Abstract
Introduction
Cachexia is often seen in end-stage cancer, 1 it accounts for up to 40–50% of cancer-associated deaths in gastrointestinal tract malignancies. 2 Cachexia is a convoluted metabolic disorder, including loss of adipose tissue due to lipolysis, loss of skeletal muscle mass, increase in reposing energy expenditure, anorexia, and reduction in oral food intake.3,4
Even though consistent studies have been conducted thus far in this field, the multifactorial pathological mechanism of cancer-associated cachexia has not been faithfully expounded, therefore conformable treatment modalities are not adequate. 5
It is not widely known that cytokine upregulation espouse results in involuntary weight loss, an influence of cancer-associated cachexia.6,7 Though the catabolism preeminently acts as intermediary by the effects of some cytokines, such as tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), interleukin 6 (IL-6), and interleukin 10 (IL-10),4,8 the mechanisms associated with cancer-cachexia are still not shown. 9
Recent studies reporting cachexia in various gastrointestinal system cancers reported that some proinflammatory cytokines, such as IL-8 and, cytokines such as vascular endothelial growth factor-A (VEGF-A) and its receptor (VEGFR-1), and midkine might be involved in the process of cachexia. 10 In addition, the proteins, such as cytokines and some hormones that import central mechanisms, are geared towards the regulation of the energy homeostasis. 11 These hormones are the adiponectin, ghrelin, and leptin.11,12
Adiponectin is a member of a group of proteins secreted from adipocytes. 13 There is significant inverse correlation between serum adiponectin levels and body weight. Thus, reduction in adiponectin levels is detected in obesity14,15 but high levels are found in anorexia nervosa 16 and at the time of weight loss. 17
There is significant association between decreased adiponectin levels and increased risk of breast, endometrial, and gastric cancers.18–20 The mechanisms responsible for the regulation of adiponectin levels have not been fully acknowledged. Eventually, recent data suggest adiponectin was downregulated by TNF-α as well as by insulin. 21
The hormone ghrelin is a 28 amino acid peptide, its third serine residue is esterified by n-octanoic acid. 22 The stomach is the master source of ghrelin.
The peptide increases the release growth hormone (GH) from the pituitary and hypothalamus. 23
Ghrelin is produced in the hypothalamic arcuate nucleus, where it stimulates the secretion of growth hormone from the anterior pituitary gland. Ghrelin also enhances the motility of gastrointestinal tract, as does motilin. Ghrelin also appears to promote gastrointestinal malignancy. 24
High ghrelin levels are related to multifarious cachectic juncture, such as anorexia nervosa and forceful heart failure. 25 Enhanced ghrelin levels were recently made evident in lung cancer-conduced cachexia, 26 and in male patients with primarily liable lung and prostate cancer. 27
Leptin is an member of the adipocytokine family. It is manufactured by the differentiation of adipocytes and affects the central nervous system to suppress food intake and stimulates energy expenditure. 12 Higher leptin serum levels are found in anorexia nervosa patients. 28
Association between acute-phase response proteins (APRPs) and weight loss in human cancer associated with cachexia has been showed exclusively in pancreatic carcinoma and in melanoma.29–31
Midkine is a heparin-binding growth factor found as the product of a retinoic acid-responsive gene. 32 Since midkine is a secreted protein, serum midkine levels are expected to increase when tumor tissues largely express midkine.
The aim of our study was to show the associations between cachexia due to weight loss and cachexia due to APRPs, albumin, C-reactive protein (CRP) and cytokines and its receptor (IL-1α, IL-1β, IL-6, IL-10, TNF-α, IL-8, VEGF-A, VEGF-C, and VEGFR1), midkine, and hormones (adiponectin, leptin, and ghrelin) in patients with various gastrointestinal system cancers.
Materials and methods
Patients
A total of 148 patients with gastrointestinal cancer (32 esophageal cancer type, 31 gastric cancer, 32 colon cancer, 18 pancreatic cancers, 35 rectum cancer) were enrolled in our study. Exclusion criteria included previous treatment by chemotherapy, radiotherapy, or a major operation history 6 months before the recovery; brain metastasis; second malignancy; acute or chronic infections; dysphagia; other primary cachectic states (i.e. congestive pulmonary disease and cirrhosis); elevated bilirubin or liver enzymes (>2 of the upper normal reference value); renal failure (creatinine >2 mg/dl); history of eating disorders; or gastrectomy.
Demographic clinical and anthropometric data were collected during the recovery period. All pathology reports were reviewed, and data on tumor histology were recorded. Stage was defined according to the 1997 American Joint Committee on Cancer Staging System. 33
We used sera from blood donors considered healthy on the basis of routine blood tests, to obtain the reference values in this study. The reference group consisted of 40 individuals, 18 females and 22 males, with a median age of 41 years (range 37–71 years). Healthy samples obtained from 40 normal healthy volunteers were used as controls. The absence of disease such as infection and asymptomatic early adenocarcinoma and adenoma was assessed by clinical history, physical examination, routine laboratory tests, including liver and renal function tests, and colonoscopy.
Body mass index (BMI) was calculated as weight (kg) divided by height (m2), and cachexia was defined as ≥5% reduction in BMI at the time of recovery. The study protocol was approved by the medical Ethics Committee of Yuzuncu Yı;l University, Medical Faculty Hospital, Van, and was in accordance with the ethical standards formulated in the Helsinki Declaration of 1975. Informed consent was obtained from all subjects.
Biochemical methods
The concentrations of all parameters in the examined samples were measured in sera obtained from blood drawn in the fasting state, clotted (15 min, room temperature) and centrifuged (15 min, 1000 g). The serum samples were then immediately frozen at −80°C until further analysis (except albumin, CRP, and midkine). The albumin concentrations were measured colorimetrically as a complex of albumin with bromocresol blue dye under acidic conditions. High-sensitive CRP was determined by the immunonephelometry (Behring Nephelometer II). Serum midkine concentrations were assayed with indirect enzyme-linked immunosorbent assay (ELISA; R&D Systems, USA) and anti-human midkine polyclonal antibodies were used. The serum concentrations of IL-1α, IL-1β, IL-6, IL-8, and TNF-α were assayed using a validated commercial ELISA (Quantikine R&D Systems, Minneapolis, MN, USA). The IL-10 levels were measured using the Endogen Inc. assay (Cambridge). The concentrations of VEGF-A, VEGF-C, and VEGFR1 were measured in duplicate with a commercially available quantitative sandwich enzyme immunoassay kit (R&D Systems, USA). Adiponectin, ghrelin, and leptin concentrations were determined using radioimmunoassay kits (Linco Research, St. Charles, MO, USA).
Statistical analysis
Data are presented as mean ± SD. Comparisons were performed with the nonparametric Mann-Whitney U test for continuous variables and with the χ2 tests for categorical data. The comparison of all cancer types and control samples was done using one-way analysis of variance (ANOVA) test. Differences between groups were determined using the log-rank test. Two-sided p values <0.05 were considered statistically significant.
Results
There were no differences in terms of age between the patients and the control groups (p > 0.05). All parameters and all groups were examined in terms of age and there were no significant differences (p > 0.05). There were no gender differences in plasma leptin levels (p > 0.05) in all patients.
We found significantly higher levels of the serum CRP, IL-1α, IL-1β, IL-6, IL-8, IL-10, TNF-α, VEGF-A, VEGF-C, VEGFR1, and leptin concentrations in patients with esophageal, gastric, pancreas, colon, and rectum cancer than the controls (p < 0.001, p < 0.0001). Otherwise, we found lower levels of the serum albumin, midkine, adiponectin, and ghrelin in patients with esophageal, gastric, pancreas, colon, and rectum compared to the controls (p < 0.05, p < 0.01, p < 0.001; Tables 1–5).
Concentrations of all parameters in patients with esophageal cancer and in controls

BMI: body mass index, CRP: C-reactive protein, IL: interleukin, TNF: tumor necrosis factor, VEGF: vascular endothelial growth factor, VEGFR1: VEGF receptor 1.
Concentrations of all parameters in patients with gastric cancer and in controls
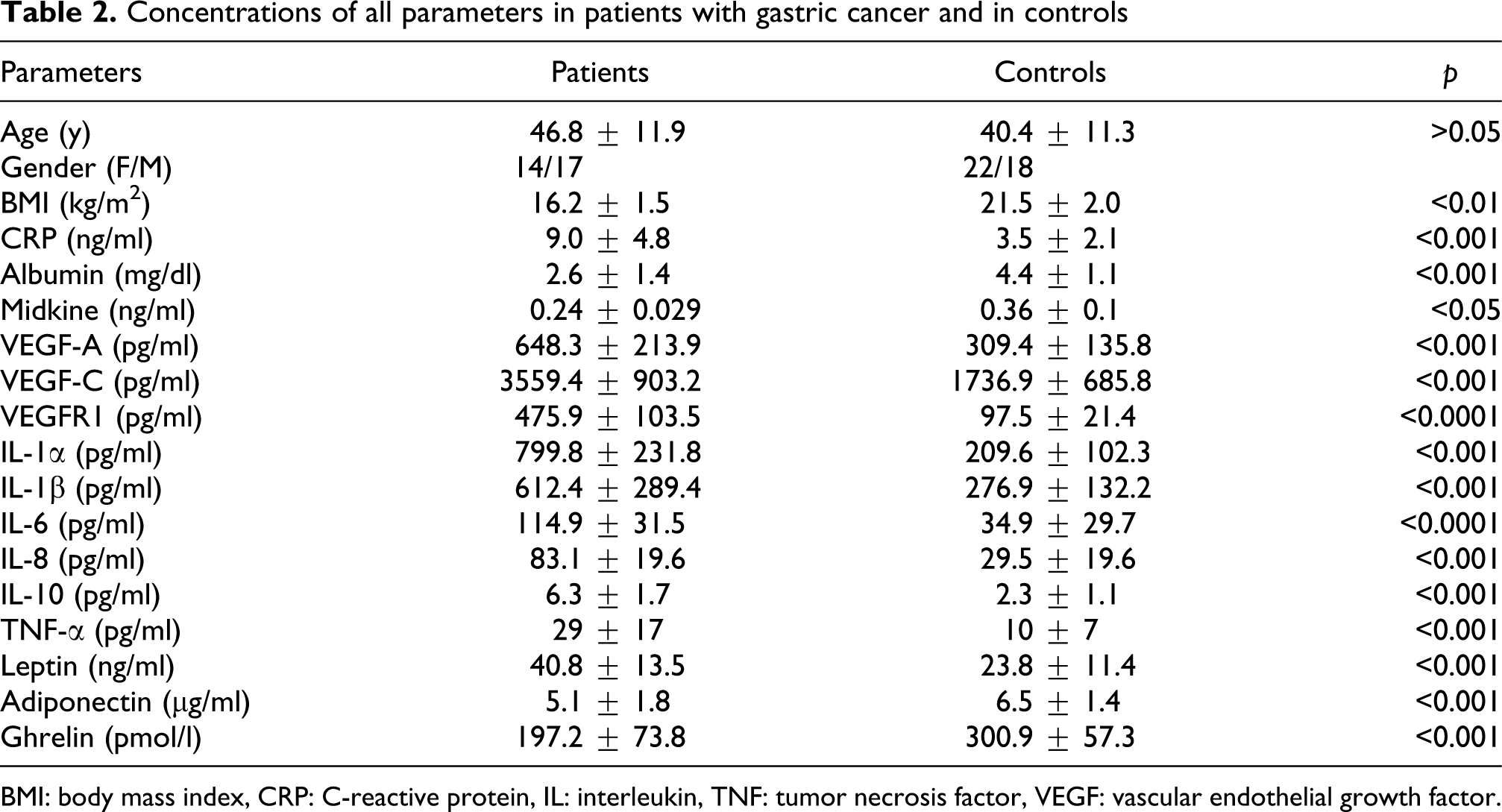
BMI: body mass index, CRP: C-reactive protein, IL: interleukin, TNF: tumor necrosis factor, VEGF: vascular endothelial growth factor.
Concentrations of all parameters in patients with pancreas cancer and in controls
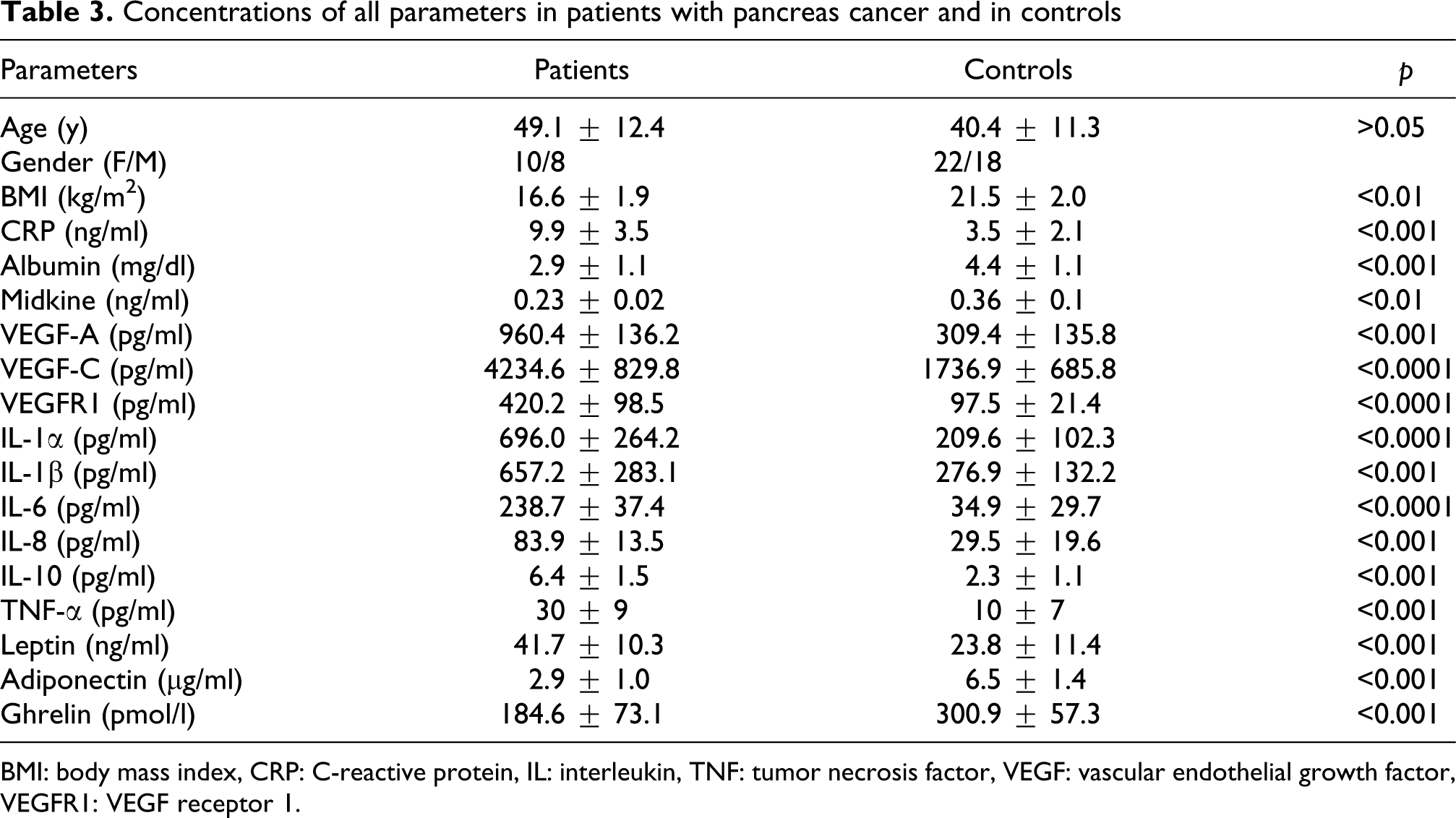
BMI: body mass index, CRP: C-reactive protein, IL: interleukin, TNF: tumor necrosis factor, VEGF: vascular endothelial growth factor, VEGFR1: VEGF receptor 1.
Concentrations of all parameters in patients with colon cancer and in controls
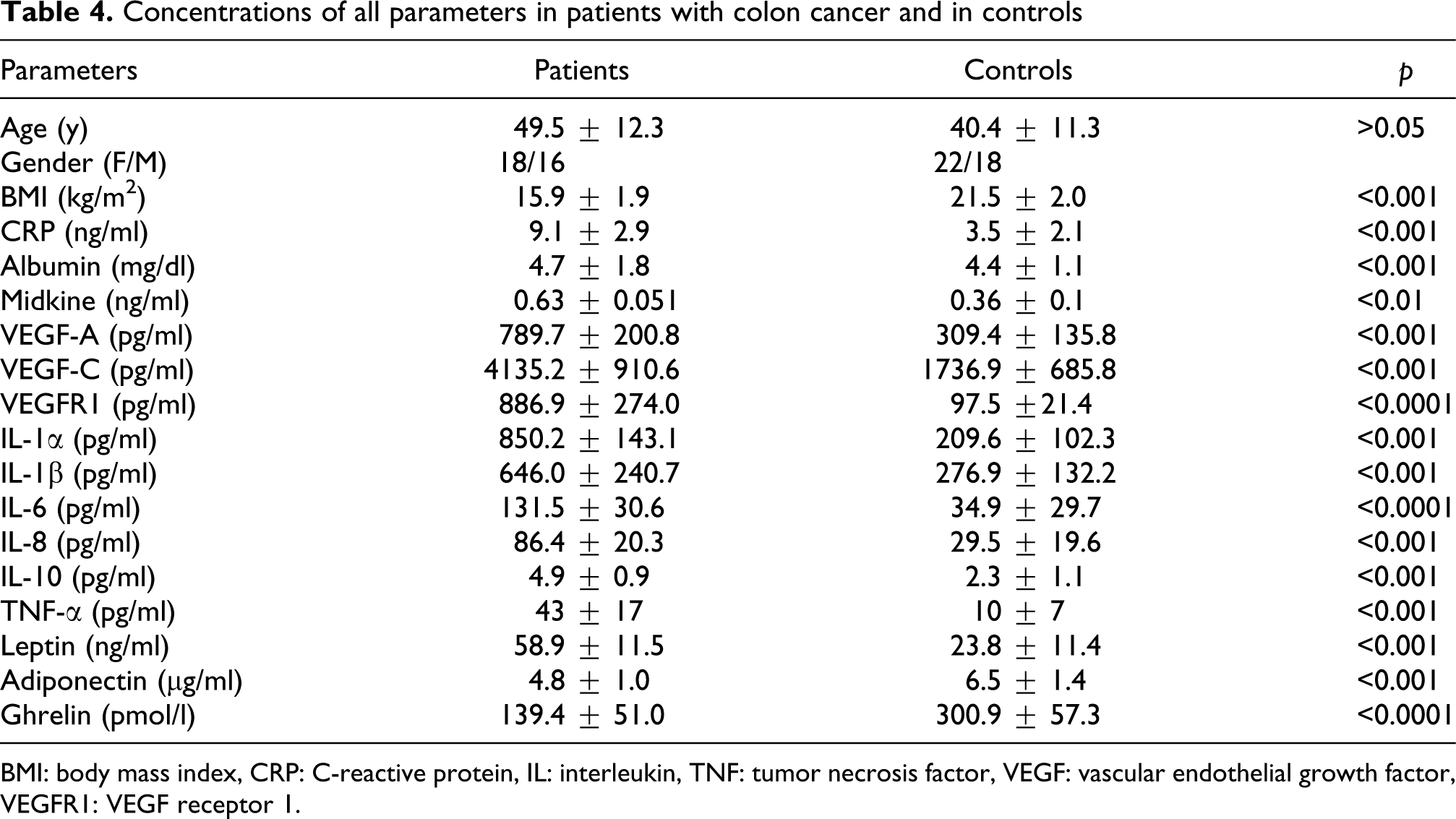
BMI: body mass index, CRP: C-reactive protein, IL: interleukin, TNF: tumor necrosis factor, VEGF: vascular endothelial growth factor, VEGFR1: VEGF receptor 1.
Concentrations of all parameters in patients with rectum cancer and in controls

BMI: body mass index, CRP: C-reactive protein, IL: interleukin, TNF: tumor necrosis factor, VEGF: vascular endothelial growth factor, VEGFR1: VEGF receptor 1.
There were no gender differences in plasma leptin, adiponectin, ghrelin levels, and BMI index (p > 0.05) in all patients (Table 6).
Hormone levels in female and male patients

BMI: body mass index.
We found the correlation between BMI loss and adiponectin levels in all patients (p < 0.01, r = 0.71). Also, we found a positive correlation between midkine and albumin; similarly between both BMI loss and plasma leptin levels; BMI loss and midkine; BMI loss and albumin; and IL-6 and TNF-α in all patients (respectively, p < 0.001, r = 0.69, r = 0.71, r = 0.59, r = 0.63, r = 0.75).
There was significant positive correlation between BMI loss and VEGF-A and VEGFR1 as well as VEGF-A and IL-1 (respectively, p < 0.001, r = 0.78, r = 0.71).
Correlation between VEGF-A and IL-6 was similarly statistically significant (p < 0.001, r = 0.74).
Discussion
In our study, we determined the associations between acute-phase response and proinflammatory cytokines, cytokine, hormones and cancer-related cachexia in patients with various gastrointestinal system cancer types.
Systemic inflammation is a nonspecific procedure of many cancer types. Association between acute-phase-related proteins and accelerated weight loss has been characterized only in a few cancer types including pancreatic and lung cancers and in melanoma. 31
Decreased albumin concentrations are associated with cachexia and gastrointestinal system cancers. Hypoalbuminemia has newly been showed to be a predictive factor of helpless responsiveness.34,35 Our results showed that a weight-loss-dependent association with cachexia may assist the association of hypoalbuminemia with cachexia and all types of gastrointestinal system cancers. The decrease in transferring concentrations seems to be specially weight loss dependent. The ongoing systemic inflammatory response described in terms of CRP concentrations has newly carried some interest as an easy-to measure and well-standardized outcome predictor.36,37
Similar to hardy weight loss, 10 an elevation in CRP concentration has been related to increased importance of primary tumor and has been associated with deficient survival.36,37 Our results may support the association of CRP concentrations and all cancer type-related cachexia.
The association of CRP with the proangiogenic environment may contribute to adverse effects together with CRP elevation. 38
Our results describe a positive correlation between CRP, IL-8 and midkine, which have proangiogenic properties in all cancer types . 39 Similarly CRP and VEGF correlation has been determined and these results may further support this hypothesis. It is also of interest that similar to CRP,36,37 circulating midkine 10 and IL-8 40 have been found to reflect lymph node involvement in esophageal squamous cell carcinoma. The concentrations of IL-6 and midkine are independently associated with patients weight status, among IL-1, IL-8 and VEGF in cachectic cancer patients. TNF-α, IL-1, and IL-6 are the key cytokines involved in cancer-related cachexia. However, apart from IL-6, alterations in their systemic levels are seldom determined. 31 As experimental cytokine-directed anti-cachectic therapies yielded moderate results, 31 there is a need for finding other mediators of cancer cachexia.5,31
We found midkine and VEGF to be the independent predictors of weight loss in patients with gastrointestinal system cancer. Our results supply evidence for an association of midkine and VEGF with systemic inflammation and malnutrition, supporting a probably involvement of these cytokines in the pathogenesis of cachexia. However, only the concentrations of VEGF and leptin, but not midkine, are associated with weight loss in the examined cohort of cancer patients. Midkine was related to inflammation and was correlated with albumin concentration, while the associations were not affected by cachexia. These together may further indicate VEGF as a procachectic cytokine, corroborating the findings of other authors, showing VEGF associations with standard procachectic cytokines. IL-1 and IL-6 have been propagated in the regulation of VEGF expression, 41 while anti-TNF-α treatment (infliximab) has been described to decrease serum VEGF concentrations. 42
We found that VEGF correlated with IL-1 and IL-6 merely in cachectic gastrointestinal cancer patients. Although the involvement of midkine in inflammation is well documented, 43 only a weak correlation between midkine and CRP in cancer patients has been reported. 44
Adiponectin levels are reported to be inversely correlated with body weight. Thus, voluntary weight loss, as well as anorexia nervosa, is associated with increased adiponectin levels.14,17,45 However, in our study, we found no correlation between BMI loss and adiponectin levels. Adiponectin levels are regulated essentially by changes in the adipose tissue. 45 The lack of association between adiponectin levels and weight loss may entirely reflect the preservation of adipose tissue. Recent studies have shown that the inhibition of adiponectin secretion from adipocytes by various cytokines, including TNF-α.21,46 The lack of elevation of adiponectin levels after cancer cachexia may reflect altered regulation of adiponectin in this condition. Interestingly, lower adiponectin levels were also found in a cohort of cachectic patients with advanced stage of lung cancer compared with healthy volunteers. 47 Elevated levels of total or active ghrelin in cancer cachexia have been reported in cohorts of primarily male lung cancer patients. 26 In our study, we report on increased ghrelin levels in a cohort of gastric and colon cancer patients. Specially, high levels of ghrelin were also found among a significant number of cachectic lung cancer patients. 26 Our results suggest that the measurement of ghrelin levels may have important clinical implications in treating cancer-related cachexia syndrome. Although leptin levels are associated with weight loss after fasting, 48 associations between leptin levels and cancer cachexia are not yet completely elucidated. Thus, lower leptin levels were found in patients with gastrointestinal system cancers, regardless of the degree of weight loss. 49 However, association between leptin levels and weight loss was noted in a cohort of lung 50 and pancreatic cancer patients. 29 We also found a correlation between leptin levels and cancer-induced weight loss, especially in patients with all types of gastrointestinal system cancers. Our results demonstrating a weight-loss-dependent association with cachexia may support the association of leptin with cachexia. Moreover, leptin levels have positive correlation with IL-6 levels and CRP in our study, in all patient groups. This situation may explain that high IL-6 levels related with the cancer progression and invasion. Pyrogenic activity of this proinflammatory cytokine may be responsible for cachexia and weight loss. In addition, IL-6 stimulates the synthesis of APRP. So, adiponectin, ghrelin, and leptin levels have been accelerated by APRP levels. Association between weight loss and levels of proinflammatory cytokines, cytokines, APRP, adiponectin, ghrelin, and leptin levels has not been explained. We found that adiponectin, ghrelin, and leptin highly regulate the energy homeostasis such as cytokines, which affect various central mechanisms. 11 Our study revealed an association between cytokine and proinflammatory cell concentrations and APRP in patients with gastrointestinal system cancer. In our study, there is an association between these parameters and in the levels of these hormones, which confirms our hypothesis and is consistent with our previous study. 51
In conclusion, our results provide evidence for an association between all types of gastrointestinal system cancer-related cachexia and changes in the concentrations of APRPs, cytokines, and hormones. More studies should be performed to confirm this association between cachexia and APRP, cytokines and hormones in patients with gastrointestinal system cancers.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
