Abstract
Nigeria is an African country where transmission of malaria occurs all year round and where most inhabitants use plants as remedies against parasitic diseases, including malaria. Some of such medicinal plants have their antimalarial efficacies already demonstrated experimentally, active compounds isolated and the mechanism of drug action suggested. Decoction of Cocos nucifera husk is used in the middle belt region of Nigeria as an antimalarial remedy. In our current studies, we tested extracts from husks of four varieties of C. nucifera, all collected in Brazil, where the plant fruit is popularly named ‘coco’. The husks of coco mestiço, amarelo, anão and gigante collected in the Northeast of Brazil were used to prepare extracts at the Chemistry Department, Federal University of Alagoas (UFAL), which were then tested for their antiplasmodial activities, cytotoxicities and hemolytic activities in vitro. Only the hexane extract of coco mestiço was active against the blood forms of Plasmodium falciparum human malaria parasite maintained in continuous culture. Most extracts presented selectivity indices of <10, while hexane extract of coco mestiço had a selectivity index of 35, meaning that the extract is not toxic. The isolation of the active compounds from coco mestiço husks has not yet been done.
Introduction
Nigeria is a West African country, which accounts for a quarter of all malaria cases in Africa, 1 mostly caused by Plasmodium falciparum, the leading cause of death worldwide in 2004 from a single infectious agent. 2 A nationwide surveillance data on drug efficacy in Nigeria showed that chloroquine and sulphadoxine-pyrimethamine are no longer viable therapeutic options for the effective treatment of human malaria. 3 Thus, the increased number of drug-resistant parasites coupled with expensive malaria treatment, resulting from the high cost of artemisinin used nowadays in combined therapies (ACT), have left the poor masses of Nigeria heavily reliant on traditional practitioners and medicinal plants for the treatment of the disease.
Some plants used popularly for malaria treatment in Nigeria have been scientifically authenticated and their active principles isolated, for example, Azadirachta indica, 4 Fagara zanthoxyloides 5 and Tithonia diversifolia. 6 However, most plants used for malaria treatment still need scientific documentation and authentication, for example, the case of Cocos nucifera Linn. (Palmae). The plant is indigenously used in the middle belt region of Nigeria and across continents for diverse medicinal purposes. Its husk fiber decoction is also used in northeastern Brazil in traditional medicine to treat diarrhea and arthritis. 7 In addition, the scanty oil which oozes out while burning coconut shell is used against ringworm infections in Indian popular medicine.
The decoction of
Materials and methods
Reagents
RPMI 1640 medium, sodium bicarbonate,
Collection of plants and preparation of extracts
Husks from four varieties of C. nucifera (mestiço, amarelo, anão and gigante) were collected at Maceió, Alagoas State, Northeast of Brazil (Figure 1). The plants were identified by Mrs R. P. de Lyra Lemos and samples were deposited at the herbarium Instituto do Meio Ambiente, Alagoas, Brazil. The husks were phytochemically processed in Maceió, Brazil, at the Federal University of Alagoas (UFAL), after being separated, dried and pulverized into powder. The hexane extracts were prepared by percolating 140 g of husk powder in 1 L hexane for 72 h twice. For aqueous extract preparation, 400 g of husk powder was percolated in 6 L of boiling water for 3 h. For ethanolic extract preparation, 400 g of husk powder was percolated in 6 L of ethanol for 72 h, twice. The hexane and ethanolic extracts were concentrated under pressure at 35°C using a rotary evaporator. The aqueous extracts were freeze-dried using a lyophilizer.

The species Cocos nucifera, variety mestiço, which grows extensively in the Northeastern States of Brazil (Photo by Dr Marilia OF Goulart, Maceio, AL, 2009).
In vitro antimalarial tests against malaria blood parasites
The in vitro tests were performed with blood parasites of P. falciparum W2 clone, which are chloroquine- resistant, kept in continuous culture at 37°C in human erythrocytes, using the candle jar method as described. 21 The antiplasmodial effects of the extracts were measured through inhibition of parasite growth, by the [3H]-hypoxanthine incorporation assay, as described 22 and modified. 23 For the test, a stock solution of each extract was diluted in complete culture medium without hypoxanthine (RPMI 1640 containing 10% human serum, 2% glutamine and 75% NaHCO3). Blood stage parasites in the ring form obtained in sorbitol-synchronized blood (180 μL/well) 24 were cultured in 96-well culture plates at 1% parasitemia and 1% hematocrit and then incubated with the extracts. Controls without extracts or with chloroquine, used as the reference antimalarial drug, were run in parallel. After a 24-h incubation period, 20 μL of medium containing [3H]-hypoxanthine (0.5 μCi/well) was added to each well, followed by incubation for 18 h at 37°C. The plates were frozen and thawed, and the cells were harvested [Tomtec 96-Harvester (Tomtec Inc., handem, CT, USA)] on pre-wet glass-fiber filters (Wallac Ou, Turku, Finland), which were placed in sample bags (Wallac) and immersed in scintillation fluid (Optiphase super mix, Wallac). Radioactive emission was counted using a 1450 Microbeta reader (Wallac).
The plant extracts were tested for their antiplasmodial activities against the erythrocytic forms of P. falciparum parasites (clone W2, chloroquine resistant) at doses of up to 50 μg/mL, tested in triplicates and half-maximal inhibitory concentrations (IC50) calculated. The inhibition of parasite growth was evaluated from the [3H]-hypoxanthine incorporation levels plotted to generate dose–response curves. The IC50 as compared to the drug-free controls were estimated using curve-fitting software (Microcal Origin Software 8.0, Inc.). Each experiment was repeated at least two times.
In vitro Cytotoxicity test using cultures of hepatoma cell line
Hep G2 A16 hepatoma cells were kept at 37°C in RPMI supplemented with 5% fetal calf serum (complete medium), in a 5% CO2 environment. Cells from confluent monolayers were trypsinized, washed, counted, resuspended in complete medium, distributed in 96-well microtiter plates (4 × 104 cells/well) and then incubated for another 18 h at 37°C. The extracts prepared as stock solutions in dimethyl sulfoxide (DMSO) were diluted in incomplete RPMI without fetal calf serum. After 24 h incubation at 37°C, 20 μL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (5 mg/mL in RPMI 1640 without phenol red) was added to each well. 25 After 4 h incubation at 37°C, the supernatant was removed and 200 μL of acidified isopropanol was added to each well. The culture plates were read using a spectrophotometer with a 570 nm filter and a background of 630 nm. The minimum lethal dose (MLD) that killed 50% of the cells was determined as reported 26 ; each assay was performed two times at least. Based on the values of cytotoxicity and antimalarial activity, the selectivity index (SI) of activity was calculated using the formula SI = MDL50/IC50.
Erythrocyte lysis assay
The hemolysis assay was performed as described 27 using washed human erythrocytes (200μL, 0.15 × 108 cells) in phosphate-buffered saline incubated with the extracts (with a range of concentrations of each extract) for 30 min at 37°C. Cells were pelleted and the absorbance of the supernatant read in a spectrophotometer at 415 nm was compared with that of a sample of cells lysed with SDS to determine the percentage hemolysis.
Results
Ten different extracts of C. nucifera (ethanolic and hexane extracts of the four varieties and the aqueous extracts of mestiço and amarelo) were tested in vitro. The hexane extract of coco mestiço showed a partial activity, with an IC50 of 10.6 µg/mL, an average of two independent experiments; all the other extracts were considered inactive at the doses of 50 µg/mL, the highest dose tested (Table 1).
In vitro activities of husk fiber extracts from four varieties of Cocos nucifera against Plasmodium falciparum blood-cultured parasites (clone W2, chloroquine resistant and mefloquine sensitive) and the cytotoxicities to the hepatoma cell line (HepG2)
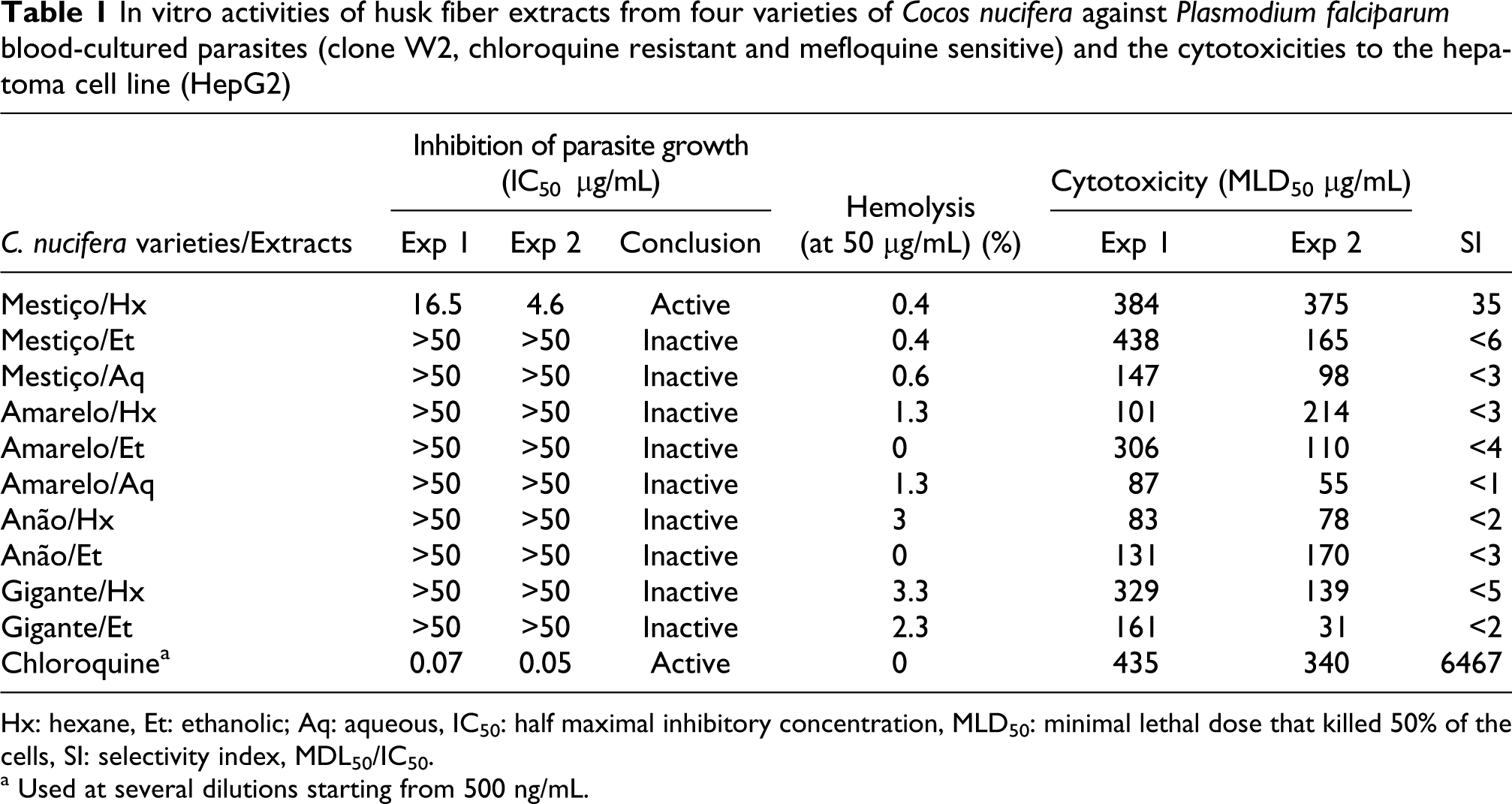
Hx: hexane, Et: ethanolic; Aq: aqueous, IC50: half maximal inhibitory concentration, MLD50: minimal lethal dose that killed 50% of the cells, SI: selectivity index, MDL50/IC50.
a Used at several dilutions starting from 500 ng/mL.
The in vitro cytotoxicity evaluated against a hepatoma cell line, Hep G2 A16, revealed that most extracts had low toxicity. The evaluation of the SI showed that only one extract was active, the mestiço hexane extract, with an SI = 35; the SI of chloroquine tested in parallel was 6,466 (Table 1). In addition, all mestiço extracts caused no significant red blood cell lysis, whereas the extracts of coco amarelo (hexane and aqueous), anão (hexane) and gigante (hexane and ethanol) caused some hemolysis (>1.3% at 50 µg/mL). Chloroquine and the ethanolic extracts of amarelo and anão caused no hemolysis
Discussion
The spread of multidrug-resistant malaria parasites and insecticide-resistant mosquitoes have led to major difficulties in the treatment and control of the disease. At present, the malaria control efforts include (i) attempts to seek for effective vaccines, a task performed by several groups including those in African countries28,29; (ii) eradication of mosquito vectors or its adequate control with indoor insecticides and use of bed nets in highly endemic areas of malaria transmission30,1 and (iii) the search for new antimalarials to treat the disease caused by multi-drug resistant parasites.30–32
The development of new antimalarial drugs based on medicinal plants has been the main goal of our group in Brazil, 32 and of other research laboratories worldwide, with a focus on ethnopharmacology, investigating the possible activities of plants used as remedies. We believe that plants proven to be active may represent good sources of new lead compounds. In addition, the specific activities of plants used in phytotherapy once confirmed may contribute to affordable treatments against malaria for the less privileged populations, who are most at risk of acquiring this devastating disease.
In this article, we studied a medicinal plant used in Nigeria, that is, the species C. nucifera, reported to be used for the treatment of human pathologies caused by virus, parasites, bacteria or inflammatory reactions. The evaluation of the in vitro antiplasmodial activities of the husk fibers of C. nucifera showed that only one of the four plant varieties tested, that is Coco mestiço, was active (Table 1). According to previous studies,32–34 the following are the criteria for extract activity: (i) extracts with IC50 ≤ 5 µg/mL are considered active; (ii) extracts with IC50 between 5 and 25 µg/mL are considered having a moderate or partial activity and (iii) extracts with IC50 ≥ 50 µg/mL are considered inactive. It is surprising that most extracts from C. nucifera, a medicinal plant largely used in Nigeria, were not active in vitro.
The fact that 50% of the extracts caused hemolysis suggests that the pharmacological activity described in the literature for extracts of other varieties of C. nucifera may not be selective enough against hemoparasites or other pathogens.7,10,11 The varieties that caused hemolysis are amarelo (hexane and aqueous), anão (hexane) and gigante (hexane and ethanolic), implying that they either act nonspecifically and should not be used as medicinal plants at all, or they have a nonspecific mechanism of erythrocyte binding. 27 Moreover, all the extracts had selectivity indices less than 10 (except mestiço hexane extract), suggesting that they are toxic and act via nonspecific mechanisms.
The mestiço hexane extract had antiplasmodial activity against P. falciparum in vitro, by a mechanism not clear, and whether this results from modifications of the red blood cell membrane is a matter for further studies. Our results showing a total absence of activity in most varieties of C. nucifera tested, in addition to their toxicities, including the hemolytic activities of some of the cocos varieties, suggest that the popular use of the species as a medicinal remedy should be limited to the correct ‘mestiço’ type.
Polyphenols, especially catechins, have been indicated as the main components of C. nucifera husk fiber and implicated in the biological activities of this plant material,7,10 but whether catechins are responsible for the antiplasmodial activity herein described, reflecting their different levels in the varieties of cocos plants, is yet to be elucidated. In conclusion, regardless of the mechanism underlying our findings about its confirmed activity against the malaria parasites, the active components of C. nucifera in the mestiço hexane extract may provide a new chemotherapeutic lead against malaria of worldwide significance.
Footnotes
Acknowledgements
The authors thank the technical help of Luisa G. Krettli, Renata C Paula and Lucas Felipe, with the parasites cultures and cytotoxicity tests, and also Dr Marília F. Goulart for the plant photo and helpful discussions.
This research received financial support from TWAS, CNPq and FIOCRUZ.
