Abstract
Heat shock proteins (Hsp) play a complex role in cytoprotection, inflammation, and function of the immune system. They may be involved in pathogenesis of various diseases. Our aim was to determine circulating Hsp70 and anti-Hsp70 antibodies concentrations in patients with chronic spontaneous urticaria (CSU). Concentrations of Hsp70 in plasma and anti-Hsp70 antibodies in serum as well as serum C-reactive protein (CRP) were measured in CSU patients and in the controls. Plasma Hsp70 concentrations were significantly higher in CSU (all) and mild CSU patients as compared with the controls. Moderate–severe CSU patients tended to show higher Hsp70 concentration as compared with the controls, but not with mild activity of the disease. There were no significant differences in Hsp70 concentration between moderate–severe and mild CSU patients. Serum anti-Hsp70 antibodies concentrations were significantly higher in CSU (all) and mild CSU in comparison to the controls. Association was observed between anti-Hsp70 antibodies and increased CRP concentration; however, no correlation between anti-Hsp70 and Hsp70 concentrations was seen in the patients. It seems that up-regulation of Hsp70 in CSU may induce marked increase in anti-Hsp70 antibodies production, which are accompanied by parallel changes in CRP concentration. We suggest that Hsp may be released in CSU in response to stressful stimuli, such as inflammation.
Keywords
Introduction
Chronic spontaneous urticaria (CSU) is associated with activation of (auto-)immune response and the inflammatory processes. The multifactorial etiopathogenesis of CSU is not completely understood; however, the environmental challenges, such as distress and infection, are among the suspected causative factors.1–5
Heat shock proteins (Hsp) are highly conserved cytoprotective and immunomodulatory molecules, which can be released from viable cells in response to several stressors, including mental and physiological stress, infection, inflammation, or other environmental challenges.6–8
Hsp play a complex role in inflammation and function of the immune system. They can promote cytokines production and expression of adhesion molecules, and activate both humoral and cellular immune response, as well the complement system.6,9,10 Overexpression of Hsp may be involved in pathogenesis of various disorders.7,11
The 70-kDa Hsp (Hsp70) is one of the best studied members of the Hsp family. Hsp70 is up-regulated in inflamed tissue presenting itself as a very sensitive indicator of cellular stress. It has been suggested that autoimmunity to Hsp70 may be present in many immune-mediated diseases.7,11,12
This has been the first study to look for the Hsp status in CSU. We investigated circulating Hsp70 and anti-Hsp70 antibody concentrations in patients with CSU and their association with C-reactive protein (CRP) concentration, a nonspecific inflammatory marker of the disease activity/severity.
Materials and methods
Active 58 CSU patients (18 men and 40 women; median age: 38 years, range: 24–52; median disease duration of 3 years) and 22 sex-, age- and body mass index (BMI)- (<30) matched healthy subjects were enrolled in the study.
Each patient underwent the tests described previously. 2 The study comprised 32 patients with mild and 26 patients with moderate–severe activity of the disease, according to Urticaria Activity Score (UAS), 13 as follows: mild (0–8), moderate (9–16), and severe (17–24), which was estimated during 4 days and on the blood sampling day. H1-antihistamines were withdrawn at least 4 days before blood collection.
The Ethics Committee of the Medical University of Silesia (KNW-1-041/K/6/K) approved the study. All the subjects gave informed, signed consent to participate in the study.
Blood collection
Blood samples were taken on fasting, using tubes with anticoagulant (EDTA) for plasma samples and without it for serum samples. Plasma and serum obtained by centrifugation were stored at −85°C until the analysis.
Assay of Hsp70 in plasma and anti-Hsp70 antibodies in serum
Concentrations of Hsp70 and anti-Hsp70 antibodies were measured by ELISA method, using commercially available kits (Hsp70 high-sensitivity ELISA kit and anti-Hsp70 IgG/A/M ELISA kit by Enzo Life Sciences, Inc., NY, USA). The variance coefficients for intra-assay and inter-assay were below 8% and 10%, respectively.
Serum CRP assay
CRP concentrations were measured by Roche/Hitachi cobas c system. Normal lab ranges: lower than 5.0 mg/L.
Statistical analysis
Normal distribution of data was determined using Shapiro–Wilk’s test. Independent data were compared with the use of non-parametric U Mann–Whitney test. The Spearman’s rank test was used for correlations. The P < 0.05 was considered statistically significant.
Results
Plasma Hsp70 concentration
We observed significantly higher Hsp70 concentration in CSU as compared with the controls (median: 0.38 vs 0.29 ng/mL; P = 0.017) (Figure 1).
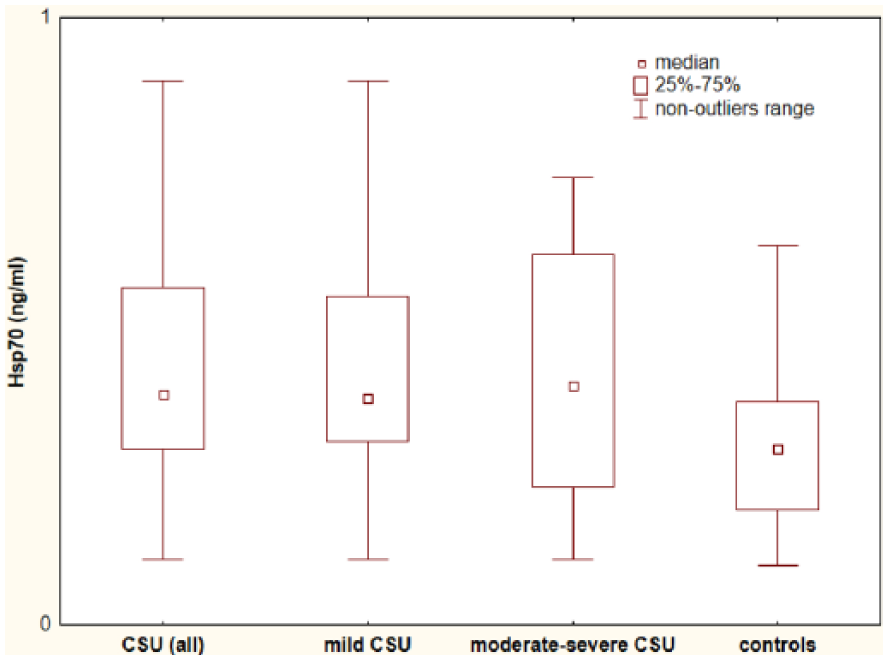
Plasma Hsp70 concentration in chronic spontaneous urticaria (CSU) patients with different disease activity and in the healthy subjects. CSU versus controls, P < 0.05; moderate–severe CSU patients versus mild CSU versus controls, P > 0.05 and P < 0.05, respectively; mild CSU versus controls, P < 0.05.
Hsp70 concentration was significantly higher in mild CSU as compared with the controls (median: 0.37 vs 0.29 ng/mL, P = 0.025).
No significant differences in Hsp70 concentration between moderate–severe CSU and mild CSU (median: 0.39 vs 0.37 ng/mL, P = 0.77) were observed; although moderate–severe CSU tended toward higher plasma Hsp70 concentration as compared with the controls (median: 0.39 vs 0.29 ng/mL, P = 0.054, near borderline significance).
We did not observe any significant differences in Hsp70 concentrations between ASST(+) and ASST(–) CSU patients (selected according to the similar UAS).
Serum anti-Hsp70 antibodies concentration
Anti-Hsp70 antibodies concentration was significantly higher in CSU (as a whole) in comparison to the controls (median: 148.5 vs 53.36 ug/mL; P = 0.0008) (Figure 2).

Serum anti-Hsp70 concentration in chronic spontaneous urticaria (CSU) patients with different disease activity and in the healthy subjects. CSU versus controls, P < 0.001; moderate–severe CSU patients versus mild CSU versus controls, P > 0.05 and P < 0.001, respectively; mild CSU versus controls, P < 0.05.
Anti-Hsp70 antibodies concentration was significantly higher in moderate–severe CSU as compared with the controls, but not with mild the disease activity (median: 188.67 vs 53.36 vs 126.72 ;ug/mL, P = 0.0006 and P = 0.069, respectively). Although, anti-Hsp70 antibodies concentration in mild CSU activity was significantly higher than in the controls (126.72 vs 53.36 ug/mL, P = 0.011).
Serum CRP concentration
We observed significantly higher CRP concentration in CSU patients (all groups) as compared with the controls (median: 3.7 vs 0.35 mg/L; P < 0.0001). In addition, there were significant differences in CRP concentration between CSU with mild, moderate–severe activity and the controls (median: 1.35 vs 9.8 vs 0.35 mg/L, respectively; P < 0.001).
Correlation between values of CRP, Hsp70, anti-Hsp70 antibodies and UAS in CSU
Serum CRP concentration correlated weakly with anti-Hsp70 antibodies concentration (R = 0.3; P = 0.03) and strongly with UAS value (R = 0.9; P = 0.000000). There were no significant correlations between concentrations of CRP and Hsp70 as well as Hsp70 and anti-Hsp70 antibodies. Also, the UAS value correlated weakly with anti-Hsp70 antibodies concentration (R = 0.3; P = 0.03), but not with Hsp70 concentration.
Discussion
In this study, we found significantly higher Hsp70 concentration in plasma of CSU (all group) and mild CSU patients as compared with the controls. There were no significant differences in plasma Hsp70 concentration between moderate–severe and mild disease activity; although moderate–severe CSU patients tended to show higher plasma Hsp70 concentration as compared with the controls (P = 0.054). These data suggest that CSU may be associated with enhanced Hsp70 expression.
Nevertheless, the mechanism and clinical significance of the increased circulating Hsp70 concentration in CSU remain unknown.
Theoretically, elevation of Hsp70 concentrations in CSU patients may be explained by mechanisms associated with the cell damage or active release in response to different stresses including inflammation, infection, or psychological stress.7,8,14
It has been reported that exosomes may contribute to the release of Hsps.15,16 The processes were facilitated by freeze thawing of samples. 15 Exosomes contribute to the release of HSP70 from different cell types including human peripheral blood mononuclear cells (PBMCs). 16
Interestingly, urticarial lesions are often infiltrated by CD4(+) lymphocytes consisting of a mixture of T(H)1 and T(H)2 subtypes and monocytes plus other cells including eosinophils. 17
It has been speculated that cellular overexpression of Hsp70 is a protective mechanism, activated to increase stress tolerance and to participate in the repair processes. 18 It addition, Hsp can induce anti-inflammatory immunoregulatory T-cell responses. 12
Recent studies have demonstrated a dual role for Hsp70 as both, a chaperone and cytokine, induce potent pro-inflammatory response in human monocytes. 19 Hsp70 may act like a cytokine to activate NF-κB, which stimulate IL-6, IL-1, and TNF-α production through a CD14-dependent pathway. 19 However, it is known that CSU is associated with up-regulation of IL-6 signaling system and of other cytokines. 20 Thus, it is tempting to speculate that Hsp70 activation may be part of a systemic inflammatory response in CSU.
It remains unclear whether up-regulation of Hsp70 in CSU is anti-inflammatory, pro-inflammatory, or merely an epiphenomenon.
In our study, serum anti-Hsp70 concentration was significantly higher in CSU patients as compared with the controls. This seems especially interesting because not all immune-inflammatory processes associated with Hsp70 overexpression are accompanied by elevated anti-Hsp70 antibodies concentrations. It has been proposed that autoantibodies against Hsp may be involved in pathogenesis and/or prognosis of some diseases. Hsp are highly conserved proteins, and there is close homology between Hsp in pathogens and in mammalian cells. Exposure to Hsp from microbial pathogens, including normal gut microflora, is very common.7,8,11,12
They may trigger humoral and cellular autoimmune response as well as provide link between autoimmunity and infections.8,21 In addition, it has been suggested that local accumulation of self Hsp in response to microbial pathogens may subsequently provide a stimulus for autoimmunity activation. 21 Although autoimmunity to Hsp70 was implicated in certain autoimmune diseases and other conditions, the exact role of anti-Hsp70 antibodies is not known. Interestingly, the relationship between infections and the origin or maintenance/amplification of the chronic urticarial processes has been proposed. 5
It has been suggested that Hsps may act as autoantigens, triggering both cell-mediated and humoral immunity. 22 Taken together, it is tempting to speculate that Hsp70 may serve as autoantigens and trigger autoimmune CSU, similarly to other immune-inflammatory diseases.
The association between higher anti-Hsp70 antibodies and CRP concentrations in CSU is an interesting observation with no definite explanation. CRP has been considered a marker of a systemic inflammatory response and CSU activity/severity. 23 It seems that systemic inflammation may be—at least in part—responsible for increased circulating anti-Hsp70 antibodies concentration in CSU patients, as indicated by the significant positive correlations between concentrations of anti-Hsp70 antibodies and CRP. These data suggest that higher anti-Hsp70 antibodies concentrations found in CSU patients compared to the controls could reflect their inflammatory status. Anti-Hsp70 might thus be considered as an important marker in a clinical context of the disease activity. Importantly, serum anti-Hsp70 concentration in mild CSU patients was significantly higher than in the health subjects. In addition, anti-Hsp70 serum concentration was significantly higher in moderate–severe CSU activity as compared with the controls, but not with mild the disease activity. Precise relation between circulating anti-Hsp70 antibodies and the disease activity should be further investigated.
It seems that Hsp70 and its antibodies may increase in CSU patients upon exposure to a number of stressors associated with urticarial inflammation, such as inflammatory mediators.
In addition, it has been suggested that local hsp70-antigen-antibody complexes may activate the pro-inflammatory cytokine cascade, which may contribute to pathological processes. 24 It is important to investigate whether such complexes accumulate at the site of urticarial inflammation. In has also been speculated that pro-inflammatory soluble heat shock protein–heat shock protein antibody immune complexes might be present in peripheral circulation. 25
It has been shown that Hsp70 expression is generally correlated with the grade of inflammation. 26 However, there was no association between concentrations of Hsp70 and CRP, probably due to a relatively small number of patients. This study did not explore the sources of Hsp70 in CSU. Hsp70 may be released from almost all types of cells. Elevated circulating Hsp70 possibly reflects the tissue damage or active release from cells in response to stressful insults. 14 It seems that circulating Hsp70 in CSU possibly reflects its overexpression in the skin and might be released from cells involved in the urticarial inflammatory response.
Conclusion
The main findings in this study were increased plasma Hsp70 and serum anti-Hsp70 concentrations in patients with CSU, as compared with the controls. Anti-Hsp70 antibodies concentration generally correlated with the grade of inflammation. It seems that up-regulation of Hsp70 in CSU may induce marked increase in anti-Hsp70 antibodies production, which are accompanied by parallel changes in CRP concentration. We suggest that the Hsp may be released in CSU as a response to stressful stimuli, such as inflammation. However, further studies should determine whether circulating Hsp70 and anti-Hsp70 antibodies play a role in the CSU pathogenesis or if they are just an epiphenomenon.
Footnotes
Acknowledgements
A.K.-Z. conceived and designed the study as well as reviewed the manuscript. A.D.-B. performed the lab and statistical data analysis and wrote the manuscript, contributed to data analysis and interpretation. K.T.-G. performed the lab. K.B. and A.S.-F. provided clinical data. A.G. collected and provided clinical data. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a research grant from the Committee for Scientific Research KNW-1-041/K/6/K (KNW-640-2-1-004/15).
