Abstract
The present study evaluated the efficacy of melatonin in cyclophosphamide (CP)-induced testicular injury, lipid peroxidative damage, and antioxidant enzymes status of the mice testis on the basis of biochemical and histological studies. Mice were pretreated with four different doses of melatonin (2.5, 5, 10, and, 20 mg/kg by body weight (b.w.)) via intraperitoneal injection for five consecutive days followed by injection with CP (200 mg/kg b.w.) 1 h after the last injection of melatonin on the 5th day. After 24 h, mice were euthanized, testes were immediately removed, and biochemical and histological studies were conducted. Treatment with melatonin significantly mitigates lipid peroxidation, superoxide dismutase, and catalase activity and the level of reduced glutathione content abnormality induced by CP in mice testis. Histological examination clearly demonstrates that pretreatment of melatonin prevented CP-induced spermatogenesis toxicity and spermatogenic cells reduction in mice testis. The protective effect of melatonin is likely due to the antioxidative properties of the indolamine existed in the chemical structure. Because melatonin is a safe, natural compound, it could be used concomitantly as a supplement to protect people undergoing chemotherapy against reproductive toxicity.
Introduction
Cyclophosphamide (CP, N,N-bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxazaphosphorin-2-amine 2-oxide) is a widely used chemotherapeutic alkylating agent with antitumor and immunosuppressant properties. 1 CP undergoes bioactivation by the hepatic microsomal cytochrome P450 mixed function oxidase system to active metabolites that enter the circulatory system. 2 The principal alkylating metabolite, phosphoramide mustard, is responsible for the therapeutic activity. It may also invoke a wide range of adverse effects including testicular toxicity in humans and animals. 3 The cytotoxic effects of CP and other chemotherapeutic drugs result in part from their interaction with DNA leading to defective DNA, abnormal cell function, and cell death. 4 However, CP is not specific for cancer cells and will affect all dividing cells, including those in the immune system, reproductive systems, and the gastrointestinal tract. CP interferes with the formation of eggs in the ovaries and the formation of sperm in the testicles and may cause infertility in both sexes. 5 Animal studies have revealed that the treatment of rats/mice with CP led to a decreased testicular weight, transitory oligospermia, decreased DNA synthesis in spermatogonia and protein synthesis in spermatids, as well as biochemical and histological alterations in the testis and epididymis. 6 –8 The mechanism by which CP causes testicular injury is unknown; however, numerous studies have shown that CP exposure can disrupt the redox balance of tissues to suggest that biochemical and physiological disturbances may result from oxidative stress. 9 –11 Human body has different methods of mitigating oxidative injuries, either by repairing injuries or by directly diminishing the occurrence of oxidative stress via enzymatic and/or nonenzymatic antioxidant systems. Nonenzymatic antioxidants such as natural compounds can also protect the body against oxidative stress via their antioxidant activities. Many reports demonstrate that natural compounds can reduce lipid peroxidation. 12 It is therefore important to continue the search for an effective model compound that will protect against CP-induced oxidative stress and reduce toxicity issues associated with chemotherapy. In the light of this, several chemopreventive strategies have been presented, which have beneficial impacts on the CP-experimental model of testicular injury. 13,14 These studies suggest that natural compounds with antioxidative properties may have the potential to ameliorate CP-mediated testicular injury. In the past decade, the bioactivities of natural compounds on human health have given rise to much attention, especially the antioxidant activity. 15 Biological compounds with antioxidant properties may protect cells and tissues from deleterious effects of reactive oxygen species (ROS) and other free radicals generated during CP exposure. 9,16
Melatonin (N-acetyl-5-methoxytryptamine; Figure 1) is synthesized mainly by the pineal gland and is suggested to have antioxidant and prophylactic properties.
17
Melatonin possesses strong antioxidant activity by which it protects cells, tissues, and organs from the oxidative damage caused by ROS, especially the hydroxyl radical (

Chemical structure of melatonin.
Since melatonin has excellent antioxidative properties, both direct and indirect mechanisms of antioxidant actions, there is a likely possibility that melatonin would protect against the toxicity of CP, that is, an elevated level of melatonin in body may act as a prophylactic against damage. The present study was designed to investigate the efficacy of melatonin in CP-induced testicular injury, lipid peroxidative damage, and antioxidant enzymes status of the mice testis on the basis of biochemical and histological studies. Lipid peroxidation was explored by determinations of malondialdehyde (MDA). The superoxide dismutase (SOD) and catalase (CAT) activity and the level of reduced glutathione (GSH) content were determined to reflect the activities of the antioxidant enzymes. Also, histological analysis in seminiferous tubuli of testis was also evaluated.
Materials and methods
Animals
The protocol for the study was approved by the Research Committee of Mazandaran University of Medical Sciences, Sari, Iran. Male Naval Medical Research Institute mice weighing 25 ± 3 g were purchased from the Pasteur Institute of Iran (Amol, Iran). The mice were kept in good condition at the university animal facility and were given standard food pellets and water ad libitum. All of the animals were maintained under a controlled 12 h light–dark cycle and temperature (23 ± 1°C). The ‘Care and Use of Laboratory Animals’ was followed by the guidelines provided by Mazandaran University of Medical Sciences.
Chemicals
CP (Endoxan ®) was obtained from Baxter Oncology GMBH (Westfalen, Germany). Melatonin and kits for measurement of biochemical parameters were purchased from Sigma Chemicals Co. (St Louis, Missouri, USA). 5,5-Dithiobis(2-nitrobenzoic acid), glacial acetic acid, nitro blue tetrazolium chloride, potassium dihydrogen phosphate, reduced GSH, sodium dihydrogen phosphate, trichloroacetic acid, thiobarbituric acid, and hydrogen peroxide (H2O2) were purchased from Sigma-Aldrich Chemical Co. Bovine serum albumin and a kit for protein measurement were purchased from Ziest Chem Co. (Tehran, Iran). All other chemicals were either at or purer than analytical grade.
Experimental treatment
Animals were divided into six groups (groups 1–6, n = 5 for each group), comprising the following: group 1 (negative control): mice that received distilled water (10 ml/kg body weight (b.w.)) via intraperitoneal (i.p.) injection for 5 days; group 2 (positive control): mice that received a single toxic dose of CP (200 mg/kg b.w, i.p.) in distilled water (10 ml/kg b.w); group 3: mice that were treated with melatonin (2.5 mg/kg b.w. by i.p. injection) in distilled water (10 ml/kg b.w) per day for 5 days followed by a single i.p. dose of CP 1 h after the last dose of melatonin; group 4: mice that were treated with melatonin (5 mg/kg b.w. by i.p. injection) in distilled water (10 ml/kg b.w.) per day for 5 days followed by a single i.p. dose of CP 1 h after the last dose of melatonin; group 5: mice that were treated with melatonin (10 mg/kg b.w. by i.p. injection) in distilled water (10 ml/kg b.w) per day for 5 days followed by a single i.p. dose of CP 1 h after the last dose of melatonin; and group 6: mice that were treated with melatonin (20 mg/kg b.w. by i.p. injection) in distilled water (10 ml/kg b.w.) per day for 5 days followed by a single i.p. dose of CP 1 h after the last dose of melatonin. Mice were pretreated with solutions of melatonin at four different doses (2.5, 5, 10, and 20 mg/kg b.w.) via i.p. injection for five consecutive days followed by injection of CP (200 mg/kg b.w.) on the 5th day 1 h after the last injection of melatonin. All animals were anesthetized with ketamine (60 mg/kg) and xylazine (5 mg/kg) and killed 24 h after the final dose of CP.
Preparation of tissue homogenate
Testes were immediately removed, cleared of adhering connective tissue, washed in 0.9% saline, blotted on filter paper, and weighed. A section was set aside for histological processing and immersed in 10% neutral formalin solution. The whole tissue was homogenized in 10 volumes of potassium dihydrogen phosphate buffer (100 mM, pH 7.4) containing 1 mM ethylene diamine tetra acetic acid and centrifuged at 16,000g for 30 min at 4°C. The clear supernatant was used for quantitative estimation of lipid peroxidation, SOD and CAT activity, and GSH levels.
Determination of protein content
The protein content was determined by the method described by Bradford using bovine serum albumin as the standard. 23
Biochemical analysis of tissue
Lipid peroxidation products
Lipid peroxidation in terms of thiobarbituric acid reactive substances (TBARS) formation was determined by the method of Esterbauer and Cheeseman. 24 Tissue homogenates (containing 1 mg protein) in the extraction buffer were incubated for 1 h in 37°C. The incubated reaction mixture was treated with trichloroacetic acid (1 ml, 20%) and thiobarbituric acid (2 ml, 0.67%) and was kept in a boiling water bath for 30 min. After cooling, the precipitate was removed by centrifugation at 1000g for 15 min. The amount of TBARS formed was measured by taking the absorbance of the supernatant at 532 nm. 25 Blank contained all the reagents except the tissue homogenates. Lipid peroxidation value was expressed as nanomolar MDA equivalent per gram of tissues.
Determination of SOD activity
SOD was examined according to the method of Misra and Fridovich. 26 Reaction mixtures contained sodium carbonate (1 ml, 50 mM), nitroblue tetrazolium (0.4 ml, 25 µm), and freshly prepared hydroxylamine hydrochloride (0.2 ml, 0.1 mM). The reaction mixtures were mixed by inversion followed by the addition of clear supernatant of tissue homogenates (0.1 ml, 1:10, w/v). The change in absorbance of samples was recorded at 560 nm.
Determination of CAT activity
The enzyme CAT converts H2O2 into oxygen and water. Briefly, tissue homogenates containing 5 µg of protein were mixed with H2O2 (2.1 ml, 7.5 mM) and a time scan was performed for 10 min at 240 nm. Temperature was 25°C. The disappearance of peroxide depending on the CAT activity was observed. One unit of CAT activity is defined as the amount of enzyme, which reduces 1 µmol of H2O2/min. 27
Determination of GSH activity
GSH level was determined according to the method of Ellman. 28 The tissue homogenate (720 µl) was double diluted and trichloroacetic acid (5%) was added to it to precipitate the protein content of the tissue homogenate. After centrifugation (at 11,112g, 5 min), the supernatant was taken, 5,5-dithiobis (2-nitrobenzoic acid) solution (Ellman’s reagent) was added to it, and the absorbance was measured at 417 nm. The GSH level of homogenate was calculated using a standard curve.
Histological analysis of testis
For light microscopic evaluation, the testes were fixed in 10% formalin. After processing, tissues were paraffin embedded and cut into 5 μm sections on a microtome and stained with hematoxylin and eosin method. Slides were viewed and photographed using a camera microscope (Olympus, Japan) at 40×, 100×, and 400× magnifications for the morphometric study. Random fields from each animal were selected for differential cell counts of seminiferous tubules. 29 For each sample, at least three equal and round seminiferous tubules were randomly selected and numbers of germinal cells were measured for each tubule. Tubules were selected at 40× magnification while stages were not considered and then measurements were carried out at 400× magnification. The mean number of spermatogenic cells such as spermatozoa, round spermatid, primary spermatocytes, and spermatogonia was counted in each tubule according to the shape of their nuclei and cytoplasm, as shown in the study by Bloom. 30
Statistical analysis
The data are presented as mean ± SD. Differences between group means were estimated using a one-way analysis of variance followed by the Tukey’s honestly significant difference test. A p < 0.05 was considered to be significant. All measurements were replicated three times.
Results
Lipid peroxidation levels of experimental groups are summarized in Figure 2. According to the results, single i.p. injection of CP significantly increases (p < 0.001) lipid peroxidation level of testis homogenates compared with the untreated control group. Five days pretreatment of 10 or 20 mg/kg of melatonin significantly reduces the levels of TBARS in testis that received CP (p < 0.001). Figure 3 shows SOD activities of the treated groups. CP reduces (p < 0.001) SOD activity of testis homogenates compared with the normal group. However, melatonin significantly increases the SOD activity compared with the CP group (p < 0.001). Also, Figure 4 shows the activity of CAT in testis homogenates of mice. The CAT activity in testis homogenates of CP-administered mice (67.64 ± 9.59 µmol/min/mg protein) is significantly (p < 0.001) lower than that of the normal group (184.08 ± 10.68 µmol/min/mg protein). In the testis homogenates of melatonin-treated mice, activities of CAT are significantly (p < 0.001) higher than those of the CP group (157.34 ± 6.05 µmol/min/mg protein for 10 mg/kg and 182.08 ± 9.13 µmol/min/mg protein for 20 mg/kg).
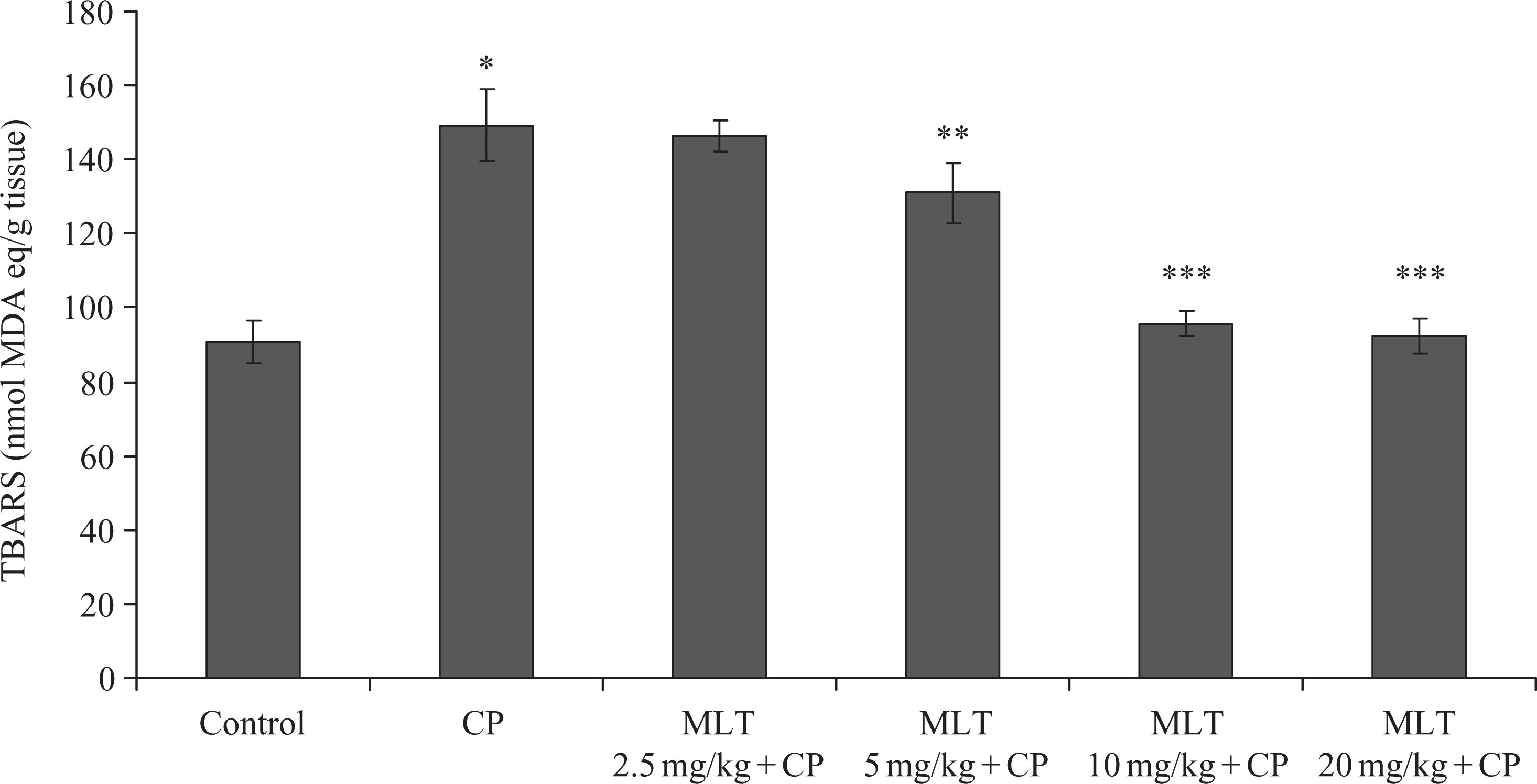
Effect of different concentrations of MLT on testis TBARS levels in CP-induced lipid peroxidation in mice. Data are expressed as mean ± SD (n = 5). *p < 0.001: control group compared to the similarly administrated mice with CP. **p < 0.01: CP group compared to MLT 5 mg + CP. ***p < 0.001: CP group compared to MLT 10 mg + CP or MLT 20 mg + CP. TBARS: thiobarbituric acid reactive substances; CP: cyclophosphamide; MLT: melatonin.

Effect of different concentrations of MLT on testis SOD activity in CP-induced SOD reduction in mice. Data are expressed as mean ± SD (n = 5). *p < 0.001: control group compared to the similarly administrated mice with CP. ** p < 0.01: CP group compared to MLT 5 mg + CP. ***p < 0.001: CP group compared to MLT 10 mg + CP or MLT 20 mg + CP. CP: cyclophosphamide; MLT: melatonin; SOD: superoxide dismutase.

Effect of different concentrations of MLT on testis CAT activity in CP-induced CAT reduction in mice. Data are expressed as mean ± SD (n = 5). *p < 0.001: control group compared to the similarly administrated mice with CP. **p < 0.01: CP group compared to MLT 5 mg + CP. ***p < 0.001: CP group compared to MLT 10 mg + CP or MLT 20 mg + CP. CP: cyclophosphamide; MLT: melatonin; CAT: catalase.
Figure 5 shows the GSH levels of treated groups. Administration of CP causes significant reduction in the GSH level in the testis homogenates of mice (6.12 ± 1.5 µg/mg protein). Melatonin treatment causes a significant increase in the level of GSH (10.62 ± 1.15 µg/mg protein for 10 mg/kg and 11.22 ± 2.61 µg/mg protein for 20 mg/kg) compared with the CP group.

Effect of different concentrations of MLT on testis GSH level in CP-induced GSH reduction in mice. Data are expressed as mean ± SD (n = 5). * p < 0.01: control group compared to the similarly administrated mice with CP. ** p < 0.01: CP group compared to MLT 10 mg + CP, or MLT 20 mg + CP. CP: cyclophosphamide; MLT: melatonin; GSH: glutathione.
A histological characteristic of the normal testis with different spermatogenic cells in seminiferous tubule is shown in Figure 6. Table 1 indicates a highly significant (p < 0.001) depletion in different spermatogenic cells of seminiferous tubules in CP-injected mice when compared with the control group. Pretreatment of 20 mg/kg of melatonin shows a significant mitigation in spermatogenic cells when compared with the CP group (p < 0.001).

Histological examination of the testis of normal group shows different spermatogenic cells in seminiferous tubules. Red arrow shows a spermatogonium, which is a 12-µm small cell close to the basal lamina. Yellow arrow shows a primary spermatocyte, derived from a spermatogonium, which is the biggest cell among spermatogenic cells. Green arrow shows a spermatid, which is the final form of spermatogenic cells formed as an early small round cell. It must undergo further maturational events to develop into spermatozoa. Blue arrow shows a spermatozoon, Plural spermatozoa, placed in the center of seminiferous tubules with a round or cylindrical nucleated cell, a short neck, and a thin motile tail, also called sperm cell.
Effects of melatonin on CP-induced reduction in different spermatogenic cells of seminiferous tubules in testis tissue of male mice.a

CP: cyclophosphamide; MLT: melatonin.
aThe data were analyzed with one-way analysis of variance and the Tukey honestly significant difference test and presented as mean ± SD.
b p < 0.001: control group compared to the similarly administrated mice with CP.
c p < 0.05: CP group compared to MLT 2.5 mg + CP, or MLT 5 mg + CP.
dNS: CP group compared to MLT 2.5 mg + CP.
e p < 0.001: CP group compared to MLT 5 mg + CP, MLT 10 mg + CP, or MLT 20 mg + CP.
fNS: control group compared to MLT 5 mg + CP, MLT 10 mg + CP, or MLT 20 mg + CP.
Discussion
In this study, to determine the toxic effect of CP and possible protective roles of melatonin in reproductive functions of healthy male mice, we examined the histopathological status of testis, lipid peroxidation level, and antioxidant enzyme activities of testis.
CP, a cytotoxic alkylating agent, is used extensively as an antineoplastic agent for the treatment of various cancers; however, its full clinical utility is limited because of several adverse effects including reproductive toxicity in humans and experimental animals. 6 The biochemical basis of CP toxicity is believed to be related to free radicals generated in these tissues. The cellular mechanisms by which CP causes testicular injury are poorly understood; however, numerous studies have shown that CP treatment is associated with induction of oxidative stress by the generation of free radicals and ROS. 9,10,31 Antioxidant substances can scavenge free radicals. The antioxidant defense system of human body includes various enzyme systems such as SOD, CAT, and antioxidant substances such as reduced GSH. 32 However, oxidative stress occurs when oxidative substances disturb the oxidant–antioxidant balance in human body and thus causing oxidative damages to deoxyribonucleic acid, proteins, and lipids. 33,34
The results of histological examination of the current study indicated that administration of a single dose of CP to the healthy male mice caused a highly significant (p < 0.001) reduction in different spermatogenic cells of seminiferous tubules such as spermatozoa, round spermatid, primary spermatocytes, and spermatogonia when compared with the control group. According to the previous studies, increased morphological defects and production of abnormal sperms may also be a result of the direct toxicity of CP, because cellular DNA is a primary target of CP in its antineoplastic and toxic activity. 35 Another mechanism for CP toxicity on sperm morphology may also be peroxidation of polyunsaturated fatty acids in plasma membranes of spermatozoa by free radicals. Many studies showed similar results to our study, CP caused histopathological reduction in size and number of the seminiferous tubules, degeneration and vacuolation in spermatogonia, spermatocytes and less number of germ cells, irregular seminiferous tubules, reduced seminiferous epithelial layers, significant maturation arrest, perivascular fibrosis, and hyalinization of intertubular tissue. 7,16,36 –38 There is a general agreement that male reproductive organs are particularly susceptible to the deleterious effects of ROS and lipid peroxidation, which ultimately lead to impaired fertility. Sperm parameters are also affected by various ROS, such as H2O2, superoxide anion (O2−), and/or hydroxyl free radical (•OH) . 39 Thus, it is concluded that ROS and lipid peroxidation may be involved in CP-induced testicular toxicities in mice. Previous studies showed that oral antioxidants treatment reduced the percentage of testicular toxicity. 40
The results of our study clearly demonstrate that pretreatment of melatonin prevented CP-induced spermatogenesis toxicity and spermatogenic cells reduction in mice (Figure 7). The protective effect of the pineal secretory product melatonin against oxidative damage has been examined in both in vivo 14,41,42 and in vitro studies. 43 –46 In a previous report, coadministration of melatonin resulted in significant protection against paraquat-induced alterations in lipid peroxidation levels and maintenance of GSH levels of the tissues. They concluded that the protective effect of melatonin was likely due to the antioxidative properties of the indolamine. 47 Melatonin scavenges a variety of ROS and nitrogen species including ·OH, H2O2, singlet oxygen, nitric oxide, and peroxynitrite anion. Based on the analyses of structure–activity relationships (Figure 1), the indole moiety of the melatonin molecule is the reactive center of interaction with oxidants due to its high resonance stability and very low activation energy barrier toward the free radical reactions. However, the methoxy and amide side chains also contribute significantly to melatonin’s antioxidant capacity. The N–C=O structure in the C3 amide side chain is the functional group. The carbonyl group in the structure of N–C=O is a key for melatonin to scavenge the second reactive species and the nitrogen in the N–C=O structure is necessary for melatonin to form the new five-membered ring after melatonin’s interaction with a reactive species. The mechanisms of melatonin’s interaction with reactive species probably involve donation of an electron to form the melatoninyl cation radical or through a radical addition at the site C3. Other possibilities include hydrogen donation from the nitrogen atom or substitution at position C2, C4, and C7 and nitrosation. Melatonin has also the ability to repair the damaged biomolecules as shown by the fact that it converts the guanosine radical to guanosine by electron transfer. Unlike the classical antioxidants, melatonin is devoid of prooxidative activity and all known intermediates generated by the interaction of melatonin with reactive species are also free radical scavengers. This phenomenon is defined as the free radical-scavenging cascade reaction of the melatonin family. Due to this cascade, one melatonin molecule has the potential to scavenge up to four or more reactive species. This makes melatonin very effective as an antioxidant. Under in vivo conditions, melatonin is often several times more potent than vitamins C and E in protecting tissues from oxidative injury when compared at an equivalent dosage (μmol/kg). 48
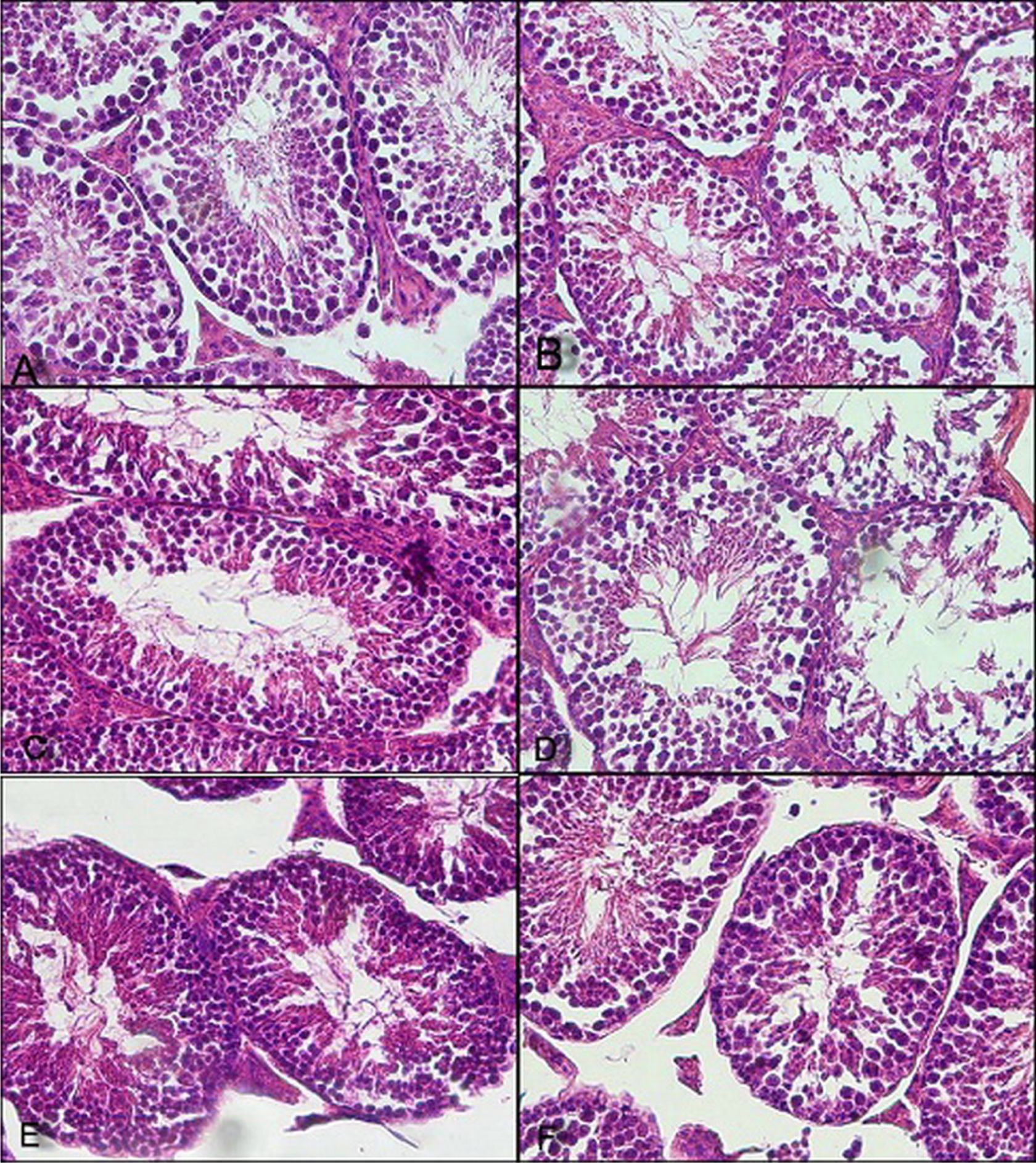
Pretreatment of MLT prevented CP-induced spermatogenesis toxicity and spermatogenic cells reduction in seminiferous tubules of mice testis. (a) Normal; (b) CP; (c) MLT 2.5 mg + CP; (d) MLT 5 mg + CP; (e) MLT 10 mg + CP; and (f) MLT 20 mg + CP. CP: cyclophosphamide; MLT: melatonin.
Melatonin receptors have been identified in the epididymis and low affinity melatonin-binding sites have been described on spermatozoa. 49 Hence, it is possible that melatonin influences sperm as they transit through the epididymis. Furthermore, the reported abnormalities in sperm seem to overcome by melatonin. 40 Unlike some other well-known antioxidants, melatonin is amphiphilic; allowing it to reduce free radical-mediated damage in both the lipid and aqueous subcellular compartments. Melatonin can also reduce nonenzymatic lipid peroxidation of rat testicular microsomes and mitochondria by changing arachidonic and docosapentaenoic acids. 50 The current study demonstrates that CP injection of animals induced testicular toxicity and oxidative stress that can be mitigated by pretreatment of melatonin before CP administration. Lipid peroxidation is the fundamental parameter of oxidative stress and MDA (or TBARS) is considered to be an index of lipid peroxidation. MDA is the breakdown product of the chain reactions of polyunsaturated fatty acid oxidation and serves as a marker of lipid peroxidation in tissues. 51 In this study, CP increased the TBARS levels and therefore the extent of lipid peroxidation in the testis tissue of the experimental animals. However, pretreatment of animals by melatonin for five consecutive days significantly reduced the TBARS levels and depleted lipid peroxidation in the testis tissue of mice.
Testicular toxicity may occur due to an increase in the intracellular levels of ROS, which are toxic at high levels and can interact with macromolecules. This study demonstrated that melatonin had potentially protective effects against the testicular toxicity induced by CP in male mice. Administration of melatonin for five consecutive days resulted in the inhibition of spermatogenic cells depletion caused by CP, as well as showed protective and antioxidative stress. Increase in the intracellular level of ROS, frequently referred to as oxidative stress, represents a potentially toxic insult, which interacts with macromolecules to induce cell membrane dysfunction and lipid peroxidation. The main characteristic of antioxidants is the ability to trap free radicals. It appears that the primary defense mechanisms of chemoprotective agents are to either directly or indirectly interact with these ROS. 52,53 In our study, melatonin had dose-dependent protective effects in reducing lipid peroxidation induced by CP in testis tissue. Administration of melatonin to mice for 5 days prior to injection of CP mitigates the antioxidant enzyme levels of SOD, CAT, and GSH in testis tissue.
In this study, melatonin is shown to reduce the testicular toxicity induced by CP in male mice through antioxidant activity and free radical-scavenging properties. With regard to the low toxicity related to its consumption and high level of antioxidant activity, melatonin is a good candidate to help defend the body against side effects, particularly reproductive toxicity, induced by hazardous chemical agents.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was approved and supported by a grant from the Student Research Committee, Mazandaran University of Medical Sciences, Sari, Iran.
