Abstract
Currently, artemisinin-based combination therapy is considered the best option in the treatment of malaria. However, toxicity of artemisinins limits their use in pregnancy. In the absence of sufficient toxicity data, the World Health Organization recommends that artemisinins are not to be used in the first trimester of pregnancy and can be used only in second and third trimesters, when other treatments are not available. We have recently observed that drugs loaded in nanolipid carriers are selectively taken up in Plasmodium-infected erythrocytes with a concomitant reduction in the dose required to cure animals. Thus, 20% of the therapeutic dose of artemether–clindamycin (ARM-CP) loaded in nanostructured lipid carriers (NLCs; mean particle size 55 ± 10 nm) resulted in complete parasite clearance and 100% survival of infected mice. Here, we investigate the teratogenicity of this formulation in rodents (dosing on alternate days from 6th day to 18th day of gestation; 12-15 animals/group). The teratogenicity of drug-free NLCs and artesunate–clindamycin (ARS-CP) solution was also evaluated. We found that the therapeutic dose of ARS-CP caused fetal resorptions (87.5% resorptions in 8 litters), suggesting its unsuitability for use in pregnancy. Artesunate–clindamycin NLCs at therapeutic doses also resulted in ∼90% fetal resorptions in 10 litters examined. However, postimplantation losses or fetal malformations were not observed at the dose of ARM-CP NLCs that was required for complete parasite clearance in preclinical trials (ie, 20% of the therapeutic dose). Our data suggest that the NLCs loaded with 20% of the therapeutic dose of ARM-CP may have potential in treating malaria during pregnancy.
Introduction
Malaria during pregnancy is a different disease than that observed in the general population. Malaria presentation tends to be atypical and more severe in pregnancy. Women become more susceptible to Plasmodium falciparum malaria during pregnancy, and the risk of disease and death is high for both the mother and her fetus. 1 In low-transmission areas, women of all parities are at risk for severe syndromes such as cerebral malaria and maternal and fetal mortality. In high-transmission areas, women are most susceptible during their first pregnancy, and severe maternal anemia and low birth weight are the frequent sequelae that can account for an enormous loss of life. 2 In Africa, 30 million women living in malaria-endemic areas become pregnant each year. 3 For these women, malaria is a threat both to themselves and to their babies. Malarial infection in pregnant mothers accounts for up to 200,000 newborn deaths each year. Treatment options for malaria during pregnancy are limited owing to severe teratogenicity of most antimalarial drugs used. Thus, there is an obvious requirement for new innovative therapeutic approaches for treating malaria during pregnancy.
Artemisinin and its derivatives (artemether [ARM], artesunate [ARS], and arteether) are the only drugs of choice to treat malaria in pregnant mothers that are included in the World Health Organization (WHO) list of essential drugs for the treatment of severe multiresistant malaria. 4,5 Although intramuscular injections of artemether (ARM) are available, they are associated with low patient compliance. 6,7 Artemisinins have their own limitations for treating pregnancy-related malaria, as they have high dose-dependent toxicity and limited safety in pregnancy. The WHO guidelines (2003) recommend that artemisinin compounds should not be used for the treatment of malaria in the first trimester because of limited safety data and that the compounds should only be used in the second and third trimesters when other treatments are considered unsuitable.
The increased incidence of drug resistance has resulted in the discontinuation of monotherapy for malaria treatment. 8 The WHO now recommends artemisinin combination therapy (ACT), wherein artemisinin or its derivatives are used in combination with another antimalarial drug, to treat falciparum infection. This has prompted the formulation of ARM–lumefantrine (AL; Coartem; Novartis Pharma AG, Basel, Switzerland), the only fixed-dose combination antimalarial drug, 9 as well as ARM–mefloquine and dihydroartemisinin–piperaquine. Although AL is recommended as a first-line drug for malaria therapy in several countries, its complex treatment regimen of 2 daily doses for 3 days severely affects adherence resulting in patient noncompliance. 10,11
In view of these treatment options and related issues, we decided to fabricate a novel drug delivery system on the principles of ACT. The aim was to reduce the dose of artemisinin, thereby reducing dose-related adverse effects and ensuring safe and rapid control of malaria in pregnant mothers. We successfully developed nanolipid carriers (NLCs) loaded with ARM and clindamycin phosphate (CP) and evaluated their antimalarial efficacy. The ARM-CP NLCs efficiently cleared malaria in infected mice at 20% of the therapeutic dose of both the actives (ie, achieving 80% dose reduction) in preclinical studies. 12 We also demonstrated prolonged drug release and reduced dosing frequency, factors vital to patient compliance (Jain et al, “unpublished data”). In this article, we assessed the teratogenicity of the fabricated NLCs to confirm their suitability to treat malaria in pregnancy. Our results are encouraging as they establish the safety of 20% therapeutic dose of ARM and CP loaded in NLCs 11 in pregnancy in rodent model, thus indicating their potential suitability to treat pregnancy-related malaria.
Methods
Materials
Artemether and ARS were obtained as a gift sample from IPCA laboratories Pvt. Ltd (India). Clindamycin was obtained from Encube Ethicals (India). Capmul MCM and glyceryl dilaurate (GDL) were obtained as gift samples from Abitec (Janesville, Wisconsin) and ISP Technologies (India), respectively. Tween 80 was procured from S.D. Fine Chemicals (India).
Animals
The Institutional Animal Ethics Committee of National Institute for Research in Reproductive Health (NIRRH) approved the study protocol. The studies were executed in compliance with the guidelines of the Committee for the Purpose of Control and Supervision of Experimental Animals (India). Holtzman rats of both sexes weighing 150 to 200 g were used. The animals were bred in premises of NIRRH and were housed in polypropylene containers with maximum of 3 animals of the same sex per cage. The cages contained autoclaved corn cobs as bedding material (ATNT Laboratories, Mumbai). The material is certified to contain minimum permissible trace levels of pesticides and was replaced twice a week. The animals were maintained in controlled temperature of 22°C ± 1°C and relative humidity of 55% to 70% in a 14-hour light/10-hour dark cycle. The animals were provided with soy-free, in-house prepared pelleted feed and filtered drinking water, ad libitum.
Fabrication, Characterization, and Antimalarial Efficacy of ARM-CP NLCs
Fabrication of NLCs was done as reported elsewhere. 12 The NLCs comprised of Capmul MCM and GDL as lipids, stabilized by a mixture of Tween 80 and Solutol HS 15. Briefly, ARM was solubilized in molten lipid mix to yield concentration of 37.5% wt/wt in the lipid load. To this preconcentrate, aqueous solution (sterile water for injection) of 2.34% wt/vol CP was added and vortexed to generate NLC dispersion (500 μL) loaded with both drugs, and the resultant aqueous-based NLC nanocoformulation was characterized for mean particle size and stability as described. 12 Briefly, ARM-CP NLCs were analyzed for mean particle size and polydispersity index on Malvern Zetasizer, Nanoseries (United Kingdom) at temperature of 25°C and scattering angle of 173°. Zeta potential of ARM-CP NLCs was determined in Malvern Zetasizer, Nanoseries with equilibration time of 120 seconds at 25°C. For parenteral administration, ARM-CP NLCs were sterilized by autoclaving. The particle size/globule size of the formulations was determined before and after autoclaving using Malvern particle size analyzer. Drug content and entrapment efficiency were also evaluated. Dilution with suitable solvent(s) was followed by bath sonication for 10 minutes to ensure complete extraction of ARM or CP. 12 Blank (drug-free) NLCs were prepared similarly but without loading drugs. 12 The fabricated NLCs were sterilized by autoclaving before administration. The efficacy of the preparation was ascertained using modified Peter’s 4-day suppression test in Swiss mice. 12 The human dose of ARM and CP is given in Table 1. The mouse equivalent of the human dose for ARM is 19.6 mg/kg/d and that for CP is 112 mg/kg/d. Briefly, mice, infected by an intraperitoneal injection of 106 red blood cells (RBCs) infected with Plasmodium berghei ANKA on day 0, were injected intravenously with saline, drug-free NLCs, or ARM-CP NLCs (20% of parenteral therapeutic dose of ARM [ie, 3.9 mg/kg/d] + 5% of parenteral therapeutic dose of CP [ie, 5.6 mg/kg/d]), 2 hours later and on day 1 to 3 and monitored for parasitemia. Slides were counted blinded, and parasitemia was reported after counting 1,000 RBCs. Survival was monitored for >30 days. All the mice treated with ARM-CP NLCs cleared the parasitemia and survived. 12
Various Treatment Groups Evaluated in the Study.
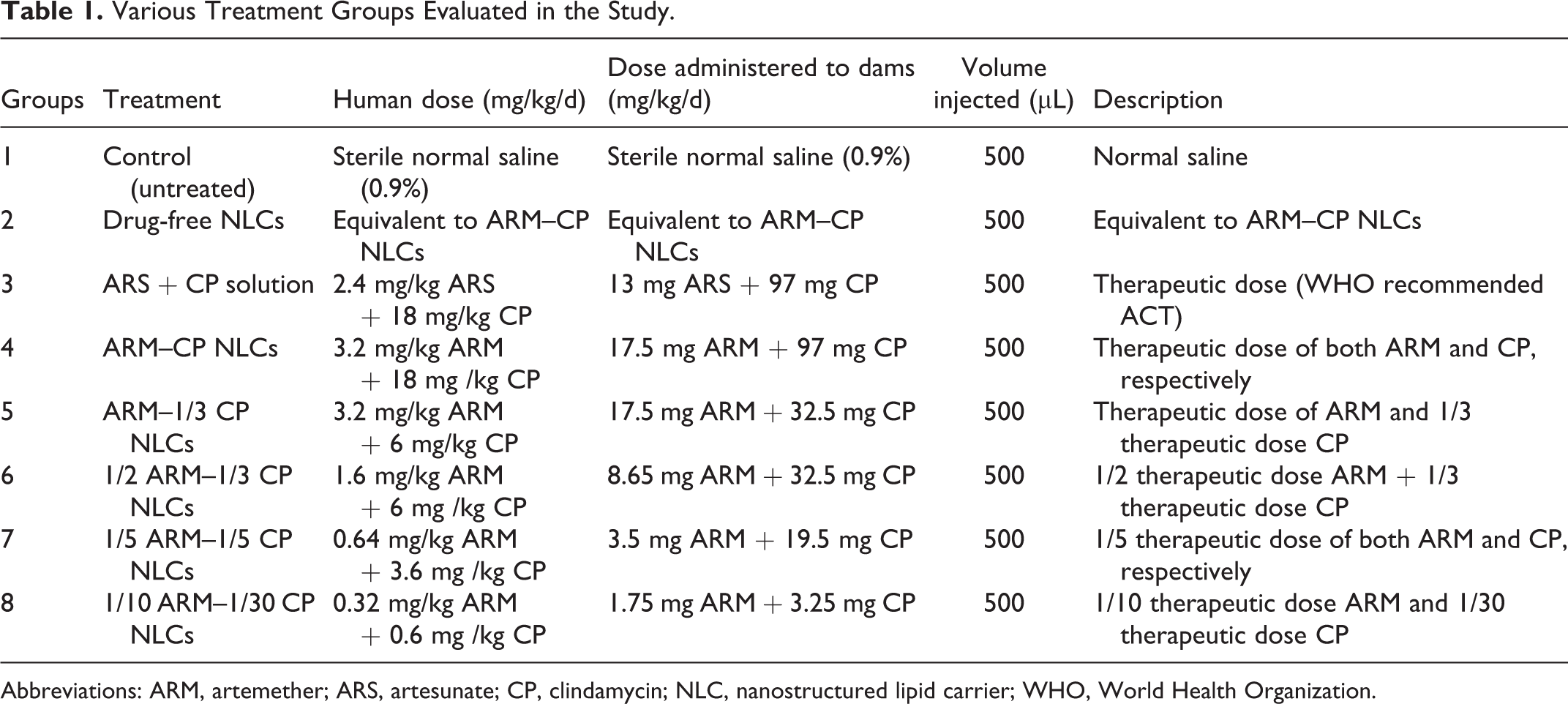
Abbreviations: ARM, artemether; ARS, artesunate; CP, clindamycin; NLC, nanostructured lipid carrier; WHO, World Health Organization.
Doses and Treatment
Given the success of ARM-CP NLCs in malaria treatment in preclinical studies (see above), we investigated them for teratogenicity. Adult male rats were cohabitated with females (1:2) overnight. Vaginal smears were collected daily early in the morning and monitored for the presence of sperm. Sperm detection in the vaginal smears established mating and was considered positive for pregnancy (day 0 of gestation). The mated females were separated and housed individually. The pregnant females (12-15 rats/group) were then divided into 8 groups and exposed to various treatment regimens by the intravenous route via tail vein as shown in Table 1. All animals were treated every alternate day throughout the period of organogenesis (from 6th day to 18th day of gestation; 6 total number of doses per animal per group) with the respective treatment regimens. 13,14 The cumulative dose of the drugs did not exceed the individual drugs’ cumulative LD50.
Maternal Observations
Animals were examined and monitored daily throughout the study for signs of toxicity, behavioral changes, vaginal bleeding, and mortality. Feed consumption was also noted. Body weights were recorded on day 0 as well as on the 6th, 9th, 12th, 15th, 18th, and 20th day of gestation.
Uterine and Litter Observations
Observations with respect to number of mated females, number of pregnant and nonpregnant females, and female fertility index (%) were recorded. All surviving dams were sacrificed 1 day before the expected day of parturition, that is, on gestation day 20, by carbon dioxide asphyxiation. Each dam was necropsied and subjected to gross pathological examination and evaluation of placenta. The uterine horns with ovaries and cervix were exteriorized via a midline abdominal incision, opened, and were further observed. Gravid uteri with ovaries and cervix were excised, weighed, and were further examined for number of implantation sites, resorbed and live fetuses per litter. The viability of the fetus was determined by spontaneous breathing and responses to tactile stimuli. Both right and left ovaries were subjected to counting of corpora lutea for each dam. 15
Evaluation of Gross Abnormalities
Fetuses from individual dams were separated from placenta, examined under magnification for gross external anomalies and malformations. 16 Fetuses were then individually weighed and sexed based on anogenital distance. The sex of individual fetus from each litter was also confirmed upon visceral examination. Biometry with respect to crown-rump (CR) length was calculated in cm. The fetuses from individual litter were divided into 2 parts—one part was used for visceral examination and the other for skeletal observation. The fetuses for visceral examination were left unfixed. After completion of visceral examination, the head of individual fetus was decapitated for razor-blade sectioning and evaluated for any anomalies. Head of each fetus was fixed in aceto–alcohol–formalin for fixation and decalcification. The heads were placed in individual labeled scintillation vials. At the time of evaluation, head was cut with razor blade in sequential manner to observe for head abnormalities, if any. The first cut was made with head turned nose upward to obtain a horizontal section beginning at mouth and coursing immediately inferior to the ears. This exposes the tongue, palate, upper lips, and lower jaw and was examined for correct closure of the palate. The flat surface produced by this cut enabled remaining cuts by stabilizing the head. The second cut was made about half way between tip of the nose and foremost corner of eye slits. The nasal passages, nasal conchae, nasal septum, palate are examined on either side of the cut. The third cut was made through the eyes; both eyes including cornea, lens, and retina were visible in cross section. The fourth cut was made well in front of the earflaps to examine the cerebral hemispheres, lateral, and third ventricles. 17
Evaluation of Skeletal Abnormalities
Fetuses were fixed in a 70% isopropyl alcohol solution, subsequently cleared and stained with alizarin red S, stored in storage solution, and subjected to analysis, as described by Staples and Schnell. 18 The stained fetuses were examined under Carl Zeiss, Germany, StereoZoom microscope. The skull was examined for size, shape, and degree of ossification of skull bones. Similarly, the vertebral centers, ribs, and sternal centers were also examined for size and shape and counted for the number of ossification centers. The cervical, thoracic, lumbar, sacral, and caudal vertebrae were counted and observed for centers and arches. Pelvic girdle, fore limbs, and hind limbs were examined for the number of bones and ossification of the bones. Any deviation from normal development and the type and/or number of bones affected were recorded for each fetus of each dam. 19
Statistical Analysis
Data on maternal weights, maternal weight gain, gravid uterus weights, resorptions, number of implantation sites per litter, number of corpus luteum, percentage postimplantation losses per litter, litters with live/dead fetuses, fetal body weights, and fertility indices were analyzed, and comparisons were done within control and treated groups using analysis of variance followed by Bonferroni test with multiple group comparisons. P value of <0.05 was considered significant.
Results
Fabrication, Characterization, and Antimalarial Efficacy of ARM-CP NLCs
We have previously successfully fabricated ARM-CP NLCs and established their antimalarial efficacy using modified Peter’s 4-day suppressive tests. 12 The mean particle size of ARM-CP NLCs was 55 ± 10 nm. Sterile water for injection was used throughout the study to generate NLC dispersion with the required drug concentrations. Artemether–CP NLCs were subjected to various storage conditions of temperature and humidity to assess their stability as per ICH guidelines Q1A (R2). They were found to be stable with respect to their drug content, particle size, and polydispersity index. 12 Our studies showed that treatment with ARM-CP NLCs at 20% of the therapeutic dose of ARM and 5% of the therapeutic dose of CP loaded in nanocarriers (3.5 mg/kg/d ARM + 5.6 mg/kg/d CP) resulted in complete parasite clearance in infected mice, 100% animal survival, and 100% antimalarial activity. 12 However, a higher concentration of the both actives (20% of the therapeutic dose of both drugs, ie, 3.9 mg/kg/d ARM + 22.4 mg/kg/d CP) was required in clinical simulation studies, wherein parasitemia in infected mice was allowed to reach 15% before the initiation of the 4-day treatment (manuscript in preparation). We therefore determined the teratogenicity of NLCs loaded with a range of ARM and CP concentrations in rats starting from the therapeutic dose (17.5 mg/kg/d ARM + 97 mg/kg/d CP) to NLCs loaded with 20% of therapeutic dose of both ARM and CP (3.5 mg/kg/d ARM + 19.5 mg/kg/d CP) and even at doses as low as 10% therapeutic dose of ARM and 3.33% therapeutic dose of CP loaded in NLCs (1.75 mg/kg/d ARM + 3.25 mg/kg/d CP), as indicated in Table 1. Since a marketed aqueous formulation of ARM-CP is not available, we used ARS + CP solution at the therapeutic dose (13 mg/kg/d ARS + 97 mg/kg/d CP) as control.
Maternal Observations
Maternal body weights are given in Figure 1. Maternal body weights of pregnant dams treated with ARS + CP solution (13 mg/kg/d ARS + 97 mg/kg/d CP), ARM-CP NLCs (17.5 mg/kg/d ARM + 97 mg/kg/d CP), ARM-1/3 CP NLCs (17.5 mg/kg/d ARM + 32.5 mg/kg/d CP), and 1/2 ARM–1/3 CP NLCs (8.65 mg/kg/d ARM + 32.5 mg/kg/d CP) were significantly reduced from day 12 to day 20 of gestation as compared to the control group. However, we did not observe any noticeable change in the feed consumption pattern in any of the experimental animals (data not shown). Thirteen of the 14 females in the untreated control group had fetuses (Table 2). This was significantly higher (P < .05) than the groups administered with therapeutic dose of ARS + CP solution (1 female with fetus per 8 pregnant females), therapeutic dose of ARM-CP NLCs (1 female with fetus per 10 pregnant females), ARM–1/3 CP NLCs (1 female with fetus per 9 pregnant females), and 1/2 ARM–1/3 CP NLCs (8 females with fetus per 10 pregnant females). By contrast, the number of females with fetuses in groups treated with 1/5 ARM–1/5 CP NLCs (ie, with 20% dose of both actives; the dose that had given complete parasite clearance in our clinical simulation efficacy studies), 1/10 ARM–1/30 CP NLCs, and drug-free NLCs were statistically indistinguishable from the control group (Table 2).
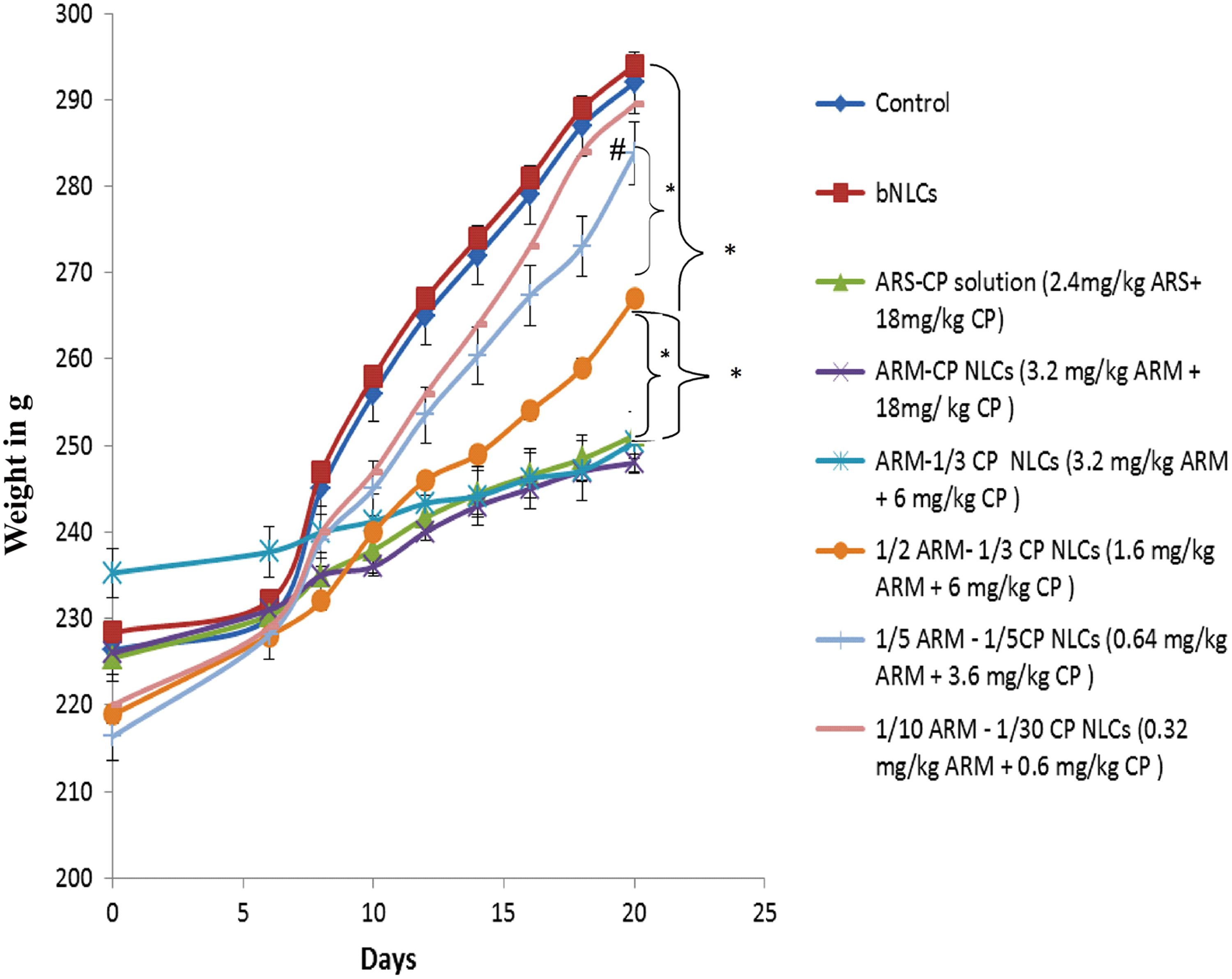
Body weight gain of dams throughout the gestation period. Maternal weight gain throughout the gestation period of animals treated with 0.64 mg/kg artemether (ARM) + 3.6 mg/kg (1/5 ARM–1/5 clindamycin [CP] nanostructured lipid carrier [NLCs]), 0.32 mg/kg ARM + 0.6 mg/kg CP (1/10 ARM–1/30 CP NLCs) was similar (statistically indistinguishable) to control group and group treated with blank NLCs (bNLCs, dose equivalent to highest dose of ARM–CP NLCs without drug load). Statistical significance at *P < 0.05 and # P < 0.01 was determined using 1-way analysis of variance (ANOVA) followed by Bonferroni test.
Evaluation of Maternal Parameters.
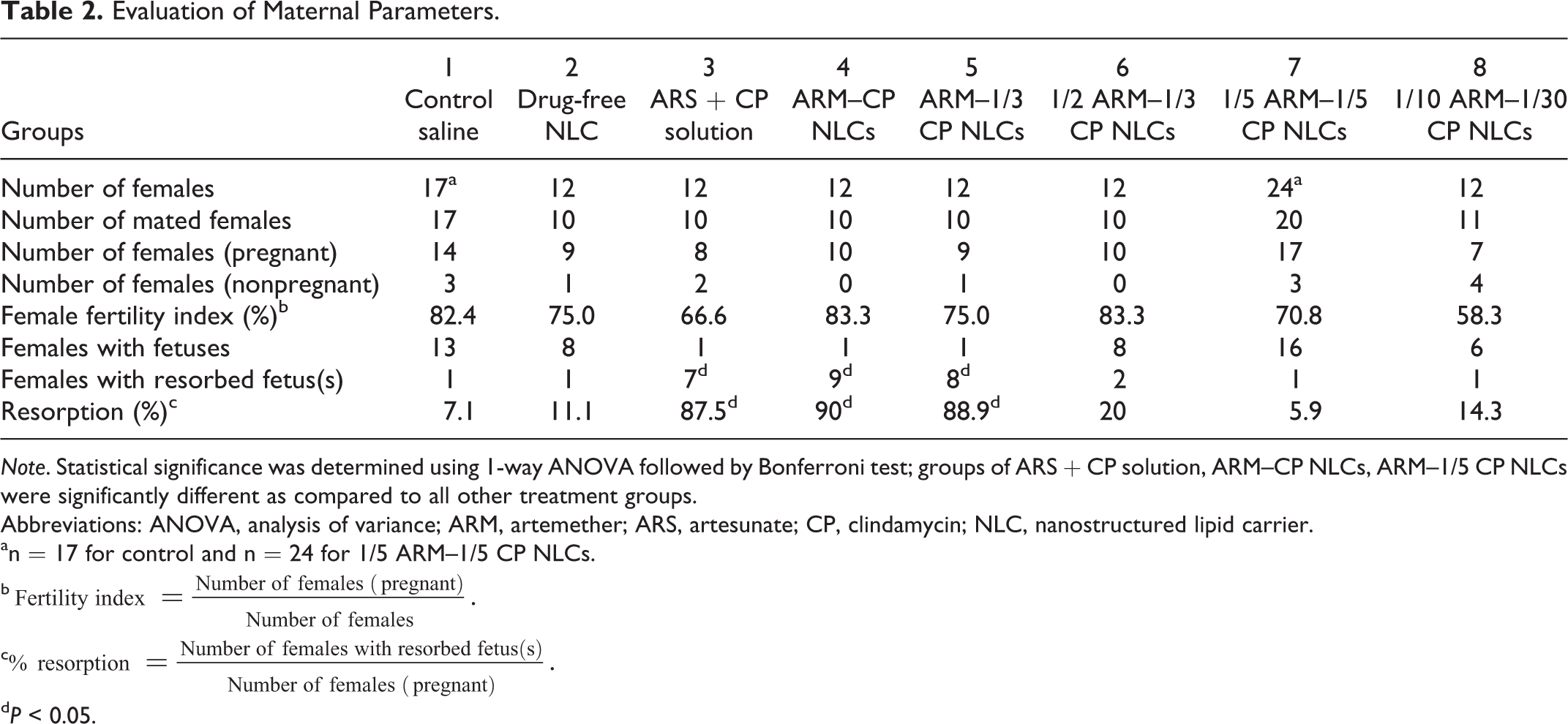
Note. Statistical significance was determined using 1-way ANOVA followed by Bonferroni test; groups of ARS + CP solution, ARM–CP NLCs, ARM–1/5 CP NLCs were significantly different as compared to all other treatment groups.
Abbreviations: ANOVA, analysis of variance; ARM, artemether; ARS, artesunate; CP, clindamycin; NLC, nanostructured lipid carrier.
an = 17 for control and n = 24 for 1/5 ARM–1/5 CP NLCs.
b
c
d P < 0.05.
Uterine and Litter Observations
Uterine observations and litter parameters of the pregnant females are presented in Table 3. A significant decrease in the number of live litters and increase in the number of resorbed fetuses were observed in the groups treated with ARS + CP solution (13 mg/kg/d ARS + 97 mg/kg/d CP), ARM–CP NLCs (17.5 mg/kg/d ARM + 97 mg/kg/d CP), ARM–1/3 CP NLCs (17.5 mg/kg/d ARM + 32.5 mg/kg/d CP), and 1/2 ARM–1/3 CP NLCs (8.65 mg/kg/d ARM + 32.5 mg/kg/d CP) as compared to untreated, drug-free NLC treated, 1/5 ARM–1/5 CP NLCs (3.5 mg/kg/d ARM + 19.5 mg/kg/d CP), and 1/10 ARM–1/30 CP NLCs (1.75 mg/kg/d ARM + 3.25 mg/kg/d CP) groups. The percentage of resorbed fetuses was significantly less in the groups treated with drug-free NLCs, 1/5 ARM–1/5 CP NLCs, and 1/10 ARM–1/30 CP NLCs as compared to all other groups in the study (Table 2). Indeed, the percentage resorption observed in groups treated with drug-free NLCs, 1/5 ARM–1/5 CP NLCs, and 1/10 ARM–1/30 CP NLCs was comparable to that observed in the untreated group. Analysis of litter parameters revealed nonteratogenic effects of 1/5 ARM–1/5 CP NLCs, and the mean litter size was comparable to the untreated control group. Thus, ARM and CP loaded in nanocarriers at concentrations used in this study have potential to be used during pregnancy, however, further investigations are needed to confirm this.
Uterine and Litter Parameters.
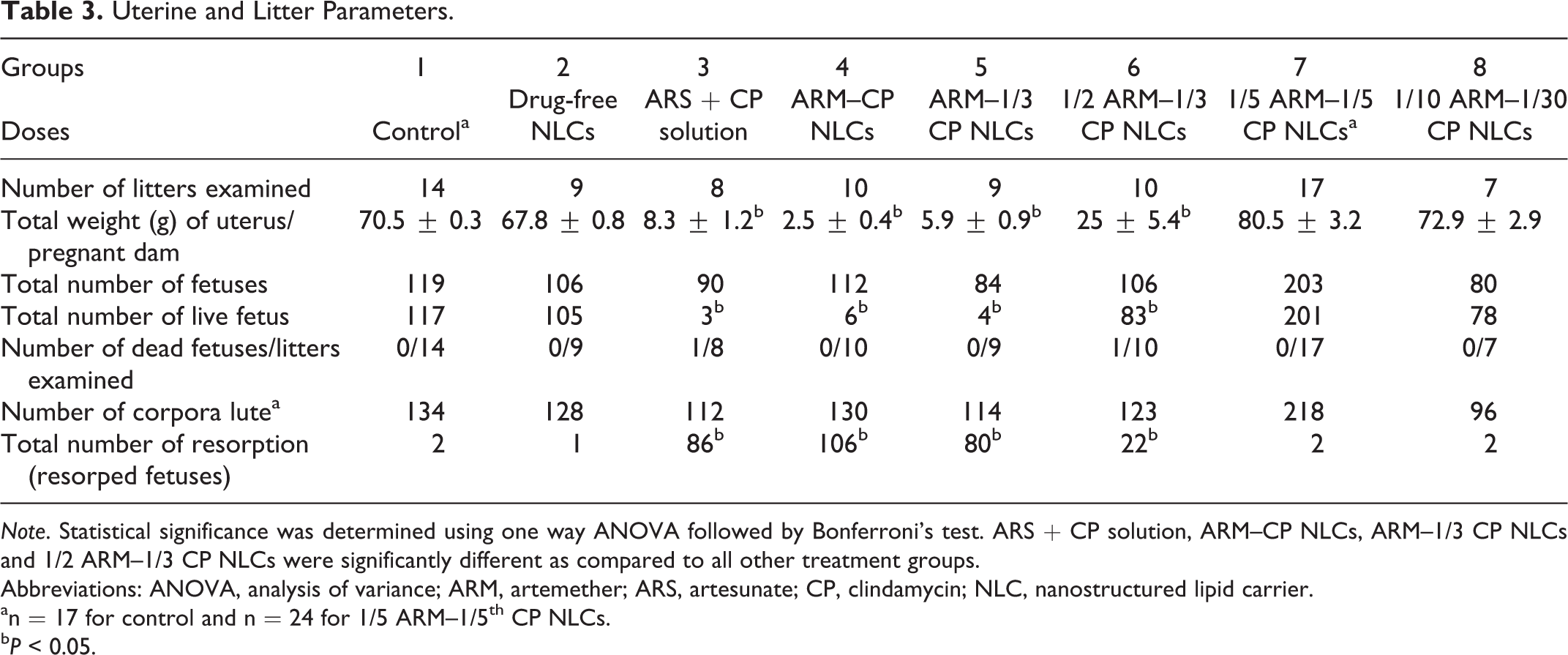
Note. Statistical significance was determined using one way ANOVA followed by Bonferroni’s test. ARS + CP solution, ARM–CP NLCs, ARM–1/3 CP NLCs and 1/2 ARM–1/3 CP NLCs were significantly different as compared to all other treatment groups.
Abbreviations: ANOVA, analysis of variance; ARM, artemether; ARS, artesunate; CP, clindamycin; NLC, nanostructured lipid carrier.
an = 17 for control and n = 24 for 1/5 ARM–1/5th CP NLCs.
b P < 0.05.
Evaluation of Gross Abnormalities
No major external gross abnormalities were detected in any of the litters during their scheduled necropsies, except that one tail-less fetus was observed in a total of 10 litters in the ARM–CP NLCs treatment group. One small-sized fetus was observed in a total of 9 litters in the ARM–1/3 CP NLCs treatment group and 3 in a total of 10 litters in the 1/2 ARM–1/3 CP NLCs treatment group. Significant differences were not observed in litter weights and CR lengths between the groups. External gross parameters are summarized in Table 4. No significant external malformations or variations were detected in control group or groups treated with drug-free NLCs, ARS–CP, 1/5 ARM–1/5 CP NLCs, and 1/10 ARM–1/30 CP NLCs. None of the fetuses from any group exhibited visceral abnormality.
Evaluation of Gross External Parameters.
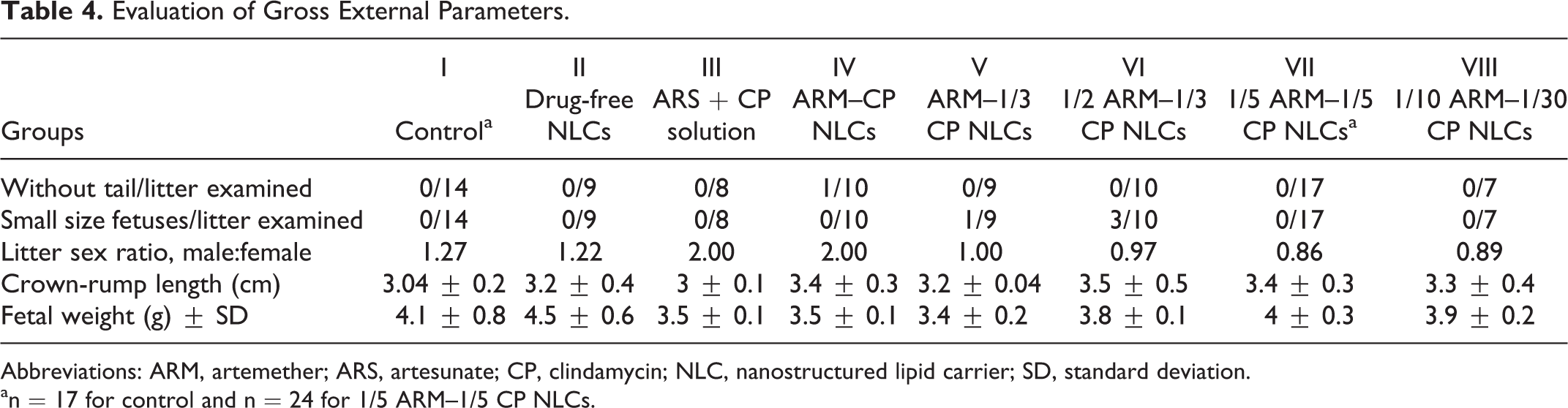
Abbreviations: ARM, artemether; ARS, artesunate; CP, clindamycin; NLC, nanostructured lipid carrier; SD, standard deviation.
an = 17 for control and n = 24 for 1/5 ARM–1/5 CP NLCs.
Skeletal Abnormalities
Fetuses exposed to ARM–CP NLCs at different doses and to ARM + CP solution were evaluated for skeletal anomalies, and the data are shown in Table 5. Fetuses exposed to therapeutic dose of ARM–CP NLCs, ARM–1/3 CP NLCs, 1/2 ARM–1/3 CP NLCs, and ARS + CP solution exhibited significant sternebrae alterations. They exhibited incomplete ossification and unossified sternebrae. Similar findings were also observed in the control group but at a lesser extent. The preliminary observations made in this study indicate that ARM-CP NLCs have potential as antimalarial drug to be used during pregnancy.
Skeletal Anomalies in Fetuses Exposed to Different Treatment Groups.a
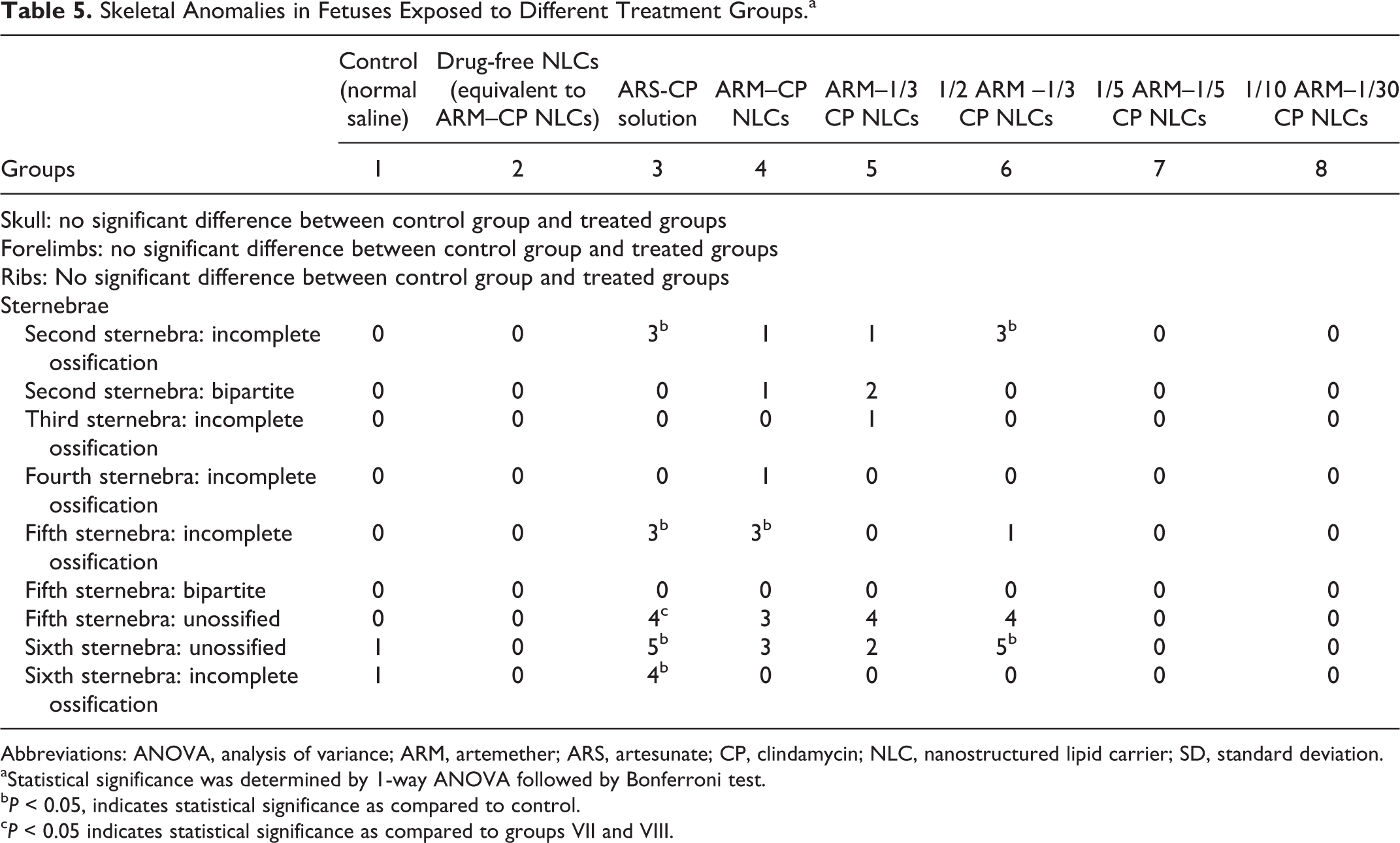
Abbreviations: ANOVA, analysis of variance; ARM, artemether; ARS, artesunate; CP, clindamycin; NLC, nanostructured lipid carrier; SD, standard deviation.
aStatistical significance was determined by 1-way ANOVA followed by Bonferroni test.
b P < 0.05, indicates statistical significance as compared to control.
c P < 0.05 indicates statistical significance as compared to groups VII and VIII.
Discussion and Conclusion
Although the WHO has recommended ACT for treatment of malaria in pregnancy, the combination antimalarial therapy has not been evaluated in detail for preclinical and clinical reproductive toxicity. Established ACT partner drugs include amodiaquine, lumefantrine, piperaquine, and antibiotics such as CP, but data on the use of these drugs in pregnancy and other special populations are limited. There is a growing evidence in favor of the use of artemisinins in pregnancy as artemisinins do appear to be safe in the second and third trimester, but data from animal studies indicate that there is a teratogenic and embryolethal effect in rats, guinea pigs, and rabbits at high doses or upon prolonged treatment. 20 It is still unclear whether this effect is reflected in humans, as there are some significant differences in human organogenesis as compared to rodent organogenesis. 20 Animal studies aimed at evaluating toxicity of artemisinins in pregnancy exhibited either dose-dependent embryotoxicity or teratogenic effects. 21 –23 Furthermore, clinical studies evaluating the safety of artemisinins also reported conflicting data regarding the first-trimester exposure but found no adverse effects in the second or third trimester. 19
Commercially available antimalarial drugs and drug combinations have been reported to have teratogenic effects. Thus, Coartem (AL) has been assigned to pregnancy category C by the Food and Drug Administration (FDA), 24 –26 since animal studies have revealed evidence of embryo–fetal loss and fetal defects. Although clinical trial data suggest that exposure to AL in pregnancy, including first trimester, is not associated with increased safety risks in terms of mortality, malformations, or developmental impairment, there is insufficient data to support its safety. 27 Although the WHO has recommended ACT to treat uncomplicated Plasmodium falciparum malaria during the second and third trimesters of pregnancy and quinine plus clindamycin during the first trimester, 28 there is an urgent need for further safety studies to determine the outcome of ACT use during pregnancy.
Artemether, a key part of ACT, is a challenging molecule because of several reasons. It has a short half-life, poor aqueous solubility, low bioavailability (∼40%), and acid instability. Our nanolipidic carrier system allows solubilization of ARM and allows it to be used for parenteral treatment. We have previously shown that this carrier system is taken up selectively in malaria-infected erythrocytes, thereby targeting the drug to the parasite. 12 Thus, ARM–CP NLCs were designed to combat uncomplicated and severe malaria and were effective at significantly low doses in the murine model. 12 We therefore expected ARM–CP NLCs treatment to have minimal side effects and dose-related toxicity. The data obtained from this study indicated that drug-free NLCs did not exhibit any teratogenic effect, and observations made throughout the gestation period were comparable to the saline control group, clearly indicating that the nanoformulation itself was not teratogenic and was suitable for use in pregnancy.
Maternal observations indicated that ARM–CP NLCs treatment, at doses shown to clear parasitemia, also did not cause noticeable reduction in the maternal weights throughout the gestation period. By contrast, therapeutic dose of ARM–CP NLCs and ARS–CP solution resulted in significant weight loss after day 12 of gestation. Xu et al have reported ARM-related teratogenic effects, viz, embryo absorption in mice and rabbits and abortion in guinea pigs and hamsters. 29 Our data with therapeutic doses of ARM–CP NLCs are in agreement with this. Reports show that all artemisinins cause the same pattern of teratogenicity, indicating that dihydroartemisinin, the common metabolite of all artemisinins, is the probable causal toxicant, likely because of its antiangiogenic properties. 30,31
On the other hand, clindamycin is a pregnancy category B drug (FDA guidelines) and is therefore supposed to be safe in pregnancy. 32 No major abnormalities in any of the live litters in all treatment groups was observed barring litters of small sizes (n = 1) and (n = 3) in ARM–CP NLCs (17.5 mg/kg/d ARM + 97 mg/kg/d CP) and 1/2 ARM–1/3 CP NLCs (8.35 mg/kg/d ARM + 32.5 mg/kg/d CP) groups, respectively, and 1 tail-less litter in the group treated with therapeutic dose of ARM–CP NLCs. Thus, the possibility of external malformations in fetuses during the gestation period cannot be denied even with NLCs loaded with therapeutic doses of ARM–CP. These observations thus provide evidence that the rat prenatal development may be sensitive mainly to ARM (pregnancy category C, FDA guidelines) rather than CP. Skeletal anomalies in fetuses exposed to therapeutic dose and higher doses of ARM–CP loaded in NLCs showed significant alterations in sternebrae. By contrast, 1/5 ARM–1/5 CP NLCs and groups treated with lower concentrations of both the actives did not show such alterations indicating dose-dependent safety of ARM–CP loaded in NLCs.
By interpretation of teratogenicity parameters, it is possible to infer that ARM–CP NLCs and doses of ARM–1/3 CP or 1/2 ARM–1/3 of CP interfere in the regular development of fetuses after implantation with significant increase in resorption rates as well as alterations in fetal growth. Surprisingly, therapeutic dose of the recommended ACT combination for malaria in pregnancy—ARS + CP—also demonstrated fetal resorptions. By contrast, administration of 1/5 ARM–1/5 CP NLCs was found to be a safe nanocombination dose exhibiting teratogenic safety with no embryotoxicity, postimplantation losses, and no gross, visceral, and skeletal alterations. Because the animal teratogenic study discussed here has shown a specific pattern of teratogenicity and fetal loss, this study raises concerns regarding safe use of artemisinins in humans. However, the mechanism of teratogenicity by which artemisinins can cause congenital malformations in rats may differ significantly from that observed in humans, and therefore, relying only on animal data may not be predictive of potential for risk to human fetuses in the first trimester. The embryo lethality and fetal losses seen with artemisinins is caused by an interruption in erythropoiesis, which is seen with short-course exposures. Although data are limited in humans, primates require longer duration of days of exposure to reach the same level of inhibition of erythropoiesis or angiogenesis. Therefore, short courses of treatment with artemisinins, as is currently prescribed for malaria in both children and adults, would be unlikely to result in pregnancy loss or congenital abnormality. 33 Nevertheless, reducing the doses of both the actives by 80% by loading them in NLCs could serve as an effective alternative to existing commercially available antimalarials for malaria treatment in pregnancy. However, further studies are required to substantiate the safety of the lower doses of ARM–CP NLCs.
Footnotes
Acknowledgments
S. A. Jain is thankful to UGC for funding formulation-based research activities. The authors are grateful to Dr S. Suryavanshi and NIRRH lab members for providing help with animal usage.
Author Contributions
S. Jain contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically the revised manuscript. M. Awale contributed to conception and design and drafted the manuscript. G. Vanage contributed to design, contributed to acquisition, and critically revised the manuscript. S. Pathak contributed to conception, contributed to analysis, and critically revised the manuscript. S. Sharma contributed to interpretation and critically revised the manuscript. V. Patravale contributed to conception and design, contributed to acquisition and analysis, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded through TIFR and NIRRH intramural funds. S. A. Jain was a recipient of UGC grant.
