Abstract
Methotrexate (MTX) is a drug used in the treatment of various types of cancer and inflammatory diseases, but its clinical use has been restricted due to its toxicity. Apigenin (API) is an effective flavonoid with antioxidant and anti-inflammatory properties. The aim of this study was to determine the protective effect of API against MTX-induced liver and kidney toxicity. Four groups with 12 male mice each were used. The control and API groups were received 0.9% saline (ip) and API (3 mg/kg ip) for 4 days, respectively. The MTX group were given a single dose of MTX (20 mg/kg ip) on the fourth day. The MTX + API group were administered API for 7 days and then MTX on fourth day. Blood, liver and kidney were collected to evaluate tissue injury markers, oxidative stress biomarkers, and histopathological and immunohistochemical assessments. In MTX-treated group, significant increases in aminotransferases activities, creatinine and malondialdehyde (MDA) levels and significant decreases in catalase (CAT), glutathione peroxidase (GSH-Px) and superoxide dismutase1 (SOD1) activities and glutathione (GSH) levels were determined compared to the control group. Furthermore, histopathological changes and significant increases in caspase-3, C-reactive protein (CRP), granulocyte colony-stimulating factor (G-CSF), and inducible nitric oxide synthase (iNOS) expressions were detected in both liver and kidney tissues of MTX-treated mice. Pretreatment with API alleviates liver and kidney toxicity by attenuating oxidative stress and tissue injury markers, histopathological alterations, and apoptosis and inflammation. These results suggest that API has a protective effect against oxidative stress and liver-kidney toxicity induced by MTX.
Introduction
Methotrexate (MTX) is a compound known as a folate antimetabolite used for the treatment of various types of cancer (such as acute lymphoblastic leukemia, lymphoma, osteosarcoma, breast and testicular cancer), autoimmune diseases, rheumatoid arthritis, psoriasis and various inflammatory disorders.1,2 Although MTX is an effective drug, its clinical use is limited due to its toxicity in various organs. 3 The toxicity of MTX depends on the disease used, the treatment dose and duration. While high doses of this drug is associated with severe hepatotoxicity and acute renal failure, chronic use of MTX at low doses can cause progressive liver fibrosis, uremia, and hematuria. 4 It is metabolized by the microsomal enzyme system in the liver, and it has been emphasized that its metabolites formed as a result of the phase 1 reaction causing oxidative stress and various organ damages (especially in the liver, kidney, intestine). Reactive metabolites of MTX interact with cellular macromolecules (proteins, lipids and nucleic acids) leading to lipid peroxidation (LPO), DNA damage, disruption of protein synthesis and oxidative stress.1,5,6 Although the mechanisms of hepatotoxicity and nephrotoxicity of MTX have not been fully elucidated yet, experimental and clinical studies support the hypothesis that hepatotoxicity may be a result of oxidative stress.7–9 It is metabolized in the liver to its main metabolite, 7-hydroxy-MTX. In liver cells, this drug is stored in the form of polyglutamate. 10 As a result of prolonged exposure, the MTX-polyglutamate complex accumulates in hepatocytes, folic acid levels decrease and ultimately liver necrosis occurs. 11 In addition, cytosolic nicotinamide adenosine diphosphate (NADP) dependent dehydrogenase is inhibited by MTX. As an antioxidant, NADP is used by the glutathione reductase (GSH-R) enzyme to scavenge reactive oxygen species (ROS). Reduction of NADP in the environment due to MTX reduces glutathione (GSH) levels, which are responsible for protecting hepatocytes against damage from reactive oxygen molecules such as superoxide anions, hydroxyl radicals, hydrogen peroxide and hypochlorite radicals.8,9,12,13 It is thought that as a result of the accumulation of MTX and its metabolites in renal tubules, renal dysfunction occurs. More than 90% of methotrexate is excreted by the kidneys. High doses cause higher drug concentrations in plasma and urine, which may increase the risk of renal dysfunction. The reason for MTX-induced renal dysfunction is that it remains in high concentrations for a long time in plasma.14,15
In recent years, the use of various natural antioxidant substances as supportive treatment agents has become widespread, as their effectiveness has been clinically proven. Flavonoids constitute a significant portion of these natural antioxidant substances. These compounds have been the focus of interest for researchers because of their very low toxicity and easy availability.
16
Apigenin (API), which chemical structure is 4’, 5, 7-trihydroxyflavone, has hydroxyl groups at the C-5 and C-7 positions of the A ring and at the C-4’ position of the B ring (Figure 1). API is one of the most abundant flavones in various natural sources, including fruits and vegetables.
17

Chemical structure of apigenin (API).
Materials and methods
Chemicals
API, MTX, NADP and NADPH were purchased from Cayman, while SOD kit was obtained from Elabscience. glutathione reductase (GR), oxidized glutathione (GSSH), GSH and thiobarbituric acid (TBA) were obtained from Sigma. Trichloroacetic acid (TCA) was purchased from Merck and 1,2 dithio-bis-nitrobenzoic acid was obtained from Tokyo Chemical Industry (TCI).
Animals
Forty-eight male CD-1 mice, 10–12 weeks old age and weighing approximately 20–30 g, were obtained from Burdur Mehmet Akif Ersoy University Experimental Animal Production and Experimental Research Center. The mice were kept in an animal care facility under room temperature (21–23°C) and humidity (55–65%) with adjustable light/dark cycle for 12 hours. The animals were given water and feed
Experimental design
In this study, a total of four groups were formed, including control, API, MTX and MTX + API (12 mice per group). The animals were treated as follows: Control group: Mice received 0.9% saline (containing 0.1% DMSO) (1 mg/kg ip) for 4 days. API group: Mice were administered API (3 mg/kg ip) for 4 days (vehicle 0.1% DMSO).
24
MTX group: Mice were given a single dose of MTX (20 mg/kg ip) on the fourth day (vehicle 0.9% saline).
4
MTX + API group: Mice were treated API (3 mg/kg ip) for 7 days and MTX (20 mg/kg ip) on the fourth day.
The study was terminated on the eighth day. All animals were anesthetized with 2–3% isoflurane (applied via inhalation), and the blood samples were collected via cardiac puncture. While animals were under anesthesia, they were euthanized by cervical dislocation method. Liver and kidney samples were taken and divided into two parts: one part was homogenized for the evaluation of tissue antioxidant status. The other part was fixed in 10% formalin for histopathological and immunohistochemical assessments.
Determination of serum aminotransferases and creatinine levels
Anticoagulant-free blood samples were centrifuged at 4000 rpm for 10 minutes and their serum was separated. The activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT), levels of creatinine in the serums were measured by an autoanalyzer (Gesan chem 200 Gesan Production srl, Campobello, Italy). The AST and ALT activities, and creatinine levels were detected according to Kinetic UV optimized IFCC methods and Jaffè method, respectively.
Evaluation of blood and tissue lipid peroxidation and antioxidant enzymes
Blood samples collected into the K3EDTA-containing tubes were centrifuged for 5 minutes at 4000 rpm at 4°C. The plasma of blood samples was separated and stored at −20°C until analysis. The erythrocyte fraction was centrifuged at 4000 rpm for 5 minutes by adding three times of its volume of saline phosphate buffer solution (pH 7.4), and the supernatant was discarded. The same procedure was repeated three times. Then 1:1 volume of saline phosphate buffer solution was added to erythrocytes. The samples were stored at −20°C until analysis.
The liver and kidney tissues were washed primarily with 0.9% ice-cold isotonic saline. The tissues were homogenized (1/10 w/v) with phosphate buffer saline (140 mM KCl, 10 mM NaHCO3, 3 mM KH2PO4 and 2 mM K2HPO4; dissolved in 950 ml of deionized water and adjusted to pH 7.2 with 5 N NaOH and completed to 1000 ml) and then centrifuged at 15000 rpm for 45 minutes (4°C), and the supernatant was separated. The samples were stored at −20°C until analysis.
The supernatant was used to evaluate the levels GSH and malondialdehyde (MDA), and the activities of SOD, CAT and GSH-Px. For evaluation of the lipid peroxidation, the methods described by Ohkawa et al. 25 were used to measure the plasma, liver and kidney content of MDA. The activities CAT and GSH-Px and levels of reduced GSH in hemolysate and tissue homogenate samples were detected according to the methods described by Aebi, 26 Paglia and Valentine, 27 and Sedlak and Lindsay, 28 respectively. SOD activities in plasma and tissue homogenate samples were analyzed using a commercial kit (Elabscience Mouse Superoxide Dismutase 1, Soluble ELISA Kit E-EL-M2398). The total protein concentrations in the kidney and liver homogenates were measured according to the Biuret method reported by Gornall et al. 29
Histopathological examination
The tissue samples taken from necropsy were fixed in 10% buffered formaldehyde solution. The tissue samples were embedded in paraffin after a routine tissue processing procedure using a Leica ASP300S model tissue processor (Leica Microsystems, Wetzlar, Germany) after 2 days of formaldehyde fixation. After cooling the blocks for 4–5 hours, the tissue samples cut with a Leica 2155 rotary microtome (Leica Microsystems, Wetzlar, Germany) with a thickness of 5 µm and then stained with Hematoxylin-Eosin (HE), coverslip and examined under a light microscope.
Immunohistochemical examination
Streptavidin biotin complex peroxidase method was applied to sections according to instruction of the manufacturer. For this purpose, primary antibodies for caspase-3 [Anti-Caspase-3 antibody ab4051), 1/100 dilution], inducible nitric oxide synthase (iNOS) [Anti-iNOS antibody (ab15323), 1/100 dilution], C-reactive protein (CRP) [Anti-C-Reactive Protein antibody-Aminoterminal end (ab65842), 1/100 dilution] and granulocyte colony-stimulating factor (G-CSF) [Anti-G-CSF antibody (ab9691), 1/100 dilution] were used. EXPOSE Mouse and Rabbit Specific HRP/DAB Detection IHC kit (ab80436) was used as the secondary kit and 3,3-diaminobenzidine (DAB) (Abcam, Cambridge, UK) was used as the chromogen. Then counterstaining was applied by Harris hematoxylin. Microphotography and morphometric analysis were performed using the Database Manual Cell Sens Life Science Imaging Software System (Olympus Corporation, Tokyo, Japan).
Statistical analysis
All data were performed with the Statistical Package for Social Sciences (SPSS 22.0 for windows). The data were expressed as mean ± standard error of the mean (SEM). Data were initially analyzed by Shapiro-Wilk test for normality of data. SOD1 and GSH-Px values in the blood, liver and kidney tissues, MDA values in kidney tissues, CAT values in liver and kidney tissues, GSH and creatinine values in the blood showed normal distribution (
Results
Effects of API on liver and kidney function
It was found that there was significantly increased (

Effects of apigenin (API), methotrexate (MTX) or their combination (MTX + API) on liver and kidney function. All values are expressed as mean ± SEM (n = 12). *Significantly different from control group (**p < 0.001).
Effect of API on oxidative stress
It was determined that there was significantly decreased (

Effects of apigenin (API), methotrexate (MTX) or their combination (MTX + API) on oxidative stress biomarkers in the blood. All values are expressed as mean ± SEM (n = 12). *Significantly different from control group (*p < 0.001).
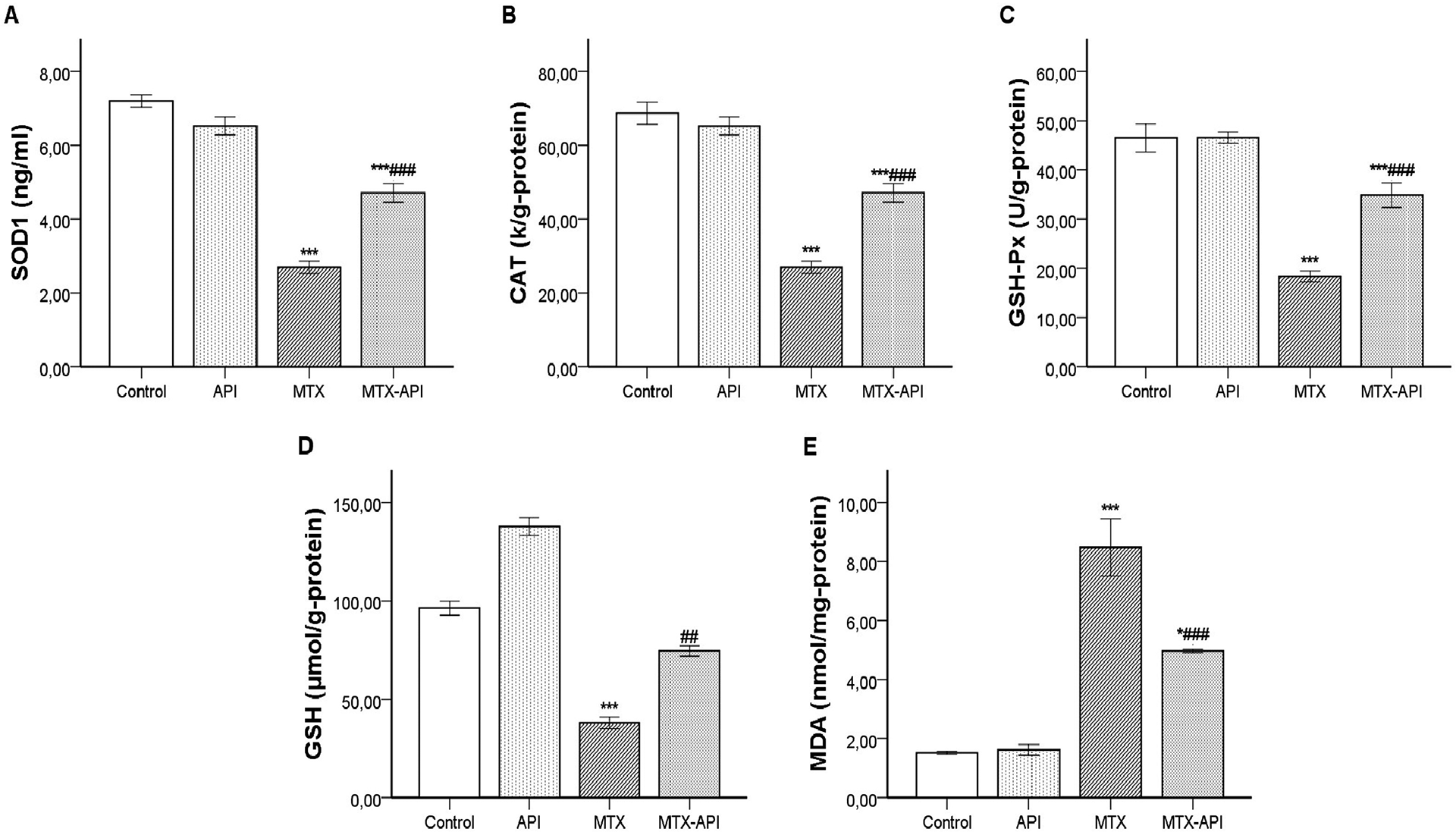
Effects of apigenin (API), methotrexate (MTX) or their combination (MTX + API) on oxidative stress biomarkers in the liver. All values are expressed as mean ± SEM (n = 12). *Significantly different from control group (*p < 0.05 and ***p < 0.001) and # MTX-treated group (##p < 0.01 and ###p < 0.001).

Effects of apigenin (API), methotrexate (MTX) or their combination (MTX + API) on oxidative stress biomarkers in the kidney. All values are expressed as mean ± SEM (n = 12). *Significantly different from control group (**p < 0.001).
It was found that there was a significant decrease (
It was found that there was a significant increase (
Histopathological findings
In histopathological examination, no pathological findings were found in liver and kidney tissues in API and control groups. Hyperemia, hemorrhage and lipidosis were observed in some hepatocytes in liver in the MTX groups. Severe hyperemia, small hemorrhagic areas and sclerosis in the glomeruli were noted in the kidney in MTX group. Both liver and kidney findings were ameliorated in the API + MTX group (Figures 6 and 7).

Histopathological appearance of the liver tissues. Normal tissue architecture in control, API and MTX-API group. Lipidosis (thick arrows) and hemorrhages (thin arrows) in the MTX group, HE. Immunohistochemical expression (arrows) of Caspase-3, CRP, G-CSF and iNOS in livers, Streptavidin biotin peroxidase method, Bars = 50µm (20× magnification).

Histopathological appearance of the kidney tissues. Normal tissue architecture in control, API and MTX-API group. Glomerulosclerosis (thick arrows) and hemorrhages (thin arrows) in the MTX group, HE. Immunohistochemical expression (arrows) of Caspase-3, CRP, G-CSF and iNOS in livers, Streptavidin biotin peroxidase method, Bars = 50µm (20× magnification).
Immunohistochemical findings
Increased caspase-3, CRP, G-CSF and iNOS expressions were observed in the liver and kidneys of animals treated by MTX. In the liver, the expressions were generally localized around the vena centralis. Tubular epithelial cells were commonly expressed the markers in MTX group in kidneys. API treatment decreased the expressions both liver and kidneys (Figures 6 and 7).
Discussion
The results of the present study demonstrate that API has a protective effect on MTX-induced hepato-renal toxicity in mice. This study determined that a single dose of 20 mg/kg MTX significantly increased aminotransferases activities and creatinine level, and changed oxidant/antioxidant balance of the tissues. Moreover, these findings were confirmed by the histopathological changes.
MTX increases ROS production and lipid peroxidation by suppressing the antioxidant defense system in tissues. 8 Lipid peroxidation is considered one of the main mechanisms of tissue damage caused by free radicals. MDA, which used as an indicator of cell membrane damage, is known as one of the secondary LPO products. 23 The SOD enzyme, which is the most important antioxidant enzyme system that form the first line of defense, catalyzes the decomposition of superoxide anion into H2O2 and molecular oxygen. 30 CAT and GSH-Px are enzymes responsible for the conversion of hydroperoxides including H2O2 to molecular oxygen and water. 31 GSH reacts with free radicals such as singlet oxygen, peroxy radicals, and is converted into GSSG and other disulfides. 1 This study demonstrated that administration of MTX caused significantly increased MDA levels, while SOD1, CAT and GSH-Px enzymes and the levels of GSH were significantly decreased in tissues of mice treated with MTX group (Figures 3–5). Results of this study revealed that MTX could induce oxidative stress in the blood, liver and kidney tissues. In previous studies, it was emphasized that MTX causes decreases in the efficiency of the antioxidant systems and increases lipid peroxidation.1,2,4,8,10,32,33 However, API showed protective effects by improving the antioxidants defense, inhibiting oxidative stress in tissues. It was determined that SOD1, CAT (excluding blood tissue) and GSH-Px activities, and GSH levels in tissues were significantly increased in MTX + API group compared to MTX group. In addition, it was found that MDA levels were found to be significantly decreased in API + MTX group compared to MTX alone (Figures 3–5 and 8). Previous studies indicate that API could have protective effect against oxidative damage induced by different agents. Similar to the current study, API increased GSH level and SOD, CAT and GSH-Px activities, and a decrease in MDA level in previous studies.30,34–40

The hypothetical pathway of the mechanism of apigenin (API) on methotrexate (MTX)-induced liver and kidney toxicity.
Significant changes in blood biochemical parameters are observed in organ failures and damages. Increases in serum ALT and AST enzyme levels are markers of liver damage, and increases in biochemical parameters such as creatinine and urea are markers of kidney damage. 1 In this study, similar to the previous studies,1–3,5,6,8,32,33 a significant increase was found in serum ALT and AST activities in the MTX group compared to the control group. In addition, this study revealed that MTX increases creatinine level, which is an indicator of kidney function. These findings are consistent with the previous studies.4,14,41 In this study, pretreatment of API significantly reduced in the ALT and AST enzyme and creatinine level compared the MTX group (Figures 2 and 8). Similar to this study, API was reported to reduce AST and ALT enzyme activities and creatinine levels to normal levels in the previous studies.21,23,24,39,42,43
At the histopathological examination, hyperemia, small hemorrhages and lipidosis in some hepatocytes were observed in the MTX group (Figure 6). In addition, severe hyperemia, hemorrhage and sclerosis in glomeruli were common findings in kidney tissues (Figure 7). Histopathological findings of previous
Inflammation plays an important role in the pathogenesis of MTX-induced toxicity. iNOS is proinflammatory biomarker 5 and CRP is acute inflammation biomarker. 41 In this study there were increased expression of iNOS and CRP in MTX-treated group compared to control group (Figures 6 and 7). These findings are in accordance with the previous investigations.5,41 In MTX-induced toxicity, mitochondrial permeability elevates as a result of increased oxidative stress, which may result in the release of cytochrome c into cytosol and expression of caspases. 45 In the present study, the expression of caspase-3 is significantly higher in MTX-treated group compared to control group which supports the previous research.5,8 G-CSF is a cytokine which mobilizes the stem cells from bone marrow. Similar to this study, MTX has been reported to significantly increase G-SCF expression in previous studies.46,47 In this study, it was determined that API decreased the expression of caspase-3, CRP, G-CSF and iNOS (Figures 6–8). In agreement with these finding Ahmad et al. 38 and Hassan et al. 24 reported that API could reduce the expression of caspase-3.
API’s ability to increase the cellular antioxidant defense system can be explained by several mechanisms. It destroys free radicals or stimulates the enzymatic antioxidant defense systems thanks to the OH groups in the 4´, 5 and 7 positions of the API structure.
48
Also, it shows antioxidant properties due to the double bond (C2=C3) between the carbon atoms (C2 and C3) of the C ring of API and the 4 oxo structures in the A and C rings (Figure 1).
49
Most of the
Conclusion
Findings of this study indicated that MTX causes severe biochemical, histopathological changes and oxidative damage by increasing MDA levels and decreasing GSH levels and antioxidant enzymes in the liver and kidney tissues. It was determined that MTX increased the expression of caspase-3, CRP, G-CSF and iNOS. Pretreatment of animals with API significantly attenuated MTX-induced liver and kidney toxicity by restoring the antioxidant defenses system (decreases MDA level and increases SOD, CAT and GSH-Px activities and GSH level) and downregulation in caspase-3, CRP, G-CSF and iNOS expressions. Finally, the results suggest that API has a protective effect against oxidative stress and liver-kidney toxicity induced by MTX.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Burdur Mehmet Akif Ersoy University (Project No: 0532-YL-18).
