharide rutinose. 17 It produces a wide range of pharmacological effects, including hypolidemic, 19 anti-inflammatory, 17,20 anticarcinogenic 21,22 and antioxidant. 23 It also can attenuate hyperglycaemia-mediated oxidative stress and pro-inflammatory cytokine production in diabetic rats. 24 It has been reported to protect against chemical-induced injuries, such as hepatotoxicity and nephrotoxicity resulting from cisplatin treatment. 25,26 The use of HSD as a supplement in the treatment of some neurodegenerative diseases has proven to be effective 27 just as it has been shown to protect against neurotoxicity. 28 HSD’s ability to protect cells from oxidative stress has been predicated on its functioning as a potent radical scavenger. 29 –31 Its attenuation of benzo[α]pyrene-induced testicular toxicity in the rat was also credited on its regulation of oxidant/antioxidant balance. 32
Although ALP is widely regarded as a multi-system toxicant, studies on its testicular effect are sparse in literature. Additionally, the effect of HSD on ALP-induced toxicity has not been established. Therefore, in this study, we investigated the effect of ALP exposure on the rat testis and the potential of HSD as a protective agent against ALP-induced oxidative damage in the rat.
Materials and methods
Chemicals
HSD, triphenylphosphine (TPP), xylenol orange and tripyrl-S-triazine were produced by Sigma-Aldrich (Munich, Germany), while trichloroacetic acid and thiobarbituric acid were by Qualigens Fine Chemicals (Mumbai, Maharashtra, India). All other chemicals were of analytical grade.
Animal treatment
Thirty male adult Wistar rats (120–130 g) aged between 10 and 12 weeks were procured from The Animal House, Faculty of Basic Medical Sciences, Ladoke Akintola University of Technology. Rats were kept in plastic cages in a ventilated room under controlled laboratory conditions of normal light–dark cycle (12 h light/dark) and temperature (25 ± 2°C). The animals had access to food and water ad libitum. Experimental design and animal handling were executed according to the guidelines approved by the Research Ethical Committee of the Faculty of Basic Medical Sciences, Ladoke Akintola University of Technology, Nigeria that is in agreement with the Guide for the Care and Use of Laboratory Animals published by the National Institute of Health.
Experimental design
After 2-week acclimation period, rats were randomly assigned to five different groups of six animals each. Group I rats, serving as the control, orally received 1 ml/kg body weight (bwt) of corn oil, 2 h after administration of same volume of saline. Group II rats were orally administered ALP at a dose of 1.15 mg/kg bwt (one-tenth LD50) in corn oil. 33 Animals in group III orally received HSD dissolved in saline, at a dose of 200 mg/kg bwt. Groups IV and V were orally administered HSD at doses of 100 and 200 mg/kg bwt, respectively, 2 h before treatment with ALP. All treatments were given for 30 days. The dose of one-tenth LD50 for ALP was chosen to produce a sub-lethal toxic effect in the animal, while that of HSD was based on its antioxidant capacity, as previously published. 32,34 Twenty-four hours after the last treatment, the rats were anaesthetized using diethyl ether and killed by cervical dislocation. The testes were excised, cleared of adhering connective tissues, weighed and subsequently processed for biochemical assays and histology.
Biochemical analysis
Protein estimation
The protein content of the testes was determined by the method of Lowry et al., 35 using bovine serum albumin as a standard.
Determination of MDA levels
The level of lipid peroxidation was quantified by MDA content in the testis. Testicular MDA was determined using the thiobarbituric acid reactive substance assay, as described by Buege and Aust 36 with slight modifications. Briefly, 10% of testis homogenate was prepared in 0.15-M KCl. One volume of homogenate was mixed thoroughly with two volumes of a stock solution of 15% w/v trichloroacetic acid, 0.375% w/v thiobarbituric acid and 0.25 M hydrochloric acid. The mixture was heated for 15 min in a boiling water bath. After cooling, the mixture was centrifuged at 1000 × g for 10 min to remove the precipitate. The absorbance of the clear supernatant was determined at 535 nm and MDA concentration calculated using an extinction coefficient of 1.56 × 105 M−1 cm−1. The MDA concentration was expressed as nmol per gram of wet tissue.
Determination of lipid hydroperoxide concentrations
The concentrations of lipid hydroperoxides (LOOH) in testicular homogenate were estimated using the method of Nourooz-Zadeh et al. 37 Tissue homogenate (90 µl) was mixed with either 10 µl of 10 mM TPP in methanol or with 10 µl of methanol and incubated for 30 min at room temperature. Then, 900 µl of FOX2 reagent (250 µM ferrous ammonium sulfate, 100 µM xylenol orange, 25 mM H2SO4 and 4 mM butylated hydroxytoluene in 90% methanol) was added and the mixture was incubated for another 30 min. Flocculated material was removed by centrifuging the mixture at 12,000 × g for 10 min after which the absorbance was read at 560 nm.
Estimation of protein oxidation in testes
Protein oxidation was estimated spectrophotometrically in the testes by measuring advanced oxidized protein products (AOPP) by the method of Witko-Sarsat et al. 38 Briefly, 20% solution of the sample homogenate was prepared with phosphate-buffered saline (PBS). After which 1.16 M potassium iodide was added, followed by acetic acid. The absorbance of the reaction mixture was read immediately at 340 nm. Concentrations of AOPP were calculated using the extinction coefficient of 26 l mM−1 cm−1.
Determination of TAC
The TAC of testis was evaluated using the ferric reducing ability of plasma (FRAP) method as described by Benzie and Strain. 39 The FRAP method evaluates the potential of antioxidants in reducing ferric-tripyridyltriazine (Fe3+-TPTZ) to its ferrous form (Fe2+). Briefly, pre-warmed FRAP working reagent (300 mM acetate buffer, pH 3.6, 10 mM 2,4,6-tripyridyl-S-triazine in 40 mM HCl and 20 mM FeCl3 in ratio 10:1:1) was mixed with 10% homogenate of the testis. The test was performed at 37°C, and the absorbance was read at 593 nm. Aqueous solutions of known Fe2+ concentration were used for calibration.
Determination of reduced GSH
Reduced GSH in the testis was determined using the method of Moron, Depierre. 40 The supernatant (1 ml) was treated with 0.5 ml Ellman’s reagent in 0.1% sodium citrate. After that, phosphate buffer (2 ml) and 0.5 ml of DTNB were added before the absorbance was read at 412 nm.
Determination of CAT activity
The activity of CAT was measured by the method of Aebi. 41 In brief, the assay mixture maintained at 20°C consisted of 0.1 ml of supernatant tissue homogenate, 1.9 ml of 50 mM phosphate buffer and 1 ml of 30 mM H2O2. The decomposition of H2O2 was continuously monitored spectrophotometrically at 240 nm for 60 s. The change in absorbance was the measure of CAT activity and was expressed as nmol/mg protein.
Determination of SOD
The method of Misra and Fridovich 42 was used in determining SOD in the rat testis. The reaction was initiated with the addition of 0.3 ml of epinephrine (0.01%) to the mixture of 2.5 ml carbonate buffer (0.05 M) and 0.2 ml sample. Change in absorbance was measured at 480 nm, and the activity was expressed as unit per milligram of protein. One unit was defined as the amount of enzyme required to inhibit the rate of autooxidation by 50%.
Determination of GPx
GPx activity was determined using H2O2 as a substrate in the presence of reduced GSH, according to the method of Rotruck et al. 43 The reaction medium contained 0.2 ml of phosphate buffer (0.4 M), 0.1 ml of sodium azide (10 mM), 0.2ml of tissue homogenate and 0.2 ml of GSH. The reaction was initiated by adding 0.2 mM H2O2 to the mixture. The GSH content was quantified using Ellman’s reagent. The activity was expressed as unit per milligram protein where a unit is µmol of GSH consumed per minute.
Sperm analysis
Determination of sperm motility and epididymal sperm count
Epididymal spermatozoa were collected as previously described by Gupta et al. 44 The epididymis of the testis was removed, trimmed of fat and placed in physiological saline at 37°C. The cauda was minced to allow dispersion of the sperm in the medium. The sperm suspension was thoroughly mixed, and a drop of the suspension was placed on a pre-warmed (37°C) microscope slide and the percentage of motile sperm was evaluated with the aid of a microscope. Epididymal sperm count was done using a Neubauer counting chamber. 45
Determination of sperm morphologic abnormalities and viability
For the evaluation of sperm morphology, a drop of the prepared sperm suspension was smeared on a microscope slide, stained with Wells and Awa’s stain and examined for morphological abnormalities. Sperm cells with head or tail defects were considered abnormal. 46 Sperm viability was determined by the eosin-nigrosin staining technique described by Rato et al. 47 The stain penetrates sperm cells with the damaged cell membrane and cells with the characteristic red colour of the stain were considered non-viable.
Histopathology
The testicular tissues were dissected, and the tissues were fixed with Bouin’s solution for 24 h. 48 The fixed tissues were processed using a graded ethanol series and embedded in paraffin. Paraffin sections were made at a thickness of 4–5 µm and subsequently stained with haematoxylin and eosin for light microscope examination by a histopathologist.
Statistical analysis
One-way analysis of variance was used for the statistical analysis after which multiple comparisons were carried out by Tukey’s test. The results were expressed as the mean ± standard deviation (SD) and values with p < 0.05 were considered as statistically significant. Data were analysed using GraphPad Prism version 6. 49
Results
Body and testicular weights
Effects of HSD on the body and testicular weights of ALP-treated rats are shown in Table 1. At the end of the 30 days of treatment, the percentage of body weight gain was significantly reduced in the ALP group when compared to the control (p < 0.05). However, concurrent treatments with HSD significantly improved the percentage body weight gain in ALP exposure. HSD administration at 200 mg/kg bwt entirely prevented the loss in body weight associated with ALP exposure and the percentage body weight gain in this group did not differ significantly from that of the control and HSD groups (p > 0.05). Although there were no treatment-related changes in absolute testicular weight among the groups, the relative weight of testis in the ALP group was significantly increased (p < 0.05) compared to the control. Co-treatment with HSD at the doses tested prevented any changes in relative testis weight of the animals. No mortality was observed during the experimental period.
Changes in body weight, percent body weight gain, absolute testis weight and relative testis weight in rats after ALP exposure and treatments with HSD.a
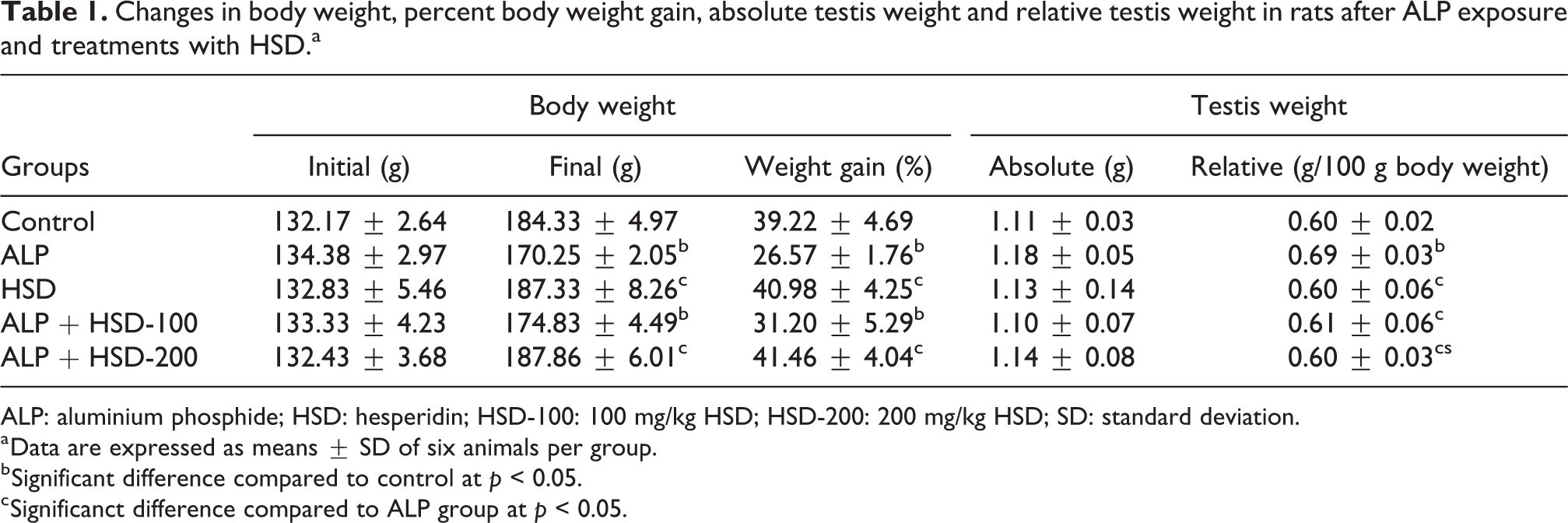
ALP: aluminium phosphide; HSD: hesperidin; HSD-100: 100 mg/kg HSD; HSD-200: 200 mg/kg HSD; SD: standard deviation.
a Data are expressed as means ± SD of six animals per group.
b Significant difference compared to control at p < 0.05.
c Significanct difference compared to ALP group at p < 0.05.
Effects of the treatment on lipid peroxidation and protein oxidation indices
The changes in the levels of lipid peroxidation and protein oxidation products in control and treated rats are depicted in Table 2. The levels of testicular MDA and LOOH were significantly increased in ALP-treated rats compared with the control group (p < 0.05). Conversely, co-administration with HSD significantly decreased the testicular levels of these lipid peroxidation products (p < 0.05), though not to the levels found in the control group. Similarly, there was a marked increase in protein oxidation products in ALP-treated rats when compared with the control. HSD administrations significantly decreased the level of AOPP, and normalcy was maintained in ALP-treated rats by the 200 mg/kg bwt treatment to a level comparable with the control. There was no significant change in lipid peroxidation and protein oxidation products levels in testes of rats administered HSD alone in comparison to the control group.
Testicular MDA, LOOH and AOPP levels in rats after ALP exposure and treatments with HSD.a
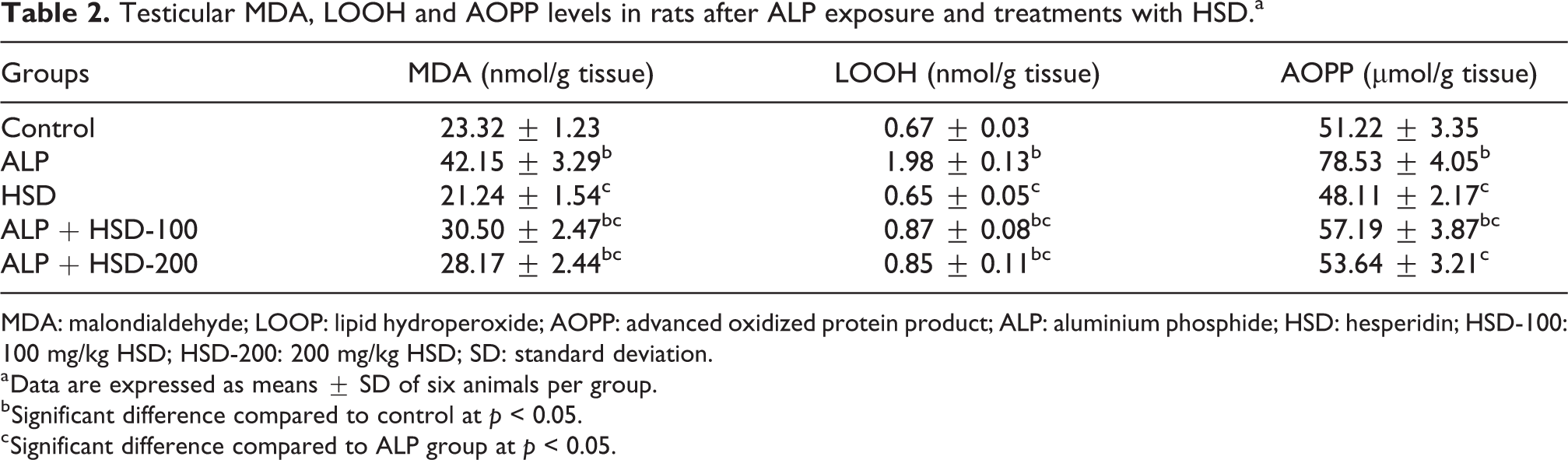
MDA: malondialdehyde; LOOP: lipid hydroperoxide; AOPP: advanced oxidized protein product; ALP: aluminium phosphide; HSD: hesperidin; HSD-100: 100 mg/kg HSD; HSD-200: 200 mg/kg HSD; SD: standard deviation.
a Data are expressed as means ± SD of six animals per group.
b Significant difference compared to control at p < 0.05.
c Significant difference compared to ALP group at p < 0.05.
Effects of the treatment on antioxidant systems
The enzymatic and non-enzymatic antioxidant status in control rats and those treated with ALP alone or in combination with HSD are presented in Table 3. There was no significant change in the activities of GPx, SOD and CAT in rats treated with HSD when compared with the control. Treatment with ALP alone resulted in a significant decrease in GPx, SOD and CAT activities when compared with the control (p < 0.05). However, co-administration with HSD significantly reversed the reduction in activity of the enzymes by stimulating the activities to near control for SOD but thoroughly for CAT and GPx in ALP-treated rats. The levels of GSH and TAC were also significantly reduced in the testes of ALP-treated rats when compared with the control (p < 0.05). HSD administration maintained normalcy of GSH and TAC concentrations in the testes of ALP-treated rats when compared to the control.
Testicular enzymatic and non-enzymatic antioxidant status in rats after ALP exposure and treatments with HSD.a
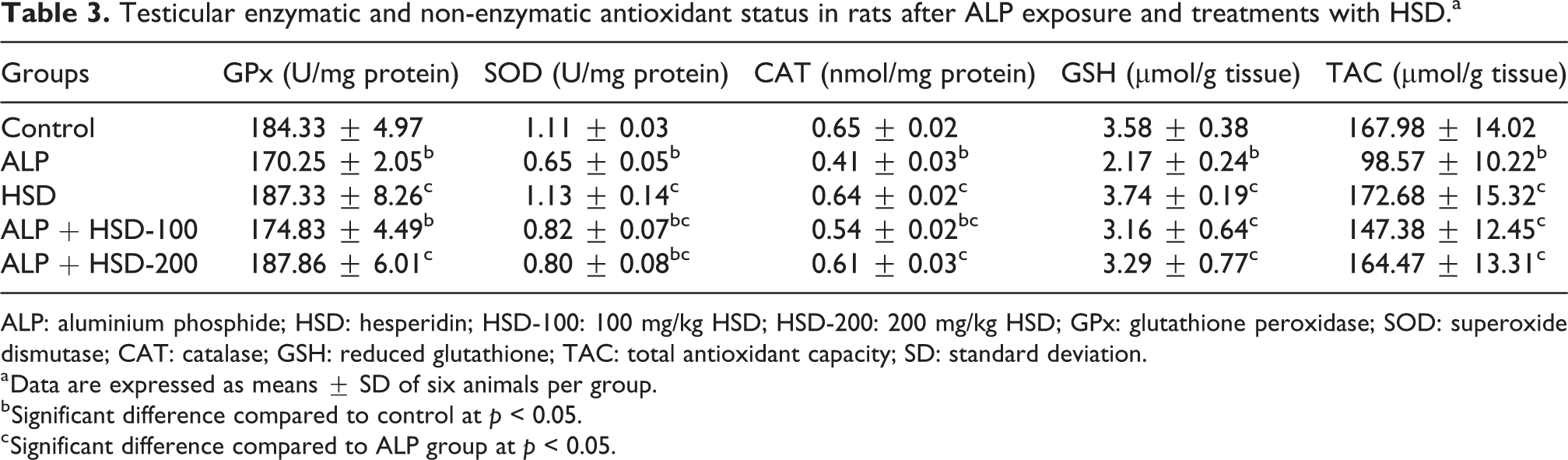
ALP: aluminium phosphide; HSD: hesperidin; HSD-100: 100 mg/kg HSD; HSD-200: 200 mg/kg HSD; GPx: glutathione peroxidase; SOD: superoxide dismutase; CAT: catalase; GSH: reduced glutathione; TAC: total antioxidant capacity; SD: standard deviation.
a Data are expressed as means ± SD of six animals per group.
b Significant difference compared to control at p < 0.05.
c Significant difference compared to ALP group at p < 0.05.
Effects of the treatment on sperm functional parameters
Changes in sperm characteristics are displayed in Table 4. A significant decrease (p < 0.05) was observed in total sperm count, sperm motility and percentage viability in rats treated with ALP alone as compared to the control rats. Moreover, in the ALP-treated group, the percentage of abnormal sperm with morphological defects was significantly increased above the control. The abnormalities observed in this group include tailless heads, bent mid-pieces and curved mid-pieces. Co-treated rats with ALP and HSD displayed a significant increase in sperm motility, sperm count and percent viability compared with the ALP group. HSD when co-administered with ALP also reduced significantly the level of abnormality caused by ALP treatment, though not to the level of the control. No significant change was observed in sperm concentration, motility, abnormal sperm count and viability in rats treated with HSD alone when compared to the control animals.
Sperm characteristics in rats after ALP exposure and treatments with HSD.a
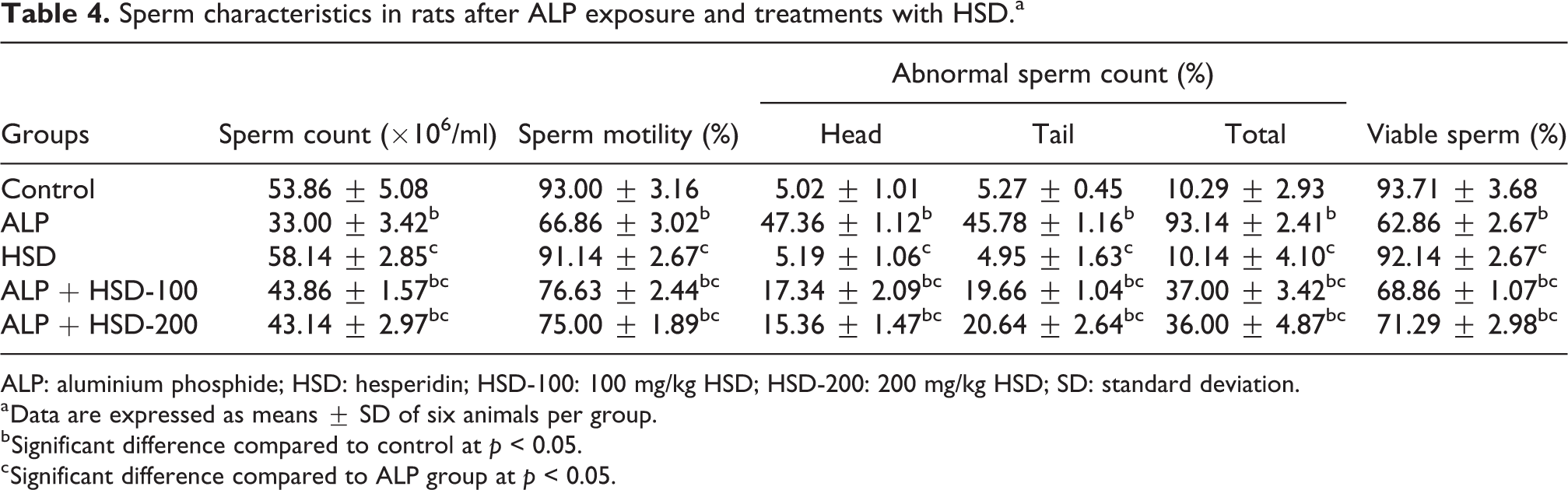
ALP: aluminium phosphide; HSD: hesperidin; HSD-100: 100 mg/kg HSD; HSD-200: 200 mg/kg HSD; SD: standard deviation.
a Data are expressed as means ± SD of six animals per group.
b Significant difference compared to control at p < 0.05.
c Significant difference compared to ALP group at p < 0.05.
Histopathological observations
The testes of control and HSD rats appeared structurally and functionally normal with uniform seminiferous tubules and regular architecture (Figure 1(a) and 1(c)). The ALP-treated testes, however, showed severe damage characterized by depletion of spermatogenic epithelium and seminiferous tubules (Figure 1(b)). The result revealed incomplete spermatogenesis, spermatogenic arrest, shrinkage of the interstitial cells, haemorrhage, enlarged lumen and thin basement membrane. On the other hand, the testes of rats co-administered with HSD at 100 mg/kg showed congestion and oedema in the interstitial tissue but with normal architecture (Figure 1(d)). Rats treated with ALP and HSD at 200 mg/kg bwt, however, displayed significantly reduced congestion and oedema but with structurally normal seminiferous tubules and the spermatogenic epithelium closely resembling those in control (Figure 1(e)).
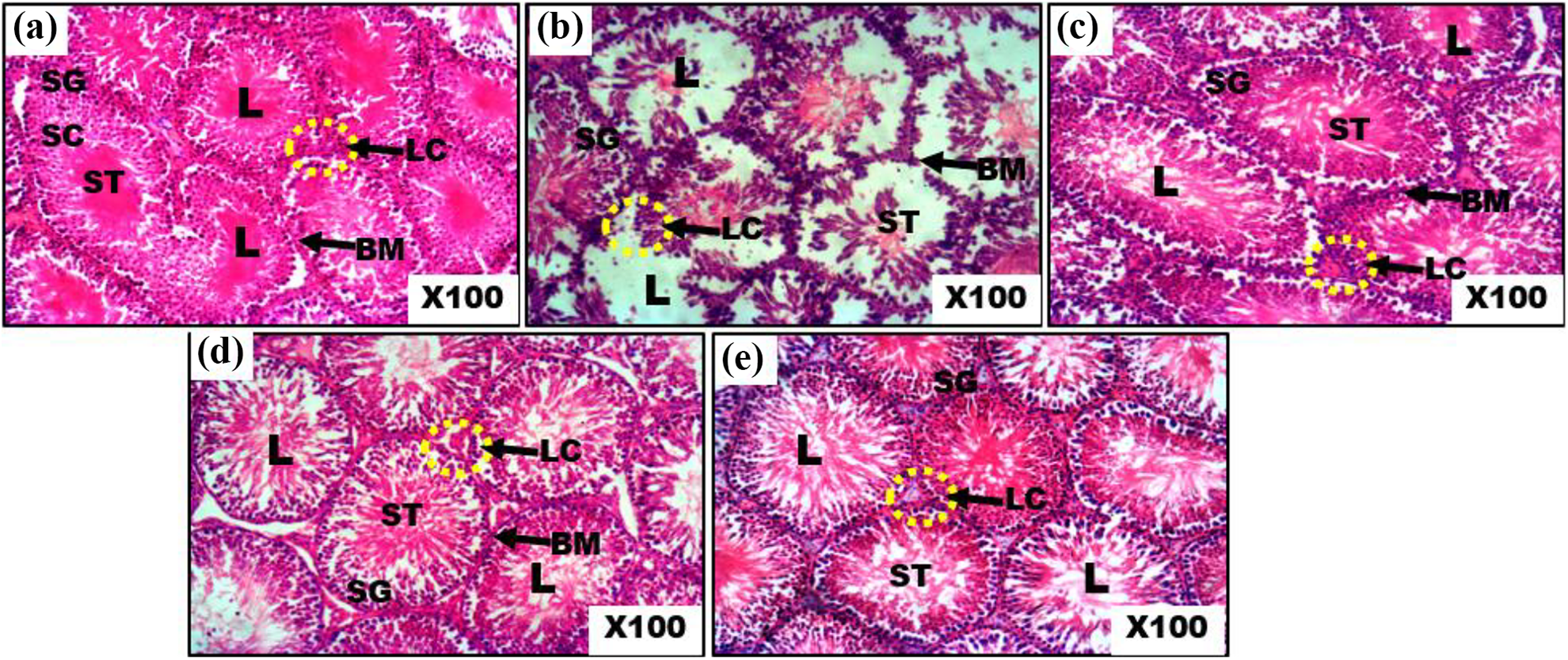
Photomicrographs of the testicular histomorphology of adult rats across treated groups. (a) Control testis showing normal seminiferous tubules and spermatogenesis; (b) ALP-treated testis showing severe damage characterized by incomplete spermatogenesis, spermatogenic arrest, shrinkage of the interstitial cells, haemorrhage and thin basement membrane; (c) HSD testis presenting normal histological architecture; (d) ALP co-treated with 100 mg/kg HSD showing a reduction in the damage to the interstitial cells and basement membrane; (e) ALP co-treated with 200 mg/kg HSD displaying a mild disruption of spermatogenesis and reduced haemorrhage. H&E stain ×100. ST: seminiferous tubule; SC: sertoli cell; BM: basement membrane; L: the lumen, SG: spermatogonium; LC: interstitial space containing interstitial cells; H&E: haematoxylin and eosin; ALP: aluminium phosphide; HSD: hesperidin.
Discussion
The oral administration of ALP at 1.15 mg/kg bwt induced a marked testicular damage in the rat. This was evident from increased testicular lipid peroxidation and protein oxidation levels coupled with disruption of the antioxidant defence system. These findings were associated with changes in testicular morphology and changes in spermatogenesis. The use of HSD ameliorated some of these toxic effects. This study, to the best of our knowledge, was the first to report ALP-induced testicular damage in rats and that the flavonone HSD could attenuate this effect.
Metabolism of xenobiotics causes increased production of electrophilic radicals that can result in the elevation of ROS in cells, giving rise to free radicals like H2O2, hydroxyl radicals, singlet oxygen and peroxynitrite. The proliferation of ROS is countered by the cell endogenous antioxidant defensive mechanism, enzymatic and non-enzymatic, that tend to convert these free radicals to non-toxic products. 50 An imbalance in these arrangements leads to oxidative stress. Lipid peroxidation, of which MDA and LOOH are indicators, signifies damage to tissues due to oxidative stress. These markers are produced as a result of ROS attacks on tissue fatty acids. Testis, because of its high polyunsaturated membrane lipid contents, is highly susceptible to oxidative stress. 51 In our study, exposure to ALP significantly increased testicular MDA, LOOH and AOPP levels, while significantly decreasing testicular TAC and GSH concentrations. Antioxidant enzyme activities, namely GPx, SOD and CAT activities, were also markedly reduced by ALP. Although the literature on ALP testicular effect is scanty, similar findings have been reported in other organ systems. 3 The ALP-induced oxidative damage in testes could have resulted from increased free radicals generation mediated by ALP inhibition of the electron transport chain. Studies have shown that PH3, a gas produced on ALP contact with moisture, impairs the activity of cytochromes and metalloproteins, 10 and these have been suggested to be responsible for its inhibitory effect on complexes I and II activities observed in rats. 52
The high concentrations of polyunsaturated fatty acid along with the low antioxidant capacity make testicular tissues highly susceptible to attack from ROS, with subsequent production of peroxides, alcohol and lipid aldehydes. 53 Increased production of ROS in the testis reportedly tends to significantly alter tissue physiology and induce DNA damage, potentially compromising male reproductive ability. 54,55 The testis, however, utilizes both enzymatic and non-enzymatic systems to protect against these effects. In tissues, SOD and CAT are considered the first-line defence against the toxic effects of ROS 56 and have been reported to exhibit synergism in their functioning. 57 Superoxide radicals are converted to H2O2 by SOD which is subsequently converted into water and oxygen by the action of CAT. In this study, the decrease in both SOD and CAT activities indicated a breakdown of the enzymatic antioxidant system against ALP-induced oxidative stress. The reduction in SOD activity in this study is contrary to the report of Mehrpour et al. 16 This could be a result of the difference in doses and duration of the study. A reduction in SOD activity usually leads to an increased level of superoxide anion, which can subsequently inactivate CAT activity. Conversely, the failure of CAT to prevent H2O2 accumulation in the cell could lead to the inactivation of SOD. 58 The reduction in CAT activity is therefore suggestive of the inactivation of the enzyme and its inability to eliminate H2O2 produced by SOD resulting to the accumulation of this free radical in the organ. 58 H2O2 is a potent oxidant capable of inducing oxidative damage in macromolecules, and its increased production may further aggravate the oxidative damage in the testes of the rats.
In biological systems, GSH and GPx act as detoxifying agents by participating in the GSH redox cycle where they convert H2O2 and lipid peroxides to nontoxic products. 50 In our study, the ALP administration significantly depleted GSH concentration and caused a decline in GPx activity in the rat testis. The decline in the GSH status could be attributed to an increased oxidative stress burden, triggered by the production of ROS that induced lipid and protein oxidation. 59 This is shown in our study by the marked increase in testicular MDA and AOPP contents in the ALP-treated animals. Increased production of lipid peroxides leads to diminished lipid soluble antioxidant system, such as GSH which could disrupt testicular GSH-dependent antioxidant enzymes as reflected in reduced GPx activity in this study. The depletion of GSH could further make the enzymatic antioxidant system such as GPx susceptible to oxidative damage, contributing to the enzyme’s reduced activity. Increase in ROS production has been associated with the generation of oxidized proteins, which explains the high testicular AOPP concentration observed in the ALP-treated animals. 60 The oxidative modification of proteins, therefore, could have contributed immensely to the decrease in activities of enzymes generally found in the present study. Oxidative stress in testis can be highly damaging because of its high rates of metabolism and cell replication. 50 The level of its antioxidant capacity is therefore critical in preventing oxidative stress induced injury in this tissue. HSD treatment significantly attenuated both the lipid peroxidation and the oxidant-mediated protein damage caused by ALP treatment.
ALP treatment resulted in reduced testicular antioxidant potential as indicated by the lowered FRAP level in ALP-exposed group. The antioxidant capacity estimation provided a value for the relative cumulative capacity for the total antioxidant system in the tissue. The reduction in TAC by ALP treatment reflected an antioxidant imbalance in the testis, indicating that the antioxidant system was over-stretched. Several studies have associated infertility in males with deficient antioxidant capacity in male reproductive tissues. 61,62 The results of the present study, however, indicated that co-treatment of ALP with HSD improved the TAC level in the rat testis. The changes effected by ALP exposure were all significantly reduced by treatment with HSD. Although studies about HSD protection against ALP-induced oxidative stress in testis are not available, several studies have shown the flavonone protects against oxidants-induced toxicity in the reproductive system. HSD has been reported to protect against cadmium, benzo[α]pyrene, arsenic and doxorubicin. 32,63 In our study, HSD restored the activities of the antioxidant enzymes SOD, CAT and GPx, while reducing MDA and AOPP levels in the testis. The antioxidant capacity, as well as GSH concentration, was significantly boosted compared with the ALP-treated rats. Flavonoids are natural antioxidants and can scavenge various types of radicals in both aqueous and organic environments. 64 The amelioration of oxidative mediated impairment in testicular tissues of ALP-treated rats by HSD administration indicated that the flavonone effectively scavenged free radicals and ameliorated the oxidative damage. Earlier studies have shown HSD as a potent antioxidant agent that protects against chemical-induced toxicity, including cyclophosphamide-induced genotoxicity, 65 doxorubicin-induced cardiotoxicity 66 and acetaminophen-induced oxidative stress. 34 From studies, it has been suggested that HSD exerts its antioxidant properties by direct radical scavenging and augmentation of cellular antioxidant defense. 17 HSD possesses the ability to neutralized ROS, and this property plays a vital role in its reported protection of macromolecules and tissues against damage induced by both intrinsic and extrinsic factors. 34,67 –69
The eventual disruption of membrane integrity by the destruction of the lipid matrix can lead to axonemal damage, decreased sperm viability, increased morphological defects and ultimately the complete inhibition of spermatogenesis. 1 Exposure to insecticides has been previously linked with a decrease in sperm count and motility and increase in abnormal sperm morphology. 70,71 In our study, ALP treatment adversely affected sperm functional parameters in rats. There were drastic reductions in sperm count, viability and sperm motility, while sperm abnormalities were markedly increased by exposure to ALP. The observed decrease in sperm count indicated ALP interference with spermatogenesis in the rats, via induced free radical generation in the tissue. Lipid peroxidation has been reported to be detrimental to germ cell membrane, eventually inducing cell death. 72 The drastic reduction in the sperm count along with the increase in abnormal sperm morphology in this study hence demonstrated the spermototoxic effects of ALP. Such toxic effects have been attributed to altered steroid metabolism and suppressed antioxidant enzyme activities. 73 Disruption of spermatogenesis and increased morphology are important indicators of genetic damage in mammalian species, possibility, thus exists that ALP is capable of inducing testicular DNA damage. 74,75 Also, the significant increase in total abnormal sperm rate is an indication of the cell’s distorted structural components. This when considered given a disrupted mitochondrial energy production both occasioned by ROS generation, could have resulted in the observed decrease in sperm motility. 76 Studies have implicated free radical generation in reduced sperm motility, 32 and this has been known to compromise the fertilizing capacity of sperm. 77 Other studies supported this notion that free radicals generation could decrease sperm motility 32,50 HSD co-treatment in this study significantly improved all sperm characteristics. HSD attenuated the adverse effect of ALP on sperm characteristics by enhancing the sperm count, motility, viability and morphology, when compared to ALP-treated animals. The results obtained suggested that HSD can ameliorate the adverse effects of ALP on testicular function in rats and this could be predicated on HSD inherent antioxidant properties.
Oxidative stress results from the excessive generation of oxidants with the consequent accumulation of oxidation products in the testis leading to tissue degeneration. ROS targets cells and leads to oxidative damage of critical cellular macromolecules. The testicular system is vulnerable to oxidative stress because of its high contents of unsaturated fatty acids. 78 ALP is known to induce free radical generation in addition to causing peroxidation, and these were demonstrated in our study by increased testicular MDA and AOPP contents along with disruption of the antioxidant system. Animals exposed to ALP in our study displayed severe disruption of the spermatogenesis. This was contrary to the testicular histomorphometric study of Fathi et al. 79 that reported no adverse effect of ALP on testicular histology. Fathi et al. 79 study was, however, a 24-h acute study in rats where the sub-lethal dose of phosphide might not have exerted a significant level of toxicity in the animals. In this study, ALP-induced disruption in spermatogenesis was characterized by incomplete spermatogenesis, spermatogenic arrest and shrinkage of the interstitial cells in testes of ALP-treated rats. This histological evaluation confirmed the increase in oxidative stress and the attendant decrease in antioxidant status resulting from ALP treatment. There were also signs of haemorrhage, enlarged lumen and thin basement membrane. The shrinkage of the interstitial cells is adduced to free radical-induced peroxidation resulting in damage to the Leydig and Sertoli cells, revealed by the haemorrhage observed in the ALP-treated rats. Damage to Leydig cells is known to result in reduced production of testosterone and eventually to the resultantly reduced testosterone protection of the sertoli cells. 80 This will ultimately result in the disruption of spermatogenesis in the testis. ROS attack on the testicular system as observed in this study can damage the germ cell membrane and cause dysfunction of the sperm, which can induce cytopathological changes 81,82 and cell death. 72 HSD treatment, however, significantly ameliorated ALP-induced testicular damage in the rats with the 200 mg/kg bwt treatment preserving the testicular architectures though not entirely to the control level.
Conclusion
In conclusion, exposure to ALP-induced significant testicular toxicity which was demonstrated by increased lipid peroxidation, generation of oxidized proteins and perturbation of the oxidant/antioxidant balance in the rat testis. These toxic effects were also associated with histopathological changes and deterioration of sperm quality. HSD administration, however, ameliorated all indices implicated in ALP-induced testicular toxicity in this study. HSD due to its antioxidant and free radical scavenging properties, therefore, presented an immense therapeutic potential in protecting against testicular toxicity resulting from ALP exposure.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
