Abstract
Effect of chlorpyrifos pesticide on testicular oxidative damage was studied in Sprague-Dawley rats at varying doses. At lower doses (5 and 10 mg/kg body weight/30 days), reduction in plasma levels of testosterone and follicular stimulating hormone (FSH) and luteinizing hormone (LH) along with significant shrinkage of seminiferous tubules and drastic changes in germ cells were seen. But these adverse changes of testes were restored with the revival of serum testosterone and FSH and LH at higher doses (20 and 30 mg/kg body weight/30 days). Similarly, levels of testicular lipid peroxidation and diene conjugates were elevated whereas activities of antioxidant enzymes (superoxide dismutase, catalase and glutathione peroxidase), steroidogenic (Δ5, 3β-HSD and Δ5, 17β-HSD) enzymes and angiotensinogen-converting enzyme and glutathione content including lipid–protein content of testes were decreased at low doses. But at higher doses, reductions in level of lipid peroxidation (as revealed by malondialdehyde [MDA] value) and conjugated dienes were found and on the contrary, revivals of testicular antiperoxidative/antioxidant enzymes defense systems, angiotensinogen-converting enzyme (ACE), steroidogenic enzymes, lipid–protein and antioxidant glutathione content were observed. Therefore, the present study indicated from the results that chlorpyrifos had a dual effect at both doses on oxidative stress changes, but at higher doses, the cells were triggering its natural defense mechanism to combat the insult of lower doses of chlorpyrifos and became operative possibly through corrective measure of antioxidant enzymes defense system and pituitary gonadotropins hormones feedback mechanisms on testes.
Introduction
Chlorpyrifos (CPF, O-diethyl–3,5,6–trichloro-2-pyridyl–phosphothionate) is a broad spectrum organophosphate pesticide widely used in agricultural and public health programme and also for domestic purposes at home for weed control. 1 It has been reported that the insecticides and pesticides enter the animal cells either directly by dermal contact or ingestion or indirectly through food chain and preferably accumulate in the membrane lipids of the cells. 2,3 Earlier studies on CPF-treated rat brain suggest that CPF alters rat brain and nervous system development by one or several mechanisms. 1,4 Possible mechanism of induction of brain damage which is caused due to oxidative stress in the adult brain is evident from the exposure to some organophosphorus pesticides. 1,4 The recent studies show that CPF causes testicular toxicity and severe degenerative changes in seminiferous tubules, which result in severe testicular damages. 5 In their study, it shows that CPF has stimulated the effects related to the reproductive toxicity which is associated with the toxic alterations in testicular histology, biochemistry, sperm dynamics and testosterone levels. 5 They also report that CPF can cause marked reduction in epidymal and testicular sperm counts in testes of exposed males. 5 But how CPF causes testicular toxicity in the light of biochemical aspect of oxidative stress and changes in lipid peroxidation and steroid biogenic enzymes, antioxidant enzymes profiles, lipids and proteins, glutathione is not resolved till date. In our previous studies, it is reported that quinalphos (one of the environmental estrogenic organophosphorus insecticides) induces testicular oxidative stress and damages and consequently degeneration of testicular tissues, and it is caused due to free radical-mediated lipid peroxidation at low doses. 3,6 It is explored that in response to that damage at low dose, an endogenous antioxidant enzyme defense system became operative at the higher dose of the treatment. 3,6
As of now, there is no information available on the effect of CPF on oxidative stress and testicular lipid peroxidation in relation to and in association with testicular damages and the role of antioxidant enzymes defense systems. The present study has been attempted and undertaken to explore the possible cause of CPF-induced testicular damages in male rat in the light of biochemical aspect of oxidative stress and the role of endogenous antioxidant enzyme defense system of testes of adult albino SD rats.
Materials and methods
Chemicals
Chlorpyrifos (CPF; 99.5% pure as per HPLC assay) was obtained from Sigma-Aldrich, GmbH, Germany, olive oil (100% pure) from Bertolli Chemical Company, Italy, thiobarbituric acid, β-nicotinamide adenine dinucleotide (NAD), reduced form of glutathione (GSH), 2-mercaptoethanol, catalase, sodium azide, bovine serum albumin (BSA), tris hydroxymethyl amino methane, 5,5-dithiobis- (2 nitrobenzoic acid), folin ciacalteu reagent, haematoxylin and eosin and 1,3,3-tetra methoxy propane (TMP) and spectroscopic grade of ethanol, glycerol, testosterone from Sigma-Aldrich Chemical Company, St. Louis, MO, USA. All other chemicals of analytical grade were from E. Merck, Germany, testosterone hormone radio-immunoassay assay kit (RIA TESTO CTC KIT) from Dia-Sorin Company, CA, USA, antioxidant enzymes assay kits such as superoxide dismutase assay kit (catalogue no.574600), catalase assay kit (catalogue no; 219263) and glutathione assay kit (catalogue no. 354102) from CNBiosciences-Calbiochem, Switzerland, and E. Merck, Germany, pituitary gonadotropins hormones such as follicular stimulating hormone (FSH) and luteinizing hormone (LH) radio-immunoassay (RIA) kits supplied from NIADDK, Bethesda, Maryland USA as generous gift were used.
Treatment of male rats with chlorpyrifos pesticide
Male Swiss albino adult rats − SD strain (body weight ranging 200−220 g) were used, bred at our Animal Physiology Research Laboratory and maintained under standard condition of light and dark (12 h: 12 h) scheduled at 24 hours with constant ambient temperature of 22 ± 2°C and humidity 40%−60% (maintained by dehumidifiers) and fed on balanced scientifically prepared diet rich in carbohydrate and protein with vitamins and minerals supplementation, and water ad libitum as per guidelines of Institutional Animals Ethics Committee. CPF (99.5% pure) dissolved in olive oil as vehicle, was injected in male rat intraperitoneally (ip) per day up to 30 days. In the study protocol, five groups each containing twelve male adult rats were considered and animals were treated as described below: Group I − Control olive oil vehicle-treated male SD rats (Gr.- I- COV-30 D), Group II − CPF 5 mg/kg body weight treated male SD rats (Gr.- II-CPF-5 mg-30 D), Group III− CPF 10 mg/kg body weight treated male SD rats (Gr.-III-CPF-10 mg-30 D), Group IV − CPF 20 mg/kg body weight treated male SD rats (Gr.-IV-CPF-20 mg-30 D), Group V − CPF 30 mg/kg body weight treated male SD rats (Gr.-V-CPF-30 mg-30 D)
Control olive oil vehicle-treated and CPF-treated rats were sacrificed under similar conditions following the method as described in our previous research reports.
3,6
Animals were killed after cessation of injection after 24 hours of the last injection, following the animal ethical rules. Rats were weighed in a top pan digital balance (Mettler Toledo make, Germany, model 705184G) just at the onset of experiment and 24 hours after the last injection to observe any final differences in the body weight changes of the rats in the groups in order to record the final body weight changes in all treatment groups.
Sacrifice of animals and experimental sample preparations
Twenty-four hours after the last injections (i.e. at 31st day), the final body weight of each groups was recorded and quick dissection was followed after killing the rats under light anesthetic ether as per animal ethics rules. The fresh blood samples for the radio-immunoassay of plasma gonadotropins and testosterone were collected from the hepatic vein under light ether anesthesia as per animal research protocols. 3,7 Immediately after the cessation of treatment, for histological and biochemical studies, both right and left testes of the control vehicle-treated and CPF-treated rats were dissected, freed of any adherent tissues, washed in ice-cold saline, blotted with dry filter paper and then weights recorded on a top pan balance sensitive to 0.0001 g to examine the testicular weight changes.
Morphological assays (histology of testis) and study of spermatogenesis
For histological studies, the right testis of each rat was fixed in Bouin’s fixative solution immediately after removal and kept overnight at 4°C, it was then dehydrated in graded alcohol. It was then processed into paraffin embedded blocks, following the routine histological methods for evaluation of histomorphological and histometric studies including quantitative analysis of spermatogenesis. These above-mentioned studies were done on paraffin sections of 5 µm to 7µm thick sections stained with haematoxylene and eosin and/or periodic acid-Schiff stain (PAS). The prepared stained slides of each group were examined under optical microscope (Leica Make, Germany) at various microscopical magnification levels (×100, ×220 and ×320) and photographed at a magnification level at ×220 by Leica make MPS photographic attachment following the routine histological procedure. Testicular histological changes were studied from the stained sections at various microscopical magnification levels (×100, ×220 and ×320) and were supplemented by routine histometric studies such as measurement of seminiferous tubular diameter and degree of testicular atrophy (extent of shrinkage) and spermatogenesis. 6,9
Hormone estimations by radio-immunoassay of serum hormones (testosterone, FSH and LH)
Serum collected from fresh blood sample was prepared by centrifugation and further processed for radio-immunoassay of testosterone and pituitary gonadotropins such as FSH and LH following research protocols as described by the supplier’s RIA assay kits. 9,10 In brief, serum testosterone levels were measured by using immunochem TM-coated tube of testosterone 125I RIA Kit following the radio-immunoassay method as prescribed by the supplier. 9,10 The sensitivity of testosterone was 0.05 ng /ml and the intra and inter assay coefficient of variations were 6.7 –8.1% and 5.7 –10.5%, respectively. Serum concentrations of FSH and LH were estimated using a double antibody radio-immunoassay, as described in the instructions provided with the kits supplied and obtained as generous gift from the NIADDK, Bethesda, MD, USA.9,10 The sensitivities of the assays were μg/L for FSH and 1 μg/L for LH. The within-assay coefficient of variation in each assay was 7%. Intra-assay variation was 3.5%. 8, 11
Biochemical estimations of lipid peroxidation (MDA value), antioxidant enzymes activities and steroidogenic enzymes levels antioxidant, GSH and lipid proteins of testicular tissues
From a portion of left testis of each rat of each group, a 10% (w/v) tissue was used to prepare as 10% tissue homogenates in chilled 100 mM TRIS-HCl buffer containing 0.16 M KCl (pH 7.4) using Remi make homogenizer with teflon pestle, following standard procedures. 3,8,10 . In brief, MDA, the end product of lipid peroxidation (as one of the biochemical parameters to study tissue degeneration and testicular damage), which was produced in the testicular tissues after CPF-treatment, was determined by the thiobarbituric acid reaction substances of an aliquot of 10% (w/v) tissue homogenate of the testes. 3,8,10
A total of 10% tissue homogenates (w/v) mentioned above was subjected to ultracentrifugation. 3
The cytosolic fraction (soluble supernatant fraction) was then run for equilibrium dialysis against Tris-HCl buffer (pH 7.4) containing 0.16 M KCl and the supernatant was used for the assays of antioxidant enzymes activities (AOE) such as superoxide dismutase (SOD) enzyme activity, catalase (CAT) enzyme activity and glutathione peroxidase (GSH-PX) following the methods described. 8 The steroid biogenic enzymes-3β-HSD and 13β-HSD enzyme activity were estimated spectrophotometrically. 7,12 Quantification of antioxidant-reduced GSH was done by monitoring the absorbance at 412 nm, which reflects the rate of reduction of 5, 5-dithiobis-(2-nitrobenzoate) to 2-nitro-5-thiobenzoate following standard method as described in the assay kit. 3,8 Testicular lipids and protein content were quantified following the method described. 3,8
Statistical analysis
Statistical analysis was performed with the analysis of variance followed by a multiple two-tailed t-test 12,13 using the Statmost software 3.5 version. One-way analysis of variance (ANOVA) is used to investigate the effects of single independent variable on the dependent variable. All statistical comparisons for biochemical assays, body weight and testicular weight changes and histometric measurements etc. were made by ANOVA and Post-hoc ANOVA tests for multiple comparison (between the groups and more than two groups) to find out whether the differences of mean value in each parameters between different group combinations are significant. Difference were considered significant when * p < 0.05 compared to the control olive oil vehicle-treated animal group I. 13,14
Results
Gross physiological changes (such as body weight and testicular weight changes) were reflected as follows: none of the rats treated with CPF at the doses described showed signs of morbidity or mortality during the studies. In comparison to the initial body weight, no significant changes were noticed in any of the CPF-treated rats, but the weights of the paired testes were reduced significantly after the treatment of CPF at dose level of 10 mg/kg body weight (Table 1 ). But results revealed a tendency of revival of testicular weight at higher doses of CPF-treated groups (Table 1), which was concomitant with the histomorphological changes of testes to the respective CPF-treated animal groups as shown in photographs of testes (Figure 1 ). Quantitative cytological studies of germ cells at Stage VII of seminiferous epithelial cell cycle revealed that CPF treatment at low dose resulted in a significant reduction of Asg, pLSc., mPSc., 7Sd, as compared to the control but at high dose treatment, there was a significant recovery very close to the control ( Table-2 ).
Effect of Chlorpyrifos (CPF) on body weight and reproductive parameters of Swiss Sprague-Dawley male rat testis (mean ± SEM from 12 male rats)

a Significant at p < 0.05, when compared with vehicle control group (i.e., group I).
Effect of chlorpyrifos (CPF) on spermatogenesis cell cycles at stage VII of the seminiferous epithelium cell cycle in SD male rat (means ± SEM for 12 rats) a

Abbreviations: ASg: type A spermatogonia, pLSc: preleptotene spermatocytes, mPSc: mid-pachytene spermatocytes.
a The value of 22.425 for the effective 7 Sd degeneration was seen in CPF-treated animals at CPF 10 mg. The value of 3.93% for the effective degeneration of the step 7 spermatid (7 Sd) represents the recovery possibly due to the synthesis of antioxidant enzymes systems at CPF 30 mg-treated group.
b p > 0.05 as compared with the corresponding olive oil vehicles-injected animal group I (Control-COV), post hoc ANOVA tests were done.
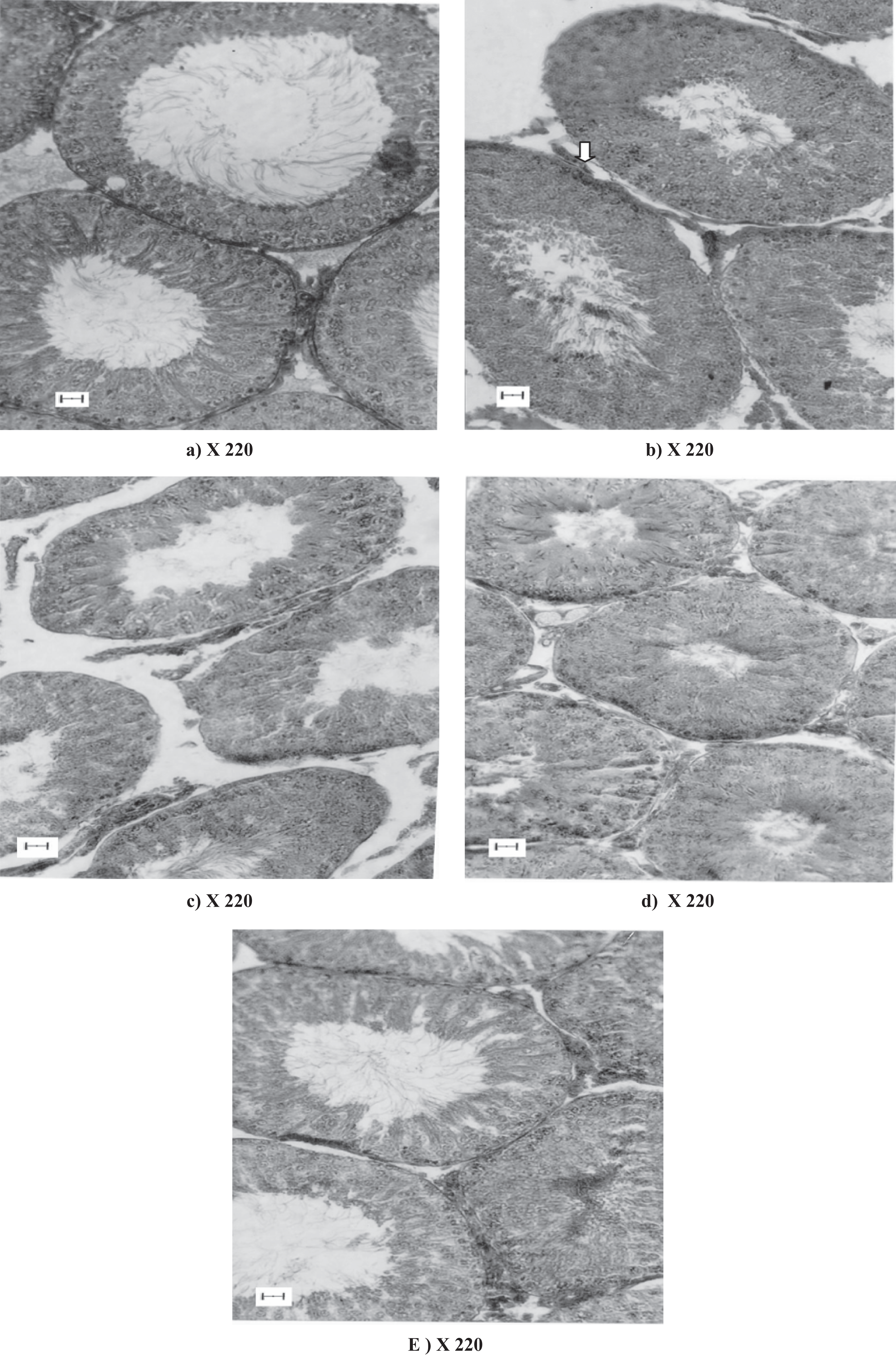
Histology also showed that at low doses of CPF treatment, there was a serious damage to the laminar basement membrane, shrinkage of the tubular diameter, degenerative changes in the germinal epithelium at low doses particularly at 10 mg/kg body weight (Table 1 and Figure 1). It was also noticed that a significant revival effect i.e. a sign of restoration function on the germ cell layers with regression of seminiferous tubules of testes (but not back to status of the normal testis) had been observed at 30mg /kg body weight of CPF treated rats (group V CPF-30; Table 1 and Figure 1). Quantitative study on spermatogenesis revealed that the detrimental effect of low doses, particularly at 10 mg of CPF treatment, was significantly maximum, but these effects were significantly less in the higher dose of CPF-30 mg (Table 1). But at higher doses of CPF treatment (groups IV and V), the detrimental effect of CPF was significantly less and gradual restoration effects on the germ cell layer were observed (Figure 1).
At low doses of CPF treatments (group II-CPF-5 and group III-CPF-10), results of hormones assays showed a noticeable reduction in serum testosterone concentration (Figure 2 ) in comparison to the control untreated group (group I-COV). But, at higher doses (group IV-CPF-20 and group V-CPF-30) of CPF treated animals, it showed a tendency of restoration and/or revival of serum testosterone level (Figure 2). There was a significant recovery effect, i.e. a sign of protective function is seen in group V-CPF-30 animals (Figure 2). The effects of CPF treatment on plasma concentrations of pituitary gonadotrophins such as FSH and LH are shown in Figure 2.

Effect of chlorpyrifos on serum concentrations of testosterone and pituitary gonadotropins (FSH and LH) in adult male rats after 30 days of treatment.
Steroidogenic enzymes (Δ5, 3β-HSD and Δ5, 17β-HSD) of testis and plasma levels of testosterone were significantly decreased in low-doses CPF-treated rats when compared to the control group (Figure 3 ), whereas CPF treatments at higher doses caused a tendency of restoration of the activities of theses enzymes almost to that of the control group (but not back to the normal) and lead to a significantly greater plasma level of testosterone than the control untreated group (Figure 2). Routine biochemical analysis of lipid peroxidation showed that at the lower doses (5 mg and 10 mg/kg body weight) of CPF treatments, significant increase of lipid peroxidation as measured by MDA value was noticed as compared to the control olive oil vehicle-treated animals (group I-COV). But this effect was lesser in higher doses of CPF (group IV-CPF-20 mg and group V-CPF-30 mg/kg body weight)-treated rats (Figure 4 ). There was a further decrease in the total lipid content of the testicular membranes significantly at low doses of CPF treatment, as compared to the control (group I-COV) male rats (Table 3 ). It had also been observed that the testicular protein contents might be deviated from this general trend: they showed a gradual increase with the amount of CPF treatment (Table 3).

Effect of chlorpyrifos (CPF) on testicular Δ5, 3β-HSD and 17β-HSD activities. Units of enzymes activity for each case is expressed as units X per mg of testicular tissue protein. Group I-COV denotes control olive oil-treated vehicle group, group-II-CPF 5 mg for chlorpyrifos treatment for 5 mg/kg body weight for 30 days, group III-CPF-10 mg depicting for chlorpyrifos treatment for 20 mg dose per kg body weight for 30 days, group IV-CPF-20 mg for chlorpyrifos at 20 mg dose per kg body weight for 30 days and lastly goup V-CPF-30 mg showing for the treatment of rats with 30 mg/kg body weight of chlorpyrifos for 30 days. Superscripts * denote means that are significantly different. Values are mean ± SEM of 12 rats in each group. *p < 0.05 as compared to the control oil vehicle.

Effect of chlorpyrifos (CPF) on malondialdehyde (MDA) value as index of testicular lipid peroxidation as measured by TBA-MDA assay and levels of the antioxidant enzymes activities(AOE) such as superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase; Post hoc ANOVA tests were done. Units of enzymes activity for each case is expressed as Units × 10-3 per mg of testicular tissue protein. Group I denotes control olive oil-treated vehicle group, group II-CPF-5 mg for chlorpyrifos treatment for 5 mg/kg body weight per day for total 30 days, Group III-CPF-10 mg depicting for chlorpyrifos treatment for 10 mg dose per kg body weight per day for total 30 days, group IV-CPF-20 mg for chlorpyrifos at 20 mg dose per kg body weight per day for total 30 days and lastly group V-CPF-30 mg showing for the treatment of rats with 30 mg/kg body weight of chlorpyrifos per day for total 30 days. Superscripts * denote means that are significantly different. Values are mean ± SEM, of 12 rats in each group.
Effect of chlorpyrifos (an estrogenic insecticide) on biochemical and physiological parameters of testes in SD rats (mean ± SEM for 12 rats)

a Significant at p < 0.05, when compared with vehicle control group-I, Post- hoc ANOVA tests and other multiple tests were done.
Biochemical analyses of antioxidant enzymes profiles showed that a marked decrease in antioxidant enzymes defense systems such as SOD, CAT and GSH-PX were noticed at low dose of CPF treatments (Figure 4). But all these antioxidant enzymes showed significantly higher activity at the higher doses in comparison to their activities at low doses of CPF (CPF-5 mg and CPF-10 mg) treatments (Figure 4). Statistical analyses indicated that all the data showed significant changes at significance level 0.05, on the basis of the studies performed with post hoc ANOVA tests. Table 3 also showed that glutathione content in the testicular tissues were gradually reduced after low dose of the treatment, but at higher doses of CPF treatments, the levels of glutathione content were slightly higher.
Discussion
Research reports clearly demonstrate the adverse effect of CPF pesticide at low doses and changes in oxidative stress at higher doses indicating the role of antioxidant enzymes defense systems in response to insult of low-dose treatment of the pesticide. Reduction in the weight of the testes is an accepted measure of testicular damage and reproductive toxicity. 3,8 Low doses of CPF-treated animals show a clear retardation in testicular growth, while for animals treated with higher doses of CPF, such a retarding trend has been partially arrested.
Histological studies show shrinkage of the seminiferous tubules and drastic damages at the laminar membranes at lower doses. But these effects are not so much pronounced at higher dose. Due to presence of inherent natural defense mechanisms, regression of the testes and recovery effects were seen in 30 mg of CPF-treated (group V-CPF-30) animals. Present observation of the degenerative changes in the testicular histology at lower doses is in congruence with previous studies on other organophosphorous insecticide/pesticide. 3,6
The decreased plasma testosterone and FSH and LH in low dose of CPF-treated rats corresponds to the observation that low dose of CPF treatment inhibits testicular steroidogenesis. 17 But these effects were lesser at higher doses and some sort of revival of the serum FSH and LH levels were noticed indicating that there had appeared a restoration and recovery effect on testis. However, these effects were lesser at higher doses showing a tendency of restoration of lipid content. Thus, it also indicated from the results that some physiological defense mechanisms were in operation at the higher doses in order to protect and combat the animals from the insult of CPF treatment and its toxicity on animals at low doses. It had also been observed that the testicular protein contents might be deviated from this general trend: they showed a gradual increase with the amount of CPF treatment. But it is presumed that FSH hormone, possibly due to feedback regulation, triggered at a particular threshold concentration of the insecticide treatment on the pituitary through hypophyseal − gonadal axis as is evident from our earlier research work on other organophosphorus insecticide/pesticide – quinalphos. 3,6,11 The exact mechanism through which CPF induces these alterations in testes is not clear.
Free radicals generated by pro-oxidant-like chemicals and other environmental xenobiotics toxicants, radiations etc. initiate lipid peroxidation of membrane-bound polyunsaturated acids of mammalian testes and other biomembranes, leading to impairment of the membrane structural and functional integrity and finally causing drastic degeneration of seminiferous tubules of testes and resulting in testicular toxicity testicular atrophy and tissue degeneration. 3,8,10,16–20,21 The testicular damage caused by the CPF is initiated by the formation of free radical as is evident from the depletion of total lipids and generation of lipid peroxide in the testicular tissues. The testicular damage caused by the CPF is initiated by the formation of free radical as is evident from the depletion of total lipids and generation of lipid peroxide in the testicular tissues. The observations indicate that the degree of these corrective measures by triggering the synthesis of antioxidant enzymes system (AOE) depend on the magnitude of stress. That is the reason for an enhanced damage at a low dose while the damages are reduced at a higher dose (Figure 5a). Simultaneously, the activities of the free radical scavenging enzymes at both doses are measured. It appears, therefore, that CPF treatment causes degenerative changes in the germ cells that inhibit androgen production. It has also been observed that an increase in testicular peroxidation causes a decrease in production of testicular androgens (testosterone) as it is evident from earlier research reports. 3,8,10 The spermatogenic inhibition in the low-dose treatment indicated in the present study may be the result of lowered plasma testosterone. Besides, the hormonal alteration, the spermatogenetic inhibition and detrimental effects on spermatogenesis may also be due to the formation of free radical products in the testicular tissues. 3,8,10,18 The post hoc ANOVA results also indicated that all these free-radical-scavenging enzymes significantly higher activities at high doses and might have a tendency to restore almost to the control untreated and in comparison to their activities than at low dose of treatment indicating some sort of repair mechanism might possibly be operated under the stress of pesticide treatment to the animals at higher dose in response to the insult at low doses. 3,8 The post-hoc ANOVA tests and best lines for the low and high doses and also for the control clearly depicts the correlation between increase of lipid peroxidation and the concomitant decrease of antioxidant enzymes at low dose, and the triggering of enhanced synthesis of antioxidant enzymes as compared to the low dose (Figure 5a). But it also revealed that SOD increased with the high dose of CPF treatment in response to the insult of low doses of CPF treatment. So it is depicted that some physiological defense mechanisms were in operation, resulting recovery of testicular tissues and extra synthesis of endogenous antioxidant enzymes defense systems of testes. Similarly, plotting CAT or GSH-PX levels against MDA leads to an identical situation (curves were not shown). Glutathione content in the testicular tissues were gradually reduced after low dose of the treatment, but at higher doses of CPF treatments, the levels of glutathione content were slightly higher, indicating some restoration effect which might be attenuated as protective defense function on testis in response to insult of CPF treatment to the animals' testicular oxidative stress and damages at lower doses. 3,6

a, Linear fit and least-squares lines describing linear regression and the correlation between testicular lipid peroxidation (as measured by MDA value) and the activity of the endogenous antioxidant enzymes system: superoxide dismutase enzyme (SOD value), statistical analysis shows the figure (a): for group I (COV), reliability (R) = −0.583, SD = 0.003 and p = 0.468 × 10−4; for group III (CPF-10 mg), R = 0.198, SD = 0.0215 and p = 0.951; for group V (CPF-30 mg) R = −0.377, SD = 0.0156, and p = 9.1235 × 10−4. Respective symbols denote for: (▪) group I (COV), the control sesame oil vehicle treated; (•), group III (CPF-10 mg), is chlorpyrifos at 10 mg/kg. body weight and (▴) group V (CPF-30 mg), chlorpyrifos treatment at 30 mg/kg body weight. Values are mean ± SEM of 12 rats in each group. * p < 0.05 as compared to the control oil vehicle-treated animals (group I-COV) group. Statistically, to accommodate the data of different orders in x axis and to illustrate the variations of superoxide dismutase activity (SOD) at various doses in a more distinct and understandable way, we had plotted the data using both top-x and right-y axes. To make it clear differentiation of effects of individual treatment, we had required to put arrow head marking pointer that was indicated for x and y axes with respect to the particular set of data so as to illustrate the situation in (a). b, Linear fit and least squares lines showing the correlation between the number of Sertoli cells/seminiferous tubules and the glutathione content of the seminiferous tubules of rat testis. Statistical analysis shows for (b): for group I (COV), reliability (R) = .24486, SD = 1.89115 and p = 4.4307 × 10–4 for group III (CPF-10 mg), R = 0.43316, SD = 8.6511 and p = 1.5955 × 10−4; for group V (CPF-30 mg) R = −0.37563, SD = 6.27301 and p = 2.5493 × 10−4; respective symbols denote for: (▪) group I (COV), the control olive oil vehicle-treated; (•), group III (CPF-10 mg), is chlorpyrifos at 10 mg/kg body weight; and (▴) group V (CPF-30 mg), chlorpyrifos treatment at 30 mg/kg body weight. Values are mean ± SEM, of 12 rats in each group. * p < 0.05 as compared to the control olive oil vehicle-treated animals (group I-COV) group. Statistically, to accommodate the data of different orders in x- axis and to illustrate the variations of glutathione content (GSH) in respect of numbers of Sertoli cells at various doses in a more distinct and understandable way, we had plotted the data using both top-x and right-y axes. To make it clear differentiation of effects of individual treatment, we had required to put arrow head marking pointer that was indicated for x and y axes with respect to the particular set of data so as to illustrate the situation in (b).
Histological studies of the insecticide-treated testicular tissues show the appearance of a large number of Sertoli cells in the seminiferous tubules at low-dose treatment, and on the contrary, the numbers of Sertoli cells are reduced in high-dose treatment. 3,16 The linear-fit and best-fit lines for the low and high doses in comparison to the control-untreated (group I-COV) group also show that the reduction in the number of the Sertoli cells after treatment with a higher dose of CPF can easily be correlated with the increase in the glutathione peroxidative enzyme activity (as shown in Figure 5b). It thus indicates that there is a positive correlation between the presence of Sertoli cells and the level of glutathione content (Table 3 and Figure 5b) as is evident from earlier reports. 3,8
The decrease in testicular Δ5, 3β-HSD and Δ5, 17β-HSD enzymes activities in low dose of CPF-treated rats may be the result of a decrease in gonadotropins or an elevation of testicular conjugates and MDA, as the enzymes activities are reduced in the presence of these free radical products. 17 It seems that total protein content increases monotonically with the dose of the insecticide. It is surprising to note that even though some proteins which are damaged by the insecticide, may be extra-synthesized or overcompensated for by the induced production of antioxidants and other enzymes at the higher doses as a part of physiological defense mechanisms.
From this study, it can be concluded that treatment to chlorpyrifos pesticide causes the reproductive toxicity in male rat and the effect is explored as the oxidative damage of testicular lipids and lipid peroxidation of testes, which is pronounced at low dose than at higher dose because the free radicals produced by the action of the insecticide are more effectively scavenged by the antioxidant defense system when subjected to a higher stress under the influence of CPF treatment. The present investigation shows that CPF has a dual effect at both doses on oxidative stress and changes in lipid peroxidation and role of antioxidant enzymes defense systems of testes.
Footnotes
Acknowledgements
The authors are grateful to AK Chatterjee of the Department of Physiology, University of Sciences and Technology, Kolkata, India, for providing animal-based physiological research materials, instrumental facilities and his helpful discussion, and to the Director, Department of Public Health Service, National hormones and Pituitary Program of National Institute of Diabetes and Digestive and Kidney Diseases (NIADDK), Bethesda, Baltimore, USA, for supplying the pituitary hormones kits as generous gifts.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
